Chapter 14 Lecture INTRODUCTORY CHEMISTRY Concepts and Critical






























![Ionization of Water • Since [H+] is 1 × 1027 mol/L at 25 C, Ionization of Water • Since [H+] is 1 × 1027 mol/L at 25 C,](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-32.jpg)
![[H+] and [OH–] Relationship • At 25 C, [H+][OH–] = 1. 0 x 10– [H+] and [OH–] Relationship • At 25 C, [H+][OH–] = 1. 0 x 10–](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-33.jpg)


![Calculating [H+] from p. H • If we rearrange the p. H equation for Calculating [H+] from p. H • If we rearrange the p. H equation for](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-36.jpg)
![Advanced p. H Calculations • What is the p. H of blood with [H+] Advanced p. H Calculations • What is the p. H of blood with [H+]](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-37.jpg)










![Chapter Summary, Continued • In an aqueous solution, [H+][OH–] = 1. 0 x 10– Chapter Summary, Continued • In an aqueous solution, [H+][OH–] = 1. 0 x 10–](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-48.jpg)
- Slides: 48

Chapter 14 Lecture INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Chapter 14 Acids and Bases by Christopher G. Hamaker Illinois State University © 2014 Pearson Education, Inc.

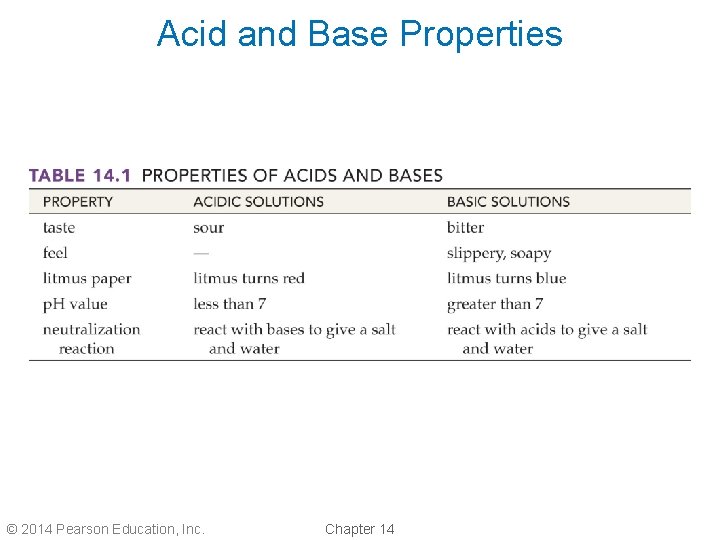
Properties of Acids • An acid is any substance that releases hydrogen ions, H+, into water. • Litmus paper turns red in the presence of hydrogen ions. Blue litmus is used to test for acids. • Acids have a sour taste; lemons, limes, and vinegar are acidic. © 2014 Pearson Education, Inc. Chapter 14

Properties of Bases • A base is a substance that releases hydroxide ions, OH –, into water. • Litmus paper turns blue in the presence of hydroxide ions. Red litmus is used to test for bases. • Bases have a slippery, soapy feel. • Bases also have a bitter taste; milk of magnesia is a base. © 2014 Pearson Education, Inc. Chapter 14

Acid and Base Properties © 2014 Pearson Education, Inc. Chapter 14

Acid–Base Neutralization • Recall that an acid and a base react with each other in a neutralization reaction. • When an acid and a base react, water and a salt are produced. • For example, nitric acid reacts with sodium hydroxide to produce sodium nitrate and water: HNO 3(aq) + Na. OH(aq) → Na. NO 3(aq) + H 2 O(l) © 2014 Pearson Education, Inc. Chapter 14

The p. H Scale • A p. H value expresses the acidity or basicity of a solution. • Most solutions have a p. H between 0 and 14. • Acidic solutions have a p. H less than 7. – As a solution becomes more acidic, the p. H decreases. • Basic solutions have a p. H greater than 7. – As a solution becomes more basic, the p. H increases. © 2014 Pearson Education, Inc. Chapter 14

Acid–Base Classifications of Solutions • A solution can be classified according to its p. H. • Strongly acidic solutions have a p. H less than 2. • Weakly acidic solutions have a p. H between 2 and 7. • Weakly basic solutions have a p. H between 7 and 12. • Strongly basic solutions have a p. H greater than 12. • Neutral solutions have a p. H of 7. © 2014 Pearson Education, Inc. Chapter 14

