Chapter 13 POLYFUNCTIONAL ACIDS DESCRIBING POLYFUNCTIONAL BASES FINDING

Chapter 13 POLYFUNCTIONAL ACIDS DESCRIBING POLYFUNCTIONAL BASES FINDING THE p. H OF SOLUTIONS OF AMPHIPROTIC SALTS CONSTRUCTING TITRATION CURVES FOR POLYFUNCTIONAL ACIDS DRAWING TITRATION CURVES FOR POLYFUNCTIONAL BASES THE COMPOSITION OF POLYPROTIC ACID SOLUTIONS AS A FUNCTION OF p. H

POLYFUNCTIONAL ACIDS This chapter, we describe methods for computing the necessary data to litration curves for acids and bases that have two or more acidic or basic functional groups. Solutions of amphiprotic salts and the composition polyprotic acid solutions as a function of p. H are also discussed DESCRIBING POLYFUNCTIONAL BASES Polyfunctional bases such as sodium carbonate, are also common. Carbonate conjugate- base of the hydrogen carbonate ion, is involved in the stepwise equilibria

When two adjacent stepwise equilibria are added, the equilibrium constant for the resulting overall reaction is the product of the two constants. Thus, . for the first two dissociation equilibria for H 3 PO 4.

Where Kal and Ka 2 are the first and second dissociation constants for carbonic acid and Kb 1 and Kb 2 are the first and second dissociation constants of base CO 32 . In some cases, we must also consider a third equilibrium namely. H 2 CO 3 CO 2(aq) + H 2 O. Because of the limited solubility of CO 2, this last reaction can limit the concentration of H 2 CO 3 in solution. For our pup however, we will neglect this reaction. The overall basic dissociation reaction of sodium carbonate is described the equations The p. H of polyfunctional systems, such as phosphoric acid or sodium carbonate, can be computed rigorously through use of the systematic approach! multiequilibrium problems. Solving the several simulitaneous equations that are involved can be difficult and time consuming, pan lady if a computer is not handy.

FINDING THE p. H OF SOLUTIONS OF AMPHIPROTIC SALTS So far it has not discussed how to calculate the p. H of solutions of salts that have both acidic and basic properties, that is, salts that are amphiprotic. Such salts are formed during neutralization titration of polyfunctional acids and bases. When 1 mol of Na. OH is added to a solution containing if 1 mol of the acid H 2 A, 1 mol of Na. HA is formed. The p. H of this solution is determined by two equilibria established between HA and water. The solution could be a basic becasue Whether a solution of Na. HA is acidic or basic depends on the relative magnitude constants for these processes

where Ka 2 and Kb 2 are the acid dissociation constants for H 2 A and Kb 2 is the basic otherwise acidic ion constant for HA. If Kb 2 is greater than Ka 2, the solution is basic or otherwise acidic. A solution of the amphiprotic salt Na. HA contains five species H 3 O+, OH , H 2 A, HA and A 2". Thus, five independent equations are needed to compute hydronium ion concentration rigorously. As we can see in picture above, the solution equations yields an approximate value of [H 3 O+] that is given by the equation CNa. HA is the molar concentration of the salt Na. HA. This equation should be used when Na. HA is the only species that contributes significantly to the p. H of a solution. This is true of every dilute solutions of Na. HA or for systems in Ka 2 or Kw / Ka 2 is relatively large.

We first write a mass balance expression. That is, The charge balance equation takes the form Since the sodium ion concentration is equal to the molar analytical concentration of the salt, the last equation can be rewritten as We now have four algebraic equations and need one additional to solve for the four knowns. The ion product constant for water serves this purpose: The rigorous computation of the hydronium ion concentration from these four equations is difficult. A reasonable approximation, applicable to solutions of most salts, however, can be obtained as follows. Steps 1. We first subtract the mass balance equation from the charge balance equation. 2. We then rearrange the acid dissociation constant expressions for H 2 A to obtain and for HA to give. 3. Substituting these expressions and the expression for Kw into equation.


