Chapter 14 Chemical Equilibrium Chemical Equilibrium In a

Chapter 14 Chemical Equilibrium

Chemical Equilibrium �In a chemical system, when the concentrations of both reactants and products reach a steady state, they are said to be in a dynamic equilibrium. �This occurs when both the forward rate and the reverse rate are equal. �Reactants are still making products, but products are also making reactants. �Static equilibrium is when no change is occurring – like in a tug-o-war.
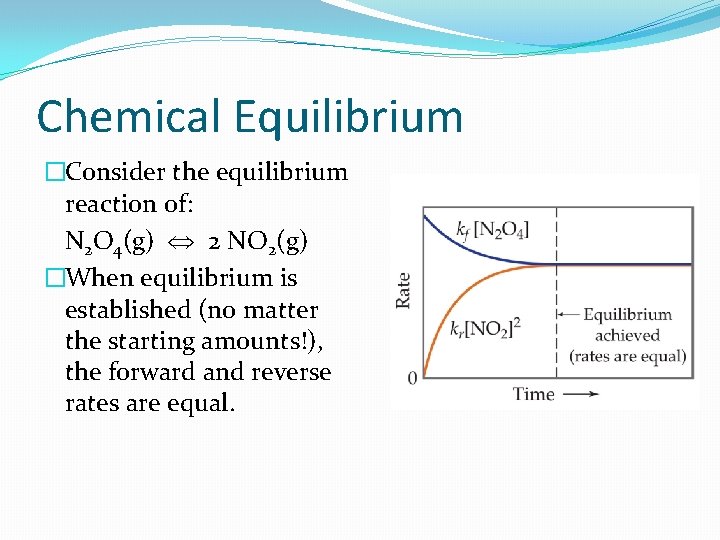
Chemical Equilibrium �Consider the equilibrium reaction of: N 2 O 4(g) 2 NO 2(g) �When equilibrium is established (no matter the starting amounts!), the forward and reverse rates are equal.
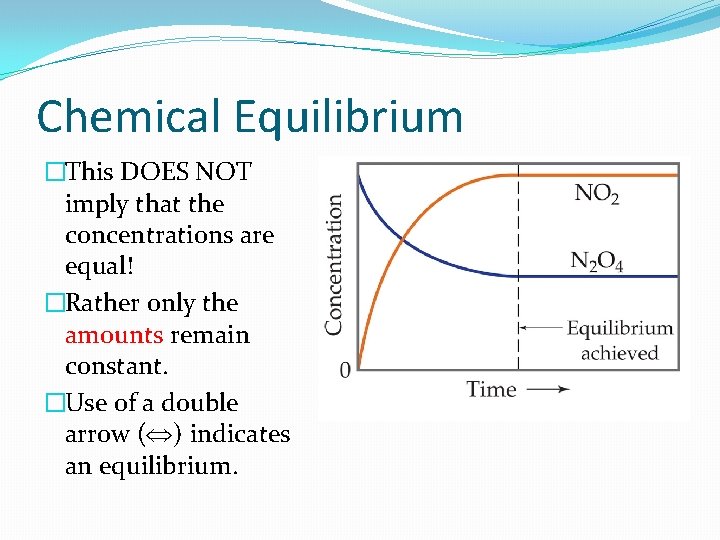
Chemical Equilibrium �This DOES NOT imply that the concentrations are equal! �Rather only the amounts remain constant. �Use of a double arrow ( ) indicates an equilibrium.

Chemical Equilibrium �Analogy with traffic flow on a bridge. �More traffic coming in to the city than leaving. �But, if the rates coming in are constant and the rates going out are constant, then it is a dynamic equilibrium.

Equilibrium Constant, K �For the N 2 O 4 reaction, the forward rate is: Ratef = kf [N 2 O 4]1 �The reverse rate is: Rater = kr [NO 2]2 �Setting these equal to each other: kf [N 2 O 4]1 = kr [NO 2]2

Equilibrium Constant, K �Re-arranging to: �Where the ratio of kr / kf = K, the equilibrium constant.

K �This concept of the equilibrium is also referred to as the law of mass action. �In general, for a reaction of the type: a. A + b. B c. C + d. D �The equilibrium constant will be:

K �Equilibrium amounts can be in moles per liter (Molarity) or as partial pressures, if gases are present. �The equilibrium constant, K, then is labeled as Kc or Kp. �As we do more types of equilibria, you will also see K a, Kb, Ksp, etc. �K is always unitless, regardless of any reaction.