Buffers • A buffer is a solution that resists changes in p. H when an acid or a base is added. • A buffer is a solution of an aqueous weak acid and one of its salts: – Citric acid and sodium citrate make a buffer solution. • When an acid is added to the buffer, the citrate reacts with the acid to neutralize it. • When a base is added to the buffer, the citric acid reacts with the base to neutralize it. © 2014 Pearson Education, Inc. Chapter 14

Arrhenius Acids and Bases • Svante Arrhenius proposed the following definitions for acids and bases in 1884: – An Arrhenius acid is a substance that ionizes in water to produce hydrogen ions. – An Arrhenius base is a substance that ionizes in water to release hydroxide ions. • For example, HCl is an Arrhenius acid and Na. OH is an Arrhenius base. © 2014 Pearson Education, Inc. Chapter 14

Strengths of Acids • Acids have varying strengths. • The strength of an Arrhenius acid is measured by the degree of ionization in solution. • Ionization is the process by which molecules of a polar compound ionize to form cations and anions. • The acid HCl ionizes into H+ and Cl– ions in solution. © 2014 Pearson Education, Inc. Chapter 14

Strengths of Bases • Bases also have varying strengths. • The strength of an Arrhenius base is measured by the degree of dissociation in solution. • Dissociation is the process whereby already existing ions of an ionic compound simply separate. • A formula unit of Na. OH dissociates into Na+ and OH– ions in solution. © 2014 Pearson Education, Inc. Chapter 14

Strong and Weak Arrhenius Acids • Strong acids ionize extensively to release hydrogen ions into solution. – HCl is a strong acid and ionizes nearly 100%. • Weak acids only ionize slightly in solution. – HF is a weak acid and ionizes only about 1%. © 2014 Pearson Education, Inc. Chapter 14

Arrhenius Acids in Solution • All Arrhenius acids have a hydrogen atom bonded to an acid molecule by a polar bond. This bond is broken when the acid ionizes. • Polar water molecules help ionize the acid by pulling the hydrogen atom away: HCl(aq) + H 2 O(l) → H 3 O+(aq) + Cl–(aq) (~100%) HC 2 H 3 O 2(aq) + H 2 O(l) → H 3 O+(aq) + C 2 H 3 O 2–(aq) (~1%) • The hydronium ion, H 3 O+, is formed when a hydrogen ion attaches to a nonbonding electron pair on a water molecule. © 2014 Pearson Education, Inc. Chapter 14

Strong and Weak Arrhenius Bases • Strong bases dissociate extensively to release hydroxide ions into solution. – Na. OH is a strong base and dissociates nearly 100%. • Weak bases only ionize slightly in solution. – NH 4 OH is a weak base and only partially dissociates. © 2014 Pearson Education, Inc. Chapter 14

Arrhenius Bases in Solution • When we dissolve Arrhenius bases in solution, they dissociate, giving a cation and a hydroxide anion. • Strong bases dissociate almost fully, and weak bases dissociate very little. Na. OH(aq) → Na+(aq) + OH–(aq) (~100%) NH 4 OH(aq) → NH 4+(aq) + OH–(aq) (~1%) © 2014 Pearson Education, Inc. Chapter 14

Neutralization Reactions • Recall that an acid neutralizes a base to produce a salt and water. HCl(aq) + Na. OH(aq) → Na. Cl(aq) + H 2 O(l) • The reaction produces the aqueous salt Na. Cl. • If we have an acid with two hydrogens (sulfuric acid, H 2 SO 4), we need two hydroxide ions to neutralize it. H 2 SO 4(aq) + 2 Na. OH(aq) → Na 2 SO 4(aq) + 2 H 2 O(l) © 2014 Pearson Education, Inc. Chapter 14

Predicting Neutralization Reactions • We can identify the Arrhenius acid and base that react in a neutralization reaction to produce a given salt such as calcium sulfate, Ca. SO 4. • The calcium must be from calcium hydroxide, Ca(OH)2; the sulfate must be from sulfuric acid, H 2 SO 4(aq) + Ca(OH)2(aq) → Ca. SO 4(aq) + 2 H 2 O(l) © 2014 Pearson Education, Inc. Chapter 14

Chemistry Connection: Svante Arrhenius • Arrhenius noted that Na. Cl solutions conducted electricity; whereas sugar solutions did not. • He also noticed that the freezing point of Na. Cl solutions were lowered twice as much as sugar solutions at the same concentration. • He proposed that Na. Cl produces ions when dissolved, whereas sugar was in solution as molecules. • It was nearly 20 years before his ideas were accepted by the scientific community. © 2014 Pearson Education, Inc. Chapter 14