These examples are self explanatory, they are straight out of the text, they work in unison with the aforementioned equations an on finding the p. H of solution.

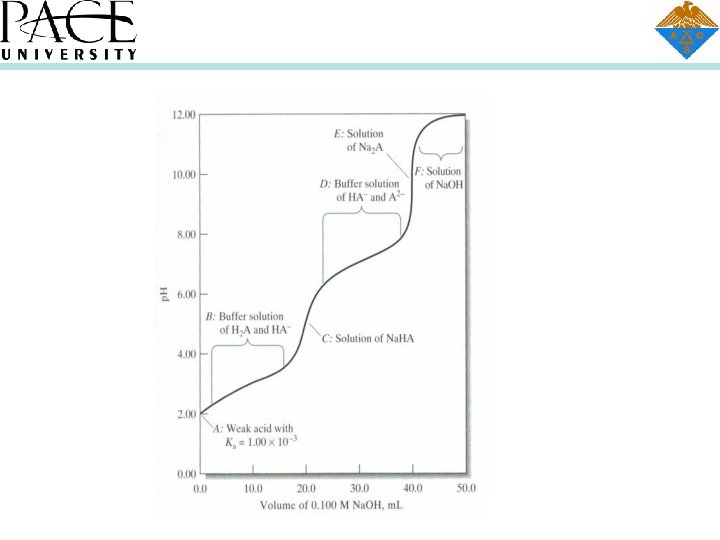
CONSTRUCTING TITRATION CURVES FOR POLYFUNCTIONAL ACIDS Figure in slide 3 shows the titration curve for a diprotic acid H 2 A with dissociation constants of Ka 1 = 1. 00 X 103 and Ka 2 = 1. 00 X 10 7. Because the ratio K al/Ka 2 is significantly greater than 10 3, we can construct this curve using the techniques developed in previous chapter for simple monoprotic weak acids. Thus, to maintain the initial p. H (point A), we treat the system as if it contained a single monoprotic acid with a dissociation constant of Kal = 1. 00 X 10 3. In region B have the equivalent of a simple buffer solution consisting of the weak acid Hi and its conjugate base Na. HA. First equivalence point, we have a solution of an acid salt and use equations or one of its simplifications to compute the hydronium ion concentration, we have a second buffer consisting of a weak acid HA~ anil gate base Na 2 A, and we calculate the p. H employing the second dissociations constant, Ka 2 = 1. 00 X 10 7. Just prior to the second equivalence point

Sulfuric acid is unusual in that one of its protons behaves as a strong acid in water and the other as a weak acid (Ka 2 = 1. 02 X 10 2). Let us consider how the hydronium ion concentration of sulfuric acid solutions is computed using a 0. 0400 M solution as an example. We will first assume that the dissociation of HSO 4 is negligible because of the large excess of H 30+ resulting from the complete dissociation of H 2 SO 4. Therefore, [H 3 O+] ≈ [HSO 4 ] ≈ 0. 0400 M An estimate of [SO 4 ] based on this approximation and the expression for Ka 2, how ever, reveals that 0. 0400[S 04 -] = 1. 02 X 10 -2 0. 0400 This result shows that [SO 4 ] is not small relative to [HSO 4 ], and a more rigorous so lution is required. From stoichiometric considerations, it is necessary that [H 3 O+] = 0. 0400 + [SO 42 -] To be continued

