Chapter 13 NMR CARBON13 13 C NMR SPECTROSCOPY
Chapter 13: NMR CARBON-13 (13 C) NMR SPECTROSCOPY Dr. Sivappa Rasapalli Chemistry and Biochemistry University of Massachusetts Dartmouth 1

Carbon-13 NMR Spectroscopy H 1 and C 13 NMR Spectroscopy both give us information about the number of chemically nonequivalent nuclei (nonequivalent hydrogens or nonequivalent carbons) both give us information about the environment of the nuclei (hybridization state, attached atoms, etc. )

SPIN PROPERTIES OF ATOMIC NUCLEI What is spin? The Simple explanation • Spin is a fundamental property of nature like electrical charge or mass. • Spin is a measure of angular momentum (rotation about an axis) hence the term • Spin comes in multiples of 1/2 (0, 1/2, 1, 3/2, 2, 5/2…) and can be + or -. • Protons, electrons, and neutrons possess spin. • Individual unpaired electrons, protons, and neutrons each possesses a spin of 1/2 • Atomic nuclei composed of neutrons and protons may also possess spin. • The spin of an atomic nucleus is determined by the number of protons and neutrons in the nucleus. • Atoms with an odd number of protons will have spin • Atoms with an odd number of neutrons will have spin • Atoms with an odd number of both protons and neutrons will have spin • Atoms with an even number of both protons and neutrons will not have spin • The value of nuclear spin is represented by the symbol I, the nuclear spin quantum number. (I = 0, 1/2, 1, 3/2, 2, 5/2…. ) • A nucleus with spin of I can exist in (2 I+1) spin states. • The shell model for the nucleus tells us that nucleons (protons and neutrons), just like electrons, fill orbitals. When the number of protons or neutrons equals 2, 8, 20, 28, 50, 82, and 126, orbitals are filled. Because nucleons have spin, just like electrons do, their spin can pair up when the orbitals are being filled and cancel out. Odd numbers mean unfilled orbitals, that do not cancel out.
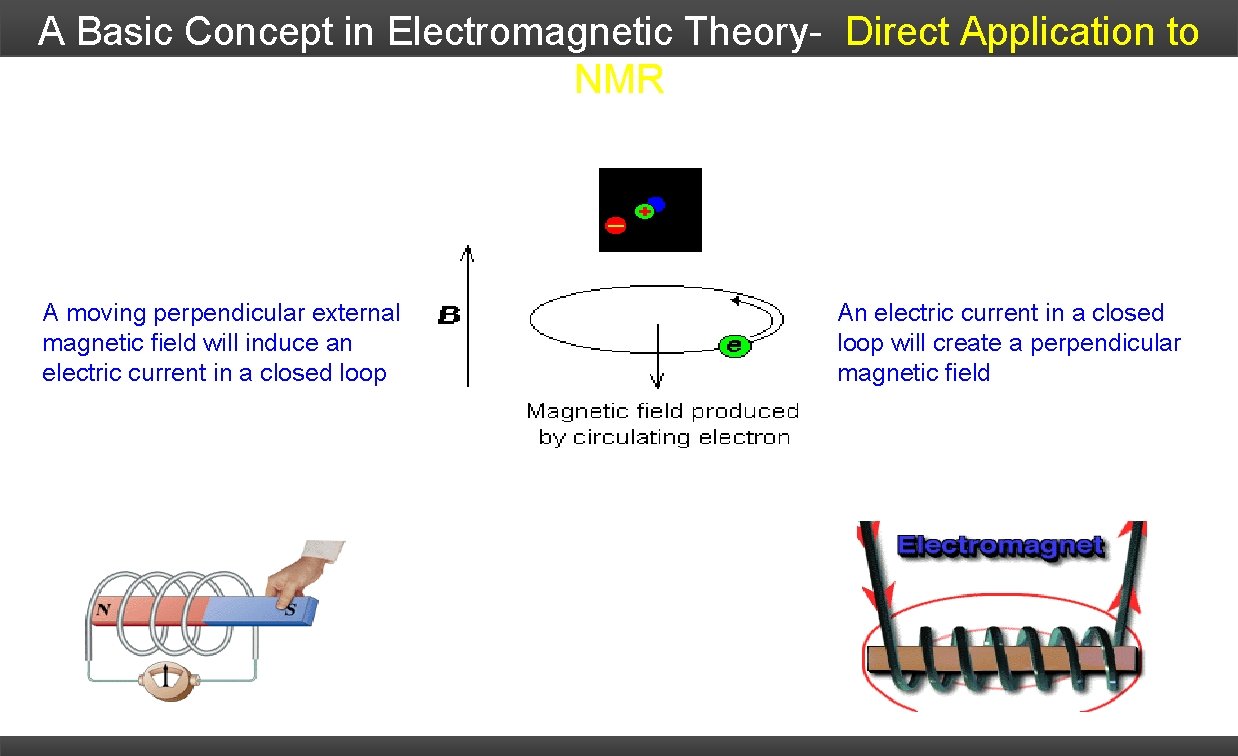
A Basic Concept in Electromagnetic Theory- Direct Application to NMR A moving perpendicular external magnetic field will induce an electric current in a closed loop An electric current in a closed loop will create a perpendicular magnetic field

