Chapter 14 Lecture Basic Chemistry Fourth Edition 14

Chapter 14 Lecture Basic Chemistry Fourth Edition 14. 3 Strengths of Acids and Bases 14. 4 Dissociation Constants for Acids and Bases Learning Goal • Write equations for the dissociation of strong and weak acids; identify the direction of reaction. • Write the expression for the dissociation constant of a weak acid or weak base. © 2014 Pearson Education, Inc. Acids produce hydrogen ions in aqueous solution.

Strength of Acids and Bases • The strength of an acid is determined by the moles of H 3 O+ produced per mole of acid that dissociates. • The strength of a base is determined by the moles of OH− produced per mole of base that dissolves. © 2014 Pearson Education, Inc.

Review: Strong and Weak Acids A solution of hydrochloric acid forms as H+ transfers from hydrogen chloride to water. Water is acting as a base in this reaction. © 2014 Pearson Education, Inc.

Strong Acids Strong acids are strong electrolytes. They • donate H+ easily • ionize completely in water HCl(g) + H 2 O(l) H 3 O+(aq) + Cl−(aq) There are six strong acids: Strong acid Hydroiodic acid HI Hydrochloric acid HCl Hydrobromic acid HBr Sulfuric acid H 2 SO 4 Perchloric acid HCl. O 4 Nitric acid © 2014 Pearson Education, Inc. HNO 3

Strong versus Weak Acids • A strong acid such as HCl completely dissociates 100%. • Weak acids such as HC 2 H 3 O 2 are only slightly ionized in water to form a solution. © 2014 Pearson Education, Inc.

Strong versus Weak Acids A weak acid, HC 2 H 3 O 2 , slightly dissociates: HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2−(aq) A strong acid, HCl, dissociates 100%: HCl (aq) + H 2 O(l) H 3 O+(aq) + Cl−(aq) **Notice the different reaction arrows used** © 2014 Pearson Education, Inc.

Weak Acids Weak acids are weak electrolytes. They • transfer only a small percentage of H+ ions • only partially ionize in water • have a strong conjugate base • have a forward and reverse reaction • have a strong reverse reaction HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2−(aq) Acetic acid © 2014 Pearson Education, Inc. Acetate ion

Weak Acids There are many weak acids: • citric acid, found in oranges and other citrus fruits • acetic acid, found in vinegar, used in salad dressings • phosphoric acid, found in many soft drinks © 2014 Pearson Education, Inc.
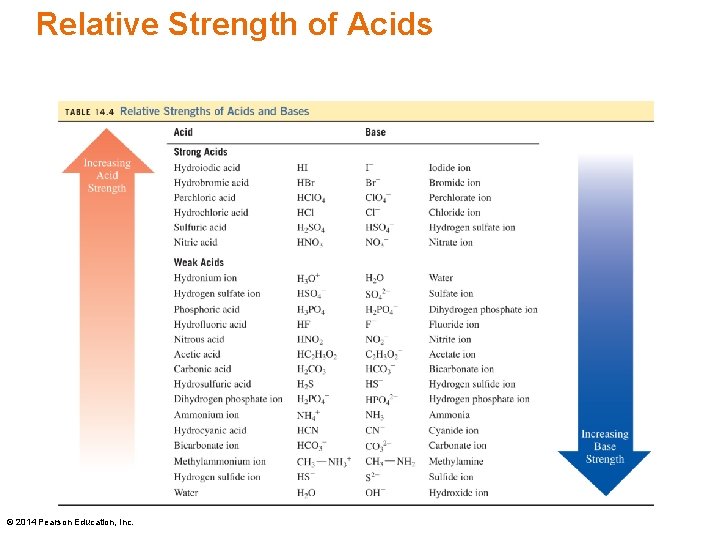
Relative Strength of Acids © 2014 Pearson Education, Inc.

Diprotic Acids Diprotic acids • can donate two H+ ions • donate one H+ ion at a time © 2014 Pearson Education, Inc.