Brønsted–Lowry Acids and Bases • The Brønsted–Lowry definitions of acids and bases are broader than that of the Arrhenius definitions. • A Brønsted–Lowry acid is a substance that donates a hydrogen ion to any other substance. It is a proton donor. • A Brønsted–Lowry base is a substance that accepts a hydrogen ion. It is a proton acceptor. © 2014 Pearson Education, Inc. Chapter 14

Brønsted–Lowry Acids and Bases, Continued • Let’s look at two acid–base reactions: 1. HCl(aq) + Na. OH(aq) → Na. Cl(aq) + H 2 O(l) 2. HCl(aq) + NH 3(aq) → NH 4 Cl(aq) 1. HCl donates a proton in both reactions and is a Brønsted–Lowry acid. 2. In the first reaction, the Na. OH accepts a proton and is the Brønsted–Lowry base. 3. In the second reaction, NH 3 accepts a proton and is the Brønsted–Lowry base. © 2014 Pearson Education, Inc. Chapter 14

Amphiprotic Compounds • A substance that is capable of both donating and accepting a proton is an amphiprotic compound. • Na. HCO 3 is an example: HCl(aq) + Na. HCO 3(aq) → Na. Cl(aq) + H 2 CO 3(aq) Na. OH(aq) + Na. HCO 3(aq) → Na 2 CO 3 (aq) + H 2 O(l) • Na. HCO 3 accepts a proton from HCl in the first reaction and donates a proton to Na. OH in the second reaction. © 2014 Pearson Education, Inc. Chapter 14

Acid–Base Indicators • A solution that is sensitive to a change in p. H by exhibiting a different color is an acid–base indicator. • Shown below are three indicators at different p. H values. © 2014 Pearson Education, Inc. Chapter 14

Acid–Base Titrations • A titration is used to analyze an acid solution using a solution of a base. • A measured volume of base is added to the acid solution. When all of the acid has been neutralized, the p. H is 7. One extra drop of base solution after the endpoint increases the p. H dramatically. • When the p. H increases above 7, phenolphthalein changes from colorless to pink, indicating the endpoint of the titration. © 2014 Pearson Education, Inc. Chapter 14

Acid–Base Titrations, Continued • The beginning, middle, and end of an acid–base, using phenolphthalein as the indicator. © 2014 Pearson Education, Inc. Chapter 14

Titration Problem • Consider the titration of acetic acid with sodium hydroxide. A 10. 0 m. L sample of acetic acid requires 37. 55 m. L of 0. 223 M Na. OH. What is the concentration of the acetic acid? HC 2 H 3 O 2(aq) + Na. OH(aq) → Na. C 2 H 3 O 2(aq) + H 2 O(l) • We want concentration acetic acid; we have concentration sodium hydroxide. conc Na. OH mol HC 2 H 3 O 2 conc HC 2 H 3 O 2 © 2014 Pearson Education, Inc. Chapter 14

Titration Problem, Continued • The molarity of Na. OH can be written as the unit factor 0. 233 mol Na. OH/1000 m. L solution. 1 mol HC 2 H 3 O 2 0. 233 mol Na. OH x 37. 55 m. L solution x 1 mol Na. OH 1000 m. L solution = 0. 00837 mol HC 2 H 3 O 2 1000 m. L solution 0. 00837 mol HC 2 H 3 O 2 x 10. 0 m. L solution 1 L solution = 0. 837 M HC 2 H 3 O 2 © 2014 Pearson Education, Inc. Chapter 14

Another Titration Problem • A 10. 0 m. L sample of 0. 555 M H 2 SO 4 is titrated with 0. 233 M Na. OH. What volume of Na. OH is required for the titration? • We want m. L of Na. OH; we have 10. 0 m. L of H 2 SO 4. • Use 0. 555 mol H 2 SO 4/1000 m. L and 0. 233 mol Na. OH/1000 m. L. © 2014 Pearson Education, Inc. Chapter 14

Another Titration Problem, Continued H 2 SO 4(aq) + 2 Na. OH(aq) → Na 2 SO 4(aq) + H 2 O(l) 10. 0 m. L H 2 SO 4 x 0. 555 mol H 2 SO 4 1000 m. L H 2 SO 4 x 1 mol H 2 SO 4 2 mol Na. OH x 0. 233 mol Na. OH 1000 m. L Na. OH = 49. 8 m. L Na. OH • 49. 8 m. L of 0. 233 M Na. OH is required to neutralize 10. 0 m. L of 0. 555 M H 2 SO 4. © 2014 Pearson Education, Inc. Chapter 14