Carbon-13 NMR Spectroscopy Where’s Waldo? 5
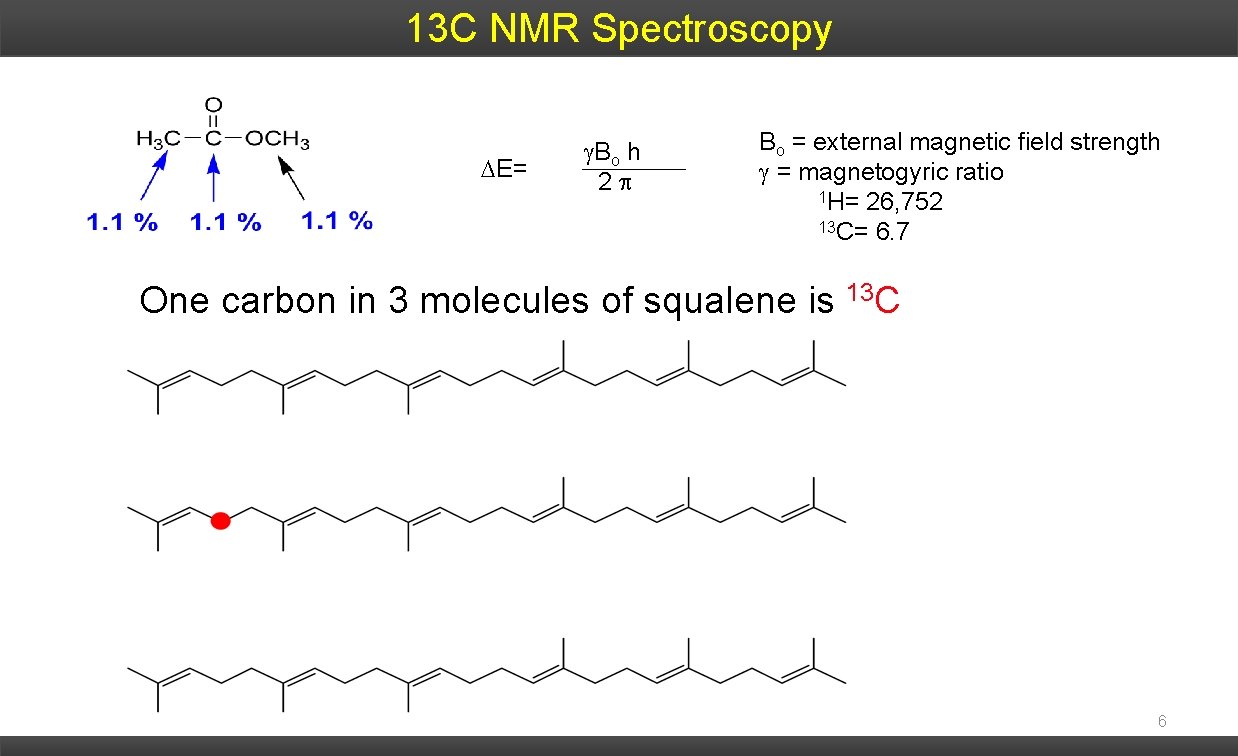
13 C NMR Spectroscopy E= Bo h 2 Bo = external magnetic field strength = magnetogyric ratio 1 H= 26, 752 13 C= 6. 7 One carbon in 3 molecules of squalene is 13 C 6
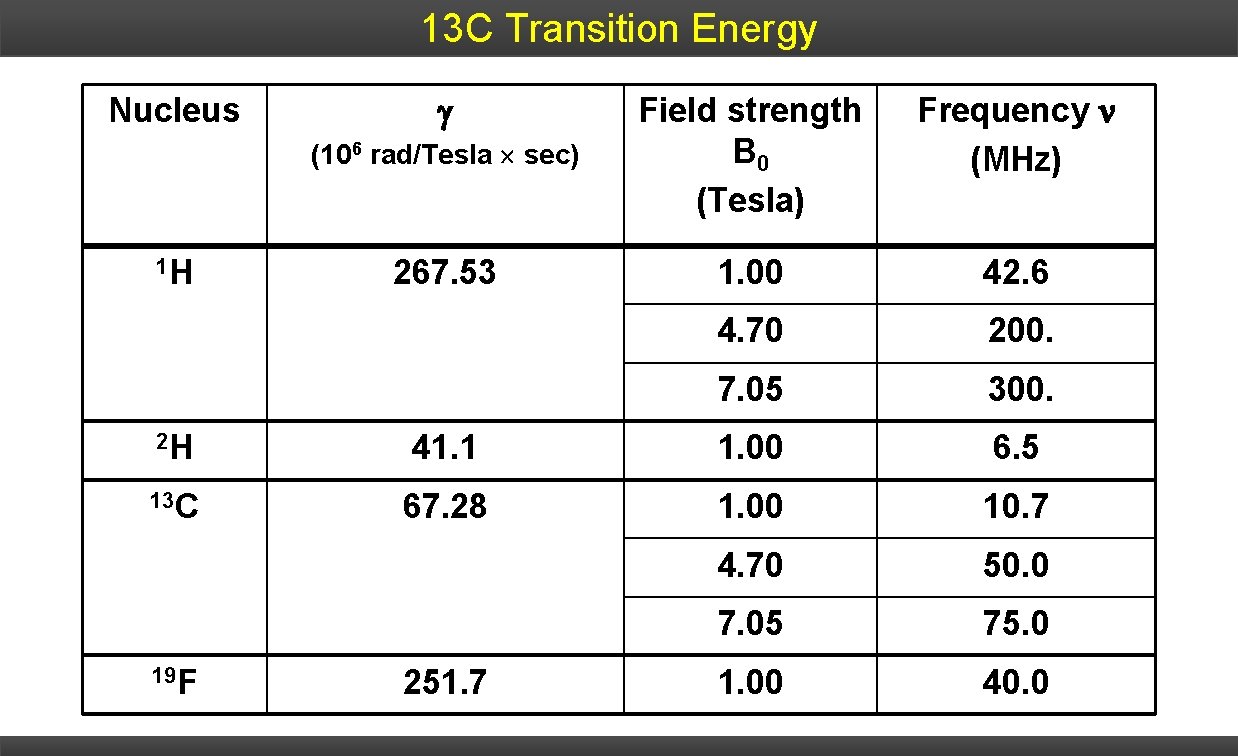
13 C Transition Energy Nucleus 1 H (106 rad/Tesla sec) Field strength B 0 (Tesla) Frequency (MHz) 267. 53 1. 00 42. 6 4. 70 200. 7. 05 300. 2 H 41. 1 1. 00 6. 5 13 C 67. 28 1. 00 10. 7 4. 70 50. 0 7. 05 75. 0 1. 00 40. 0 19 F 251. 7
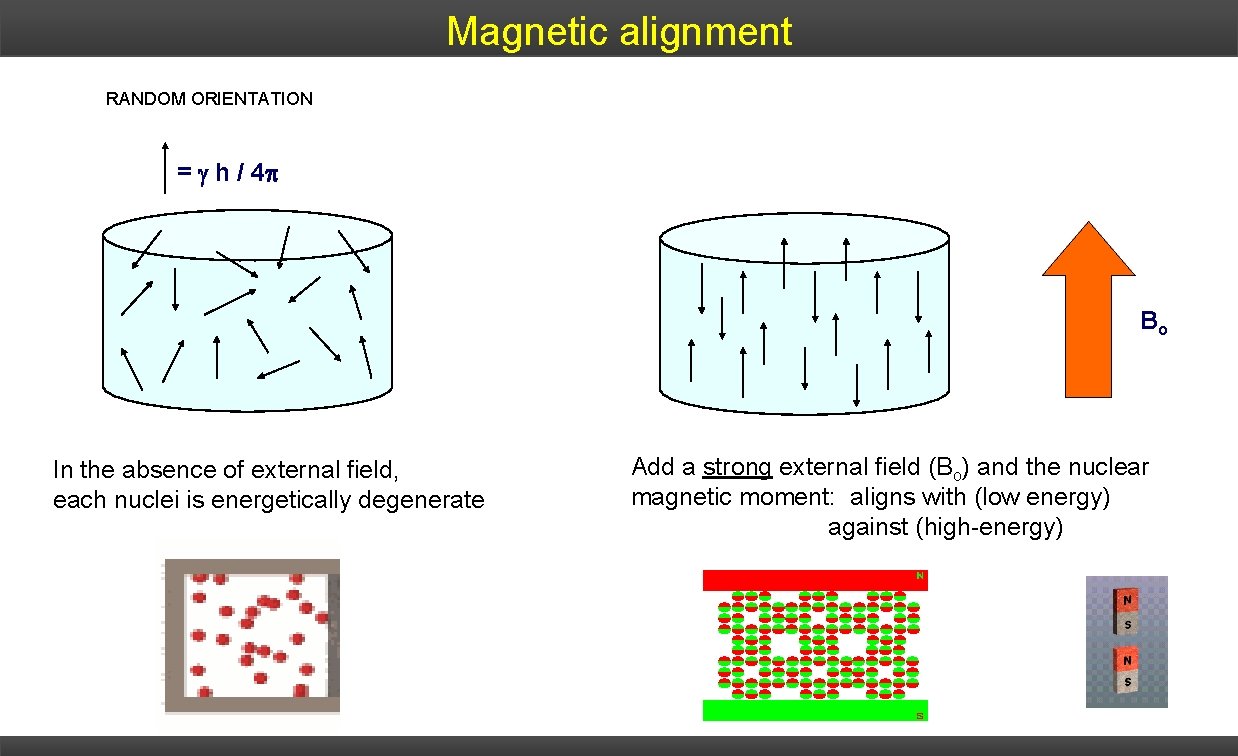
Magnetic alignment RANDOM ORIENTATION = h / 4 p Bo In the absence of external field, each nuclei is energetically degenerate Add a strong external field (Bo) and the nuclear magnetic moment: aligns with (low energy) against (high-energy)
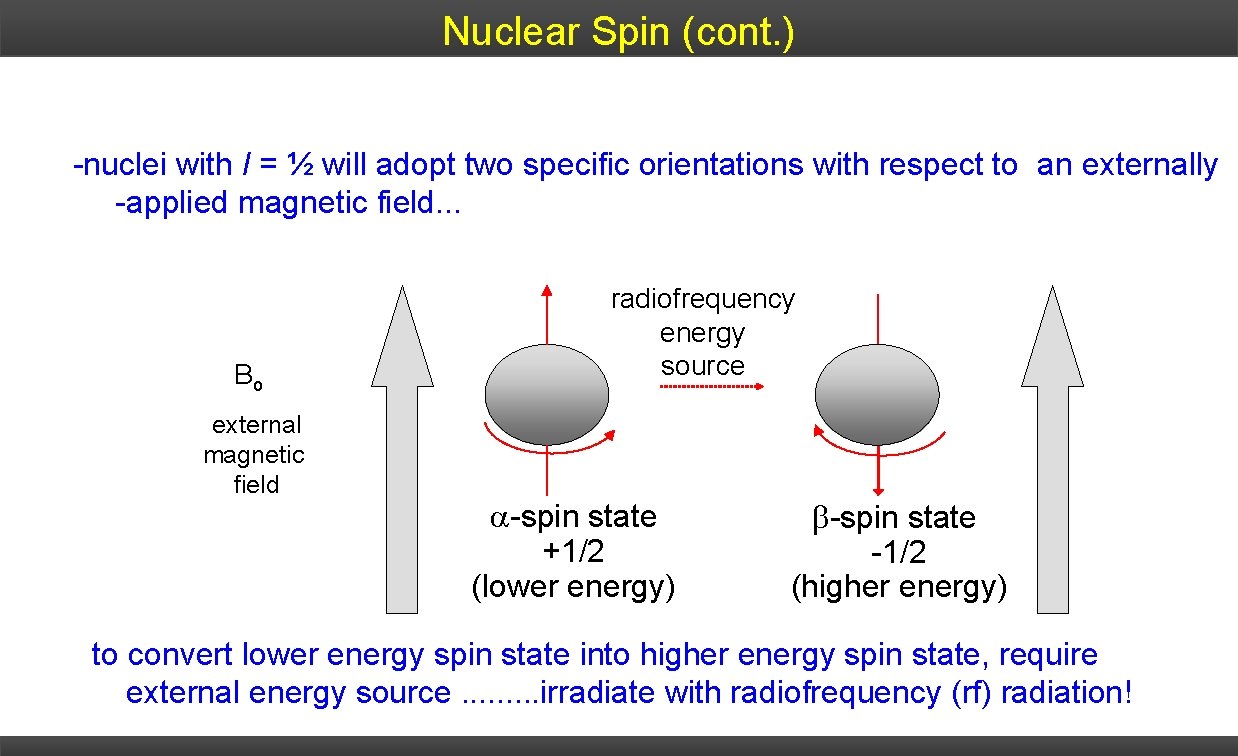
Nuclear Spin (cont. ) 1. At zero external magnetic field, spins are degenerate! 2. Apply an external magnetic field – spins states will differ in energy -nuclei with I = ½ will adopt two specific orientations with respect to an externally depending upon relative orientation with respect to external field. -applied magnetic field. . . Bo external magnetic field radiofrequency energy source -spin state +1/2 (lower energy) -spin state -1/2 (higher energy) to convert lower energy spin state into higher energy spin state, require external energy source. . irradiate with radiofrequency (rf) radiation!
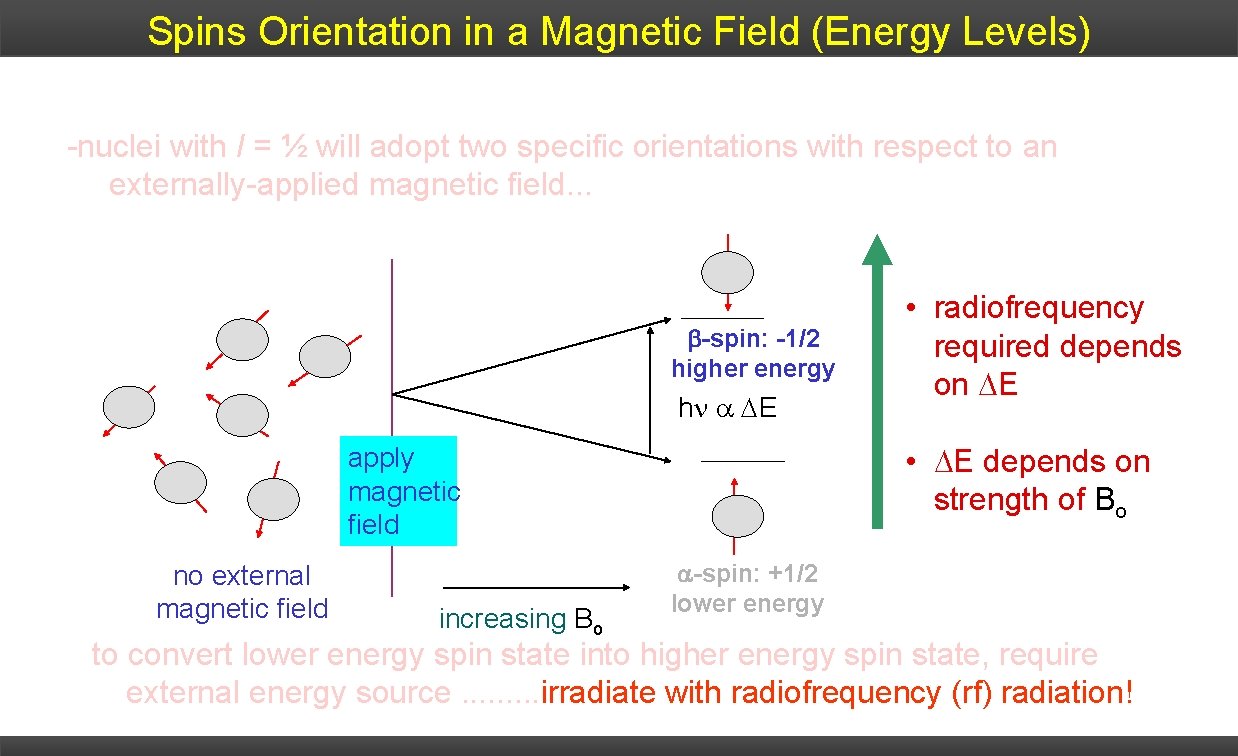
Spins Orientation in a Magnetic Field (Energy Levels) 1. At zero external magnetic field, spins are degenerate! 2. Apply an. I external magnetic field – spins states will differ in energy -nuclei with = ½ will adopt two specific orientations with respect to an depending upon relative orientation externally-applied magnetic field. . . with respect to external field. -spin: -1/2 higher energy h E • E depends on strength of Bo apply magnetic field no external magnetic field increasing Bo • radiofrequency required depends on E -spin: +1/2 lower energy to convert lower energy spin state into higher energy spin state, require external energy source. . irradiate with radiofrequency (rf) radiation!

