CHAPTER 14 Chemical Equilibrium 14 1 Equilibrium Constant

CHAPTER 14 Chemical Equilibrium

14. 1: Equilibrium Constant, Keq Objective: (1) To write the equilibrium constant expression for a chemical reaction.

Reversible Reactions and Equilibrium Reversible Reaction: A chemical reactions in which products re-form the original reactants. Arrows that point in opposite directions are used to indicate a reaction is reversible. Example: H 2(g) + I 2(g) 2 HI(g) Chemical Equilibrium: A state of balance in which the rate of a forward reaction equals the rate of the reverse reactions and the concentrations of products and reactants remain unchanged.

Equilibrium Constant, Keq Equilibrium Constant, Keq: a number that relates that concentrations of starting materials and products of a reversible chemical reaction to one another at a given temperature. a. A + b. B c. C + d. D coefficient concentration

Writing an Equilibrium Constant Expression • • Step 1: Balance the chemical equation. Step 2: Set up your Keq expression with the products on the top of a fraction and the reactants on the bottom of a fraction. Step 3: Raise each substance's concentration to the power equal to the substance’s coefficient in the balanced equation. Note: Solids (s) and pure liquids (l) are not used in the expression because their concentrations do not change.

Example Write the equilibrium constant expression for the following reaction: Ca. CO 3(s) + CO 2(aq) + H 2 O(l) Ca 2+(aq) + 2 HCO 3 -(aq)

Practice Write the equilibrium constant expression for the following chemical reactions at equilibrium (don’t forget to balance the equation): 1. ) H 2 CO 3(aq) + H 2 O(l) HCO 3 -(aq) + H 3 O+ (aq) 2. ) COCl 2 (g) 3. ) CO(g) + Cl 2 (g) C(s) + CO 2 (g)
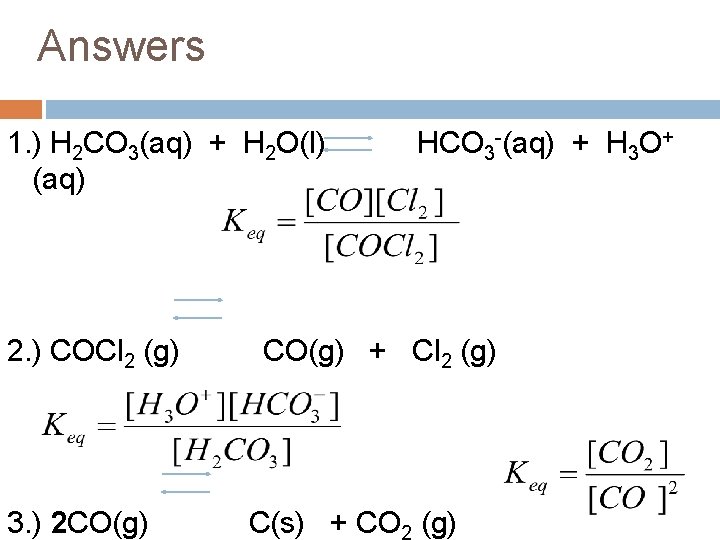
Answers 1. ) H 2 CO 3(aq) + H 2 O(l) (aq) 2. ) COCl 2 (g) 3. ) 2 CO(g) HCO 3 -(aq) + H 3 O+ CO(g) + Cl 2 (g) C(s) + CO (g)

14. 1: Equilibrium Constant, Keq Objective: (1) To calculate the equilibrium constant.

What does the Keq tell us? Keq < 1 Favors Reactants Keq = 1 Same amount of Reactants and Products Keq > 1 Favors Products Practice: Determine if the following Keq values favor the reactants, products, or neither. 1. ) Keq = 0. 02 2. ) Keq = 1 3. ) Keq = 50

Calculating Keq Step 1: Write the balanced chemical equation. Step 2: Set up your Keq expression. Step 3: Substitute concentrations. Step 4: Calculate!

Example An aqueous solution of carbonic acid reacts to reach equilibrium as described below: H 2 CO 3(aq) + H 2 O(l) HCO 3 -(aq) + H 3 O+ (aq) The solution contains the following solute concentrations: H 2 CO 3 = 3. 3 x 10 -2 M; HCO 3 - = 1. 19 x 10 -4 M; H 3 O+ = 1. 19 x 10 -4 M. Determine the Keq. Note: Keq does not have units!

Practice 1. a. Calculate the equilibrium constant for the following reaction: COCl 2(g) CO(g) + Cl 2(g) [CO] = 0. 0178 M [Cl 2] = 0. 0178 M [COCl 2] = 0. 00740 M b. Are the reactants for products favored?

Practice 2. a. For the system involving dinitrogen tetraoxide and nitrogen dioxide at equilibrium at a temperature of 100⁰C, the product concentration of N 2 O 4 is 4. 0 x 10 -2 M and the reactant concentration of NO 2 is 1. 4 x 10 -1 M. What is the Keq value for this reaction? NO 2(g) N 2 O 4(g) b. Are the reactants or products favored?

