Chapter 13 States of Matter Chemistry Adapted from





































- Slides: 40

Chapter 13 States of Matter Chemistry Adapted from Stephen L. Cotton

The Nature of Gases n Kinetic refers to motion. n The energy an object has because of its motion is called kinetic energy. n The kinetic theory states that the tiny particles in all forms of matter are in constant motion!

Three basic assumptions of the kinetic theory as it applies to gases: #1. Gas is composed of particlesusually molecules or atoms. n Small, hard spheres n Insignificant volume; relatively far apart from each other n No attraction or repulsion between particles

#2. Particles in a gas move rapidly in constant random motion. n Move in straight paths, changing direction only when colliding with one another or other objects n Example of Speed: Average speed of O 2 in air at 20 o. C is an amazing 1700 km/h!


#3. Collisions are perfectly elastic. n meaning kinetic energy is transferred without loss from one particle to another n the total kinetic energy remains constant

n Gas Pressure – defined as the force exerted by a gas per unit surface area of an object. n Due to: a) force of collisions, and b) number of collisions n No particles present? Then there cannot be any collisions, and thus no pressure – called a vacuum

n Atmospheric pressure results from the collisions of air molecules with objects. n Decreases as you climb a mountain because the air layer thins out as elevation increases n Barometer is the measuring device for atmospheric pressure, which is dependent upon weather & altitude


- Page 394 Not Boiling Normal Boiling Point Boiling, but @ @ 101. 3 k. Pa = 100 o. C 34 k. Pa = 70 o. C

Measuring Pressure The first device for measuring atmospheric pressure was developed by Evangelista Torricelli during the 17 th century. The device was called a barometer. Baro = weight Meter = measure Torricelli

Units of Pressure n The SI unit of pressure is the pascal (Pa) n At sea level, atmospheric pressure is about 101. 3 kilopascals (k. Pa) n Older units of pressure include millimeters of mercury (mm Hg), and atmospheres (atm) – both of which came from using a mercury barometer.

Pressure Standards For gases, it is important to relate measured values to standards. n Standard values are defined as a temperature of 0 o. C and a pressure of 101. 3 k. Pa, 760 mm Hg, or 1 atm n This is called Standard Temperature and Pressure, or STP. n Equal pressures: 1 atm = 760 mm Hg = 101. 3 k. Pa

Calculating Pressure A gas is at a pressure of 6. 1 atm. What is the pressure in kilopascals? G= 6. 1 atm F= k. Pa F= 101. 3 k. Pa 1 atm S= 6. 1 atm 101. 3 k. Pa = 617. 93 k. Pa 1 atm n Now convert 312 mm Hg to units of atm. G= 312 mm Hg F= atm F= 1 atm 760 mm Hg n S= 312 mm Hg 1 atm =. 41 atm 760 mm Hg

What happens when a substance is heated? Particles absorb energy! n Some of the energy is stored within the particles- this is potential energy, and does not raise the temperature. n Remaining energy speeds up the particles (increases kinetic energy)which increases temperature.

n The particles in any collection have a wide range of kinetic energies, from very low to very high- but most are somewhere in the middle, thus the term average kinetic energy is used. n The higher the temperature, the wider the range of kinetic energies.

n An increase in the kinetic energy of particles causes the temperature to rise. n As it cools, the particles tend to move more slowly, and the average K. E. declines.

n When particles have no kinetic energy, they would have no motion. n Absolute zero (0 K, or – 273 o. C) is the temperature at which the motion of particles theoretically ceases. n This has never been reached, but about. 000005 K has been achieved.

n The Kelvin temperature scale reflects a direct relationship between temperature and average kinetic energy. n Particles of He gas at 200 K have twice the average kinetic energy as particles of He gas at 100 K.

n Solids and liquids differ in their response to temperature. n However, at any given temperature the particles of all substances, regardless of their physical state, have the same average kinetic energy.

The Nature of Liquids n Liquid particles are also in motion. n Liquid particles are free to slide past one another. n Gases and liquids can both flow, as seen in Fig. 13. 5, p. 390 n However, liquid particles are attracted to each other, whereas gases are not.

n Particles of a liquid spin and vibrate while they move, thus contributing to their kinetic energy. n But, most of the particles do not have enough energy to escape into the gaseous state; they would have to overcome their intermolecular attractions with other particles.

n The intermolecular attractions also reduce the amount of space between particles of a liquid. n Thus, liquids are more dense than gases. n Increasing pressure on liquid has hardly any effect on it’s volume.

n Increasing the pressure also has little effect on the volume of a solid. n For that reason, liquids and solids are known as the condensed states of matter. n Water in an open vessel or puddle eventually goes into the air.

n The conversion of a liquid to a gas or vapor is called vaporization. n When this occurs at the surface of a liquid that is not boiling, the process is called evaporation. n Some of the particles break away and enter the gas or vapor state; but only those with the minimum kinetic energy.

n. A liquid will also evaporate faster when heated. n Because the added heat increases the average kinetic energy needed to overcome the attractive forces. n But, evaporation is a cooling process. n Cooling occurs because those with the highest energy escape first.

n Particles left behind have lower kinetic energies; thus the temperature decreases n Similar to removing the fastest runner from a race- the remaining runners have a lower average speed n Evaporation helps to keep our skin cooler on a hot day, unless it is very humid on that day.

