Chapter 14 Chemical Kinetics Overview Reaction Rates Stoichiometry




![Reaction Rates (graphical): • Average Rate = [M] Dt for reaction A B D[M] Reaction Rates (graphical): • Average Rate = [M] Dt for reaction A B D[M]](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-5.jpg)
![Rates • for A B - D[A] = D[B] Dt Dt Rate of the Rates • for A B - D[A] = D[B] Dt Dt Rate of the](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-6.jpg)
![Average Rate = [A]final time - [A]initial time Dtfinal - Dtinitial for A B Average Rate = [A]final time - [A]initial time Dtfinal - Dtinitial for A B](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-8.jpg)
![Instantaneous Ratetime, t = slope of the tangent at time = t [M] tangent Instantaneous Ratetime, t = slope of the tangent at time = t [M] tangent](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-9.jpg)
![Stoichiometry 4 PH 3 - 1 D[PH 3] 4 Dt - D[PH 3] Dt Stoichiometry 4 PH 3 - 1 D[PH 3] 4 Dt - D[PH 3] Dt](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-10.jpg)
![General Relationship Rate = - 1 D[A] = - 1 D[B] = + 1 General Relationship Rate = - 1 D[A] = - 1 D[B] = + 1](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-11.jpg)




![Exponent Values Relative to DRate Exponent Value 0 1 2 3 4 [conc] rate Exponent Values Relative to DRate Exponent Value 0 1 2 3 4 [conc] rate](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-16.jpg)



![Conversion to base-10 logarithms: ln [A]t = - kt [A]0 to log [A]t = Conversion to base-10 logarithms: ln [A]t = - kt [A]0 to log [A]t =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-20.jpg)

![Concentration vs. Time • Second Order: rate = - D[A] = k[A]2 Dt • Concentration vs. Time • Second Order: rate = - D[A] = k[A]2 Dt •](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-22.jpg)

![Graphical Methods • Equation for a Straight Line • y = • ln[A]t = Graphical Methods • Equation for a Straight Line • y = • ln[A]t =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-24.jpg)
![First Order: 2 H 2 O 2(aq) [H 2 O 2] time 2 H First Order: 2 H 2 O 2(aq) [H 2 O 2] time 2 H](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-25.jpg)
![Second Order: 2 NO 2 2 NO 1/[NO 2] slope, b = +k time Second Order: 2 NO 2 2 NO 1/[NO 2] slope, b = +k time](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-27.jpg)








![Rate Equations • Molecularity unimolecular bimolecular termolecular Rate Law rate = k[A][B] rate = Rate Equations • Molecularity unimolecular bimolecular termolecular Rate Law rate = k[A][B] rate =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-39.jpg)





![Substitution Method at equilibrium k 1[O 3] = k 2[O 2][O] rate 3 =k Substitution Method at equilibrium k 1[O 3] = k 2[O 2][O] rate 3 =k](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-45.jpg)

![Cont’d rate 1 = k 1 [OCl -][H 2 O] = rate 2 = Cont’d rate 1 = k 1 [OCl -][H 2 O] = rate 2 =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-47.jpg)


- Slides: 51

Chapter 14 Chemical Kinetics

Overview: • Reaction Rates – Stoichiometry, Conditions, Concentration • Rate Equations – Order – Initial Rate – Concentration vs. Time – First Order Rxns. – Second Order Rxns. • Graphical Methods

Cont’d • Molecular Theory – Activation Energy – Concentration – Molecular Orientation – Temperature – Arrhenius Equation • Reaction Mechanisms – Elementary Steps, Reaction Order, Intermediates • Catalysts

