Chapter 13 Types and Applications of Metals Ceramics

















- Slides: 26

Chapter 13: Types and Applications of Metals, Ceramics, and Polymers ISSUES TO ADDRESS. . . • How are metal alloys classified and what are their common applications ? • How do we classify ceramics? • What are some applications of ceramics? • What are the various types/classifications of polymers? Chapter 13 - 1

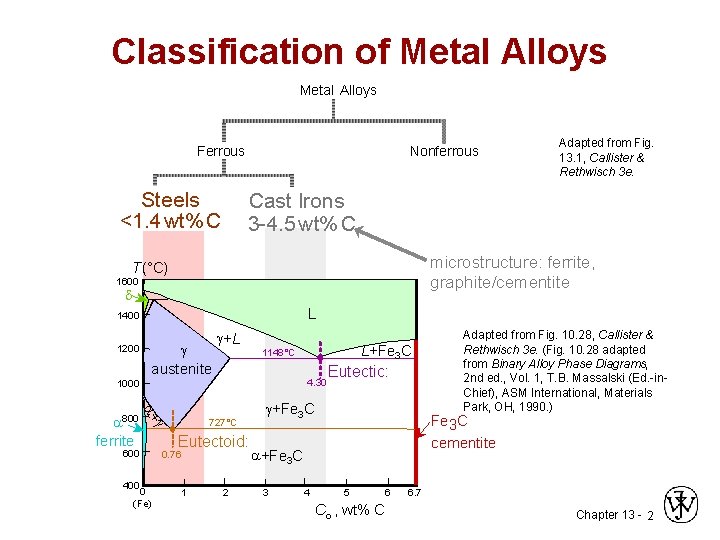
Classification of Metal Alloys Ferrous Steels <1. 4 wt%C <1. 4 wt% C Nonferrous Cast Irons 3 -4. 5 wt%C 3 -4. 5 wt% C microstructure: ferrite, graphite/cementite T(°C) 1600 d L 1400 1200 austenite +L 4. 30 + 600 400 L+Fe 3 C 1148°C 1000 800 ferrite Adapted from Fig. 13. 1, Callister & Rethwisch 3 e. 0 (Fe) 727°C Eutectoid: 0. 76 1 2 Eutectic: +Fe 3 C cementite +Fe 3 C 3 4 Adapted from Fig. 10. 28, Callister & Rethwisch 3 e. (Fig. 10. 28 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in. Chief), ASM International, Materials Park, OH, 1990. ) 5 6 Co , wt% C 6. 7 Chapter 13 - 2

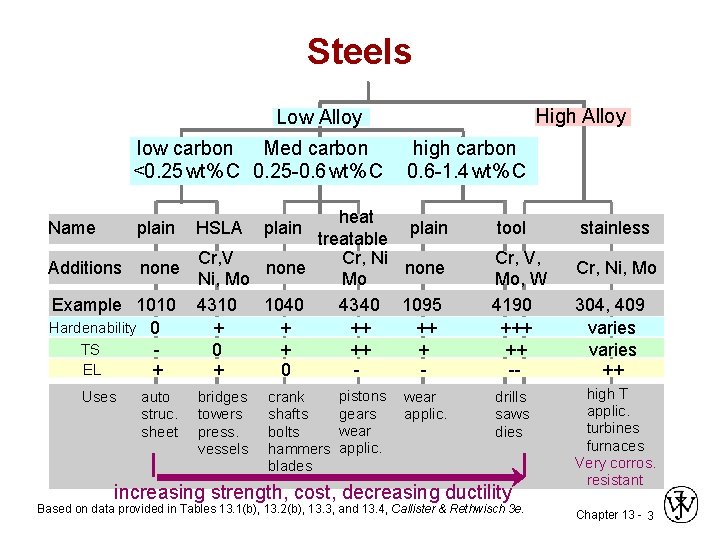
Steels High Alloy Low Alloy low carbon Med carbon <0. 25 wt% C 0. 25 -0. 6 wt% C high carbon 0. 6 -1. 4 wt% C heat plain treatable Cr, V Cr, Ni Additions none Ni, Mo Mo Example 1010 4310 1040 4340 1095 Hardenability 0 + + ++ ++ TS 0 + ++ + EL + + 0 - Name plain Uses auto struc. sheet HSLA bridges towers press. vessels plain crank shafts bolts hammers blades pistons gears wear applic. tool Cr, V, Mo, W 4190 +++ ++ -drills saws dies increasing strength, cost, decreasing ductility Based on data provided in Tables 13. 1(b), 13. 2(b), 13. 3, and 13. 4, Callister & Rethwisch 3 e. stainless Cr, Ni, Mo 304, 409 varies ++ high T applic. turbines furnaces Very corros. resistant Chapter 13 - 3