K �How do we prove that the law of mass action works? �Experiments, experiments.
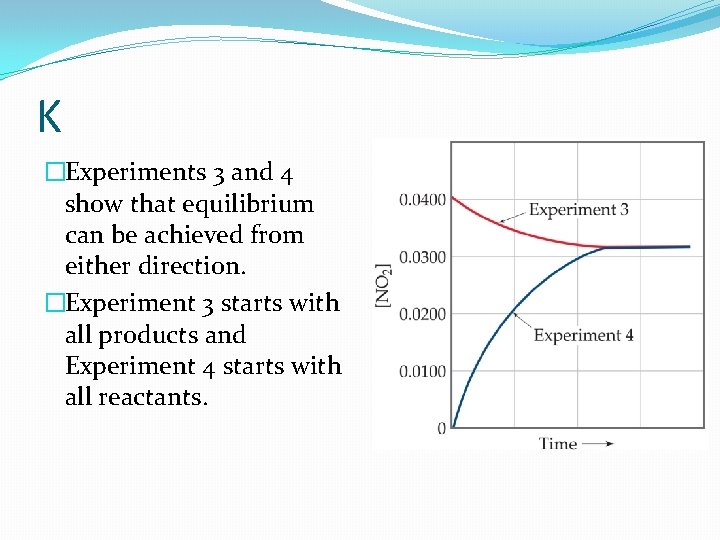
K �Experiments 3 and 4 show that equilibrium can be achieved from either direction. �Experiment 3 starts with all products and Experiment 4 starts with all reactants.

K �Are Kc and Kp the same? �Recall that PV = n. RT from Chapter 10. �Molarity = n / V, so… �P = M(RT) �Resulting in: �Kp = Kc(RT)Dn �Where Dn =
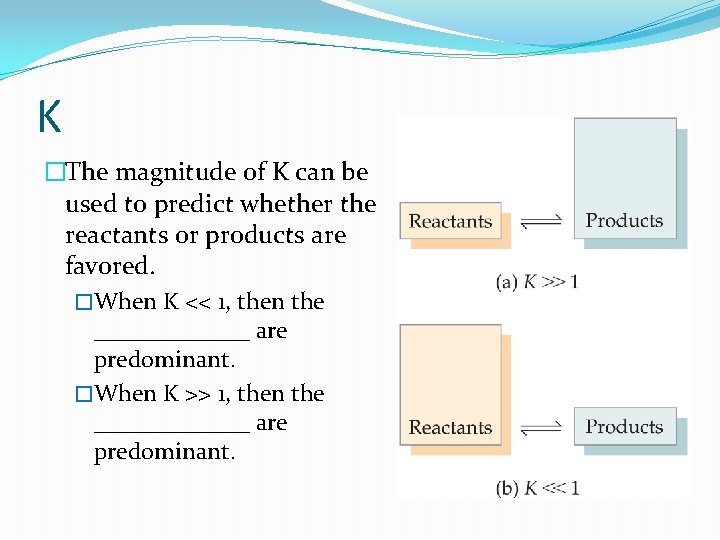
K �The magnitude of K can be used to predict whether the reactants or products are favored. �When K << 1, then the _______ are predominant. �When K >> 1, then the _______ are predominant.

K �What happens to K if a reaction is reversed? �N 2 O 4 2 NO 2 ; Kc = 0. 212 � 2 NO 2 N 2 O 4 ; Kc = ? ? ? �What happens to K if a reaction is multiplied by a factor? � 2 N 2 O 4 4 NO 2 ; Kc = ? ? ?

K �What happens when two reactions are added together like in Hess’ Law? � 2 NOBr 2 NO + Br 2 ; Kc = 0. 014 �Br 2 + Cl 2 2 Br. Cl ; Kc = 7. 2 �__________________ � 2 NOBr + Cl 2 2 NO + 2 Br. Cl ; Kc = ? ? ?

K �When all substances in an equilibrium are of the same phase, then that equilibrium is homogeneous. �When two different phases are present, the equilibrium is said to be heterogeneous. �The amounts of solids and/or liquids in the presence of gases or aqueous compounds are in large excess. �Thus, their concentrations do not change.

K Consider the equilibrium for: Ca. CO 3(s) Ca. O(s) + CO 2(g)

Evaluating K �If given equilibrium amounts of all reactants and products, then a value for Kc can be found. �Or – can find an unknown concentration if Kc is known. �Can also use basic stoichiometric relationships in some problems. �These require an ICE table.

Evaluating K �ICE stands for Initial, Change, and Equilibrium. �Set-up as a table under the reaction. �Initial = starting amounts in Molarity. �Change = includes a sign plus a variable and reflects mole-to-mole amounts. �Equilibrium = combines the I and C lines. �Suppose a 4. 0 L flask has 0. 20 moles of N 2 O 4 initially added to it, the ICE table set-up would be:

Evaluating K N 2 O 4 2 NO 2 N 2 O 4 Initial Change Equilibrium NO 2

Evaluating K �If the equilibrium amounts of the reactants and products are unknown, then an equation with a variable must be set-up and solved. �Must be given the value of K. �Two common problems are the perfect square problem and the quadratic problem.

