Chapter 14 Chemical Equilibrium Copyright Cengage Learning All

Chapter 14 Chemical Equilibrium Copyright © Cengage Learning. All rights reserved. 14 | 1

Contents and Concepts Describing Chemical Equilibrium 1. Chemical Equilibrium—A Dynamic Equilibrium 2. The Equilibrium Constant 3. Heterogeneous Equilibria; Solvents in Homogeneous Equilibria Using the Equilibrium Constant 4. Qualitatively Interpreting the Equilibrium Constant 5. Predicting the Direction of Reaction 6. Calculating Equilibrium Concentrations Copyright © Cengage Learning. All rights reserved. 14 | 2

Changing Reaction Conditions: Le Châtelier’s Principle 7. Removing Products or Adding Reactants 8. Changing the Pressure and Temperature 9. Effect of a Catalyst Copyright © Cengage Learning. All rights reserved. 14 | 3

Learning Objectives Describing Chemical Equilibrium 1. Chemical Equilibrium—A Dynamic Equilibrium a. Define dynamic equilibrium and chemical equilibrium. b. Apply stoichiometry to an equilibrium mixture. Copyright © Cengage Learning. All rights reserved. 14 | 4

2. The Equilibrium Constant a. Define equilibrium-constant expression and equilibrium constant. b. State the law of mass action. c. Write equilibrium-constant expressions. d. Describe the kinetics argument for the approach to chemical equilibrium. e. Obtain an equilibrium constant from reaction composition. Copyright © Cengage Learning. All rights reserved. 14 | 5

2. The Equilibrium Constant (cont. ) f. Describe the equilibrium constant Kp; indicate how Kp and Kc are related. State the law of mass action. g. Obtain Kc for a reaction that can be written as a sum of other reactions of known Kc values. Copyright © Cengage Learning. All rights reserved. 14 | 6

3. Heterogeneous Equilibria; Solvents in Homogeneous Equilibria a. Define homogeneous equilibrium and heterogeneous equilibrium. b. Write Kc for a reaction with pure solids or liquids. Using the Equilibrium Constant 4. Qualitatively Interpreting the Equilibrium Constant a. Give a qualitative interpretation of the equilibrium constant based on its value. Copyright © Cengage Learning. All rights reserved. 14 | 7

5. Predicting the Direction of Reaction a. Define reaction quotient, Q. b. Describe the direction of reaction after comparing Q with Kc. c. Use the reaction quotient. Copyright © Cengage Learning. All rights reserved. 14 | 8

6. Calculating Equilibrium Concentrations a. Obtain one equilibrium concentration given the others. b. Solve an equilibrium problem (involving a linear equation in x). c. Solve an equilibrium problem (involving a quadratic equation in x). Copyright © Cengage Learning. All rights reserved. 14 | 9

Changing the Reaction Conditions; Le Châtelier’s Principle 7. Removing Products or Adding Reactants a. State Le Châtelier’s principle. b. State what happens to an equilibrium when a reactant or product is added or removed. c. Apply Le Châtelier’s principle when a concentration is altered. Copyright © Cengage Learning. All rights reserved. 14 | 10

8. Changing the Pressure and Temperature a. Describe the effect of a pressure change on chemical equilibrium. b. Apply Le Châtelier’s principle when the pressure is altered. c. Describe the effect of a temperature change on chemical equilibrium. d. Apply Le Châtelier’s principle when the temperature is altered. e. Describe how the optimum conditions for a reaction are chosen. Copyright © Cengage Learning. All rights reserved. 14 | 11

9. Effect of a Catalyst a. Define catalyst. b. Compare the effect of a catalyst on rate of reaction with its effect on equilibrium. c. Describe how a catalyst can affect the product formed. Copyright © Cengage Learning. All rights reserved. 14 | 12