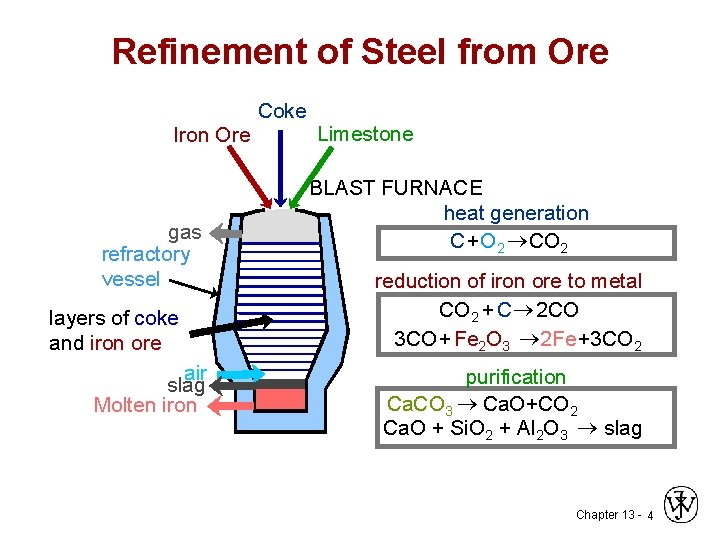
Refinement of Steel from Ore Coke Iron Ore gas refractory vessel layers of coke and iron ore air slag Molten iron Limestone BLAST FURNACE heat generation C+O 2 ®CO 2 reduction of iron ore to metal CO 2 + C ® 2 CO 3 CO + Fe 2 O 3 ® 2 Fe+3 CO 2 purification Ca. CO 3 ® Ca. O+CO 2 Ca. O + Si. O 2 + Al 2 O 3 ® slag Chapter 13 - 4

Ferrous Alloys Iron-based alloys • Steels • Cast Irons Nomenclature for steels (AISI/SAE) 10 xx Plain Carbon Steels 11 xx Plain Carbon Steels (resulfurized for machinability) 15 xx Mn (1. 00 - 1. 65%) 40 xx Mo (0. 20 ~ 0. 30%) 43 xx Ni (1. 65 - 2. 00%), Cr (0. 40 - 0. 90%), Mo (0. 20 - 0. 30%) 44 xx Mo (0. 5%) where xx is wt% C x 100 example: 1060 steel – plain carbon steel with 0. 60 wt% C Stainless Steel -- >11% Cr Chapter 13 - 5

Cast Irons • Ferrous alloys with > 2. 1 wt% C – more commonly 3 - 4. 5 wt% C • Low melting – relatively easy to cast • Generally brittle • Cementite decomposes to ferrite + graphite Fe 3 C 3 Fe ( ) + C (graphite) – generally a slow process Chapter 13 - 6