Evaluating K �Problems with partial pressures are based on Dalton’s Law of Partial Pressures. �Ptotal = Pa + Pb + Pc + … �Thus, if the intital total pressure is known, then one gas has a pressure of “x” and the other is this pressure minus “x”.

Reaction Quotient �Given amounts of all reactants and products, which direction will the reaction move? �The Q expression is identical to K. �Can think of Q as a continuum. Q = 0, all reactants Q = , all products

Reaction Quotient �When Q < K, the system has too many reactants and not enough products.

Reaction Quotient �When Q > K, the system has too many products and not enough reactants.
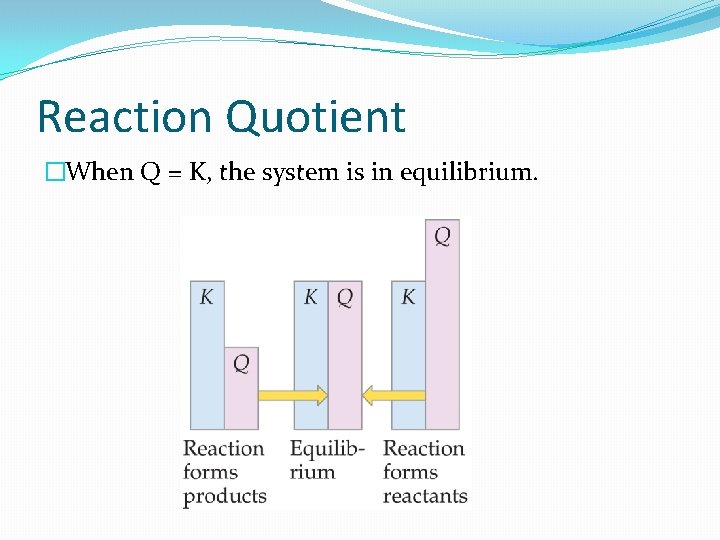
Reaction Quotient �When Q = K, the system is in equilibrium.

Le. Chatelier’s Principle �If a system in equilibrium is disturbed by a change in temperature, pressure, or the concentration of one of the components, the system will shift its equilibrium position to counteract the disturbance. �Certain reactions, like the production of NH 3 require the manipulation of an equilibrium to maximize the product.

Le. Chatelier’s Principle �Haber process – Fritz Haber looked at P, T effects on making NH 3. �Reaction favors lower T and higher P. �Why?

Le. Chatelier’s Principle �Changes in reactant or product concentrations will force the equilibrium to re-balance just like a two pan balance. �Can add or remove components in the equilibrium system.
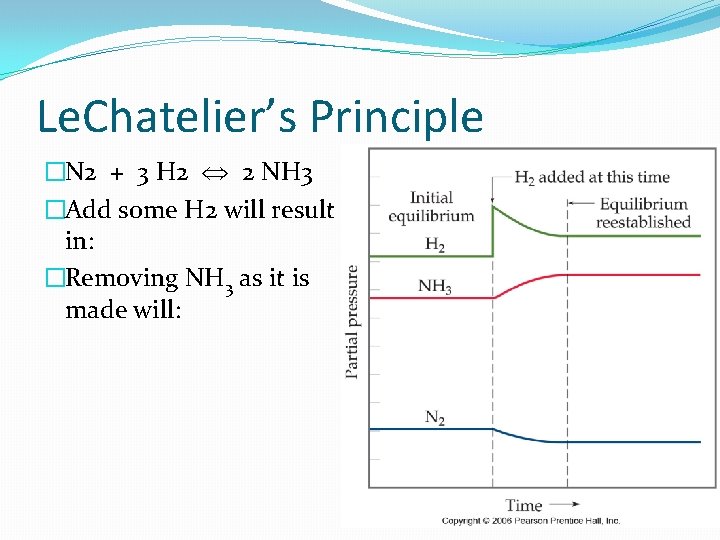
Le. Chatelier’s Principle �N 2 + 3 H 2 2 NH 3 �Add some H 2 will result in: �Removing NH 3 as it is made will:

Le. Chatelier’s Principle �Pressure changes �As Boyle’s Law predicts, as P increases, V decreases. �For a system to reduce its volume, it must reduce the moles of gas present. �For reactions with different moles of products and moles of reactants, increasing the pressure will favor the side with fewer total moles of gases. �Of course, the opposite is true if the pressure is decreased. �Does this agree with Haber results?
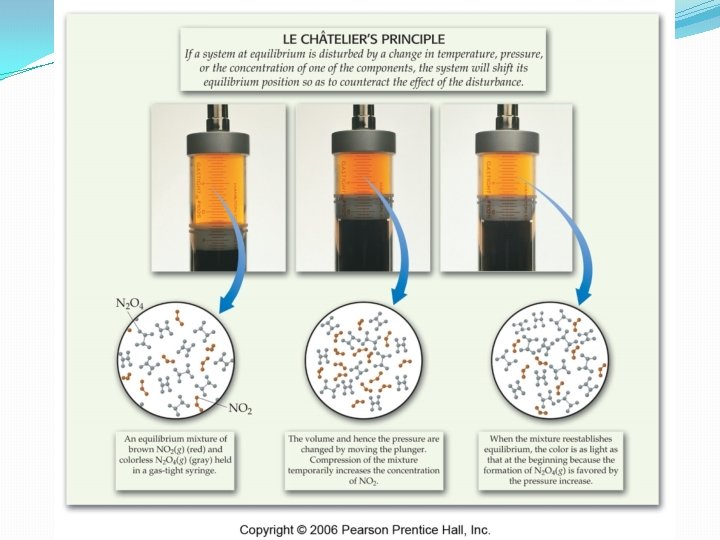

Le. Chatelier’s Principle �Changing the pressure at constant temperature does NOT change the value of K. �As the total pressure is increased, the relative partial pressures increase, but not equally. �Thus, the K value remains the same.

Le. Chatelier’s Principle �Temperature changes �Depends on whether the reaction is endo- or exothermic. �Endothermic: Reactants + Heat Products � As T increases, reaction shifts to right and K will ______ �Exothermic: Reactants Products + Heat � As T increases, reaction shifts to left and K will ______
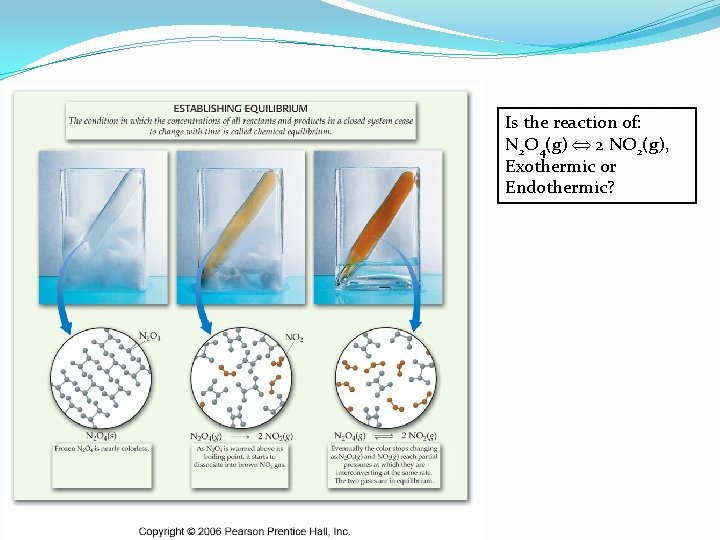
Is the reaction of: N 2 O 4(g) 2 NO 2(g), Exothermic or Endothermic?
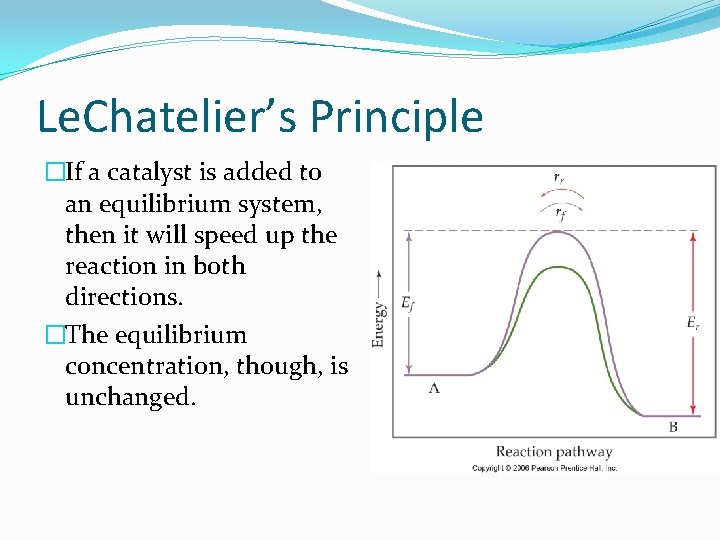
Le. Chatelier’s Principle �If a catalyst is added to an equilibrium system, then it will speed up the reaction in both directions. �The equilibrium concentration, though, is unchanged.

Ammonia Production �N 2(g) + 3 H 2(g) 2 NH 3(g); DH = -92 k. J �Favors high or low temperatures? �Favors high or low pressures? �Removal of NH 3 as it is made �Catalyst – iron oxides �Over 100 million tons produced per year using 1 -2% of the annual world’s energy supply. �Plants and some bacteria can perform the same reaction at much lower temperatures and pressures.

Ammonia Production
- Slides: 38