Spins Orientation in a Magnetic Field (Energy Levels) • Difference in energy between the two states is given by: DE = h Bo / 2 p (DE = h ) where: Bo = external magnetic field h = Planck’s constant; – gyromagnetic ratio -spin: -1/2 higher energy h E • E depends on strength of Bo apply magnetic field no external magnetic field Bo =0 increasing H 0 • radiofrequency required depends on E -spin: +1/2 lower energy Bo >0
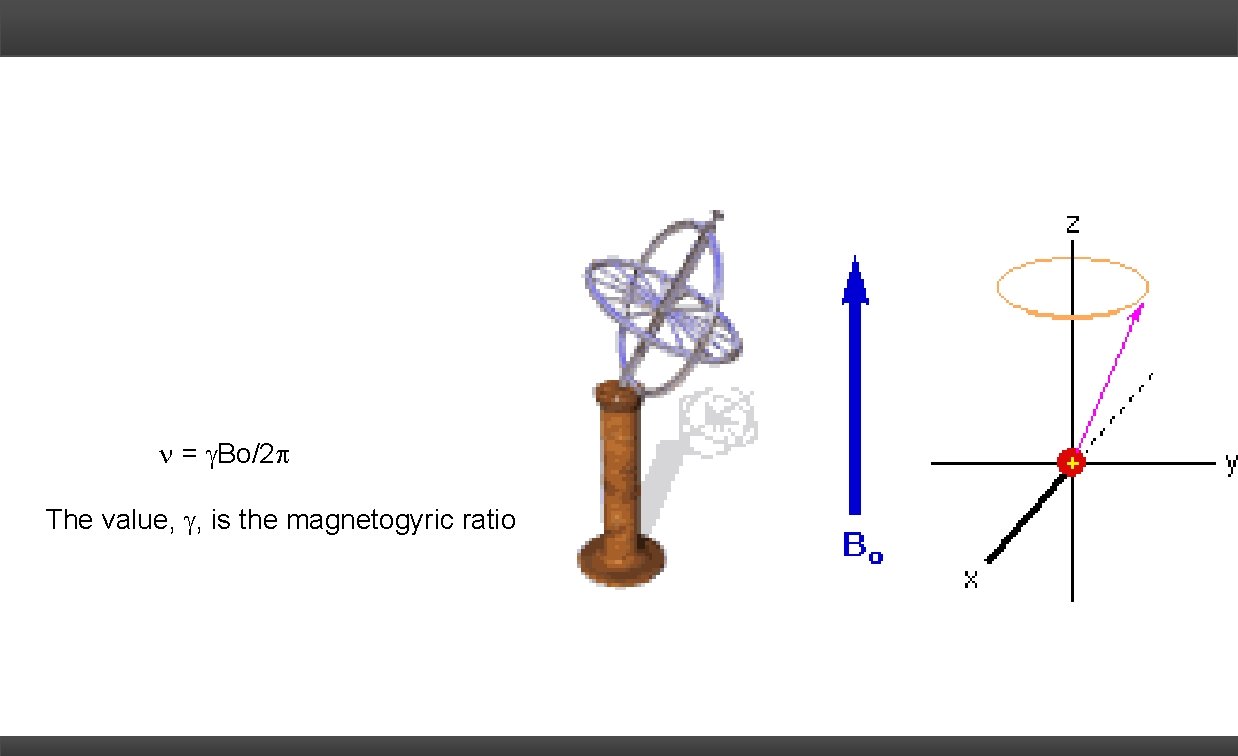
= Bo/2 The value, , is the magnetogyric ratio
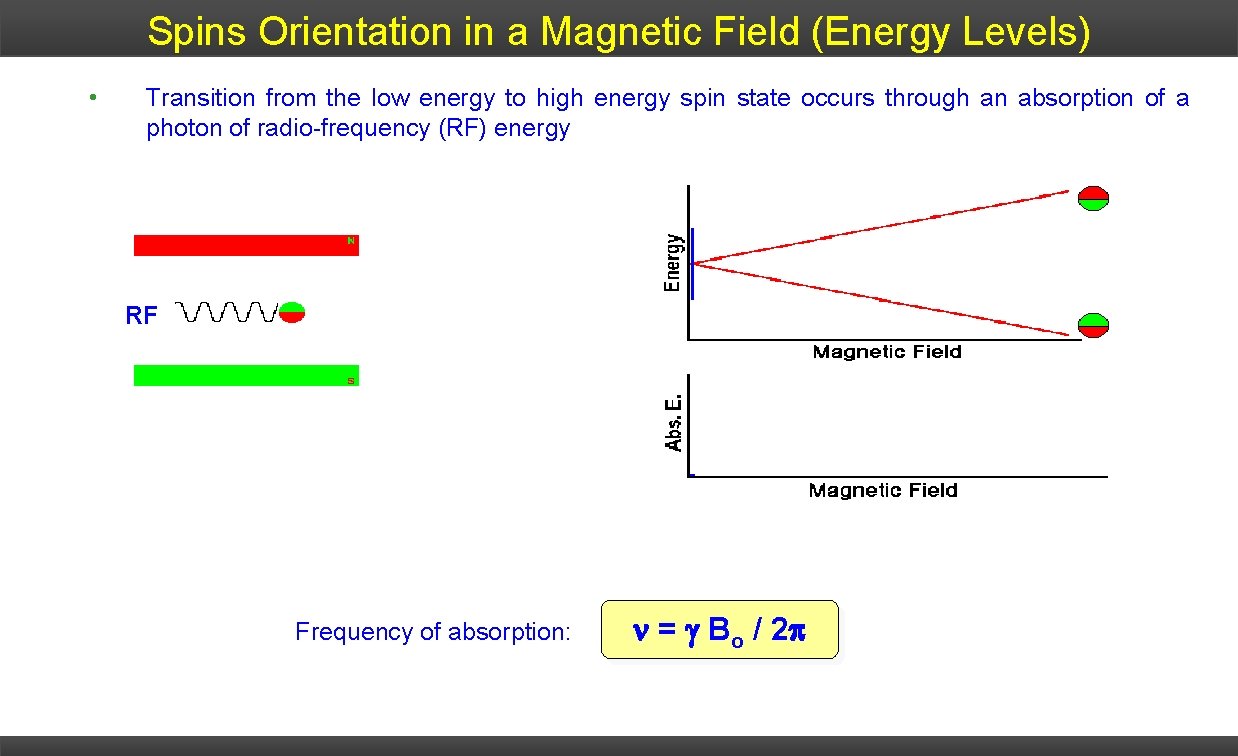
Spins Orientation in a Magnetic Field (Energy Levels) • Transition from the low energy to high energy spin state occurs through an absorption of a photon of radio-frequency (RF) energy RF Frequency of absorption: = Bo / 2 p
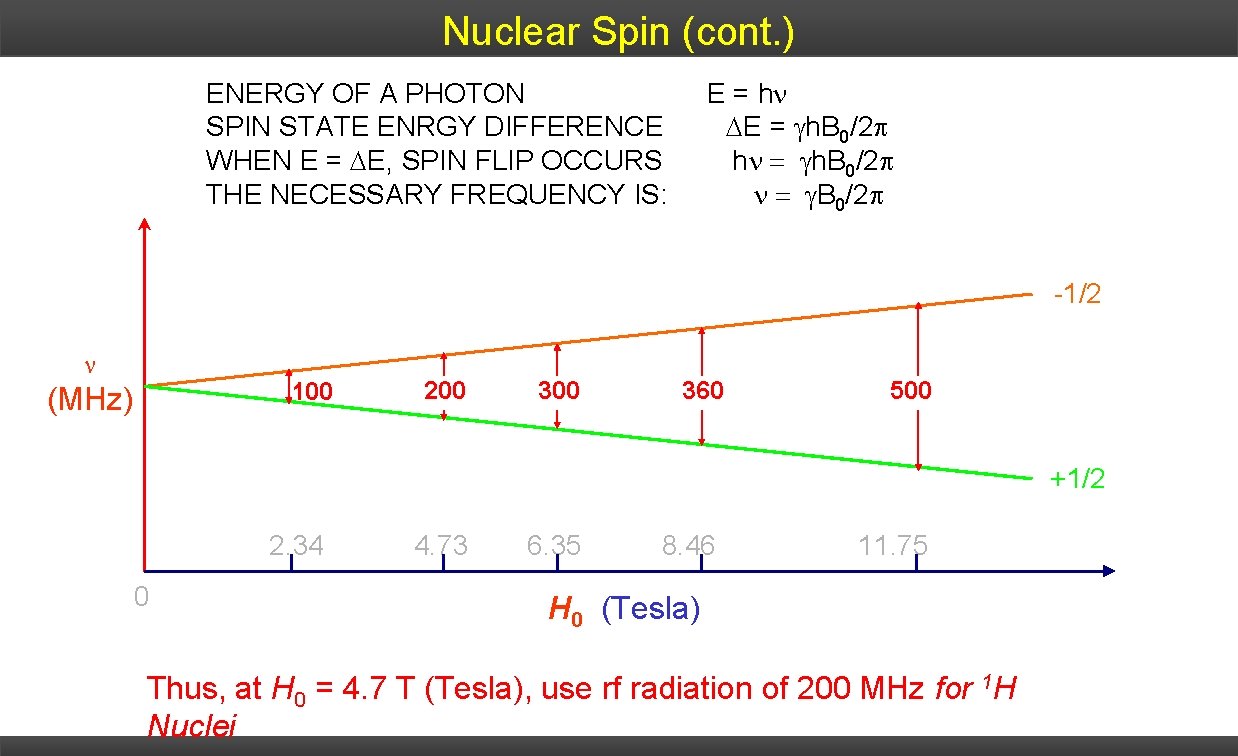
Nuclear Spin (cont. ) ENERGY OF A PHOTON E = h • Difference in energy between the two nuclear spin states: depends SPIN STATE ENRGY DIFFERENCE E = h. B 0/2 on strength external magnetic field WHEN Eof = E, SPIN FLIP OCCURS h h. B 0/2 • For THE nucleus of H atom (proton), spin differences: NECESSARY FREQUENCY IS: energy B 0/2 -1/2 100 (MHz) 200 360 500 +1/2 2. 34 0 4. 73 6. 35 8. 46 11. 75 H 0 (Tesla) Thus, at H 0 = 4. 7 T (Tesla), use rf radiation of 200 MHz for 1 H Nuclei
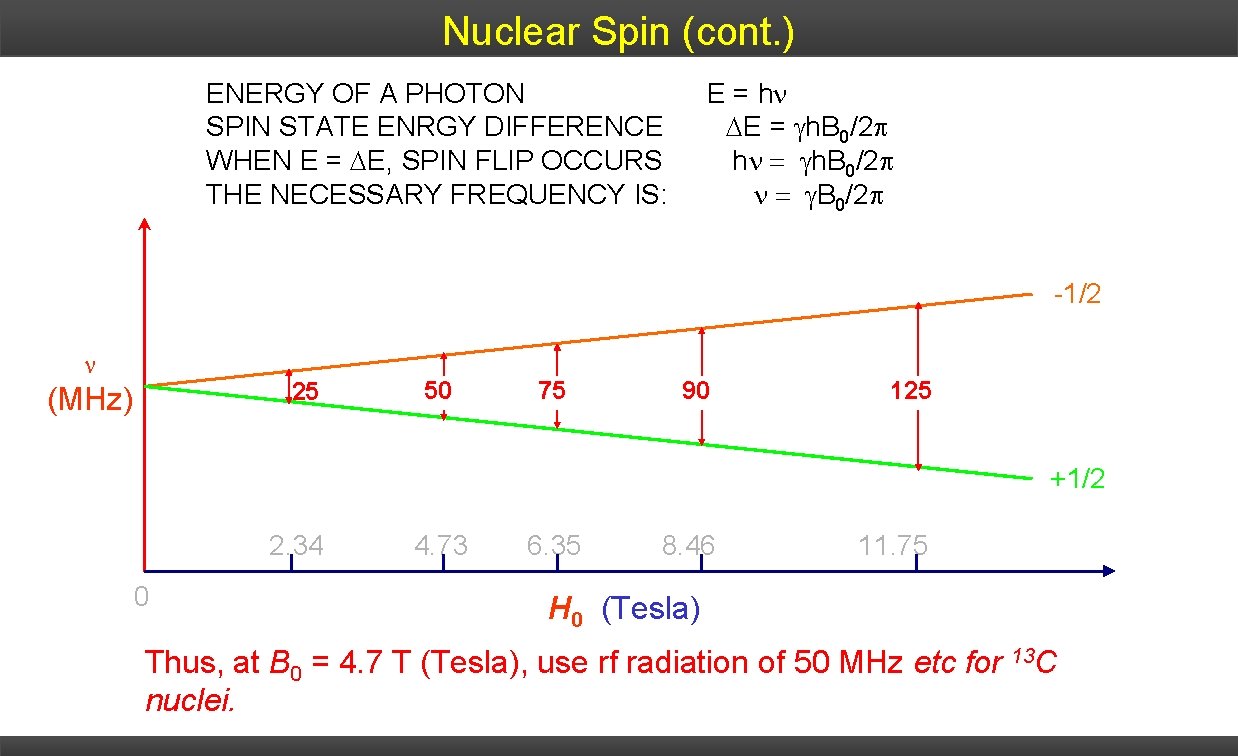
Nuclear Spin (cont. ) ENERGY OF A PHOTON E = h • Difference in energy between the two nuclear spin states: depends SPIN STATE ENRGY DIFFERENCE E = h. B 0/2 on strength external magnetic field WHEN Eof = E, SPIN FLIP OCCURS h h. B 0/2 • For THE nucleus of H atom (proton), spin differences: NECESSARY FREQUENCY IS: energy B 0/2 -1/2 25 (MHz) 50 75 90 125 +1/2 2. 34 0 4. 73 6. 35 8. 46 11. 75 H 0 (Tesla) Thus, at B 0 = 4. 7 T (Tesla), use rf radiation of 50 MHz etc for 13 C nuclei.