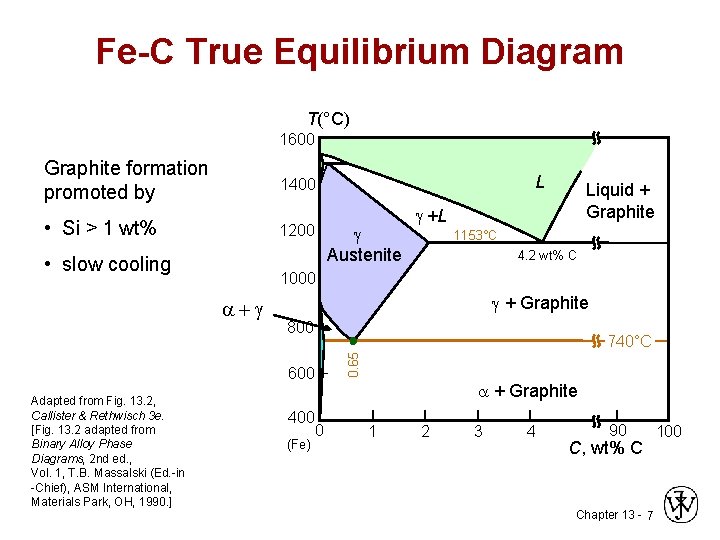
Fe-C True Equilibrium Diagram T(°C) 1600 Graphite formation promoted by 1400 • Si > 1 wt% 1200 • slow cooling Austenite Liquid + Graphite +L 1153°C 4. 2 wt% C 1000 + Graphite 800 740°C 0. 65 + 600 Adapted from Fig. 13. 2, Callister & Rethwisch 3 e. [Fig. 13. 2 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in -Chief), ASM International, Materials Park, OH, 1990. ] L 400 (Fe) + Graphite 0 1 2 3 4 90 C, wt% C Chapter 13 - 7 100

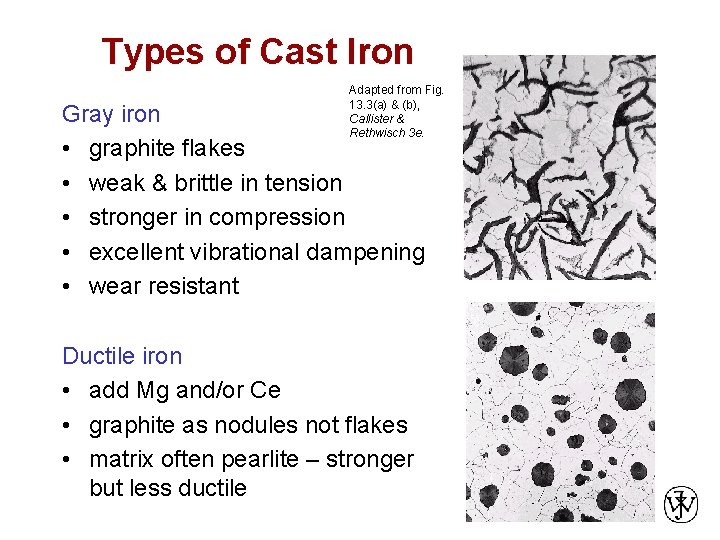
Types of Cast Iron Adapted from Fig. 13. 3(a) & (b), Callister & Rethwisch 3 e. Gray iron • graphite flakes • weak & brittle in tension • stronger in compression • excellent vibrational dampening • wear resistant Ductile iron • add Mg and/or Ce • graphite as nodules not flakes • matrix often pearlite – stronger but less ductile Chapter 13 - 8

Types of Cast Iron (cont. ) White iron • < 1 wt% Si • pearlite + cementite • very hard and brittle Adapted from Fig. 13. 3(c) & (d), Callister & Rethwisch 3 e. Malleable iron • heat treat white iron at 800 -900ºC • graphite in rosettes • reasonably strong and ductile Chapter 13 - 9

Types of Cast Iron (cont. ) Compacted graphite iron • relatively high thermal conductivity • good resistance to thermal shock • lower oxidation at elevated temperatures Adapted from Fig. 13. 3(e), Callister & Rethwisch 3 e. Chapter 13 - 10