Diprotic Acids Sulfuric acid, H 2 SO 4 , a strong acid, ionizes 100% to form HSO 4− and H+. Bisulfate, HSO 4−, is considered a weak acid; it only partially ionizes to form SO 42−. © 2014 Pearson Education, Inc.
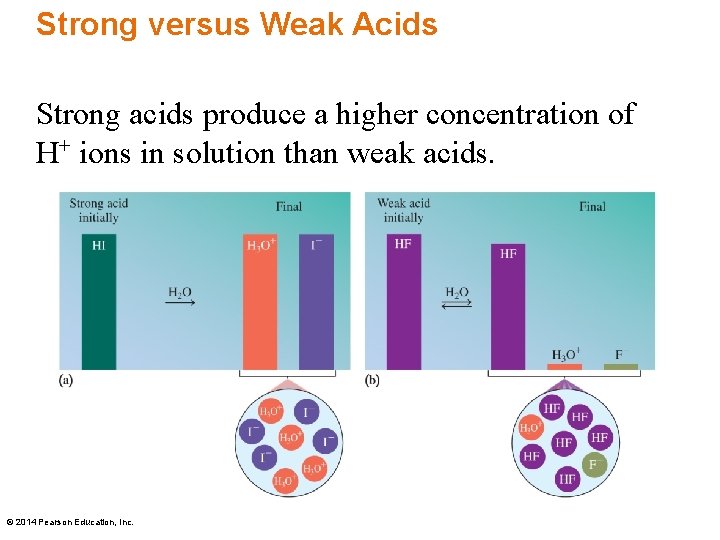
Strong versus Weak Acids Strong acids produce a higher concentration of H+ ions in solution than weak acids. © 2014 Pearson Education, Inc.

Strong versus Weak Bases are found in many household products, such as those that remove grease and open drains. © 2014 Pearson Education, Inc.

Strong Bases Strong bases • are strong electrolytes • dissociate completely in water to give metal ions and hydroxide ions H 2 O KOH(s) K+(aq) + OH−(aq) © 2014 Pearson Education, Inc.

Strong Bases Strong bases include Group 1 A (I) hydroxides and some Group 2 A (II) hydroxides. Strong Bases Lithium hydroxide Li. OH Sodium hydroxide Na. OH Potassium hydroxide KOH Strontium hydroxide Sr(OH)2 Calcium hydroxide Ca(OH)2 Barium hydroxide Ba(OH)2 © 2014 Pearson Education, Inc.

Strong Bases High concentrations of hydroxide ions in household products can cause severe damage to skin and eyes. It is important to follow directions carefully when hydroxide ions are used in the home or a chemistry laboratory. © 2014 Pearson Education, Inc.

Weak Bases Weak bases produce very few ions in solution and are • poor electrolytes • poor acceptors of hydrogen ions A typical weak base ammonia, NH 3, used in window cleaners, reacts with water to form NH 4+ and OH−. NH 3(g) + H 2 O(l) NH 4+(aq) + OH−(aq) © 2014 Pearson Education, Inc.

Direction of Reaction In acid−base reactions, • one acid is stronger than the other acid • one base is stronger than the other base In a conjugate acid−base pair, • the stronger the acid, the weaker its conjugate base • the stronger the base, the weaker its conjugate acid © 2014 Pearson Education, Inc.

Direction of Reaction Comparing the strengths of acids and bases in a chemical reaction, we can determine the direction of the reaction. © 2014 Pearson Education, Inc.

Learning Check Identify each of the following as a strong or weak acid or base: A. HBr B. HNO 2 C. Na. OH D. H 2 SO 4 E. Cu(OH)2 © 2014 Pearson Education, Inc.

Solution Identify each of the following as a strong or weak acid or base: A. HBr strong acid B. HNO 2 weak acid C. Na. OH strong base D. H 2 SO 4 strong acid E. Cu(OH)2 weak base © 2014 Pearson Education, Inc.

Learning Check A. Identify the stronger acid in each pair. 1. HNO 2 or H 2 S 2. HCO 3− or HBr B. Identify the stronger base in each pair. 1. CO 32− or NO 2− 2. OH− or H 2 O © 2014 Pearson Education, Inc.

Solution A. Identify the stronger acid in each pair. 1. HNO 2 or H 2 S HNO 2 2. HCO 3− or HBr HCO 3− B. Identify the stronger base in each pair. 1. CO 32− or NO 2− CO 32− 2. OH− or H 2 O OH− © 2014 Pearson Education, Inc.

Strong Acids and Bases Strong acids and bases • dissociate completely in water, producing high concentrations of ions HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl−(aq) Na. OH(aq) Na+(aq) + OH−(aq) • have very large dissociation constants that are not usually given © 2014 Pearson Education, Inc.

