Chapter 14 Electrochemical Cells Cells and Batteries Zn

Chapter 14 Electrochemical Cells

Cells and Batteries Zn Alessandro Volta (1745 -1827) An electric cell is a device that continuously converts chemical potential energy to electrical energy. A battery is a group of two or more electric cells connected to each other in series. Cu
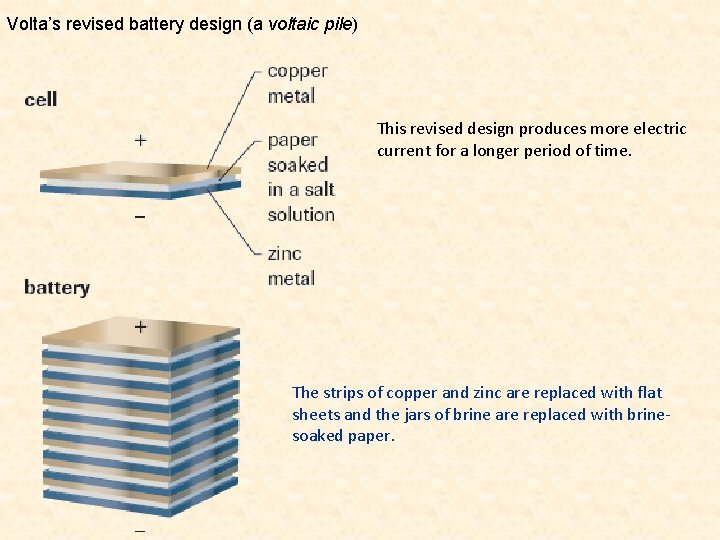
Volta’s revised battery design (a voltaic pile) This revised design produces more electric current for a longer period of time. The strips of copper and zinc are replaced with flat sheets and the jars of brine are replaced with brinesoaked paper.
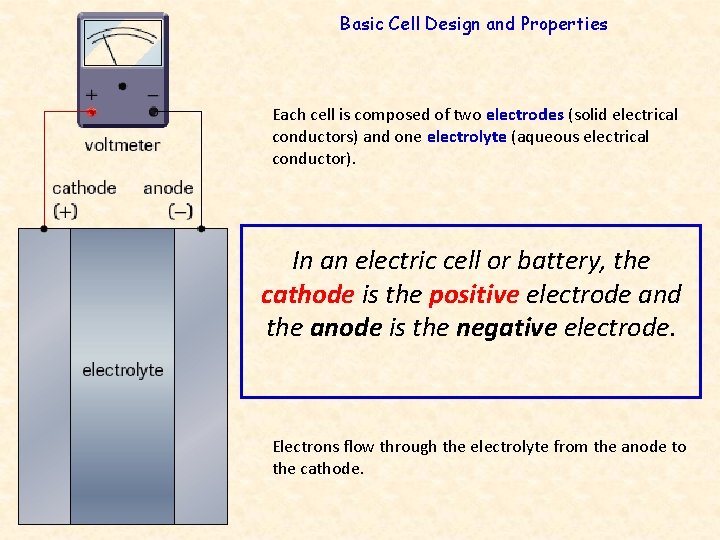
Basic Cell Design and Properties Each cell is composed of two electrodes (solid electrical conductors) and one electrolyte (aqueous electrical conductor). In an electric cell or battery, the cathode is the positive electrode and the anode is the negative electrode. Electrons flow through the electrolyte from the anode to the cathode.
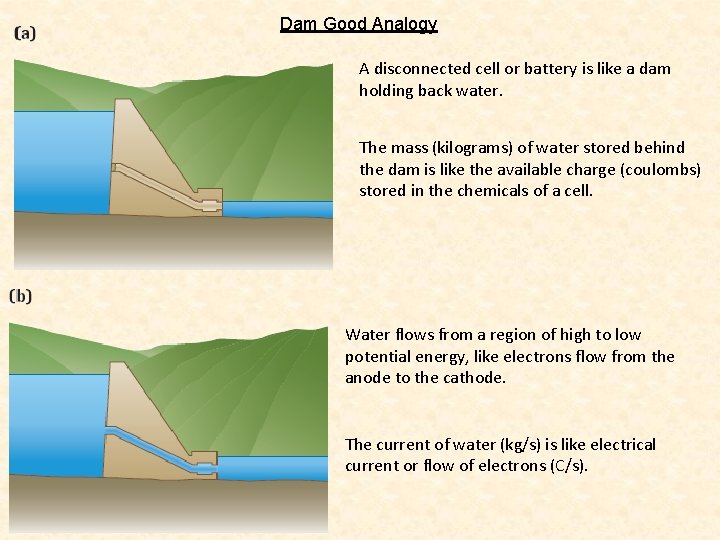
Dam Good Analogy A disconnected cell or battery is like a dam holding back water. The mass (kilograms) of water stored behind the dam is like the available charge (coulombs) stored in the chemicals of a