Chemical reactions often seem to stop before they are complete. Actually, such reactions are reversible. That is, the original reactants form products, but then the products react with themselves to give back the original reactants. When these two reactions—forward and reverse— occur at the same rate, a chemical equilibrium exists. Copyright © Cengage Learning. All rights reserved. 14 | 13
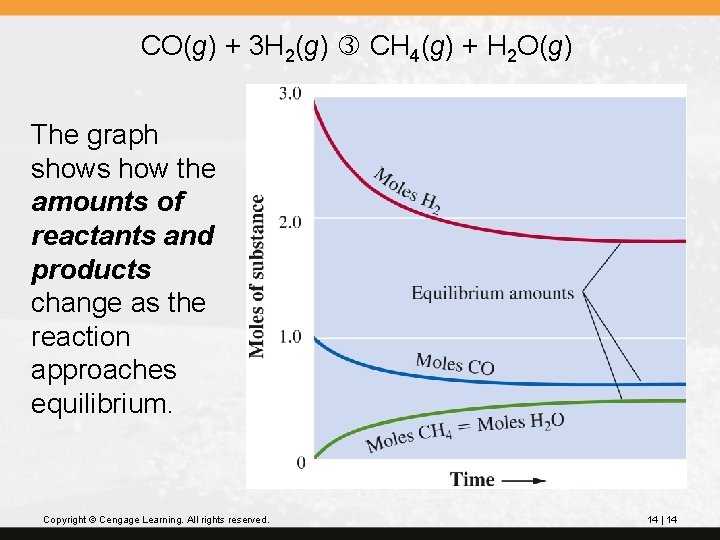
CO(g) + 3 H 2(g) CH 4(g) + H 2 O(g) The graph shows how the amounts of reactants and products change as the reaction approaches equilibrium. Copyright © Cengage Learning. All rights reserved. 14 | 14
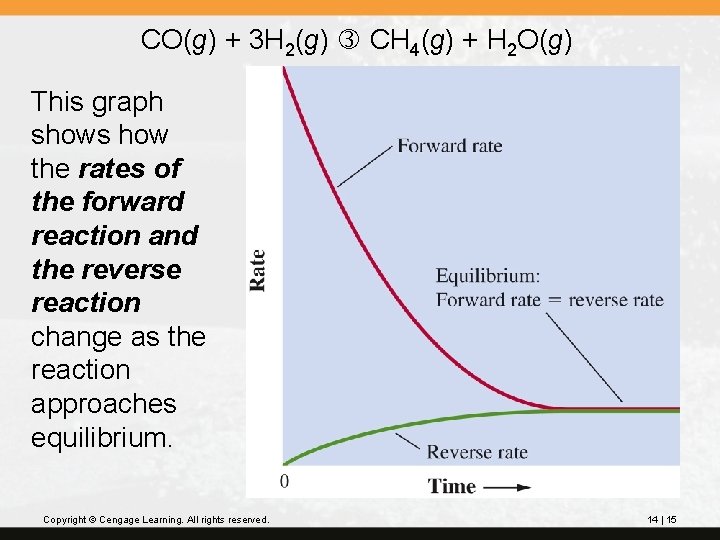
CO(g) + 3 H 2(g) CH 4(g) + H 2 O(g) This graph shows how the rates of the forward reaction and the reverse reaction change as the reaction approaches equilibrium. Copyright © Cengage Learning. All rights reserved. 14 | 15

Chemical equilibrium is the state reached by a reaction mixture when the rates of the forward and reverse reactions have become equal. Copyright © Cengage Learning. All rights reserved. 14 | 16

We can apply stoichiometry to compute the content of the reaction mixture at equilibrium. Copyright © Cengage Learning. All rights reserved. 14 | 17

? When heated PCl 5, phosphorus pentachloride, forms PCl 3 and Cl 2 as follows: PCl 5(g) PCl 3(g) + Cl 2(g) When 1. 00 mol PCl 5 in a 1. 00 -L container is allowed to come to equilibrium at a given temperature, the mixture is found to contain 0. 135 mol PCl 3. What is the molar composition of the mixture? Copyright © Cengage Learning. All rights reserved. 14 | 18

We will organize this problem by using the chemical reaction to set up a table of initial, change, and equilibrium amounts. Initially we had 1. 00 mol PCl 5 and no PCl 3 or Cl 2. The change in each is stoichiometric: If x moles of PCl 5 react, then x moles of PCl 3 and x moles of Cl 2 are produced. For reactants, this amount is subtracted from the original amount; for products, it is added to the original amount. Copyright © Cengage Learning. All rights reserved. 14 | 19