Reaction Rates • What Affects Rates of Reactions? – Concentration of the Reactants – Temperature of Reaction – Presence of a Catalyst – Surface Area of Solid or Liquid Reactants
![Reaction Rates graphical Average Rate M Dt for reaction A B DM Reaction Rates (graphical): • Average Rate = [M] Dt for reaction A B D[M]](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-5.jpg)
Reaction Rates (graphical): • Average Rate = [M] Dt for reaction A B D[M] Dt time
![Rates for A B DA DB Dt Dt Rate of the Rates • for A B - D[A] = D[B] Dt Dt Rate of the](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-6.jpg)
Rates • for A B - D[A] = D[B] Dt Dt Rate of the disappearance of A is equal in magnitude but opposite in sign to the rate of the appearance of B

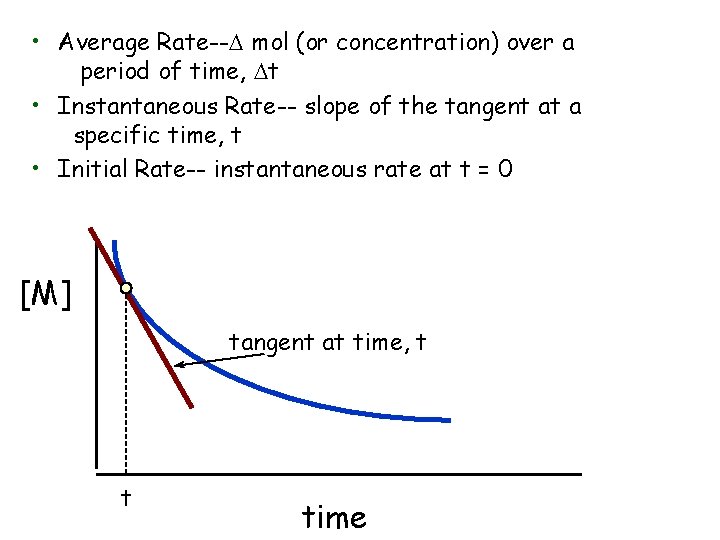
• Average Rate--D mol (or concentration) over a period of time, Dt • Instantaneous Rate-- slope of the tangent at a specific time, t • Initial Rate-- instantaneous rate at t = 0 [M] tangent at time, t t time
![Average Rate Afinal time Ainitial time Dtfinal Dtinitial for A B Average Rate = [A]final time - [A]initial time Dtfinal - Dtinitial for A B](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-8.jpg)
Average Rate = [A]final time - [A]initial time Dtfinal - Dtinitial for A B
![Instantaneous Ratetime t slope of the tangent at time t M tangent Instantaneous Ratetime, t = slope of the tangent at time = t [M] tangent](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-9.jpg)
Instantaneous Ratetime, t = slope of the tangent at time = t [M] tangent at time, t t time
![Stoichiometry 4 PH 3 1 DPH 3 4 Dt DPH 3 Dt Stoichiometry 4 PH 3 - 1 D[PH 3] 4 Dt - D[PH 3] Dt](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-10.jpg)
Stoichiometry 4 PH 3 - 1 D[PH 3] 4 Dt - D[PH 3] Dt => P 4 + 6 H 2 = + 1 D[P 4] = 1 Dt = + 4 D[P 4] Dt + 1 D[H 2] 6 Dt = + 2 D[H 2] 3 Dt
![General Relationship Rate 1 DA 1 DB 1 General Relationship Rate = - 1 D[A] = - 1 D[B] = + 1](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-11.jpg)
General Relationship Rate = - 1 D[A] = - 1 D[B] = + 1 D[C] = + 1 D[D] a. Dt b Dt c Dt d Dt a. A + b. B c. C + d. D

Conditions which affect rates • Concentration – concentration rate • Temperature – temperature rate • Catalyst – substance which increases rate but itself remains unchanged

Rate Equations: • a. A + b. B x. X rate law rate = k[A]m[B]n • m, n are orders of the reactants – extent to which rate depends on concentration – m + n = overall rxn order • k is the rate constant for the reaction

Examples: • 2 N 2 O 5 => 4 NO 2 + rate = k[N 2 O 5] • 2 NO + Cl 2 1 st order => 2 NOCl rate = k[NO]2[Cl 2] • 2 NH 3 => N 2 O 2 + 3 rd order 3 H 2 rate = k[NH 3]0 = k 0 th order