The first term on the right is the concentration of H 3 O+ from dissociation of the H 2 SO 4 to HSO 4. The second term is the contribution of the dissociation of HS 04. Rearrangement yields [SO 4] = [H 30+] - 0. 0400 Mass balance considerations require that CH 2 SO 4, = 0. 0400 = [HS 04 - ] + [SO 42 -] Combining the last two equations and rearranging yields [HSO 4] = 0. 0800 - [H 3 O+] Introduction of these equations for [SO 4 ] and HSO 4 into the expression for yields
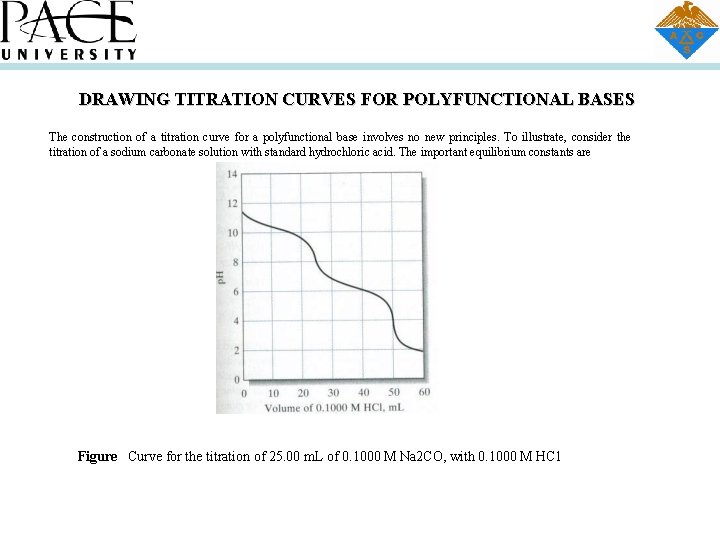
DRAWING TITRATION CURVES FOR POLYFUNCTIONAL BASES The construction of a titration curve for a polyfunctional base involves no new principles. To illustrate, consider the titration of a sodium carbonate solution with standard hydrochloric acid. The important equilibrium constants are Figure Curve for the titration of 25. 00 m. L of 0. 1000 M Na 2 CO, with 0. 1000 M HC 1

The molecular structure of glycine. Glycine is one of the so called nonessential ammo acids; it is nonessential in the sense that it is synthesized in the bodies of mammals and so is not generally essential in the diet. Because of its compact structure, glycine acts as a versatile building block in protein synthesis and in the biosynthesis of hemo globin. A significant fraction of the collagen, or the fibrous protein constituent of bone, cartilage, tendon, and other connective tis sue, in the human body is made up ofglycine. Glycine is also an inhibitory neurotransmitter and, as a result has been sug gested as a possible therapeutic agent in dis eases of the central nervous system such as multiple sclerosis and epilepsy. The calming effects of glycine are currently being investi gated to assess its utility in the treatment of schizophrenia.


• The zwitterion of an amino acid, containing as it does a positive and a negative charge, has no tendency to migrate in an electric field, whereas the singly charged anionic and cationic species are attracted to electrodes of opposite charge. • No net migration of the amino acid occurs in an electric field when the p. H of the solvent is such that the concentration of the anionic and cationic forms are identical. • The p. H at which no net migration occurs is called the isoelectric point; this point is an important physical constant for characterizing amino acids. The isoelectric point is readily related to the ionization constants for the species. Thus, for glycine,

A titration curve for the zwitterion of alanine was presented in another chapter illustrate the determination of p. K values. For simple amino acids, Ka and Kb are generally so small that their quantitative determination by neutralization titrations is impos sible. Amino acids that contain more than one carboxyl or amine group can sometimes be determined. If the Ka values are different enough (104 or more), stepwise end points can be obtained just like other polyfunctional acids or bases as long as the Ka values

THE COMPOSITION OF POLYPROTIC ACID SOLUTIONS AS A FUNCTION OF p. H I has now been shown how alpha values are useful in visualizing the changes in the concentration of various species that occur in a titration of a sim pleweak acid. Alpha values can also be calculated for polyfunctional acids and bases. For example, if we let ct be the sum of the molar concentrations of the maleate containing species in the solution throughout the titration described in Example 13 4, the alpha value for the free acid au is defined as The alpha values for HM and M 2 - are given by similar equations: The sum of the alpha values for a system must equal unity:

The alpha values for the maleic acid system are readily expressed in terms of [H 3 O+], Ka 1, and Kb 1. We follow the method used to obtain equations
- Slides: 20