PCl 5(g) PCl 3(g) + Cl 2(g) Initial 1. 00 mol 0 0 –x +x +x 1. 00 – x x x Change Equilibrium We were told that the equilibrium amount of PCl 3 is 0. 135 mol. That means x = 0. 135 mol. We can now find the amounts of the other substances. Copyright © Cengage Learning. All rights reserved. 14 | 20

Moles PCl 5 = 1. 00 – 0. 135 = 0. 87 mol (2 decimal places) Moles PCl 3 = 0. 135 mol (given with 3 significant figures) Moles Cl 2 = 0. 135 mol Copyright © Cengage Learning. All rights reserved. 14 | 21

The Equilibrium Constant, Kc The equilibrium constant expression for a reaction is obtained by multiplying the concentrations of products, dividing by the concentrations of reactants, and raising each concentration term to a power equal to its coefficient in the balanced chemical equation. The equilibrium constant, Kc, is the value obtained for the Kc expression when equilibrium concentrations are substituted. Copyright © Cengage Learning. All rights reserved. 14 | 22

For the reaction a. A + b. B c. C + d. D The equilibrium constant expression is Kc = Copyright © Cengage Learning. All rights reserved. 14 | 23

? Methanol (also called wood alcohol) is made commercially by hydrogenation of carbon monoxide at elevated temperature and pressure in the presence of a catalyst: 2 H 2(g) + CO(g) CH 3 OH(g) What is the Kc expression for this reaction? Copyright © Cengage Learning. All rights reserved. 14 | 24

When we are given some information about equilibrium amounts, we are able to calculate the value of Kc. We need to take care to remember that the Kc expression uses molar concentrations. Copyright © Cengage Learning. All rights reserved. 14 | 25

? Carbon dioxide decomposes at elevated temperatures to carbon monoxide and oxygen: 2 CO 2(g) 2 CO(g) + O 2(g) At 3000 K, 2. 00 mol CO 2 is placed into a 1. 00 -L container and allowed to come to equilibrium. At equilibrium, 0. 90 mol CO 2 remains. What is the value of Kc at this temperature? Copyright © Cengage Learning. All rights reserved. 14 | 26

2 CO 2(g) 2 CO(g) + O 2(g) Initial 2. 00 mol 0 0 – 2 x +x 2. 00 – 2 x 2 x x Change Equilibrium 0. 90 mol 1. 10 mol 0. 55 mol We can find the value of x. 2. 00 – 2 x = 0. 90 2 x = 2(0. 55) = 1. 10 mol 1. 10 = 2 x x = 0. 55 mol Copyright © Cengage Learning. All rights reserved. 14 | 27

2 CO 2(g) 2 CO(g) + O 2(g) Copyright © Cengage Learning. All rights reserved. 14 | 28

In a heterogeneous equilibrium, in the Kc expression, the concentrations of solids and pure liquids are constant (due to these substances’ constant density). As a result, we incorporate those constants into the value of Kc, thereby making a new constant, Kc. In other words, equilibrium is not affected by solids and pure liquids as long as some of each is present. More simply, we write the Kc expression by replacing the concentration of a solid or pure liquid with 1. Copyright © Cengage Learning. All rights reserved. 14 | 29

? Write the Kc expression for the following reaction: H 2 O(g) + C(s) CO(g) + H 2(g) Copyright © Cengage Learning. All rights reserved. 14 | 30

Given: a. A + b. B c. C + d. D; K 1 When the reaction is reversed: c. C + d. D a. A + b. B; K 2 The equilibrium constant expression is inverted: K 2 = Copyright © Cengage Learning. All rights reserved. 14 | 31

Given: a. A + b. B c. C + d. D; K 1 When the reaction is doubled: 2 a. A + 2 b. B 2 c. C + 2 d. D; K 2 The equilibrium constant expression, K 2 , is the square of the equilibrium constant expression, K 1: K 2 = Copyright © Cengage Learning. All rights reserved. 14 | 32

For the reaction a. A(g) + b. B(g) c. C(g) + d. D(g) The equilibrium constant expressions are Kc = Copyright © Cengage Learning. All rights reserved. and Kp = 14 | 33

How are these related? We know From the ideal gas law, we know that So, Copyright © Cengage Learning. All rights reserved. 14 | 34