Determination of Rate Equations: Data for: Expt. # 1 2 3 A + B (0. 10)2 M 2 C [A] [B] initial rate 0. 10 0. 20 0. 10 4. 0 16. 0 rate = k[A]2[B]0 = k[A]2 k = 4. 0 Ms-1 => = 400 M-1 s-1
![Exponent Values Relative to DRate Exponent Value 0 1 2 3 4 conc rate Exponent Values Relative to DRate Exponent Value 0 1 2 3 4 [conc] rate](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-16.jpg)
Exponent Values Relative to DRate Exponent Value 0 1 2 3 4 [conc] rate double double same double x 4 x 8 x 16

Problem: Data for: 2 NO + H 2 => N 2 O + H 2 O Expt. # 1 2 3 [NO] [H 2] rate 6. 4 x 10 -3 2. 2 x 10 -3 2. 6 x 10 -5 12. 8 x 10 -3 2. 2 x 10 -3 1. 0 x 10 -4 6. 4 x 10 -3 4. 5 x 10 -3 5. 0 x 10 -5 rate = k[NO]2[H 2]

Units of Rate Constants • units of rates M/s • units of rate constants will vary depending on order of rxn M = 1 (M)2 (M) s s. M 2 rate = for k [A]2 [B] • rate constants are independent of the concentration

Concentration vs. Time 1 st and 2 nd order integrated rate equations • First Order: rate = - D[A] = k [A] Dt • ln [A]t = - kt [A]0 A = reactant • or ln [A]t - ln [A]0 = - kt
![Conversion to base10 logarithms ln At kt A0 to log At Conversion to base-10 logarithms: ln [A]t = - kt [A]0 to log [A]t =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-20.jpg)
Conversion to base-10 logarithms: ln [A]t = - kt [A]0 to log [A]t = - kt [A]0 2. 303

Problem: The rate equation for the reaction of sucrose in water is, rate = k[C 12 H 22 O 11]. After 2. 57 h at 27°C, 5. 00 g/L of sucrose has decreased to 4. 50 g/L. Find k. C 12 H 22 O 11(aq) ln 4. 50 g/L 5. 00 g/L + H 2 O(l) => = - k (2. 57 h) k = 0. 0410 h-1 2 C 6 H 12 O 6
![Concentration vs Time Second Order rate DA kA2 Dt Concentration vs. Time • Second Order: rate = - D[A] = k[A]2 Dt •](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-22.jpg)
Concentration vs. Time • Second Order: rate = - D[A] = k[A]2 Dt • 1 - 1 = kt [A]0 • second order rxn with one reactant: rate = k [A]2