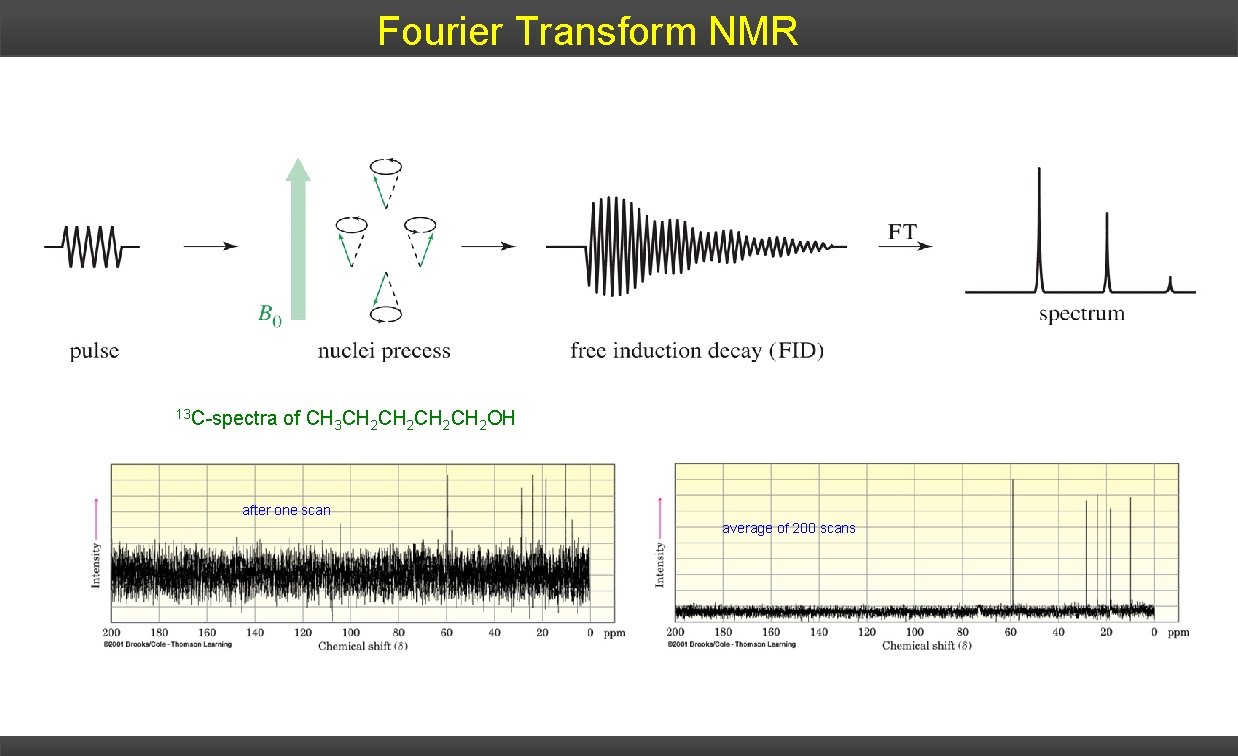
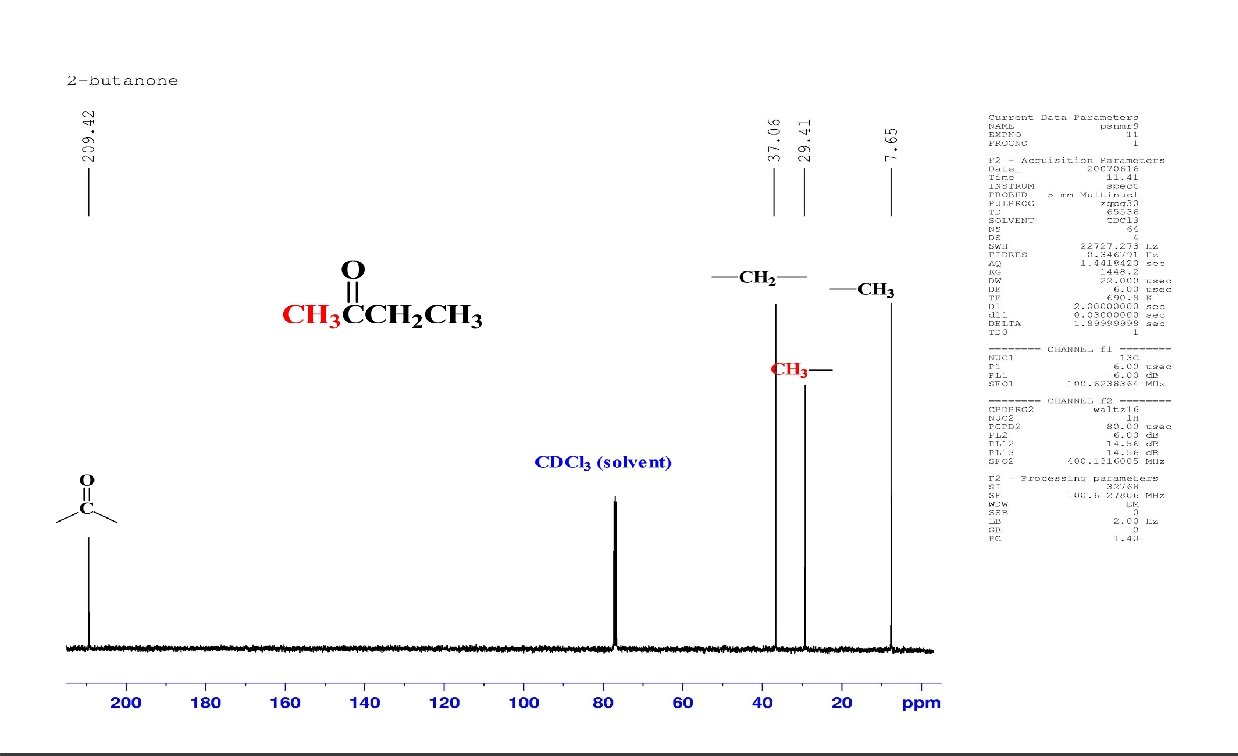
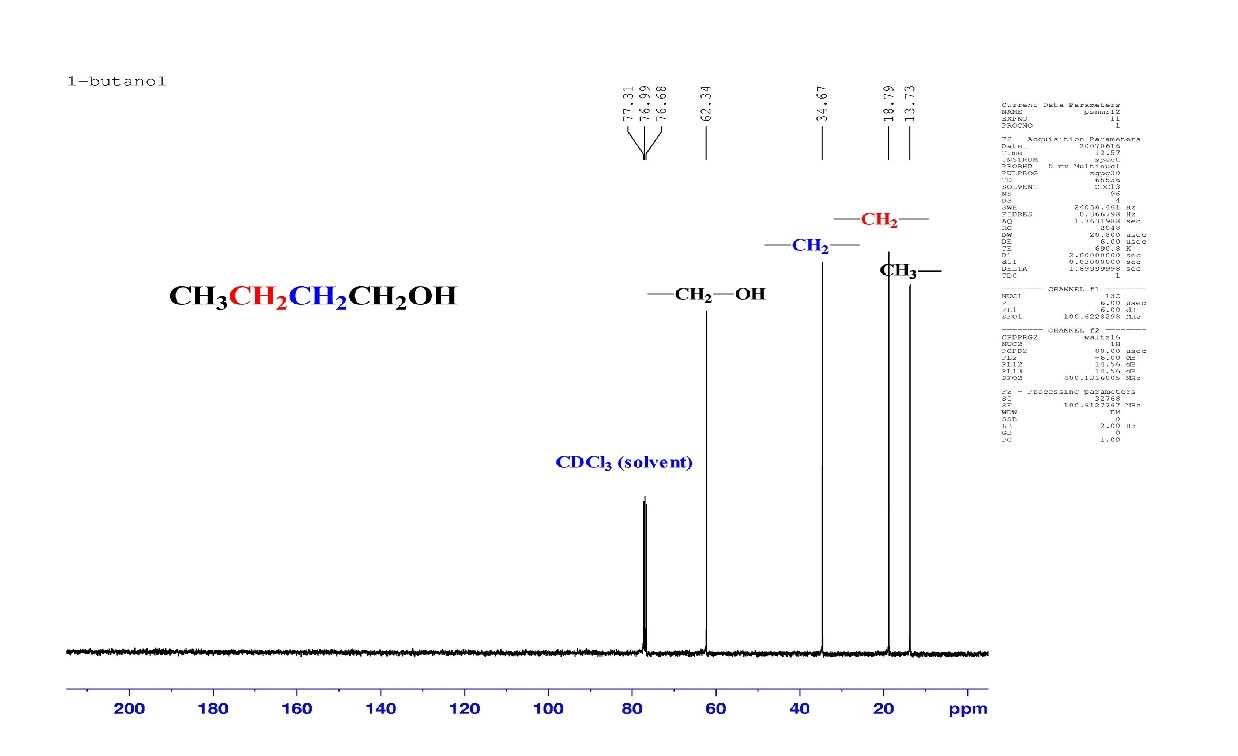
Fourier Transform NMR 13 C-spectra of CH 3 CH 2 CH 2 OH after one scan average of 200 scans

Features of 13 C NMR Spectra Each unique C in a structure gives a single peak in the spectrum; there is rarely any overlap. – The C NMR spectrum spans over 200 ppm; chemical shifts only 0. 001 ppm apart can be distinguished; this allows for over 2 x 105 possible chemical shifts for carbon. The intensity (size) of each peak is NOT directly related to the number of that type of carbon. Other factors contribute to the size of a peak: – Peaks from carbon atoms that have attached hydrogen atoms are bigger than those that don’t have hydrogens attached. Carbon chemical shifts are usually reported as downfield from the carbon signal of tetramethylsilane (TMS).