Practice 3. a. An equilibrium mixture at 852 K is found to contain 3. 61 x 10 -3 M of SO 2, 6. 11 x 10 -4 M of O 2, and 1. 01 x 10 -2 M of SO 3. Calculate the equilibrium constant for the reaction. SO 2 (g) + O 2 (g) SO 3 (g) b. Are the reactants or products favored?

Calculating Concentrations from Keq 4. Keq for the equilibrium below is 1. 8 x 10 -5 at a temperature of 25⁰C. Calculate [NH 3] when [NH 4+] and [OH-] are 3. 5 x 10 -4 M. NH 3(aq) + H 2 O(l) NH 4+ (aq) + OH-(aq) [NH 3] = 6. 8 x 10 -3 M

Practice 5. a. If the equilibrium constant is 1. 65 x 10 -3 at 2027⁰C for the reaction below, what is the equilibrium concentration of NO when [N 2] = 1. 8 x 10 -3 M and [O 2] = 4. 2 x 10 -3 M. N 2(g) + O 2(g) NO(g) b. Are the reactants for products favored?

Practice 6. a. At 600⁰C, the Keq for the reaction below is 4. 32 when [SO 3] = 0. 260 M and [O 2] = 0. 045 M. Calculate the equilibrium concentration for sulfur dioxide. SO 2(g) + O 2(g) SO 3(g) b. Are the reactants or products favored?

14. 2 Solubility Product Constant, Ksp Objective: (1) To calculate the solubility product constant, Ksp.

Solubility The maximum concentration of a salt in an aqueous solution is called the solubility of the salt in water.

Solubility Product Constant, Ksp Solubility Product Constant, Ksp: the equilibrium constant for a solid that is in equilibrium with the solid’s dissolved ions. How much of a partially soluble salt will dissolve? Aa. Bb (s) a. A (aq) + b. B (aq)

Calculating Ksp The lower the value of Ksp, the less soluble the substance. Practice: Rank the following substances from least soluble to most soluble: Salt Ag 2 CO 3 Ba. SO 4 Ca 3(PO 4)2 Cu. S Ksp 8. 4 x 10 -12 1. 1 x 10 -10 2. 1 x 10 -33 1. 3 x 10 -36

Calculating Ksp The lower the value of Ksp, the less soluble the substance. Practice: Rank the following substances from least soluble to most soluble: Salt Ag 2 CO 3 Ba. SO 4 Ca 3(PO 4)2 Cu. S Ksp 8. 4 x 10 -12 1. 1 x 10 -10 2. 1 x 10 -33 1. 3 x 10 -36 Cu. S Ca 3(PO 4)2 Ag 2 CO 3 Ba. SO 4 Least soluble Most soluble

Calculating Ksp Step 1: Write and Balance the equation. Step 2: Determine the concentration of the ions. Step 3: Write the solubility product expression. Step 4: Substitute values and calculate.

Example Most parts of oceans are nearly saturated with calcium fluoride. A saturated solution of Ca. F 2 at 25⁰C has a solubility of 3. 4 x 10 -4 M. Calculate the solubility product constant for Ca. F 2(s) Ca 2+(aq) + F-(aq)

Solution Balance equation: Ca. F 2(s) Ca 2+(aq) + 2 F(aq) 2. Determine Concentrations: Ca. F 2(s) Ca 2+(aq) + 2 F-(aq) 3. 4 x 10 -4 6. 8 x 10 -4 3. Write solubility product expression: 4. Substitute values and calculate: 1. Note: Ksp does not have units!

Practice 1. Copper(I) bromide is dissolved in water to saturation at 25⁰C. The concentration of Cu+ and Br- ions in solution is 7. 9 x 10 -5 M. Calculate the Ksp for copper(I) bromide at this temperature.

Practice 2. What is the Ksp value for calcium phosphate at 298 K if the concentrations in a solution at equilibrium with excess solid are 3. 42 x 10 -7 M for Ca 2+ and 2. 28 x 10 -7 M for PO 43 - ions?

Practice 3. If a saturated solution of silver chloride contains an Ag. Cl concentration of 1. 34 x 10 -5 M, what is the solubility product constant?

Practice 4. A saturated solution of magnesium fluoride contains a Mg. Cl 2 concentration of 1. 19 x 10 -3 M. What is the Ksp for magnesium fluoride?

Calculating Concentration from Ksp 5. What is the concentration of Ca 2+ in a saturated solution of Ca. F 2 if the concentration of F- is 2. 20 x 10 -3 M and Ksp = 5. 30 x 10 -9.

Practice 6. What is the concentration of Al 3+ in a saturated solution of Al(OH)3 if the OHconcentration is 7. 90 x 10 -9 M. Ksp = 1. 30 x 1033.

Practice: Chem 331 7. The Ksp for lead(II) iodide is 7. 08 x 10 -9 at 25⁰C. What is the molar concentration of Pb. I 2 in a saturated solution?