When you express an equilibrium constant for a gaseous reaction in terms of partial pressures, you call it the equilibrium constant, Kp. In general, the value of Kp is different from that of Kc. We will explore this relationship on the next slides. Recall the ideal gas law and the relationship between pressure and molarity of a gas: Copyright © Cengage Learning. All rights reserved. 14 | 35

Kp = Kc (RT)Dn Copyright © Cengage Learning. All rights reserved. 14 | 36

For catalytic methanation, CO(g) + 3 H 2(g) CH 4(g) + H 2 O(g) the equilibrium expression in terms of partial pressures becomes and Copyright © Cengage Learning. All rights reserved. 14 | 37

? The value of Kc at 227°C is 0. 0952 for the following reaction: CH 3 OH(g) CO(g) + 2 H 2(g) What is Kp at this temperature? Kp = 0. 0952(RT)Dn where T = 227 + 273 = 500. K R = 0. 08206 L atm/(mol K) Dn = 2 Kp = 1. 60 × 102 Copyright © Cengage Learning. All rights reserved. 14 | 38

We can use the value of the equilibrium constant in several ways. First, we can qualitatively describe the content of the reaction mixture by looking at the magnitude of Kc. Second, we can determine the direction in which a reaction will proceed by comparing Kc to the value of the reaction quotient, Q, which has the same expression as Kc but uses nonequilibrium values. Copyright © Cengage Learning. All rights reserved. 14 | 39

Finally, we can determine equilibrium concentrations given the initial concentrations and the value of Kc. Copyright © Cengage Learning. All rights reserved. 14 | 40

When Kc is very large (>102), the equilibrium mixture is mostly products. When Kc is very small (<10 -2), the equilibrium mixture is mostly reactants. When Kc approaches 1, the equilibrium mixture contains appreciable amounts of both reactants and products. Copyright © Cengage Learning. All rights reserved. 14 | 41

? Kc = 0. 82 for a reaction. Describe the composition of the equilibrium mixture. Because Kc < 100 and > 0. 01, at equilibrium there will be substantial amounts of both reactants and products. Copyright © Cengage Learning. All rights reserved. 14 | 42

Reaction Quotient, Q The reaction quotient has the same form as the equilibrium constant, but uses initial concentrations for its value. When Kc > Q, the reaction proceeds to the right. When Kc < Q, the reaction proceeds to the left. When Kc = Q, the reaction is at equilibrium. Copyright © Cengage Learning. All rights reserved. 14 | 43
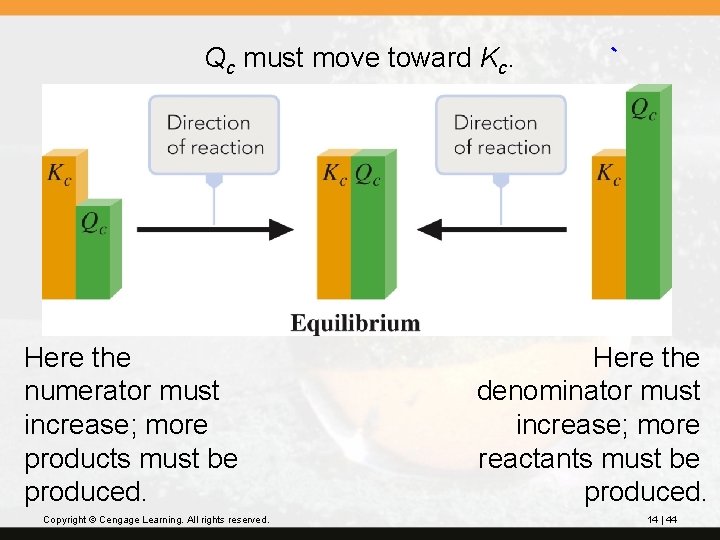
Qc must move toward Kc. Here the numerator must increase; more products must be produced. Copyright © Cengage Learning. All rights reserved. Here the denominator must increase; more reactants must be produced. 14 | 44

Calculating Equilibrium Concentrations 1. When all but one equilibrium concentration and the value of Kc are known. 2. When the value of Kc and the initial concentrations are known. a. When the Kc expression is a perfect square: solving a linear equation. b. When the Kc expression is not a perfect square: solving a quadratic equation. Copyright © Cengage Learning. All rights reserved. 14 | 45