Problem: Ammonium cyanate, NH 4 NCO, rearranges in water to give urea, (NH 2)2 CO. If the original concentration of NH 4 NCO is 0. 458 mol/L and k = 0. 0113 L/mol min, how much time elapses before the concentration is reduced to 0. 300 mol/L? NH 4 NCO(aq) => 1 (0. 300) - (NH 2)2 CO(aq) 1 (0. 458) = rate = k[NH 4 NCO] (0. 0113) t t = 102 min
![Graphical Methods Equation for a Straight Line y lnAt Graphical Methods • Equation for a Straight Line • y = • ln[A]t =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-24.jpg)
Graphical Methods • Equation for a Straight Line • y = • ln[A]t = - kt • 1 [A]t = bx kt + + a ln[A]0 + 1 [A]0 b = slope a = y intercept x = time 1 st order 2 nd order
![First Order 2 H 2 O 2aq H 2 O 2 time 2 H First Order: 2 H 2 O 2(aq) [H 2 O 2] time 2 H](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-25.jpg)
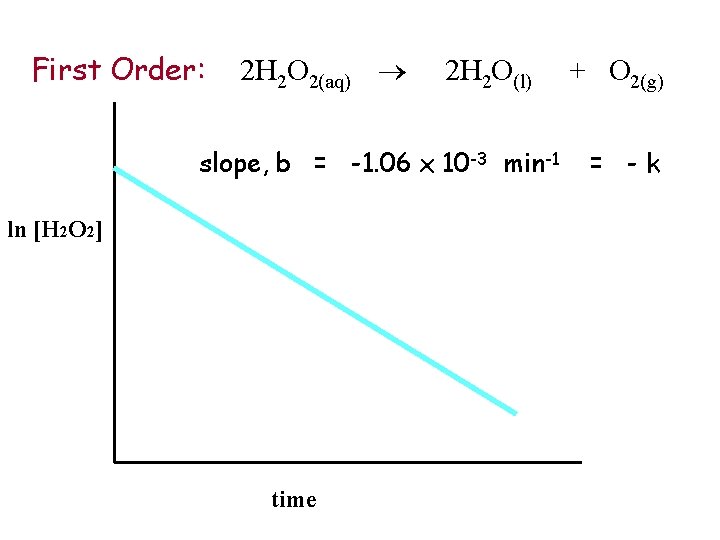
First Order: 2 H 2 O 2(aq) [H 2 O 2] time 2 H 2 O(l) + O 2(g)

First Order: 2 H 2 O 2(aq) 2 H 2 O(l) slope, b = -1. 06 x 10 -3 min-1 ln [H 2 O 2] time + O 2(g) = -k
![Second Order 2 NO 2 2 NO 1NO 2 slope b k time Second Order: 2 NO 2 2 NO 1/[NO 2] slope, b = +k time](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-27.jpg)
Second Order: 2 NO 2 2 NO 1/[NO 2] slope, b = +k time + O 2

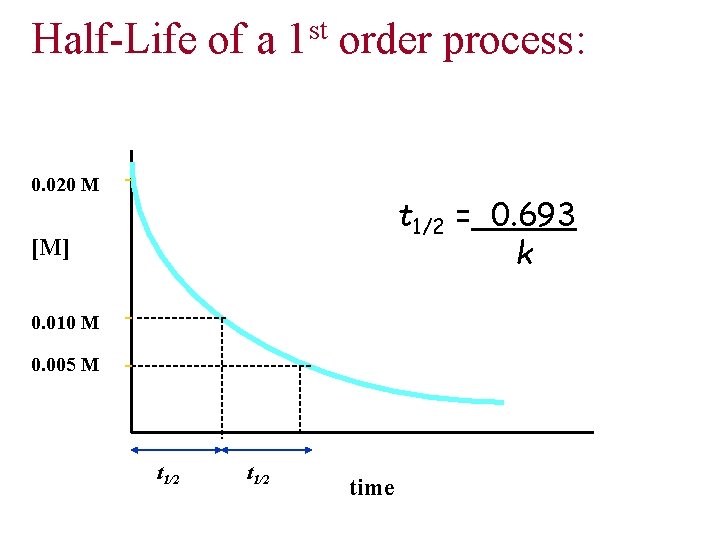
Half-Life of a st 1 order process: 0. 020 M t 1/2 = 0. 693 k [M] 0. 010 M 0. 005 M t 1/2 time

Problem: The decomposition of SO 2 Cl 2 is first order in SO 2 Cl 2 and has a half-life of 4. 1 hr. If you begin with 1. 6 x 10 -3 mol of SO 2 Cl 2 in a flask, how many hours elapse before the quantity of SO 2 Cl 2 has decreased to 2. 00 x 10 -4 mol? SO 2 Cl 2(g) => SO 2(g) + Cl 2(g)