The Nature of Solids n Particles in a liquid are relatively free to move. n. Solid particles are not. n Figure 13. 10, page 396 shows solid particles tend to vibrate about fixed points, rather than sliding from place to place.

n Most solids have particles packed against one another in a highly organized pattern. n Tend to be dense and incompressible. n Do not flow, nor take the shape of their container. n Are still able to move, unless they reach absolute zero.

n When a solid is heated, the particles vibrate more rapidly as the kinetic energy increases. n The organization of particles within the solid breaks down, and eventually the solid melts. n The melting point (mp) is the temperature when a solid turns to liquid.

n Generally, most ionic solids have high melting points, due to the relatively strong forces holding them together. n Sodium chloride (an ionic compound) has a melting point of 801 o. C. n Molecular compounds have relatively low melting points.

n Hydrogen chloride (a molecular compound) has a melting point of -112 o. C. n Not all solids melt- wood and cane sugar decompose when heated. n Most solid substances are crystalline in structure.

n Amorphous solids lack an ordered internal structure. n Rubber, plastic, and asphalt are all amorphous solids- their atoms are randomly arranged. n Another example is glasssubstances cooled to a rigid state without crystallizing.

n Glasses can be called super-cooled liquids. n The irregular internal structures of glasses are intermediate between those of a crystalline solid and a freeflowing liquid. n Do not melt at a definite mp, but gradually soften when heated.

Changes of State n Sublimation- the change of a substance from a solid directly to a vapor, without passing through the liquid state. n. Examples: (Fig. 13. 14, p. 401); iodine; dry ice; mothballs; solid air fresheners

n Sublimation is useful in situations such as freeze-drying foods- such as by freezing the freshly brewed coffee, and then removing the water vapor by a vacuum pump. n Also useful in separating substances organic chemists use it to separate mixtures and purify materials.

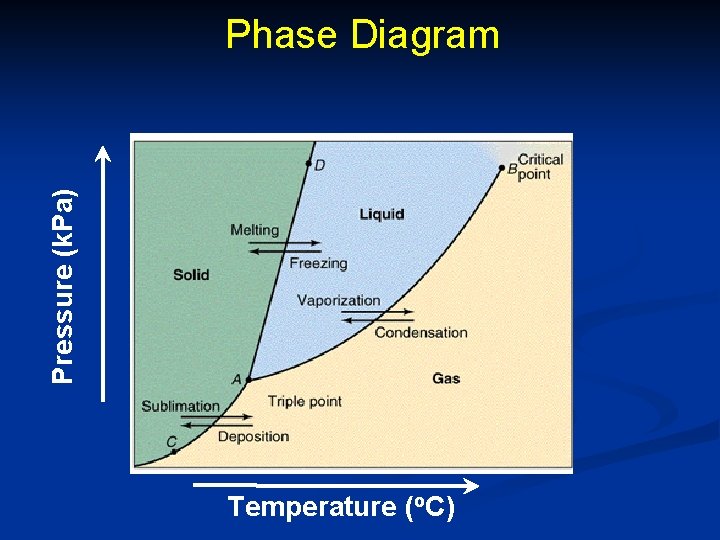
n The relationship among the solid, liquid, and vapor states (or phases) of a substance in a sealed container are best represented in a single graph called a phase diagram. n Phase diagram- gives the temperature and pressure at which a substance exists as solid, liquid, or gas (vapor).

Pressure (k. Pa) Phase Diagram Temperature (o. C)

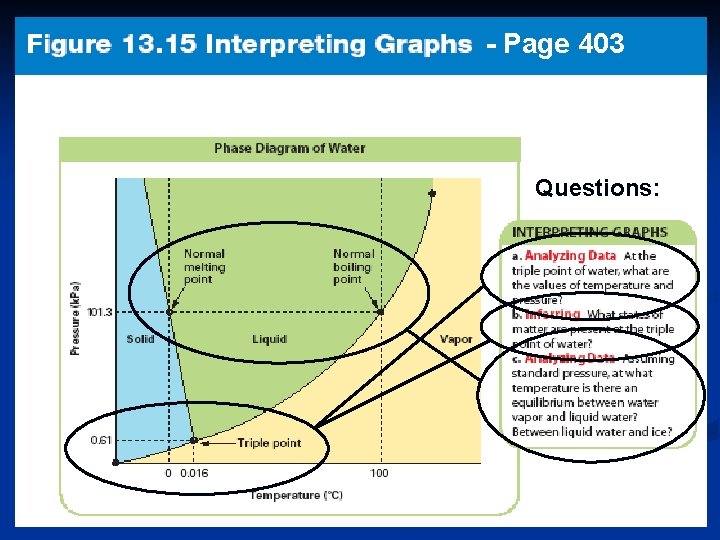
- Page 403 Questions:

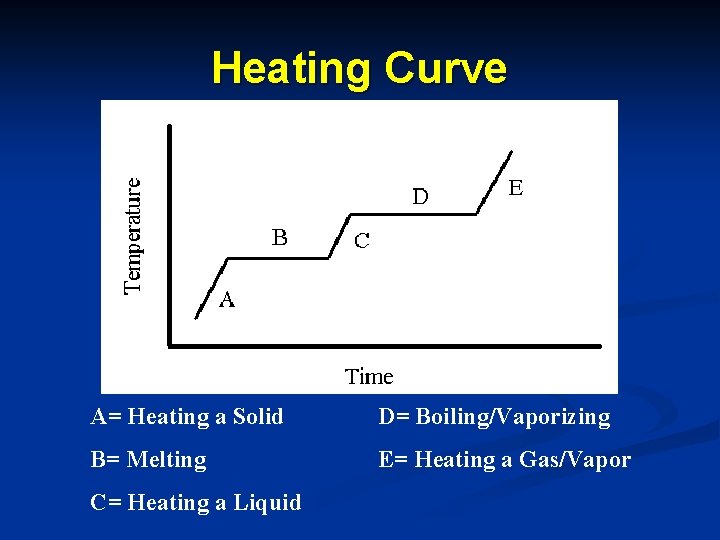
Heating Curve A= Heating a Solid D= Boiling/Vaporizing B= Melting E= Heating a Gas/Vapor C= Heating a Liquid