H 1 and C 13 NMR Spectroscopy Number of peaks Chemical shifts Integration Spin-Spin Splitting
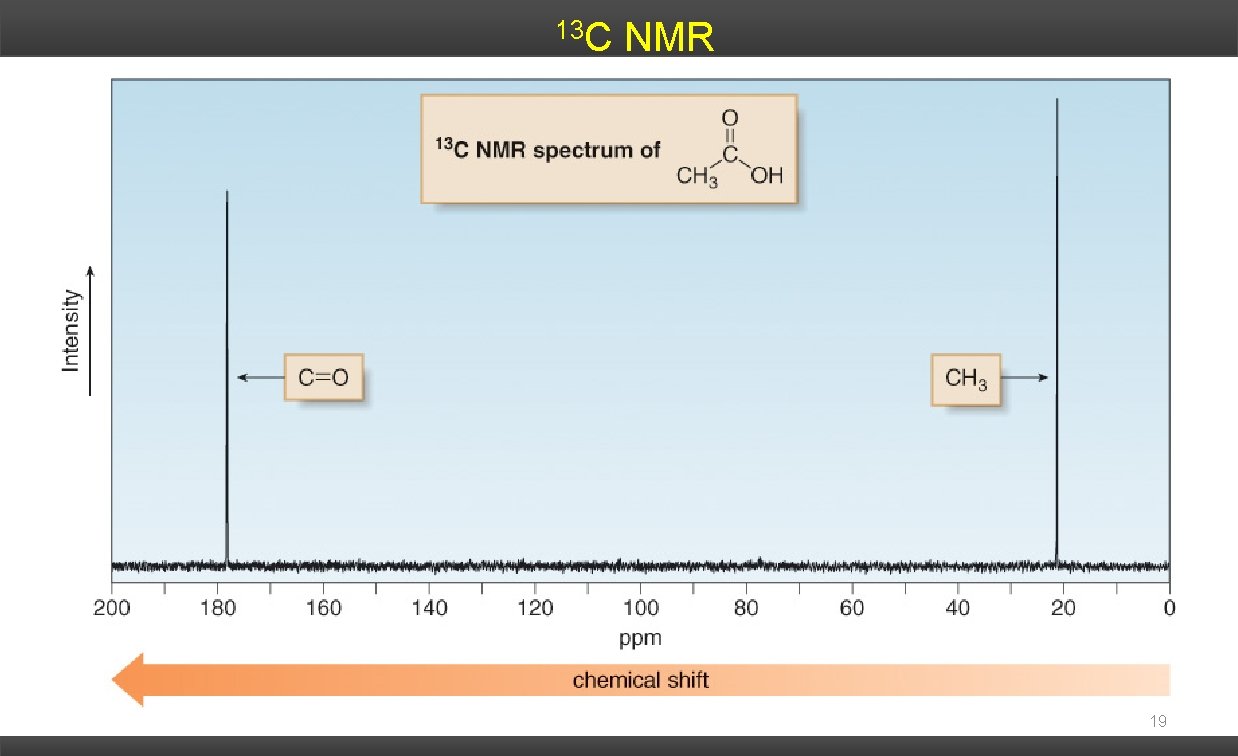
13 C NMR 19
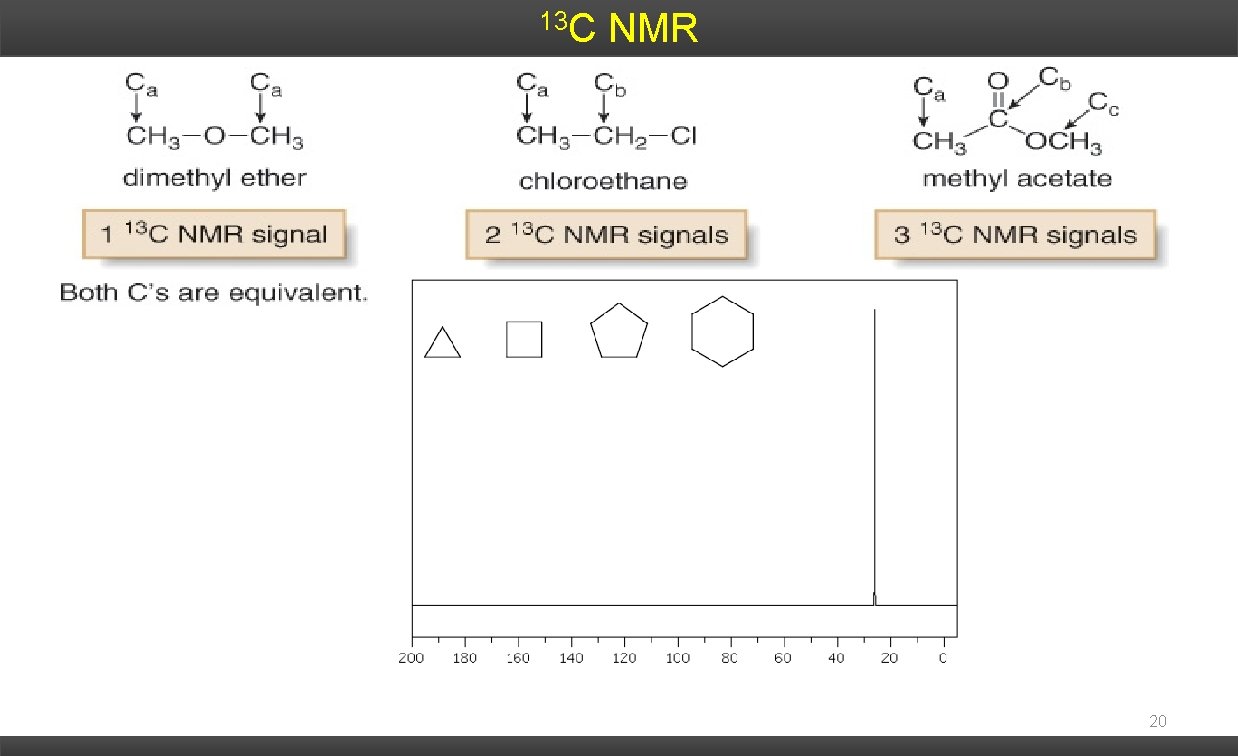
13 C NMR 20
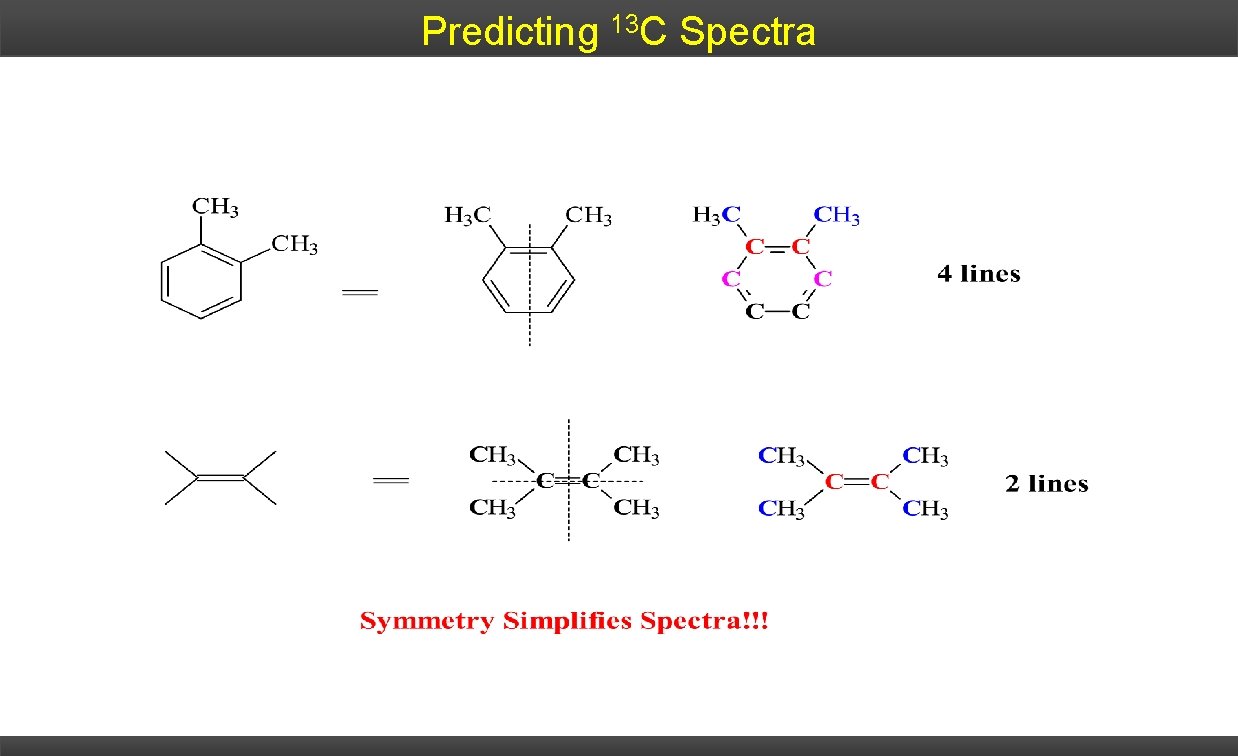
Predicting 13 C Spectra
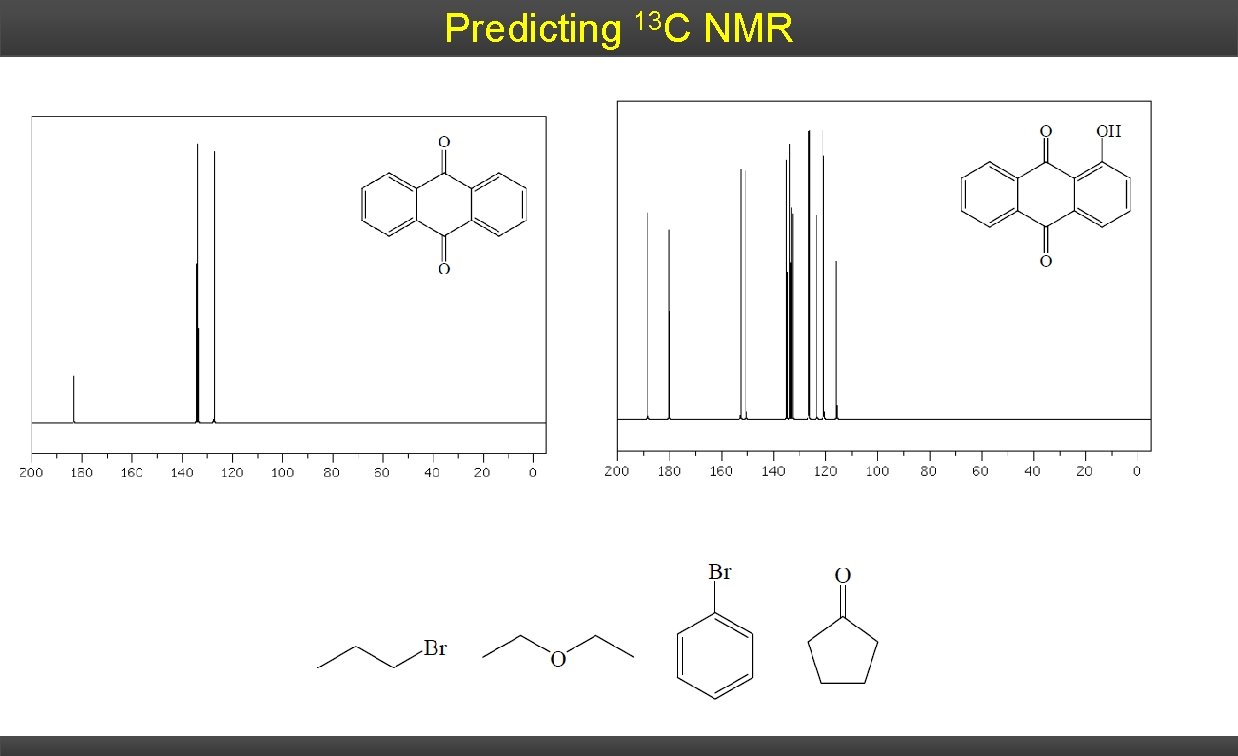
Predicting 13 C NMR
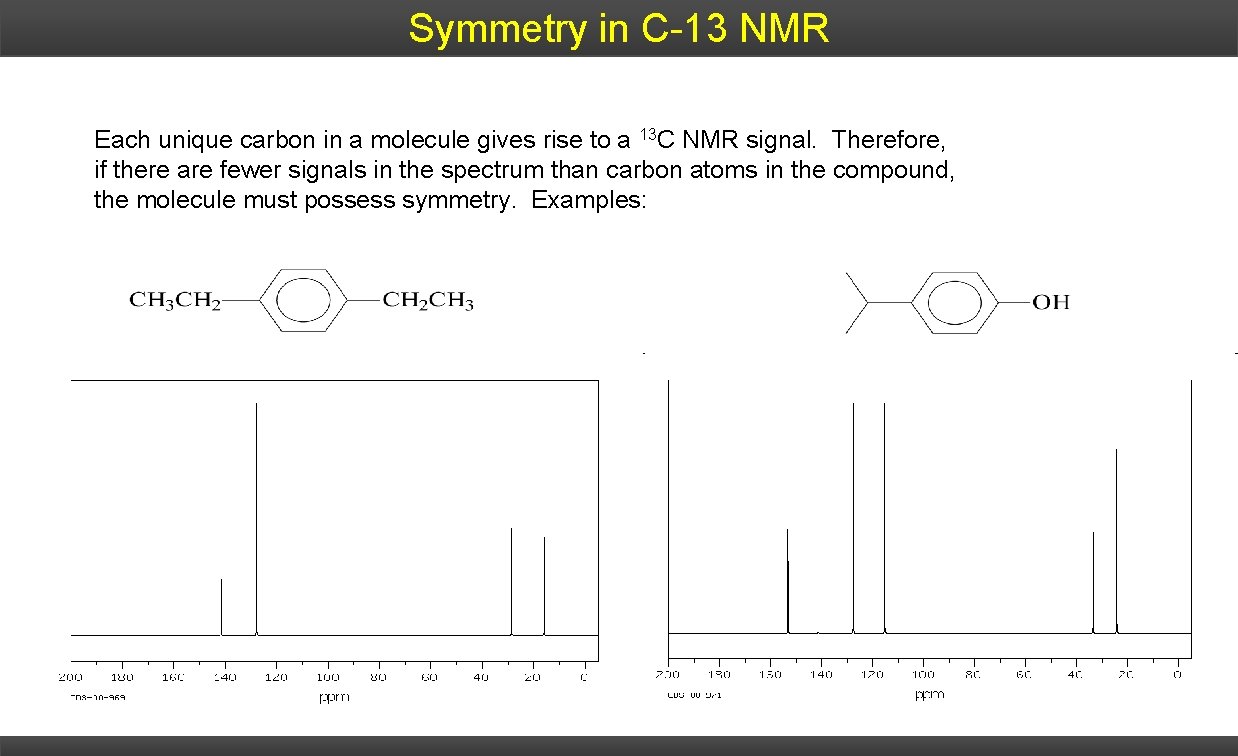
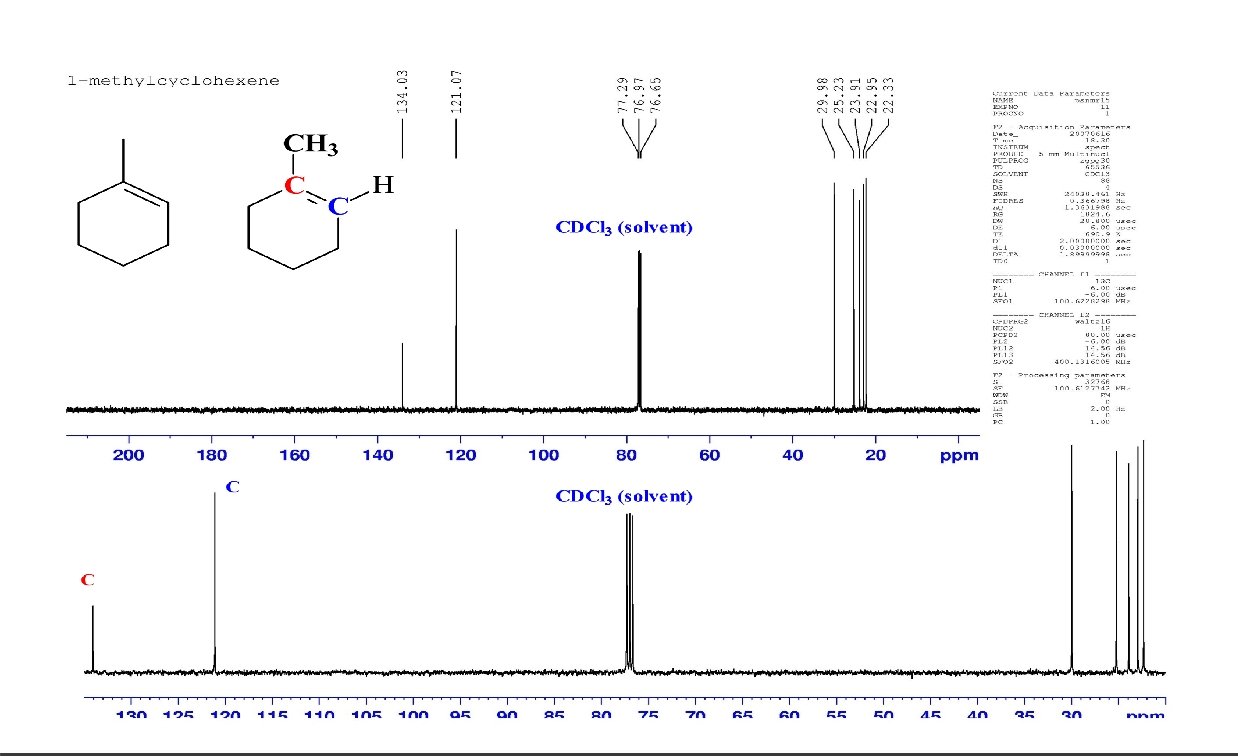