cell. Water flows from a region of high to low potential energy, like electrons flow from the anode to the cathode. The current of water (kg/s) is like electrical current or flow of electrons (C/s).
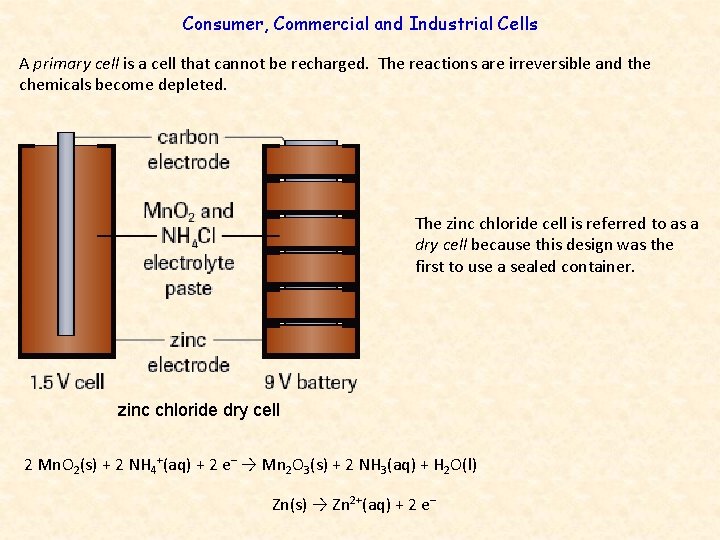
Consumer, Commercial and Industrial Cells A primary cell is a cell that cannot be recharged. The reactions are irreversible and the chemicals become depleted. The zinc chloride cell is referred to as a dry cell because this design was the first to use a sealed container. zinc chloride dry cell 2 Mn. O 2(s) + 2 NH 4+(aq) + 2 e– → Mn 2 O 3(s) + 2 NH 3(aq) + H 2 O(l) Zn(s) → Zn 2+(aq) + 2 e–
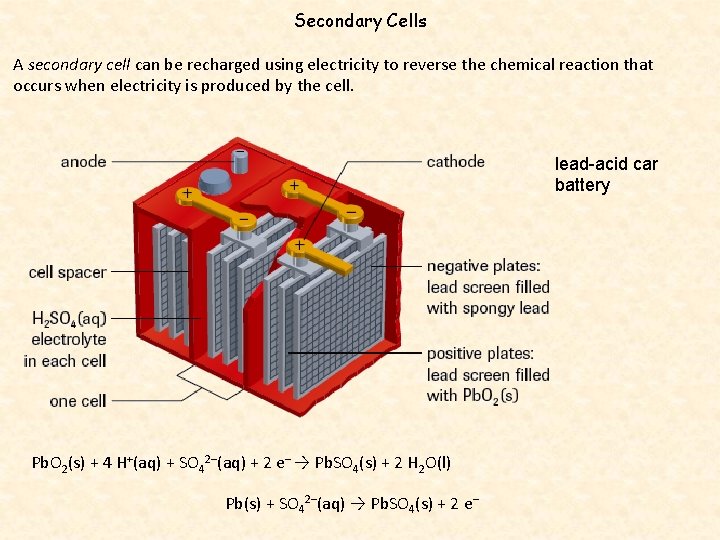
Secondary Cells A secondary cell can be recharged using electricity to reverse the chemical reaction that occurs when electricity is produced by the cell. lead-acid car battery Pb. O 2(s) + 4 H+(aq) + SO 42–(aq) + 2 e– → Pb. SO 4(s) + 2 H 2 O(l) Pb(s) + SO 42–(aq) → Pb. SO 4(s) + 2 e–
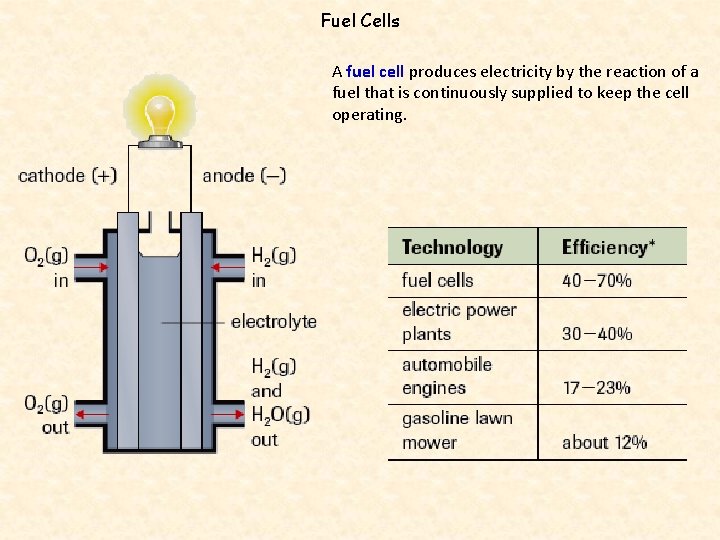
Fuel Cells A fuel cell produces electricity by the reaction of a fuel that is continuously supplied to keep the cell operating.

Homework • • Read pages 610 – 620 Questions 1 -4, page 614 Questions 10, 11, 14, page 619 Questions 1 – 5, page 621
- Slides: 10