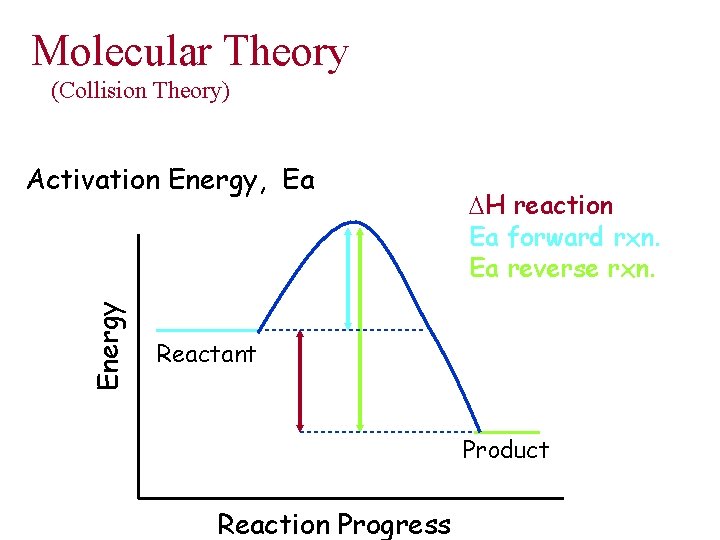
Temperature Effects • Rates typically increase with T increase • Collisions between molecules increase • Energy of collisions increase • Even though only a small fraction of collisions lead to reaction • Minimum Energy necessary for reaction is the Activation Energy

Molecular Theory (Collision Theory) Energy Activation Energy, Ea DH reaction Ea forward rxn. Ea reverse rxn. Reactant Product Reaction Progress

Activation Energy • Activation Energy varies greatly – almost zero to hundreds of k. J – size of Ea affects reaction rates • Concentration – more molecules, more collisions • Molecular Orientation – collisions must occur “sterically”

The Arrhenius Equation • increase temperature, inc. reaction rates • rxn rates are a to energy, collisions, temp. & orient • k = Ae-Ea/RT k = rxn rate constant A = frequency of collisions -Ea/RT = fraction of molecules with energy necessary for reaction

Graphical Determination of Ea rearrange eqtn to give straight-line eqtn y = bx + a ln k = -Ea 1 + ln A R T ln k slope = -Ea/R 1/T

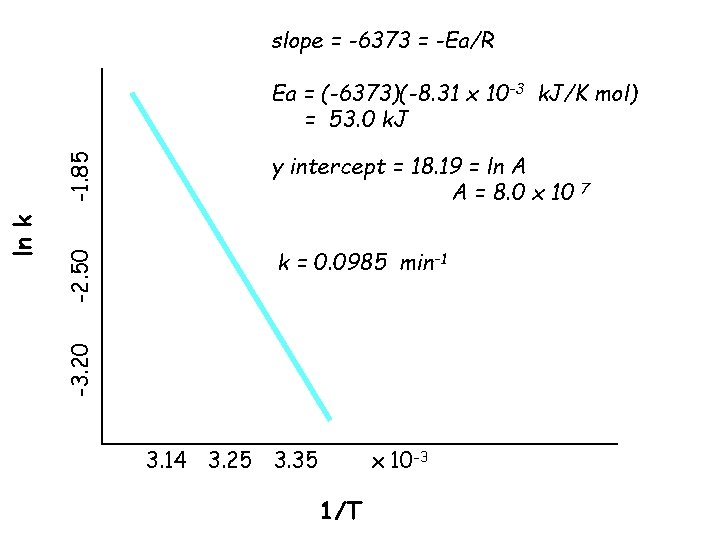
Problem: Data for the following rxn are listed in the table. Calculate Ea graphically, calculate A and find k at 311 K. Mn(CO)5(CH 3 CN)+ + NC 5 H 5 => Mn(CO)5(NC 5 H 5)+ + CH 3 CN ln k k, min-1 T (K) -3. 20 -2. 50 -1. 85 0. 0409 0. 0818 0. 157 298 308 318 1/T x 10 -3 3. 35 3. 25 3. 14

slope = -6373 = -Ea/R -1. 85 y intercept = 18. 19 = ln A A = 8. 0 x 10 -2. 50 k = 0. 0985 min-1 -3. 20 ln k Ea = (-6373)(-8. 31 x 10 -3 k. J/K mol) = 53. 0 k. J 3. 14 3. 25 3. 35 x 10 -3 1/T 7