Acid–Base Standardization • A standard solution is a solution in which the concentration is known precisely. • Acid solutions are standardized by neutralizing a weighed quantity of a solid base. • What is the molarity of a hydrochloric acid solution if 25. 50 m. L are required to neutralize 0. 375 g Na 2 CO 3? 2 HCl(aq) + Na 2 CO 3(aq) → 2 Na. Cl(aq) + H 2 O(l) + CO 2(g) © 2014 Pearson Education, Inc. Chapter 14

Acid–Base Standardization, Continued 2 mol HCl 1 mol Na 2 CO 3 0. 375 g Na 2 CO 3 × × 105. 99 g Na 2 CO 3 1 mol Na 2 CO 3 = 0. 00708 mol HCl 1000 m. L solution 0. 00708 mol HCl × = 0. 277 M HCl 25. 50 m. L solution 1 L solution © 2014 Pearson Education, Inc. Chapter 14

Ionization of Water • Occassionally two water molecules react to produce a hydronium ion and a hydroxide ion. H 2 O(l) + H 2 O(l) → H 3 O+(aq) + OH–(aq) or H 2 O(l) → H+(aq) + OH–(aq) • Only about 1 in 500 million water molecules is present as an ion, so water is a weak conductor. • The concentration of hydrogen ions, [H+], in pure water is about 1 × 1027 mol/L at 25 C. © 2014 Pearson Education, Inc. Chapter 14
![Ionization of Water Since H is 1 1027 molL at 25 C Ionization of Water • Since [H+] is 1 × 1027 mol/L at 25 C,](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-32.jpg)
Ionization of Water • Since [H+] is 1 × 1027 mol/L at 25 C, the hydroxide ion concentration [OH–] must also be 1 × 10– 7 mol/L at 25 C. H 2 O(l) → H+(aq) + OH–(aq) • At 25 C: [H+][OH–] = (1 × 10– 7)(1 x 10– 7) = 1. 0 x 10– 14 • This is the ionization constant of water, Kw. © 2014 Pearson Education, Inc. Chapter 14
![H and OH Relationship At 25 C HOH 1 0 x 10 [H+] and [OH–] Relationship • At 25 C, [H+][OH–] = 1. 0 x 10–](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-33.jpg)
[H+] and [OH–] Relationship • At 25 C, [H+][OH–] = 1. 0 x 10– 14. So if we know the [H+], we can calculate [OH–]. • What is the [OH–] if [H+] = 0. 1 M ? [H+][OH–] = 1. 0 x 10– 14 (0. 1)[OH–] = 1. 0 x 10– 14 [OH–] = 1. 0 x 10– 13 © 2014 Pearson Education, Inc. Chapter 14

The p. H Concept • Recall that p. H is a measure of the acidity of a solution. • A neutral solution has a p. H of 7, an acidic solution has a p. H less than 7, and a basic solution has a p. H greater than 7. • The p. H scale uses powers of 10 to express the hydrogen ion concentration. • Mathematically, p. H = –log[H+] – [H+] is the molar hydrogen ion concentration. © 2014 Pearson Education, Inc. Chapter 14

Calculating p. H • What is the p. H if the hydrogen ion concentration in a vinegar solution is 0. 001 M? • p. H = –log[H+] • p. H = –log(0. 001) • p. H = – ( – 3) = 3 • The p. H of the vinegar is 3, so the vinegar is acidic. © 2014 Pearson Education, Inc. Chapter 14
![Calculating H from p H If we rearrange the p H equation for Calculating [H+] from p. H • If we rearrange the p. H equation for](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-36.jpg)
Calculating [H+] from p. H • If we rearrange the p. H equation for [H+], we get: [H+] = 10–p. H • Milk has a p. H of 6. What is the concentration of hydrogen ion in milk? [H+] = 10–p. H = 10– 6 = 0. 000001 M [H+] = 1 x 10– 6 M © 2014 Pearson Education, Inc. Chapter 14
![Advanced p H Calculations What is the p H of blood with H Advanced p. H Calculations • What is the p. H of blood with [H+]](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-37.jpg)
Advanced p. H Calculations • What is the p. H of blood with [H+] = 4. 8 x 10– 8 M? p. H = –log[H+] = –log(4. 8 x 10– 8) = – (– 3. 82) p. H = 3. 82 • What is the [H+] in orange juice with a p. H of 2. 75? [H+] = 10–p. H = 10– 2. 75 = 0. 0018 M [H+] = 2. 75 x 10– 3 M © 2014 Pearson Education, Inc. Chapter 14

