Chapter 14 Solid Fuel Combustion Mechanisms ME 460

Chapter 14: Solid Fuel Combustion Mechanisms ME 460 Fuels and Combustion Drying Devolatilization Char Combustion Ash Formation

Drying Devolatilization Char Combustion Ash Formation Solid Fuel Combustion • There are three stages of solid fuel combustion: ▫ drying ▫ devolatilization (pyrolysis) ▫ char combustion • The relative significance of each stage depends upon the fuel and is a function of the proximate analysis (re: proximate vs. ultimate analysis). • Examples are peat, wood, lignite, bituminous coal, finely pulverized anthracite coal.

Drying pores containing free water (0. 01 -30 μm) absorbed water lignite <40% wood 5 - 50% Bi = hd/kp < 0. 2 for wood: b=bound water content fsp = fiber saturation point

Drying Devolatilization Char Combustion Ash Formation Drying, Small Particles note that the fiber saturation point for wood is about 30% of the dry mass! • • hsorp is neglected when MC exceeds 30% (why? ) fuel moisture is a significant heat cost hfg ≈ 2400 k. J/kg at atmospheric pressure if the fuel has a high water content it is much better to dry the fuel before combustion

Drying Devolatilization Char Combustion Ash Formation Drying, Small Particles - Analysis • Utilizing first law analysis, ideal gas assumptions ( du = cpdt), and simple heat transfer substitutions for radiation and convection yields the above relationship for ‘time to dry’ • It is also important to note that with high temperatures with combustion the vapor moving away from the particle is superheated. Thus corrections must be applied (Bird, 2007)







Drying Devolatilization Char Combustion Ash Formation Drying, Large Particles • The assumption of uniform particle temperature and uniform drying is not valid. • Because of size, moisture inside the particle is driven off while volatiles are evaporating on the surface and charring may be occurring elsewhere. • High pressure may force water into the center of the structure. • Pyrolysis occurs at the outer edges and works inward. • The entire process is complicated and difficult to model with simple relationships.

wood - top left coal – bottom left pulverized coal – bottom left
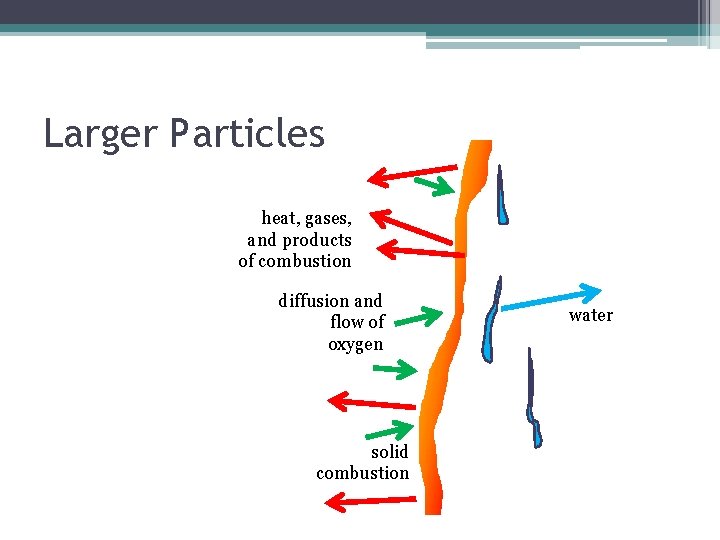
Larger Particles heat, gases, and products of combustion diffusion and flow of oxygen solid combustion water
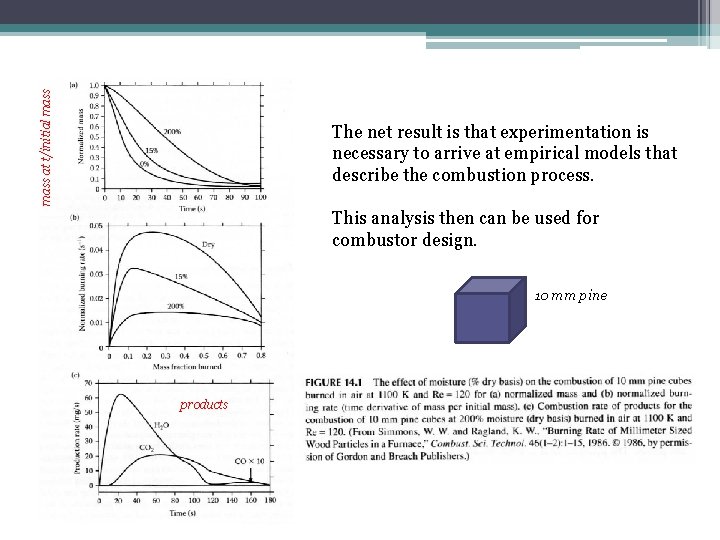
mass at t/initial mass The net result is that experimentation is necessary to arrive at empirical models that describe the combustion process. This analysis then can be used for combustor design. 10 mm pine products