Problem: The energy of activation for C 4 H 8(g) => 2 C 2 H 4(g) is 260 k. J/mol at 800 K and k = 0. 0315 sec Find k at 850 K. ln k 2 = - Ea (1/T 2 - 1/T 1) k 1 R k at 850 K = 0. 314 sec-1

Reaction Mechanisms • Elementary Step – equation describing a single molecular event • Molecularity – unimolecular – bimolecular – termolecular • 2 O 3 (1) (2) => 3 O 2 O 3 => O 3 + O O 2 + O => 2 O 2 unimolecular bimolecular
![Rate Equations Molecularity unimolecular bimolecular termolecular Rate Law rate kAB rate Rate Equations • Molecularity unimolecular bimolecular termolecular Rate Law rate = k[A][B] rate =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-39.jpg)
Rate Equations • Molecularity unimolecular bimolecular termolecular Rate Law rate = k[A][B] rate = k[A]2[B] – notice that molecularity for an elementary step is the same as the order

2 O 3 => O 3 3 O 2 => O 2 + O 3 + O => O 2 O 2 2 O 3 + O => 3 O 2 + O O is an intermediate rate = k[O 3] rate = k’[O 3][O]

Problem: • Write the rate equation and give the molecularity of the following elementary steps: NO(g) + NO 3(g) => 2 NO 2(g) rate = k[NO][NO 3] bimolecular (CH 3)3 CBr(aq) => (CH 3)3 C+(aq) rate = k[(CH 3)3 CBr] unimolecular + Br- (aq)

Mechanisms and Rate Equations rate determining step is the slow step -the overall rate is limited by the rate determining step 1 step 2 overall NO 2 + F 2 => FNO 2 + F k 1 NO 2 + F => FNO 2 k 2 rate = k 1[NO 2][F 2] slow rate = k 2[NO 2][F] fast 2 NO 2 + F 2 => 2 FNO 2 rate = k 1[NO 2][F 2]

Problem: • Given the following reaction and rate law: NO 2(g) + CO(g) => CO 2(g) + NO(g) rate = k[NO 2]2 – Does the reaction occur in a single step? – Given the two mechanisms, which is most likely: NO 2 + NO 2 =>NO 3 + NO NO 3 + CO => NO 2 + CO 2 NO 2 => NO + O CO + O => CO 2

Reaction Mechanisms & Equilibria 2 O 3(g) 1: 2: 3 O 2(g) k 1 O 3(g) O(g) k 2 + O 2(g) + O(g) O 3(g) overall rxn fast equil. rate 1 = k 1[O 3] rate 2 = k 2[O 2][O] k 3 2 O 2(g) slow rate 3 = k 3[O][O 3] rate 3 includes the conc. of an intermediate and the exptl. rate law will include only species that are present in measurable quantities
![Substitution Method at equilibrium k 1O 3 k 2O 2O rate 3 k Substitution Method at equilibrium k 1[O 3] = k 2[O 2][O] rate 3 =k](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-45.jpg)
Substitution Method at equilibrium k 1[O 3] = k 2[O 2][O] rate 3 =k 3[O][O 3] [O] = k 1 [O 3] k 2 [O 2] substitute rate 3 = k 3 k 1 [O 3]2 k 2 [O 2] overall rate = k’ [O 3]2 [O 2] or