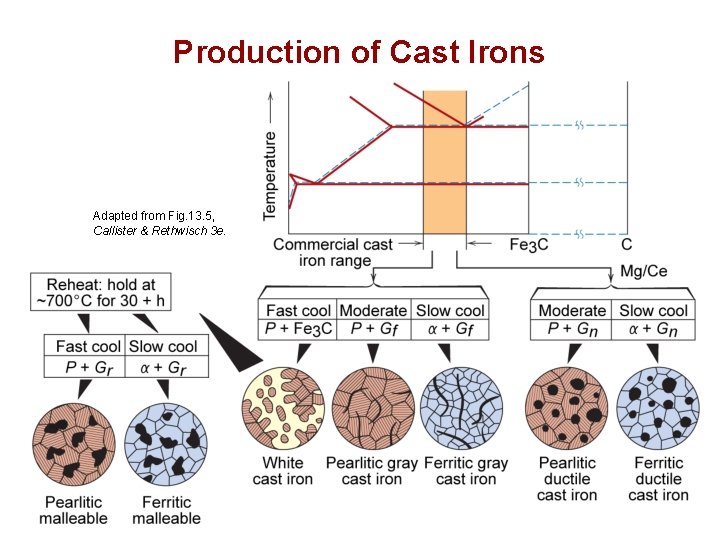
Production of Cast Irons Adapted from Fig. 13. 5, Callister & Rethwisch 3 e. Chapter 13 - 11

Limitations of Ferrous Alloys 1) Relatively high densities 2) Relatively low electrical conductivities 3) Generally poor corrosion resistance Chapter 13 - 12

Nonferrous Alloys • Cu Alloys • Al Alloys -low r: 2. 7 g/cm 3 Brass: Zn is subst. impurity (costume jewelry, coins, -Cu, Mg, Si, Mn, Zn additions corrosion resistant) -solid sol. or precip. Bronze : Sn, Al, Si, Ni are strengthened (struct. subst. impurities aircraft parts (bushings, landing & packaging) gear) • Mg Alloys Non. Ferrous Cu-Be: -very low r: 1. 7 g/cm 3 Alloys precip. hardened -ignites easily for strength -aircraft, missiles • Ti Alloys -relatively low r: 4. 5 g/cm 3 • Refractory metals vs 7. 9 for steel • Noble metals -reactive at high T’s -Ag, Au, Pt -oxid. /corr. resistant -space applic. -high melting T’s -Nb, Mo, W, Ta Based on discussion and data provided in Section 13. 3, Callister & Rethwisch 3 e. Chapter 13 - 13

Classification of Ceramics Ceramic Materials Glasses Clay Refractories products Abrasives Cements Advanced ceramics -optical -whiteware -bricks for -sandpaper -composites -engine -composite -structural high T -cutting -structural rotors (furnaces) -polishing reinforce valves -containers/ bearings Adapted from Fig. 13. 7 and discussion in -sensors household Section 13. 4 -10, Callister & Rethwisch 3 e. Chapter 13 - 14

Ceramics Application: Die Blanks • Die blanks: -- Need wear resistant properties! • Die surface: -- 4 mm polycrystalline diamond particles that are sintered onto a cemented tungsten carbide substrate. -- polycrystalline diamond gives uniform hardness in all directions to reduce wear. die Ao die Ad tensile force Adapted from Fig. 14. 2 (d), Callister & Rethwisch 3 e. Courtesy Martin Deakins, GE Superabrasives, Worthington, OH. Used with permission. Chapter 13 - 15

Ceramics Application: Cutting Tools • Tools: -- for grinding glass, tungsten, carbide, ceramics -- for cutting Si wafers -- for oil drilling • Materials: -- manufactured single crystal or polycrystalline diamonds in a metal or resin matrix. -- polycrystalline diamonds resharpen by microfracturing along cleavage planes. oil drill bits blades Single crystal diamonds polycrystalline diamonds in a resin matrix. Photos courtesy Martin Deakins, GE Superabrasives, Worthington, OH. Used with permission. Chapter 13 - 16