Critical Thinking: Acid Rain • Nitrogen oxides and sulfur oxides, produced from the combustion of fossil fuels, react with rainwater to produce nitric and sulfuric acids. • These strong acids reduce the p. H of rainwater. • Acid rain refers to rain with a p. H 5 or below. • Acid rain cause corrosion of metal and degradation of limestone and marble statues. © 2014 Pearson Education, Inc. Chapter 14

Strong and Weak Electrolytes • An aqueous solution that is a good conductor of electricity is a strong electrolyte. • An aqueous solution that is a poor conductor of electricity is a weak electrolyte. • The greater the degree of ionization or dissociation is, the greater the conductivity of the solution. © 2014 Pearson Education, Inc. Chapter 14

Electrolyte Strength • Weak acids and bases are weak electrolytes. • Strong acids and bases are strong electrolytes. • Insoluble ionic compounds are weak electrolytes. • Soluble ionic compounds are strong electrolytes. © 2014 Pearson Education, Inc. Chapter 14

Strengths of Electrolytes © 2014 Pearson Education, Inc. Chapter 14

Total Ionic Equations • The concept of ionization allows us to portray ionic solutions more accurately by showing strong electrolytes in their ionized form. HCl(aq) + Na. OH(aq) → Na. Cl(aq) + H 2 O(l) • Write strong acids and bases and soluble ionic compounds as ions. H+(aq) + Cl–(aq) + Na+(aq) + OH–(aq) → Na+(aq) + Cl–(aq) + H 2 O(l) • This is the total ionic equation. Each species is written as it predominantly exists in solution. © 2014 Pearson Education, Inc. Chapter 14

Net Ionic Equations H+(aq) + Cl–(aq) + Na+(aq) + OH–(aq) → Na+(aq) + Cl–(aq) + H 2 O(l) • Notice that Na+ and Cl– appear on both sides of the above-noted equation. They are spectator ions. • Spectator ions are in the solution, but do not participate in the overall reaction. We cancel out the spectator ions to give the net ionic equation. • The net ionic equation is as follows: H+(aq) + OH–(aq) → H 2 O(l) © 2014 Pearson Education, Inc. Chapter 14

Guidelines for Writing Net Ionic Equations • Complete and balance the nonionized chemical equation. • Convert the nonionized equation into the total ionic equation. – Write strong electrolytes in the ionized form. – Write water and dissolved gases in the nonionized form. • Cancel spectator ions to obtain the net ionic equation. – If all species are eliminated, there is no reaction. © 2014 Pearson Education, Inc. Chapter 14

Guidelines for Writing Net Ionic Equations, Continued – If all species are eliminated, there is no reaction. • Check each ion or atom on both sides of the equation. The total charge (positive or negative) on the reactants side of the equation must equal the total charge on the products side of the equation. © 2014 Pearson Education, Inc. Chapter 14

Chapter Summary • p. H is a measure of the acidity of a solution. The typical range for p. H is 0 to 14. • Neutral solutions have a p. H of 7. • Below are some properties of acids and bases: © 2014 Pearson Education, Inc. Chapter 14

Chapter Summary, Continued • An Arrhenius acid is a substance that ionizes in water to produce hydrogen ions. • An Arrhenius base is a substance that ionizes in water to release hydroxide ions. • A Brønsted–Lowry acid is a substance that donates a hydrogen ion to any other substance. It is a proton donor. • A Brønsted–Lowry base is a substance that accepts a hydrogen ion. It is a proton acceptor. © 2014 Pearson Education, Inc. Chapter 14
![Chapter Summary Continued In an aqueous solution HOH 1 0 x 10 Chapter Summary, Continued • In an aqueous solution, [H+][OH–] = 1. 0 x 10–](https://slidetodoc.com/presentation_image_h2/7818eb02d8c4fb1b83f525549dda8318/image-48.jpg)
Chapter Summary, Continued • In an aqueous solution, [H+][OH–] = 1. 0 x 10– 14. This is the ionization constant of water, Kw. • p. H = –log[H+] • [H+] = 10–p. H • Strong electrolytes are mostly dissociated in solution. • Weak electrolytes are slightly dissociated in solution. © 2014 Pearson Education, Inc. Chapter 14