CHAPTER 14 CARBOXYLIC ACIDS Lecture 33 Carboxylic acids

CHAPTER 14 CARBOXYLIC ACIDS

Lecture 33: Carboxylic acids Subject: Organic Chemistry CLO LLO By the end of this Lecture, students should be able to: K 1. 2 L 33. 1 Write the IUPAC nomenclature of Carboxylic acids and their reaction products C 2. 1 L 33. 2 Recognize the medicinal importance of carboxylic acids Essential reading: General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caretorr

The functional group of carboxylic acids is -COOH. It is called the carboxy group. The term carboxylic is taken from two structural units carbonyl and hydroxyl. Its suffix is “oic acid”. The word acid tells us they are acidic in nature; they dissociate in water to release protons. Carboxylic acids and their derivatives ester, are common part of our daily lives. They are present in your salad. General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 482 -484

The tartness in Italian dressing on your salad. Propionic acid gives tangy flavor to the swiss cheese on your salad. Long chain carboxylic acids are called fatty acids, and they form esters when they react with alcohol such as glycerol. The olive oil in your salad dressing and fat in the meat are examples of triglycerides. The derivatives of carboxylic acid RCOOH are: RCONH 2 RCOCl RCOOR Amide Acid chloride Ester RCOOCOR Acid anhydride

Importance of carboxylic acids Fruit salad for breakfast have tangy esters 2 -methylbutyl ethanoate (bananas) or methylthiobutanoate (strawberries). Some carboxylic acids are key elements in exercise physiology, and others are used in treatment of diseases. Lactic acid is metabolic product built up in muscles and blood. Butyric acid is used to treat sickle cell anemia. General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 482 -484

Structure and Physical Properties -COOH group- Boiling Point o Carboxylic acids boil at a higher temperature than alcohols or aldehydes or ketones because of presence of two polar groups: carbonyl and hydroxyl. – CO and - OH o In Chapter 13 we saw that the carbonyl group is polar because oxygen is more electronegative than carbon. This produces a dipole and hence allows dipole-dipole attractions. o In Chapter 12, we saw that the hydroxyl group is very polar because the oxygen atom has a significantly higher electronegativity than the hydrogen atom. In addition, the oxygen atom has two unshared pairs of electrons. This results in a very polar bond in which the oxygen atom carries a partially negative charge and the hydrogen carries a partially positive charge. This structure allows hydrogen bonds, the attractive force between a hydrogen atom covalently bonded to a highly electronegative atom with unshared pairs of electrons, to form: H-O-H……. . O-H 2. General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 482 -484

Physical Characteristics Ø The lower-molar-mass carboxylic acids have sharp, sour tastes and unpleasant aromas. Ø Formic acid, HCOOH, is used as a chemical defense by ants and causes the burning sensation of the ant bite. Acetic acid, CH 3 COOH, is found in vinegar; propionic acid, CH 3 CH 2 COOH, is responsible for the tangy flavor of Swiss cheese; and butyric acid, CH 3 CH 2 COOH, causes the stench associated with rancid butter and gas gangrene. Ø The longer-chain carboxylic acids are generally called fatty acids and are important components of biological membranes and triglycerides, the major lipid storage form in the body. General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 482 -484

Solubility Carboxylic acids are highly soluble due to presence of hydrogen bond. As with alcohols, the smaller carboxylic acids are soluble in water. However, solubility falls off dramatically as the carbon content of the carboxylic acid increases because the molecules become more hydrocarbon like and less polar. For example, acetic acid (the two-carbon carboxylic acid found in vinegar) is completely soluble in water, but hexadecanoic acid (a sixteen-carbon carboxylic acid found in palm oil) is insoluble in water. General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 482 -484

Structure and Physical Properties -COOH group- Boiling Point Ø Thus, carboxylic acids can hydrogen bond to one another and form dimeric Hydrogen Bond. Ø The boiling point of different compounds is as given below for different compounds with the same molecular mass (MM) CH 3 CH 2 CH 3 Butane M. M. 58 b. p. 0. 5 C CH 3 CH 2 CHO Propanal M. M. 58 b. p. 49 C CH 3 CH 2 -O-CH 3 Methoxyethane M. M 60 b. p. 7. 0 C CH 3 CH 2 OH 1 -Propanol M. M. 60 b. p. 97. 2 C CH 3 CO CH 3 Propanone M. M. 58 b. p 56 C. CH 3 COOH Ethanoic acid M. M. 60. b. p 118 C General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 482 -484

