Chapter 14 reversible reaction reaction that can proceed

Chapter 14 reversible reaction- reaction that can proceed in both directions (forward and reverse) dynamic equilibrium- when the rate of the forward reaction equals the rate of the reverse reaction -does not mean the concentrations of reactants and products are equal they just remain constant -the rates are equal

a. A + b. B c. C + d. D equilibrium constant Kc = [C]c[D]d [A]a[B]b Kc= equilibrium constant with concentrations [] = concentration of reactant or product a, b, c, d = coefficient from balanced equation Example 14. 1 page 622 FP 14. 1 page 622

-if K is large (K >> 1), forward reaction is favored -more products than reactants when equilibrium is reached -if K is small (K <<1), reverse reaction is favored -more reactants than products when equilibrium is reached -if K ≈ 1 neither reaction is favored *at equilibrium

1) if equation is reversed, invert the Kc 2) if the coefficients are multiplied by a factor, raise the Kc to the same factor 3) if you add two or more individual equations to obtain an overall equation, multiply the equilibrium constants by each other to obtain the overall Kc Koverall = K 1 x K 2 **Koverall is the final equation given

Equilibrium constant and pressure -used with gaseous reactions -partial pressures (P) are used in place of concentrations Kp = (Pc. CPd. D) / (Pa. APb. B) Kp = Kc(RT)∆n Kp = equilibrium constant with partial pressures Kc = equilibrium constant with concentrations R = ideal gas constant (0. 08206 Latm/Kmol) T = temp (in kelvin) ∆n = c + d – (a +b) *coefficients from products and reactants *if ∆n = 0 then Kp = Kc

Reactions Involving Solids and Liquids -concentrations of solids and liquids do not change b/c they do not expand to fill a container -these values are not included in the equilibrium expression Ex- Ca. CO 3(s) ↔ Ca. O(s) + CO 2(g) Write an expression for the Kc of this equation. Kc = [CO 2] FP 14. 4 pg 630 Kc = [Cℓ 2]2 / ([HCℓ]4[O 2])
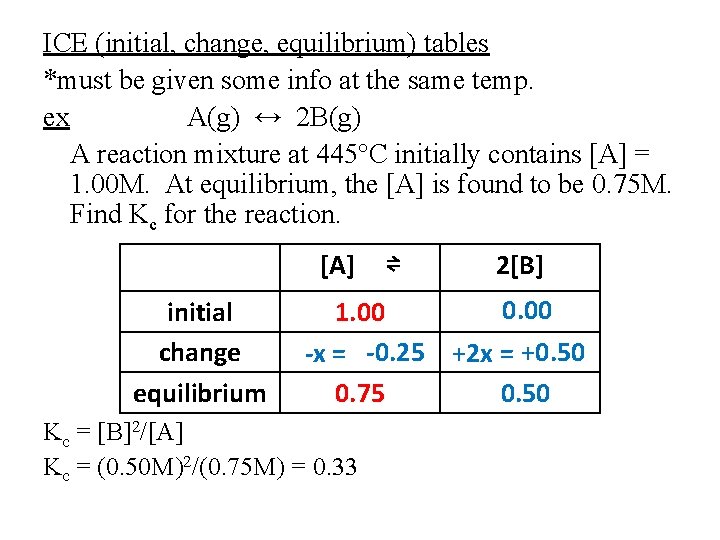
ICE (initial, change, equilibrium) tables *must be given some info at the same temp. ex A(g) ↔ 2 B(g) A reaction mixture at 445°C initially contains [A] = 1. 00 M. At equilibrium, the [A] is found to be 0. 75 M. Find Kc for the reaction. [A] initial change equilibrium ⇌ 2[B] 0. 00 1. 00 -x = -0. 25 +2 x = +0. 50 0. 75 0. 50 Kc = [B]2/[A] Kc = (0. 50 M)2/(0. 75 M) = 0. 33

Reaction Quotient (Qc or Qp) -does not have to be at equilibrium -will have different values as the reaction proceeds Qc = [C]c[D]d Qp = Pc. CPd. D [A]a[B]b Pa. APb. B Q < K reaction favors products Q > K reaction favors reactants Q = K reaction is at equilibrium page 635

Le Chatelier’s Principle -when a system at equilibrium is disturbed, the system shifts in a direction that minimizes the disturbance Ex- page 646 figure 14. 9 Effect of Concentration on Equilibrium -if [ ] reactants inc. , reaction shifts to right -if [ ] products inc. , reaction shifts to left -if [ ] reactants dec. , reaction shifts to left -if [ ] products dec. , reaction shifts to right

Ca. CO 3(s) ↔ Ca. O(s) + CO 2(g) What is the effect of adding additional CO 2 to the reaction mixture? -will shift to the left What is the effect of adding additional Ca. CO 3? -does not play a factor b/c solid no effect Try page 648 For Practice 14. 14

Effect of Volume on Equilibrium -if volume is dec. , reaction shifts to side with fewer moles of gas particles -if volume is inc. , reaction shifts to side with greater # of moles of gas particles -if equal # of moles and volume changes, no effect -adding inert gas to mixed has no effect Page 650

Effect of Temperature on Equilibrium In exothermic, heat is a product: -inc. temp. causes shift to left -dec. temp. causes shift to right In endothermic, heat is a reactant: -inc. temp. causes shift to right -dec. temp. causes shift to left
- Slides: 12