Symmetry in C-13 NMR Each unique carbon in a molecule gives rise to a 13 C NMR signal. Therefore, if there are fewer signals in the spectrum than carbon atoms in the compound, the molecule must possess symmetry. Examples:

How many signals would you expect?
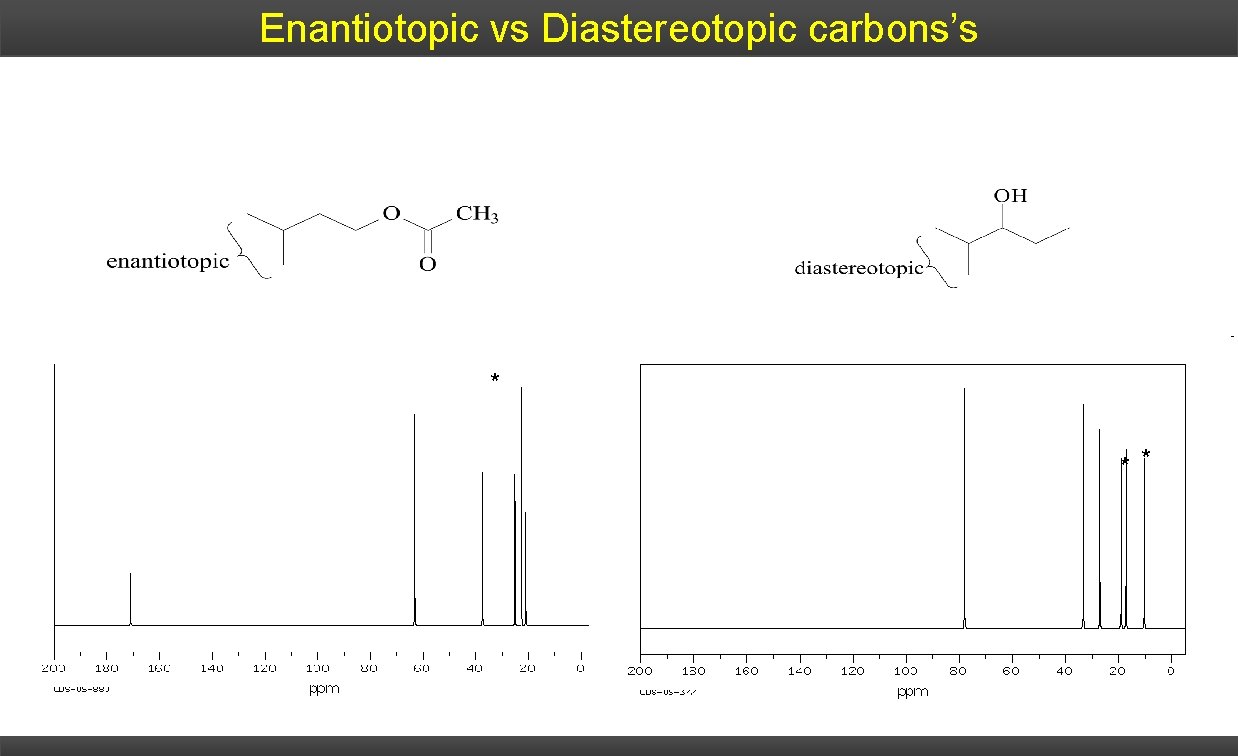
Enantiotopic vs Diastereotopic carbons’s * * *
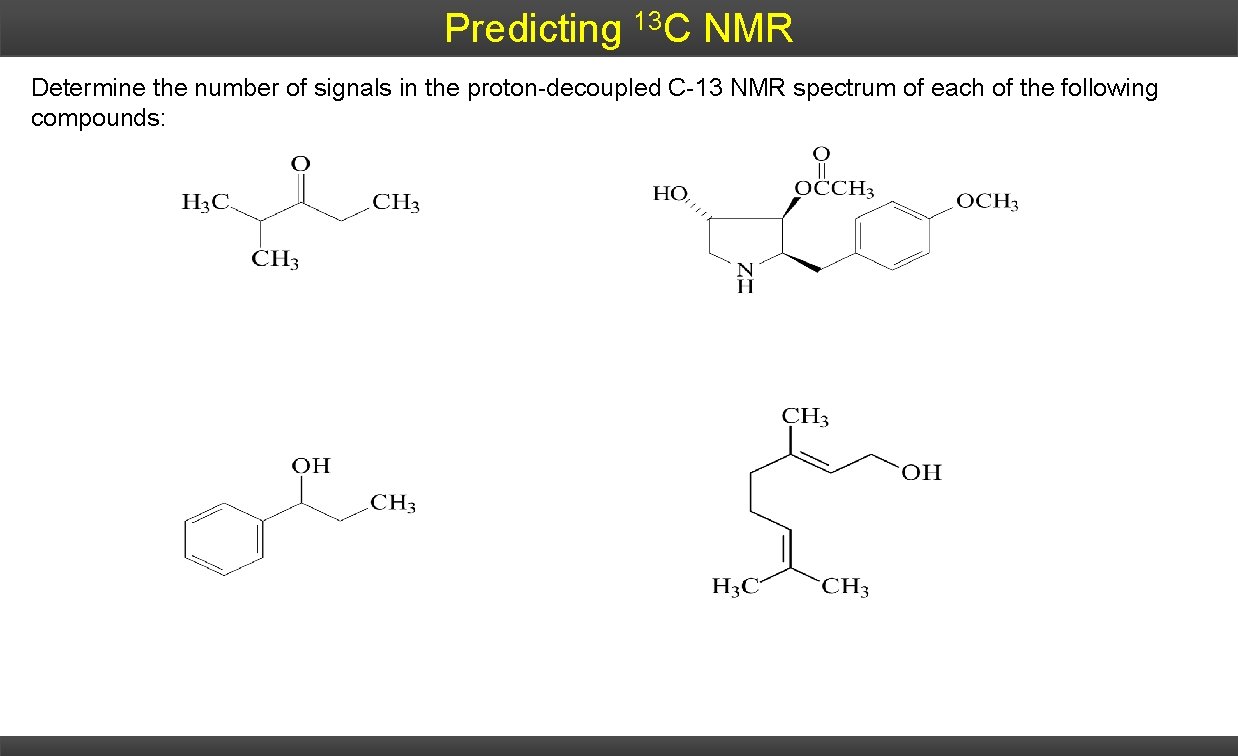
Predicting 13 C NMR Determine the number of signals in the proton-decoupled C-13 NMR spectrum of each of the following compounds:

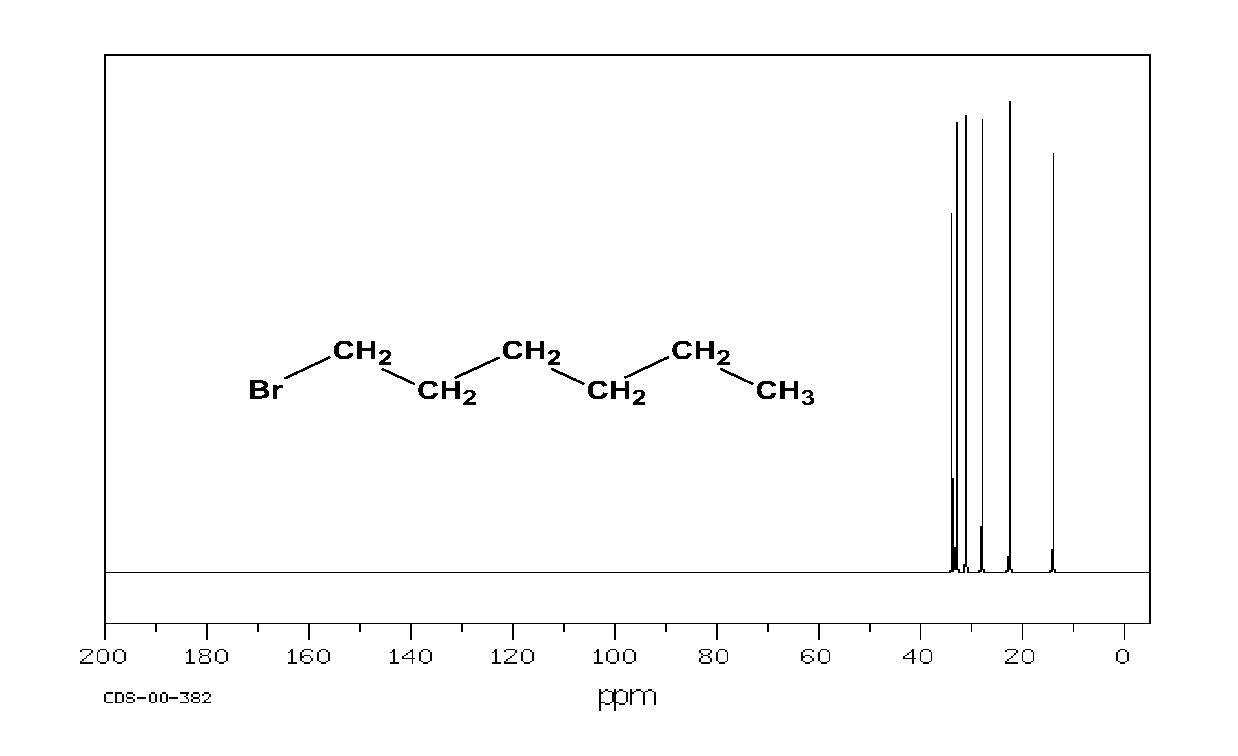
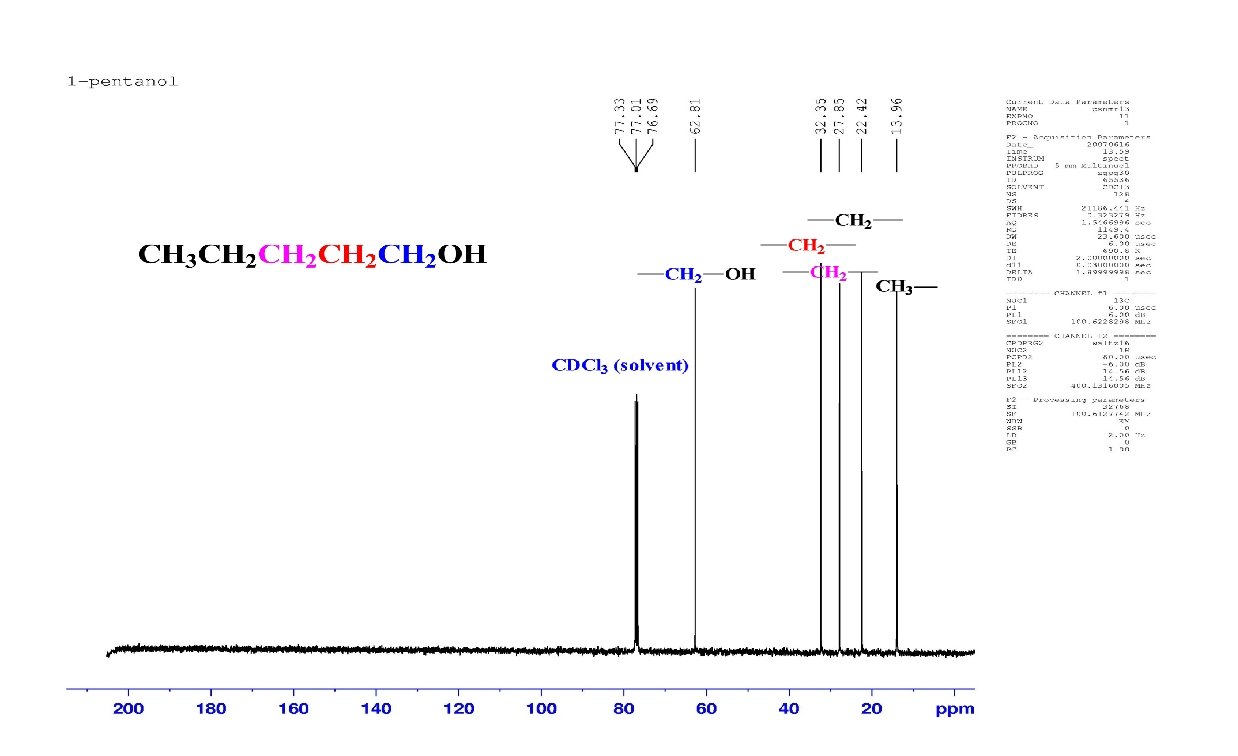
1 H and 13 C NMR compared: 13 C signals are spread over a much wider range than 1 H signals making it easier to identify and count individual nuclei The following slides show the 1 H NMR and the 13 C spectrum of 1 -chloropentane. It is much easier to identify the compound as 1 -chloropentane by its 13 C spectrum than by its 1 H spectrum.