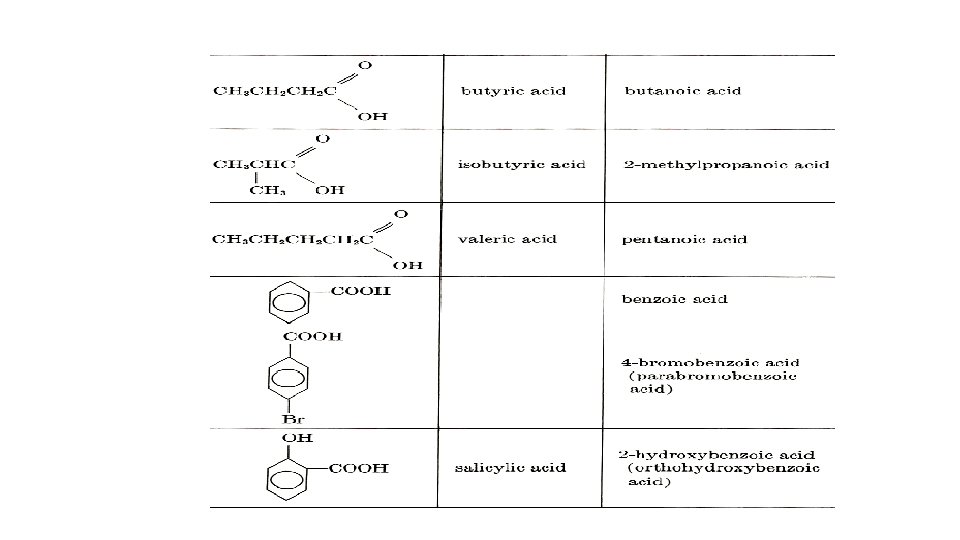
IUPAC Nomenclature • All organic acids have the function group –COOH. This group is given the number 1 in naming compounds. • The -e in the root name is dropped and oic plus the word acid is added.

Polyfunctional Carboxylic Acids : When functional groups like carboxylic acids, aldehydes, ketones, alcohols and amines are present in a molecule, carboxylic acid is given the highest priority and named with a suffix and the other functional groups are named with prefix. This can be summarized in the following table : Functional Group General Formula Suffix Carboxylic acids - COOH Aldehyde - CHO al oxo Ketone - CO - one oxo Alcohol - OH ol hydroxy Amine - NH 2 amine amino oic acid Prefix carboxy

IUPAC Nomenclature of Carboxylic acids Ø Carboxyl carbon is numbered 1 and its suffix is “oic acid” Draw the structures of: 2 -Hydroxypropanoic acid 3, 5, 7 -Tribromooctanoic acid 3 -Methylbutanoic acid Hexanedioic acid Propanedioic acid 2 -bromo-3 -chloro-4 -methylhexanoic acid 2 -Bromo-3 -chloro-4 -methylhexanoic acid 2, 4 -Dichlorobutanoic acid 2, 3 -Dihydroxy Butanoic acid 1, 4 -Cyclohexanedicarboxylic acid General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 485 -488

Reactions involving Carboxylic acids 1. Oxidation of appropriate primary alcohol or aldehyde using oxidizing agents KMn. O 4 / K 2 Cr. O 4 RCH 2 -OH RCHO RCOOH It is a commercial method for preparation of acetic acid found in vinegar Primary alcohols on oxidation give carboxylic acids as end product 1. Write the equation showing preparation of vinegar 2. Write the equation showing preparation of propanoic acid 2. Tollen’s Test: Oxidation with Tollen’s reagent Ag(NH 3)2+ Aldehydes are oxidized to carboxylic acids using Tollen’s reagent. RCHO + Ag(NH 3)2+ RCOOH + Ag o General, Organic, and Biochemistry 9 th Edition By Katherine J. Denniston, Joseph J. Topping and Danae R. Quirk Dorr, Robert L. Caret Page Number : 493 -495
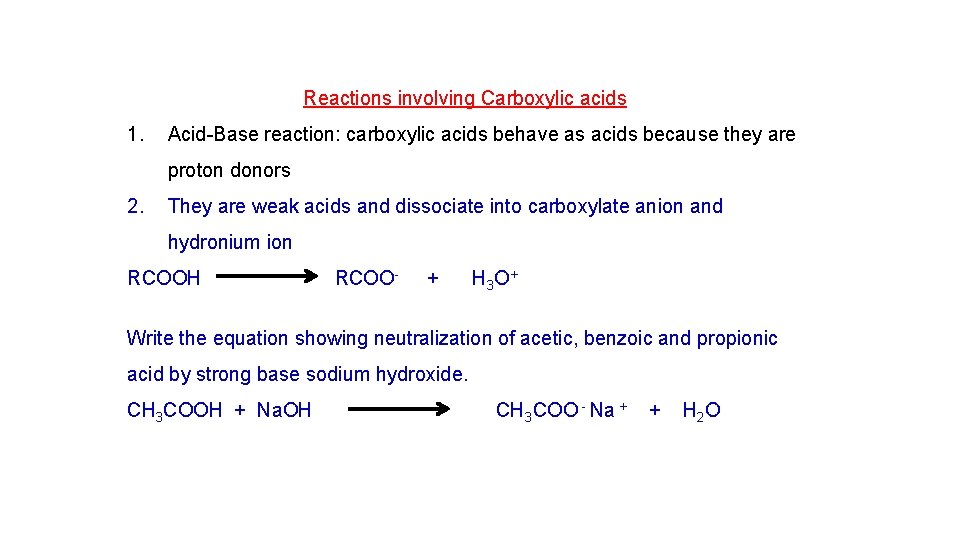
Reactions involving Carboxylic acids 1. Acid-Base reaction: carboxylic acids behave as acids because they are proton donors 2. They are weak acids and dissociate into carboxylate anion and hydronium ion RCOOH RCOO- + H 3 O+ Write the equation showing neutralization of acetic, benzoic and propionic acid by strong base sodium hydroxide. CH 3 COOH + Na. OH CH 3 COO - Na + + H 2 O
- Slides: 15