? Nickel(II) oxide can be reduced to the metal by treatment with carbon monoxide. CO(g) + Ni. O(s) CO 2(g) + Ni(s) If the partial pressure of CO is 100. mm. Hg and the total pressure of CO and CO 2 does not exceed 1. 0 atm, will this reaction occur at 1500 K at equilibrium? (Kp = 700. at 1500 K. ) Copyright © Cengage Learning. All rights reserved. 14 | 46

Copyright © Cengage Learning. All rights reserved. 14 | 47

? Nitrogen and oxygen form nitric oxide. N 2(g) + O 2(g) 2 NO(g) If an equilibrium mixture at 25°C contains 0. 040 M N 2 and 0. 010 M O 2, what is the concentration of NO in this mixture? The equilibrium constant at 25°C is 1. 0 × 10− 30. Copyright © Cengage Learning. All rights reserved. 14 | 48

N 2(g) + O 2(g) 2 NO(g) Copyright © Cengage Learning. All rights reserved. 14 | 49

When the initial concentration and the value of Kc are known, we return to the stoichiometric chart of initial, change, and equilibrium (ICE) amounts or concentrations to find the equilibrium concentrations. Copyright © Cengage Learning. All rights reserved. 14 | 50

? Hydrogen iodide decomposes to hydrogen gas and iodine gas. 2 HI(g) H 2(g) + I 2(g) At 800 K, the equilibrium constant, Kc, for this reaction is 0. 016. If 0. 50 mol HI is placed in a 5. 0 -L flask, what will be the composition of the equilibrium mixture in molarities? Copyright © Cengage Learning. All rights reserved. 14 | 51
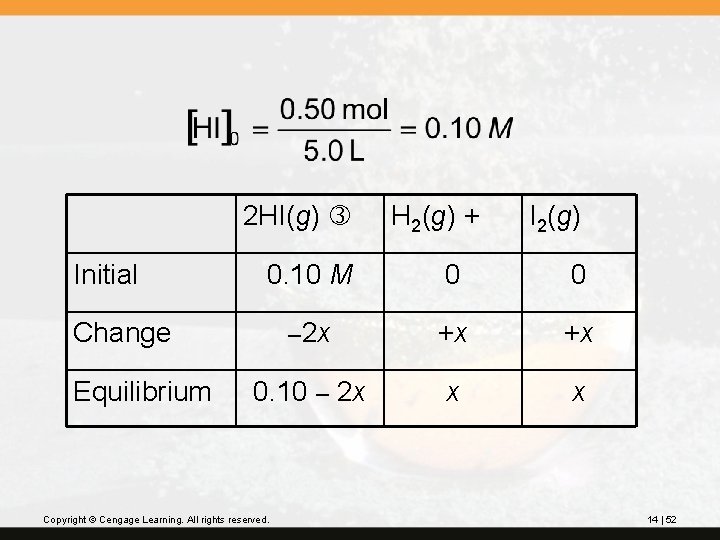
2 HI(g) Initial I 2(g) 0. 10 M 0 0 – 2 x +x +x 0. 10 – 2 x x x Change Equilibrium H 2(g) + Copyright © Cengage Learning. All rights reserved. 14 | 52

The Kc expression is, Kc = Substituting: Because the right side of the equation is a perfect square, we can take the square root of both sides. Copyright © Cengage Learning. All rights reserved. 14 | 53

Solving: 0. 126(0. 10 – 2 x) = x 0. 0126 – 0. 252 x = x 0. 0126 = 1. 252 x x = 0. 010 M Substituting: Copyright © Cengage Learning. All rights reserved. 14 | 54

When the Kc expression is not a perfect square, the equation must be rearranged to fit the quadratic format: ax 2 + bx + c = 0 The solution is Copyright © Cengage Learning. All rights reserved. 14 | 55