The Ksp for lead(II) iodide is 7. 08 x 10 -9 at 25⁰C. What is the molar concentration of Pb. I 2 in a saturated solution? Step 1: Write and Balance Equation Pb. I 2 (s) Pb 2+ (aq) + 2 I- (aq)

The Ksp for lead(II) iodide is 7. 08 x 10 -9 at 25⁰C. What is the molar concentration of Pb. I 2 in a saturated solution? Step 1: Write and Balance Equation Pb. I 2 (s) Pb 2+ (aq) + 2 I- (aq) Step 2: Write the Ksp expression

The Ksp for lead(II) iodide is 7. 08 x 10 -9 at 25⁰C. What is the molar concentration of Pb. I 2 in a saturated solution? Step 1: Write and Balance Equation Pb. I 2 (s) Pb 2+ (aq) + 2 I- (aq) Step 2: Write the Ksp expression Step 3: Assign x values to concentrations Pb. I 2 (s) Pb 2+ (aq) + 2 I- (aq) x x 2 x

The Ksp for lead(II) iodide is 7. 08 x 10 -9 at 25⁰C. What is the molar concentration of Pb. I 2 in a saturated solution? Step 1: Write and Balance Equation Pb. I 2 (s) Pb 2+ (aq) + 2 I- (aq) Step 2: Write the Ksp expression Step 3: Assign x values to concentrations Pb. I 2 (s) Pb 2+ (aq) + 2 I- (aq) x x 2 x Step 4: Solve x = [Pb. I 2] = 1. 21 x 10 -3 M

Practice 8. The Ksp of calcium sulfate is 9. 1 x 10 -6. What is the molar concentration of calcium sulfate in a saturated solution?

Practice 9. The Ksp of Cd. F 2 is 6. 4 x 10 -3. What is the molar concentration of cadmium fluoride in a saturated solution?

14. 3 Le. Chatelier’s Principle Objective: (1) To use Le. Chatelier’s Principle to determine how a system at equilibrium will respond to an external stress.

Le. Chatelier’s Principle Le. Chatelier’s Principle: When a system at equilibrium is disturbed, the system adjusts in a way to reduce the change. There are 3 possible disturbances: Change in (1) concentration, (2) temperature, or (3) pressure

1. Change in Concentration Increase concentration of reactant Equilibrium shifts toward products Decrease concentration of reactant Equilibrium shifts toward reactants Increase concentration of product Equilibrium shifts toward reactants Decrease concentration of product Equilibrium shifts toward products

Use the following reaction to answer the questions below: H 2 (g) + I 2 (g) 2 HI (g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase H 2 2. ) Decrease I 2 3. ) Increase HI 4. ) Decrease HI

Use the following reaction to answer the questions below: H 2 (g) + I 2 (g) 2 HI (g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase H 2 RIGHT 2. ) Decrease I 2 LEFT 3. ) Increase HI LEFT 4. ) Decrease HI RIGHT

2. Change in Temperature Think of heat as a reactant or product Exothermic: heat is a product Endothermic: heat is a reactant For an exothermic reaction: Increasing temperature equilibrium favors reactants Decreasing temperature equilibrium favors products For an endothermic reaction Increasing temperature equilibrium favors products Decreasing temperature equilibrium favors reactants

Use the following reaction to answer the questions below: 2 SO 3(g) + CO 2 (g) + heat CS 2 (g) + 4 O 2(g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase the temperature 2. ) Decrease the temperature

Use the following reaction to answer the questions below: 2 SO 3(g) + CO 2 (g) + heat CS 2 (g) + 4 O 2(g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase the temperature 2. ) Decrease the temperature RIGHT LEFT

3. Change in Pressure Only affects gases! Increasing pressure Equilibrium shifts toward the side with fewer moles of gas Decreasing pressure Equilibrium shifts toward the side with more moles of gas

Use the following reaction to answer the questions below: 2 SO 3(g) + CO 2 (g) + heat CS 2 (g) + 4 O 2(g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase the pressure 2. ) Decrease the pressure

Use the following reaction to answer the questions below: 2 SO 3(g) + CO 2 (g) + heat CS 2 (g) + 4 O 2(g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase the pressure LEFT (3 moles gas) 2. ) Decrease the pressure RIGHT (5 moles gas)

Use the following reaction to answer the questions below: H 2 (g) + I 2 (g) 2 HI (g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase Pressure 2. ) Decrease Pressure

Use the following reaction to answer the questions below: H 2 (g) + I 2 (g) 2 HI (g) In which direction (left or right) does the equilibrium shift in each of the following situations: 1. ) Increase Pressure NO CHANGE 2. ) Decrease Pressure NO CHANGE

Practice What direction will the equilibrium shift (left or right) in the reaction: ___POCl 3(g) ___PCl 3(g) + ___O 2 (g) + heat 1. ) Add PCl 3 2. ) Increase Pressure 3. ) Increase Temperature

Practice What direction will the equilibrium shift (left or right) in the reaction: _2_POCl 3(g) _2_PCl 3(g) + _1_O 2 (g) + heat 1. ) Add PCl 3 2. ) Increase Pressure 3. ) Increase Temperature LEFT
- Slides: 54