Devolatilization – qualitative description ide bon car particle swelling HE A T combustion s ile t ola v les ati vol Temperatures noted are a function of fuel type with wood (cellulose, hemicelulose, lignin, and terpines) x dio ter wa en g oxy • Decomposition and devolatilization begins about 250 -400 ⁰C. • Referred to pyrolysis stage because of lack of oxygen due to flow of volatiles. • Volatiles surrounding particle ignite if temperature exceeds auto-ignition temperature of 400 – 600 ⁰ C. • Flame temperature initially low due to vaporization of water, increasing with time as water is eliminated reaching 900 ⁰ C until only char (fixed carbon) and ash remain.

Devolatilization –what to expect time to ‘devolatilize’ = f (initial mass, water content, volatile content, ash content, volatile energy in particle, temperature of particle. . . )

Devolatilization first order Arrhenius form rate constant continuity upon integration of 14. 10 analysis

Devolatilization - Example
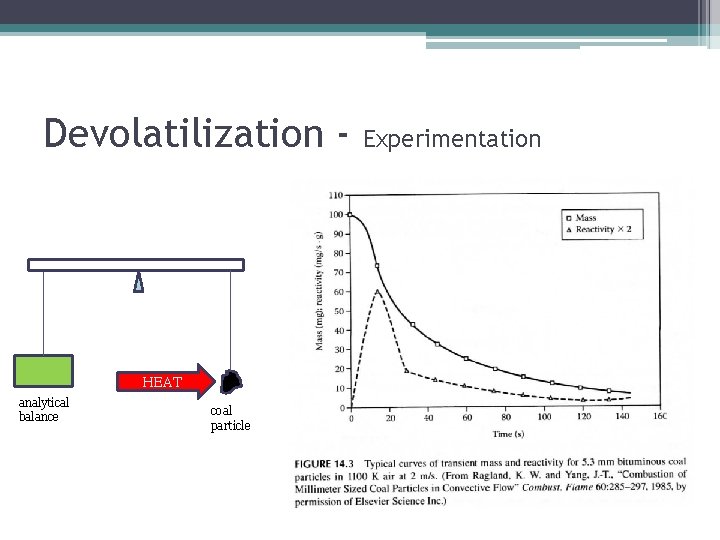
Devolatilization - HEAT analytical balance coal particle Experimentation

Devolatilization - Ignition 450 ⁰F

Char Combustion – what to expect f (ratio of carbon to oxygen, surface area of particle, kinetic rate constant, oxygen density)
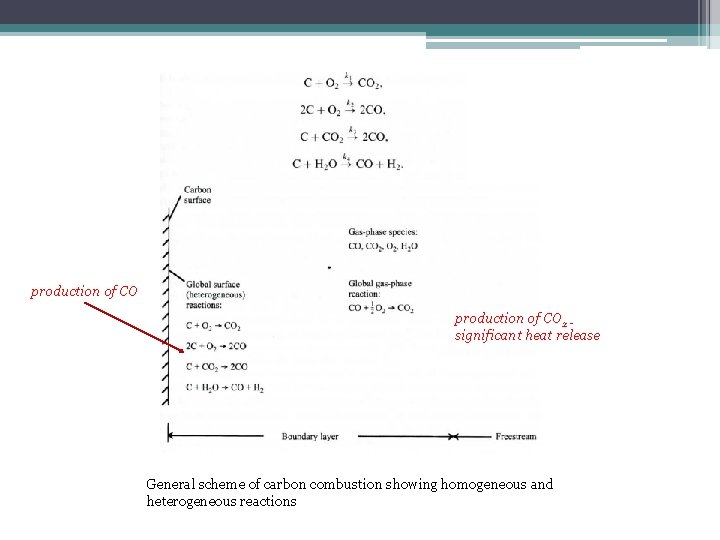
production of CO 2 significant heat release General scheme of carbon combustion showing homogeneous and heterogeneous reactions