? N 2 O 4 decomposes to NO 2. The equilibrium reaction in the gas phase is N 2 O 4(g) 2 NO 2(g) At 100°C, Kc = 0. 36. If a 1. 00 -L flask initially contains 0. 100 M N 2 O 4, what will be the equilibrium concentration of NO 2? Copyright © Cengage Learning. All rights reserved. 14 | 56
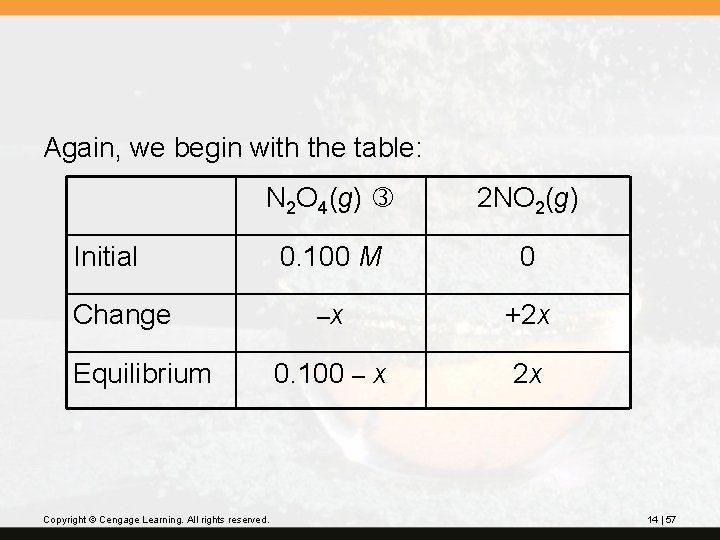
Again, we begin with the table: N 2 O 4(g) 2 NO 2(g) 0. 100 M 0 –x +2 x 0. 100 – x 2 x Initial Change Equilibrium Copyright © Cengage Learning. All rights reserved. 14 | 57

The Kc expression is, Kc = Substitute: Rearrange: Substitute: Copyright © Cengage Learning. All rights reserved. 14 | 58

Solve: We eliminate the negative value because it is impossible to have a negative concentration. Substitute to find the equilibrium concentration of NO 2: Copyright © Cengage Learning. All rights reserved. 14 | 59

? Given: H 2(g) + F 2(g) 2 HF(g); Kc = 1. 15 × 102 3. 000 mol of each species is put in a 1. 500 -L vessel. What is the equilibrium concentration of each species? First calculate the initial concentrations: Copyright © Cengage Learning. All rights reserved. 14 | 60

Initial Change Equilibrium H 2(g) + F 2(g) 2 HF(g) 2. 000 M –x –x +2 x 2. 000 – x 2. 000 + 2 x Copyright © Cengage Learning. All rights reserved. 14 | 61

Copyright © Cengage Learning. All rights reserved. 14 | 62

Now compute the equilibrium concentrations: Double-check by substituting these equilibrium concentrations into the Kc expression and solving. The answer should be the value of Kc. This is within round-off error. Copyright © Cengage Learning. All rights reserved. 14 | 63

? The value of Kc at 227°C is 0. 0952 for the following reaction: CH 3 OH(g) CO(g) + 2 H 2(g) What is Kp at this temperature? Kp = 0. 0952(RT)Dn T = 227 + 273 = 500. K R = 0. 08206 L atm/(mol K) Dn = 2 Kp = 1. 60 × 102 Copyright © Cengage Learning. All rights reserved. 14 | 64

Le Châtelier’s Principle When a system in chemical equilibrium is disturbed by a change in • temperature, • pressure, or • concentration, the system shifts in equilibrium composition in a way that tends to counteract this change of variable. Copyright © Cengage Learning. All rights reserved. 14 | 65

When a substance that is part of the equilibrium is added to the mixture, the equilibrium shifts to use it (in a direction that makes the substance a reactant). When a substance that is part of the equilibrium is removed from the mixture, the equilibrium shifts to produce it (in a direction that makes the substance a product). Copyright © Cengage Learning. All rights reserved. 14 | 66

Changes in the partial pressure of substances that are part of the equilibrium are handled in the same way as adding or removing a substance. Copyright © Cengage Learning. All rights reserved. 14 | 67

? The following reaction is at equilibrium: COCl 2(g) CO(g) + Cl 2(g) a. Predict the direction of reaction when chlorine gas is added to the reaction mixture. b. Predict the direction of reaction when carbon monoxide gas is removed from the mixture. Copyright © Cengage Learning. All rights reserved. 14 | 68