Weak Acids, Dissociation Constants Weak acids and bases • produce small amounts of ions in water HCHO 2(aq) + H 2 O(l) H 3 O+(aq) + CHO 2−(aq) • partially dissociate in water and have a dissociation constant The acid dissociation constant for HCHO 2 is: × © 2014 Pearson Education, Inc.

Weak Bases, Dissociation Constant Weak bases also have a dissociation constant, called the base dissociation constant, Kb. CH 3 NH 2(aq) + H 2 O(l) CH 3 NH 3+(aq) + OH−(aq) × © 2014 Pearson Education, Inc.

Ka, Kb Strength of Weak Acids, Bases The larger the Ka, Kb value for an acid or base, • the larger the ratio of products to reactants • the larger the concentration of ions produced • the stronger the acid or base © 2014 Pearson Education, Inc.
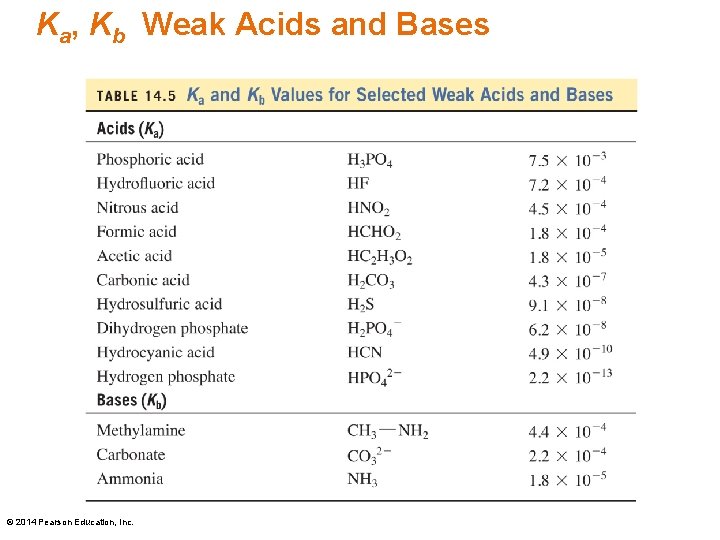
Ka, Kb Weak Acids and Bases © 2014 Pearson Education, Inc.
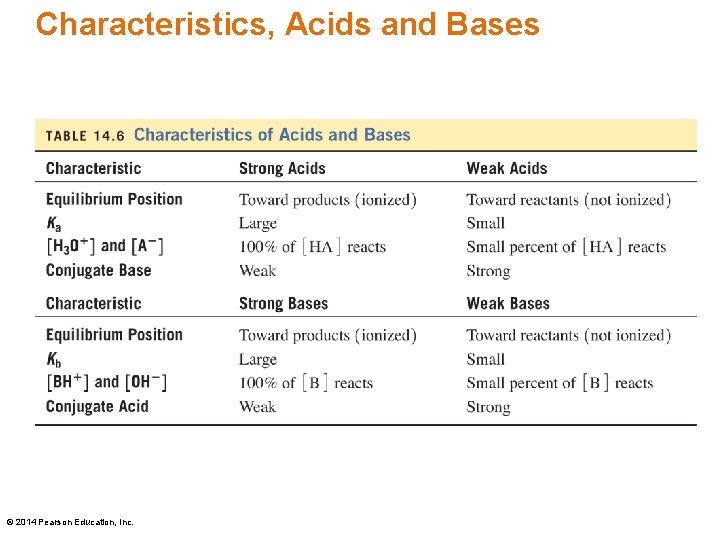
Characteristics, Acids and Bases © 2014 Pearson Education, Inc.

Learning Check Write the equilibrium reaction and dissociation constant for the weak acid, HCN. Remember, products over reactants (liquids are never included) © 2014 Pearson Education, Inc.

Solution Write the equilibrium reaction and dissociation constant for the weak acid, HCN(aq) + H 2 O(l) © 2014 Pearson Education, Inc. H 3 O+(aq) + CN−(aq)

Learning Check Write the equilibrium reaction and Kb for the weak base CH 3—CH 2 –NH 2. © 2014 Pearson Education, Inc.

Solution Write the equilibrium reaction and Kb for the weak base CH 3—CH 2 –NH 2(aq) + H 2 O(l) OH−(aq) + CH 3—CH 2 –NH 3+(aq) © 2014 Pearson Education, Inc.
- Slides: 33