Problem: Derive the rate law for the following reaction given the mechanism step below: OCl - (aq) + I -(aq) OI -(aq) + Cl -(aq) OCl - + H 2 O I - + HOCl HOI + OH - k 1 k 3 k 2 k 4 HOCl + OH fast HOI + Cl slow H 2 O + OI fast
![Contd rate 1 k 1 OCl H 2 O rate 2 Cont’d rate 1 = k 1 [OCl -][H 2 O] = rate 2 =](https://slidetodoc.com/presentation_image/c637dc84c342958ad7d41d9dea548085/image-47.jpg)
Cont’d rate 1 = k 1 [OCl -][H 2 O] = rate 2 = k 2 [HOCl][OH -] [HOCl] = k 1[OCl -][H 2 O] k 2[OH -] solvent rate 3 = k 3 [HOCl][I -] rate 3 = k 3 k 1[OCl -][H 2 O][I -] k 2 [OH -] overall rate = k’ [OCl -][I -] [OH -]

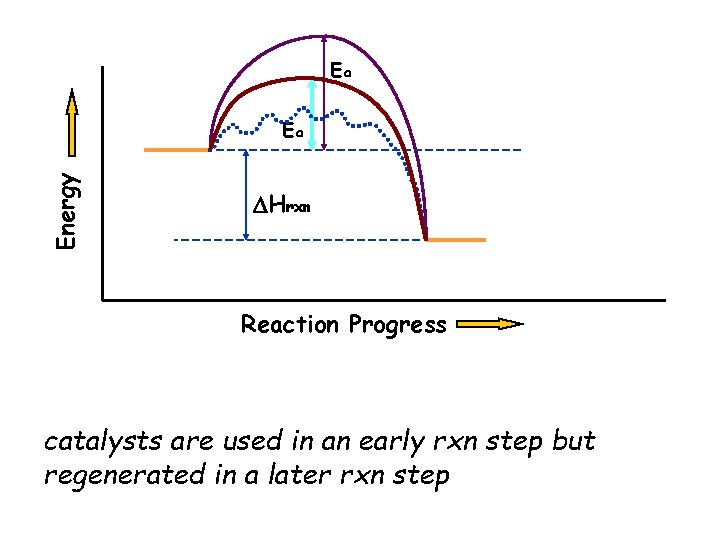
Catalyst • Facilitates the progress of a reaction by lowering the overall activation energy – homogeneous – heterogeneous

Ea Energy Ea DHrxn Reaction Progress catalysts are used in an early rxn step but regenerated in a later rxn step

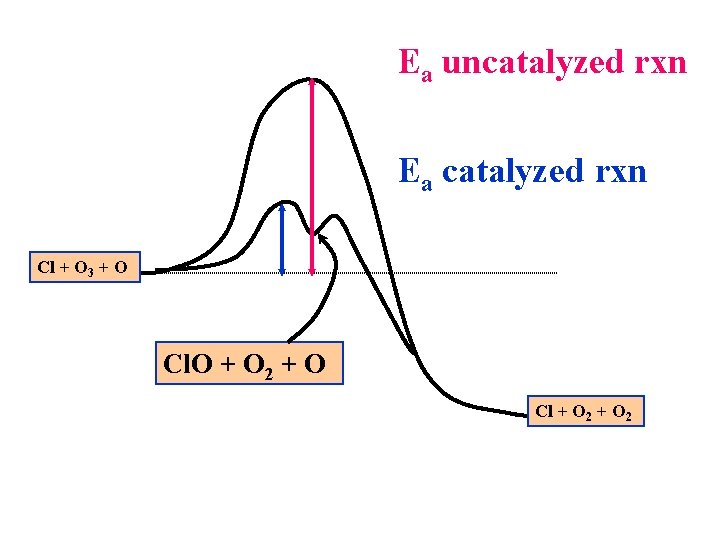
Uncatalyzed Reaction: O 3(g) <=> O(g) + O 2(g) + O(g) O 3(g) => 2 O 2(g) Catalyzed Reaction: Step 1: Cl(g) + O 3(g) + O(g) => Step 2: Cl. O(g) + O 2(g) + O(g) => Cl(g) + 2 O 2(g) Overall rxn: O 3(g) + O(g) Cl. O(g) + O 2(g) + O(g) => 2 O 2(g)

Ea uncatalyzed rxn Ea catalyzed rxn Cl + O 3 + O Cl. O + O 2 + O Cl + O 2