COCl 2(g) CO(g) + Cl 2(g) a. When we add Cl 2, the reaction will shift in the reverse direction to use it. Note: reverse = left = . b. When we remove CO, the reaction will shift in the forward direction to produce it. Note: forward = right = . Copyright © Cengage Learning. All rights reserved. 14 | 69

A change in the total pressure occurs because of a change in the volume of the reaction container. When the size of the container decreases, the overall pressure increases. The reaction will shift to reduce the pressure—that is, it will shift toward the side of the reaction with fewer gas molecules. Copyright © Cengage Learning. All rights reserved. 14 | 70

When the size of the container increases, the overall pressure decreases. The reaction will shift to increase the pressure—that is, it will shift toward the side with more gas molecules. In the event that both sides of the equilibrium reaction have the same number of moles of gas, pressure has no effect on the equilibrium. Copyright © Cengage Learning. All rights reserved. 14 | 71

? In which direction will each reaction shift when the volume of the reaction container is increased? a. CO(g) + 2 H 2(g) CH 3 OH(g) b. 2 SO 2(g) + O 2(g) 2 SO 3(g) c. COCl 2(g) CO(g) + Cl 2(g) Copyright © Cengage Learning. All rights reserved. 14 | 72

When the container volume is increased, the total pressure is decreased. Each system will shift to produce more gas by shifting toward the side with more moles of gas. a. CO(g) + 2 H 2(g) CH 3 OH(g) This reaction shifts reverse = left = b. 2 SO 2(g) + O 2(g) 2 SO 3(g) This reaction shifts reverse = left = c. COCl 2(g) CO(g) + Cl 2(g) This reaction shifts forward = right = Copyright © Cengage Learning. All rights reserved. 14 | 73

Changing the temperature changes the value of the equilibrium constant. Changing the temperature can also cause a shift in the equilibrium. The direction of each of these changes depends on the sign of DHo. Copyright © Cengage Learning. All rights reserved. 14 | 74

For an endothermic reaction, DHo > 0 (positive), we consider that heat is a reactant. For an exothermic reaction, DHo < 0 (negative), we consider that heat is a product. Copyright © Cengage Learning. All rights reserved. 14 | 75

For an endothermic reaction, increasing the temperature increases the value of Kc. For an exothermic reaction, increasing the temperature decreases the value of Kc. Decreasing the temperature has the opposite effect. Copyright © Cengage Learning. All rights reserved. 14 | 76

In addition to the value of Kc, we can consider the direction in which the equilibrium will shift. When heat is added (temperature increased), the reaction will shift to use heat. When heat is removed (temperature decreased), the reaction will shift to produce heat. Copyright © Cengage Learning. All rights reserved. 14 | 77

? Given: 2 H 2 O(g) 2 H 2(g) + O 2(g); DH° = 484 k. J Would you expect this reaction to be favorable at high or low temperatures? We rewrite the reaction to include heat: Heat + 2 H 2 O(g) 2 H 2(g) + O 2(g) When heat is added, the reaction shifts forward = right = . The reaction is favorable at high temperatures. Copyright © Cengage Learning. All rights reserved. 14 | 78

? The Fischer–Tropsch process for the synthesis of gasoline consists of passing a mixture of carbon monoxide and hydrogen over an iron–cobalt catalyst. A typical reaction that occurs in the process is 8 CO(g) + 17 H 2(g) C 8 H 18(g) + 8 H 2 O(g) Suppose the reaction is at equilibrium at 200°C, then is suddenly cooled to condense the octane, and then the remaining gases are reheated to 200°C. In which direction will the equilibrium shift? Copyright © Cengage Learning. All rights reserved. 14 | 79

This is essentially removing octane, a product. This change causes the reaction to produce octane by shifting forward = right = . Copyright © Cengage Learning. All rights reserved. 14 | 80

? A typical reaction that occurs in the Fischer– Tropsch process is 8 CO(g) + 17 H 2(g) C 8 H 18(g) + 8 H 2 O(g) In which direction will the equilibrium shift when the pressure is increased? When the overall pressure is increased, the reaction will shift to reduce the pressure—that is, the reaction shifts to fewer gas molecules. In this case, the reaction will shift forward = right = Copyright © Cengage Learning. All rights reserved. 14 | 81
- Slides: 81