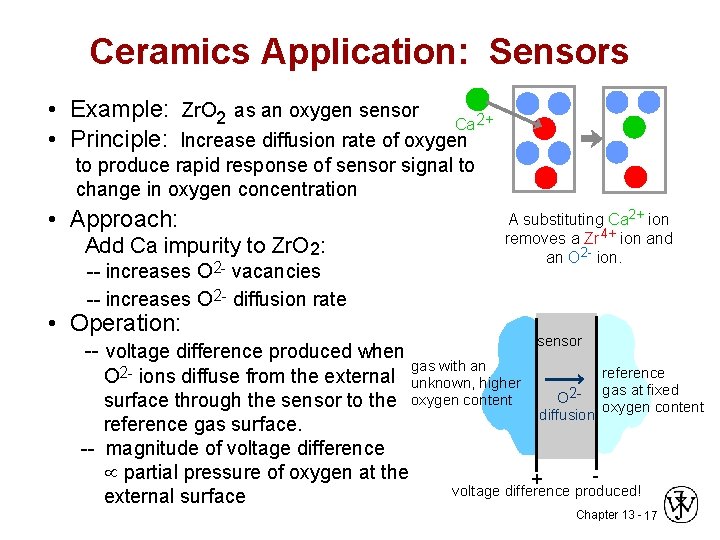
Ceramics Application: Sensors • Example: Zr. O 2 as an oxygen sensor Ca 2+ • Principle: Increase diffusion rate of oxygen to produce rapid response of sensor signal to change in oxygen concentration • Approach: A substituting Ca 2+ ion removes a Zr 4+ ion and an O 2 - ion. Add Ca impurity to Zr. O 2: -- increases O 2 - vacancies -- increases O 2 - diffusion rate • Operation: -- voltage difference produced when sensor gas with an reference O 2 - ions diffuse from the external unknown, higher gas at fixed O 2 surface through the sensor to the oxygen content diffusion reference gas surface. -- magnitude of voltage difference partial pressure of oxygen at the + voltage difference produced! external surface Chapter 13 - 17

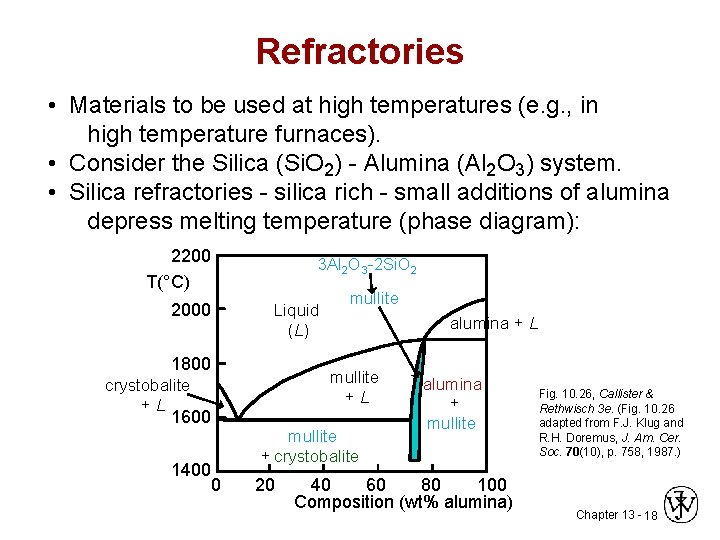
Refractories • Materials to be used at high temperatures (e. g. , in high temperature furnaces). • Consider the Silica (Si. O 2) - Alumina (Al 2 O 3) system. • Silica refractories - silica rich - small additions of alumina depress melting temperature (phase diagram): 2200 T(°C) 2000 3 Al 2 O 3 -2 Si. O 2 Liquid (L) 1800 mullite alumina + L mullite +L crystobalite +L 1600 1400 0 mullite + crystobalite 20 alumina + mullite 40 60 80 100 Composition (wt% alumina) Fig. 10. 26, Callister & Rethwisch 3 e. (Fig. 10. 26 adapted from F. J. Klug and R. H. Doremus, J. Am. Cer. Soc. 70(10), p. 758, 1987. ) Chapter 13 - 18

Advanced Ceramics: Materials for Automobile Engines • Advantages: – Operate at high temperatures – high efficiencies – Low frictional losses – Operate without a cooling system – Lower weights than current engines • Disadvantages: – Ceramic materials are brittle – Difficult to remove internal voids (that weaken structures) – Ceramic parts are difficult to form and machine • Potential candidate materials: Si 3 N 4, Si. C, & Zr. O 2 • Possible engine parts: engine block & piston coatings Chapter 13 - 19