Char Combustion Char combustion is the oxidation of free carbon producing carbon monoxide f (ratio of carbon to oxygen, surface area of particle, kinetic rate constant, oxygen density)

Char Combustion - burnout re: equation 14. 14 – previous overhead
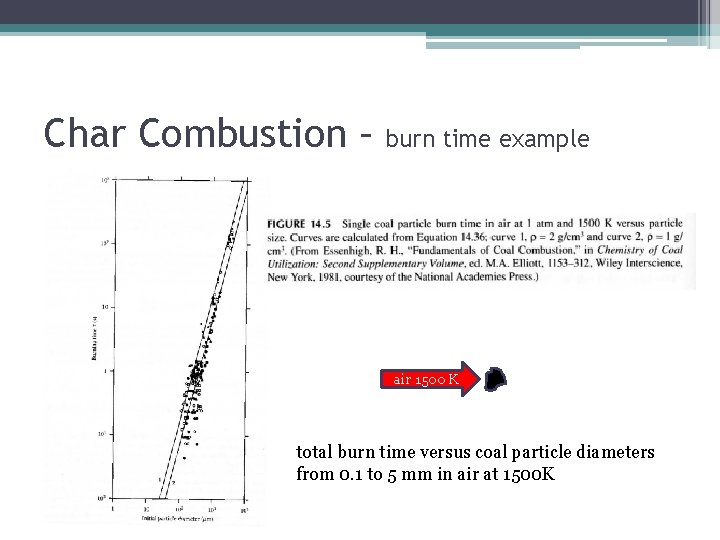
Char Combustion – burn time example air 1500 K total burn time versus coal particle diameters from 0. 1 to 5 mm in air at 1500 K
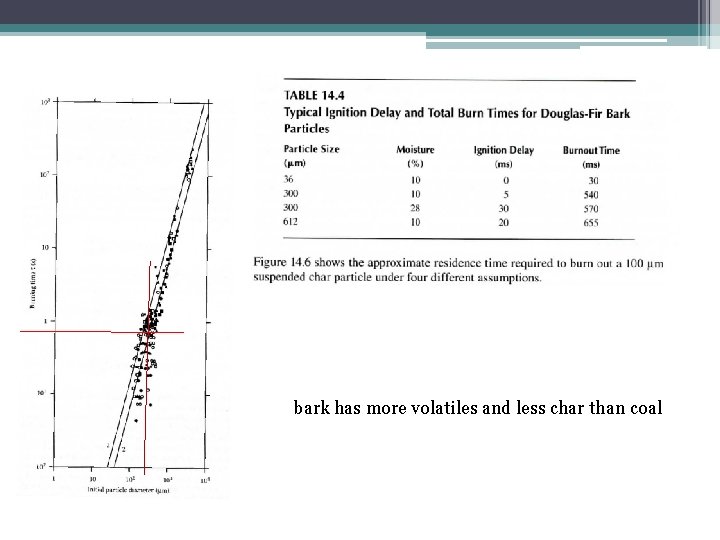
bark has more volatiles and less char than coal

Char Surface Temperature steady state energy balance heat generation at the surface to heat loss by convection and radiation


burnout rate

Recapitulation 1. 2. 3. 4. Drying De-volatilization Char Combustion Ash Formation

Drying Devolatilization Char Combustion Ash Formation • Mineral mass in biomass 1 -6%, coal up to 50% • As char burns the minerals are converted to a layer of ash on the surface • Ash layer can have a significant effect on heat capacity, radiation heat transfer, and catalysis • mineral matter can deposit on heat transfer surfaces and foul heat transfer process • minerals can also significantly influence the nature of emissions.

End
- Slides: 34