Advanced Ceramics: Materials for Ceramic Armor Components: -- Outer facing plates -- Backing sheet Properties/Materials: -- Facing plates -- hard and brittle — fracture high-velocity projectile — Al 2 O 3, B 4 C, Si. C, Ti. B 2 -- Backing sheets -- soft and ductile — deform and absorb remaining energy — aluminum, synthetic fiber laminates Chapter 13 - 20

Polymer Types – Fibers - length/diameter >100 • Primary use is in textiles. • Fiber characteristics: – high tensile strengths – high degrees of crystallinity – structures containing polar groups • Formed by spinning – extrude polymer through a spinneret (a die containing many small orifices) – the spun fibers are drawn under tension – leads to highly aligned chains - fibrillar structure Chapter 13 - 21

Polymer Types – Miscellaneous • • Coatings – thin polymer films applied to surfaces – i. e. , paints, varnishes – protects from corrosion/degradation – decorative – improves appearance – can provide electrical insulation Adhesives – bonds two solid materials (adherands) – bonding types: 1. Secondary – van der Waals forces 2. Mechanical – penetration into pores/crevices • • Films – produced by blown film extrusion Foams – gas bubbles incorporated into plastic Chapter 13 - 22

Advanced Polymers Ultrahigh Molecular Weight Polyethylene (UHMWPE) • Molecular weight ca. 4 x 106 g/mol • Outstanding properties – – high impact strength resistance to wear/abrasion low coefficient of friction self-lubricating surface UHMWPE • Important applications – bullet-proof vests – golf ball covers – hip implants (acetabular cup) Adapted from chapteropening photograph, Chapter 22, Callister 7 e. Chapter 13 - 23

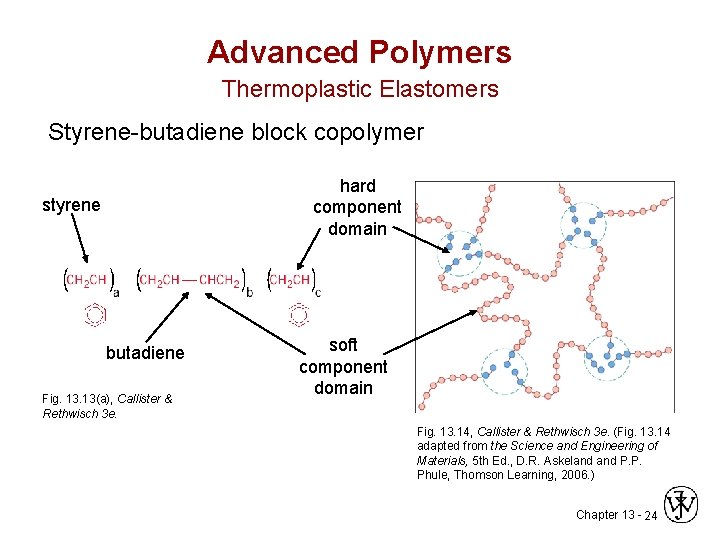
Advanced Polymers Thermoplastic Elastomers Styrene-butadiene block copolymer hard component domain styrene butadiene Fig. 13(a), Callister & Rethwisch 3 e. soft component domain Fig. 13. 14, Callister & Rethwisch 3 e. (Fig. 13. 14 adapted from the Science and Engineering of Materials, 5 th Ed. , D. R. Askeland P. P. Phule, Thomson Learning, 2006. ) Chapter 13 - 24

Summary • Ferrous alloys: steels and cast irons • Non-ferrous alloys: -- Cu, Al, Ti, and Mg alloys; refractory alloys; and noble metals. • Categories of ceramics: -- glasses -- clay products -- refractories -- cements -- advanced ceramics • Polymer applications -- elastomers -- fibers -- coatings -- adhesives -- films -- foams -- advanced polymeric materials Chapter 13 - 25

ANNOUNCEMENTS Reading: Core Problems: Self-help Problems: Chapter 13 - 26