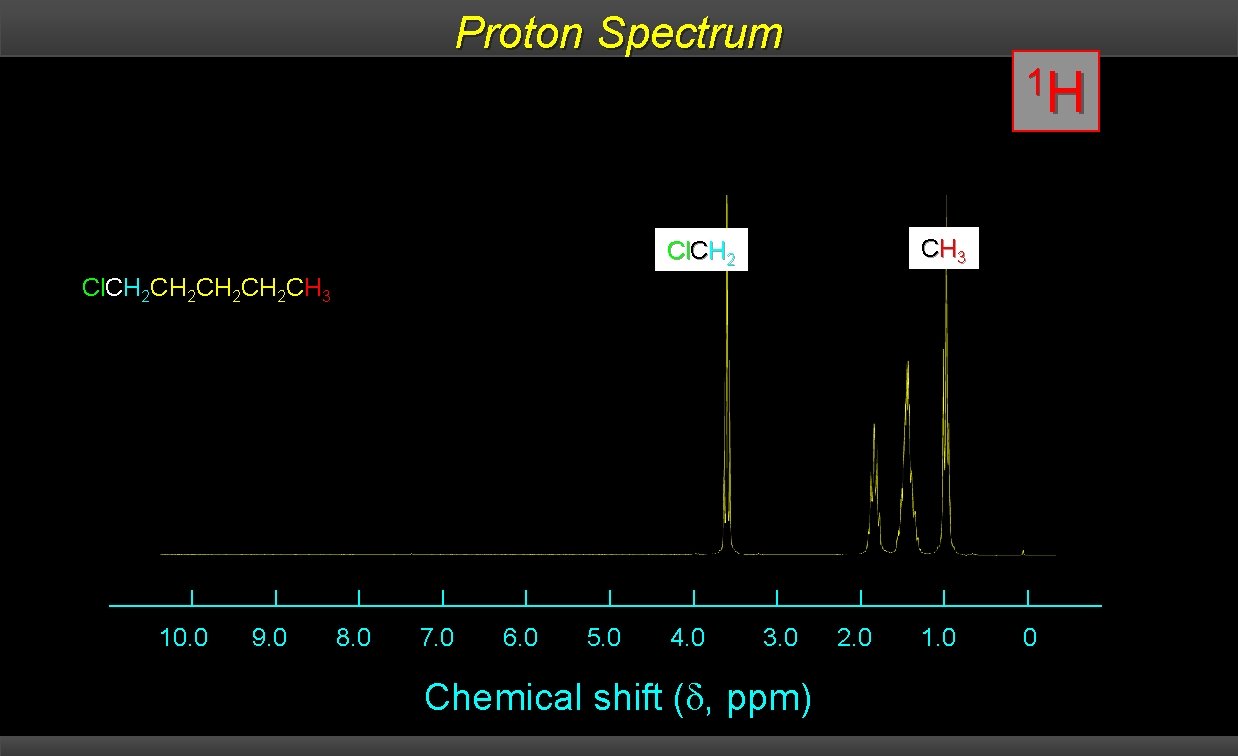
Proton Spectrum 1 H CH 3 Cl. CH 2 CH 2 CH 2 CH 3 10. 0 9. 0 8. 0 7. 0 6. 0 5. 0 4. 0 3. 0 Chemical shift ( , ppm) 2. 0 1. 0 0
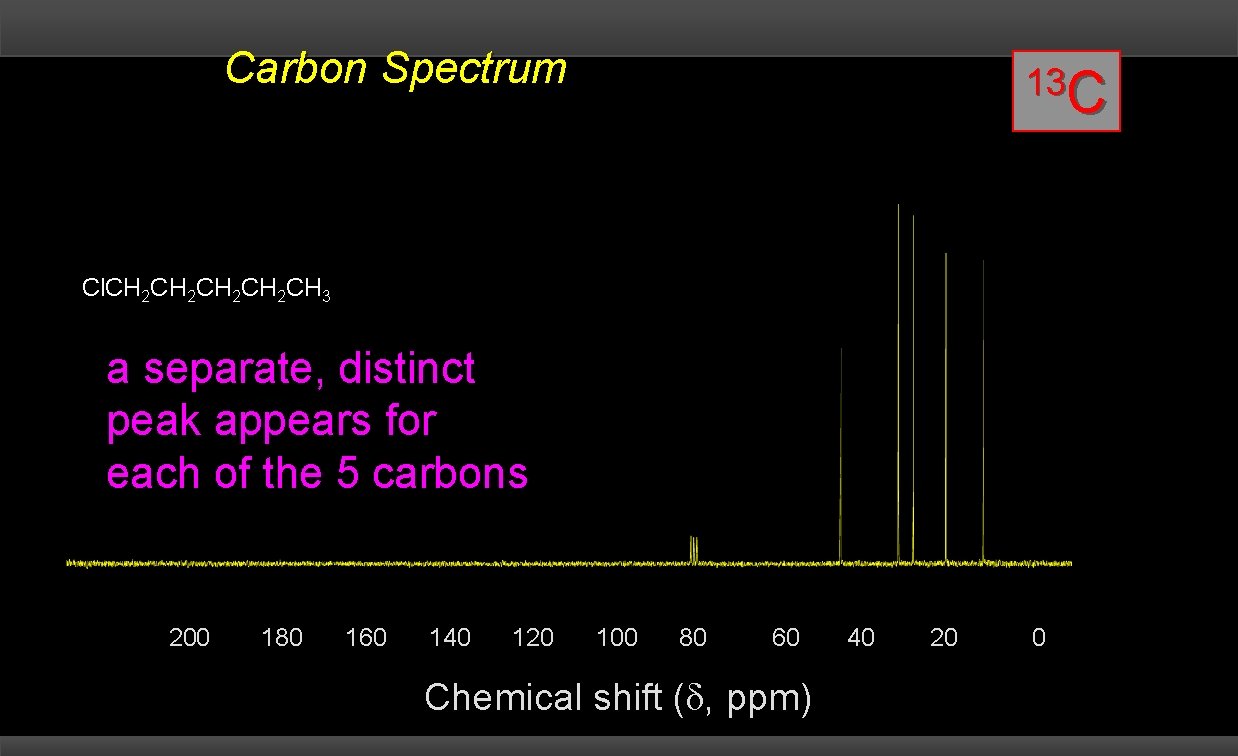
Carbon Spectrum 13 C Cl. CH 2 CH 2 CH 3 a separate, distinct peak appears for each of the 5 carbons 200 180 160 140 120 CDCl 3 100 80 60 Chemical shift ( , ppm) 40 20 0
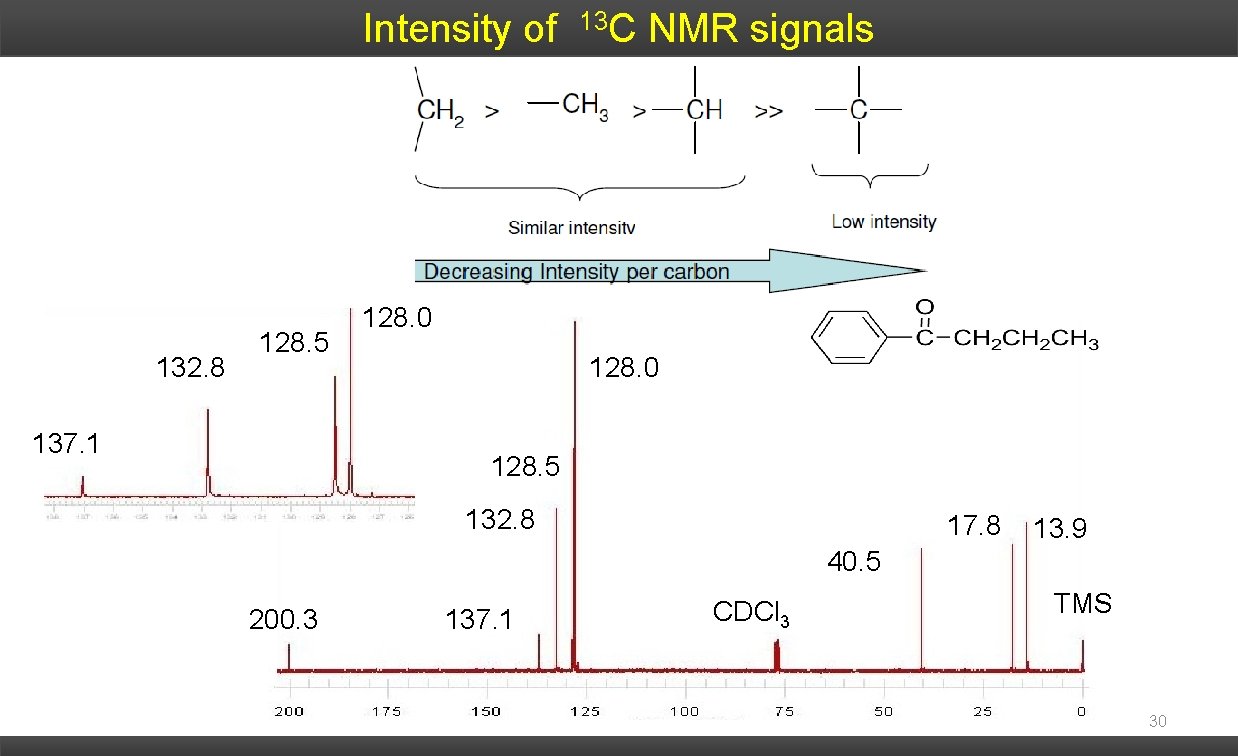
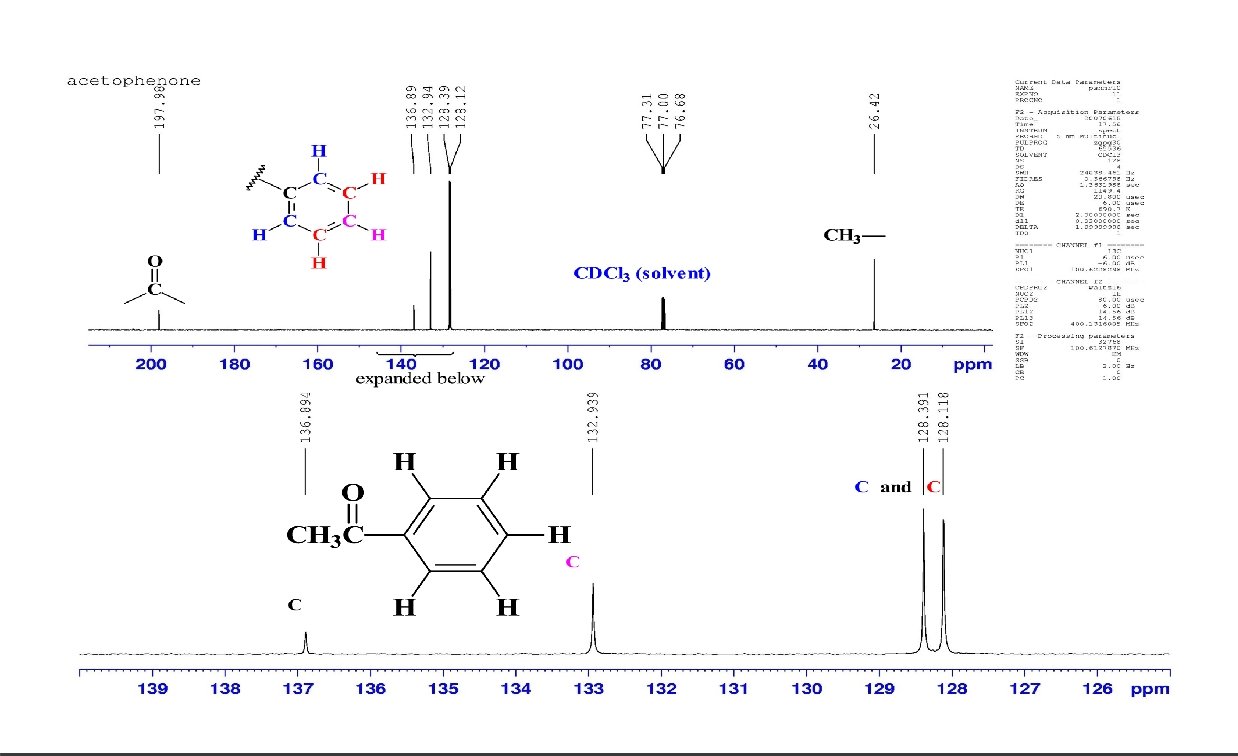
Intensity of 132. 8 128. 5 137. 1 13 C NMR signals 128. 0 128. 5 132. 8 17. 8 13. 9 40. 5 200. 3 137. 1 CDCl 3 TMS 30

13 C-NMR: Integration 1 H-NMR: Integration reveals relative number of hydrogens per signal 13 C-NMR: Integration reveals relative number of carbons per signal • Rarely useful due to slow relaxation time for 13 C time for nucleus to relax from excited spin state to ground state

13 C Chemical shifts are most affected by: electronegativity of groups attached to carbon hybridization state of carbon

13 C NMR Chemical Shifts Several functionalities appear directly on 13 C NMR which are not ‘visible’ in 1 H NMR: - Quaternary carbons sp carbon - ipso carbons sp EWG - Carbonyl carbons alkyne carbons 3 3 - alkene carbons aromatic carbons carbonyl carbons 220 200 180 160 downfield deshielded higher DE 140 120 (ppm) 100 80 upfield shielded lower DE 60 40 20 0. 0
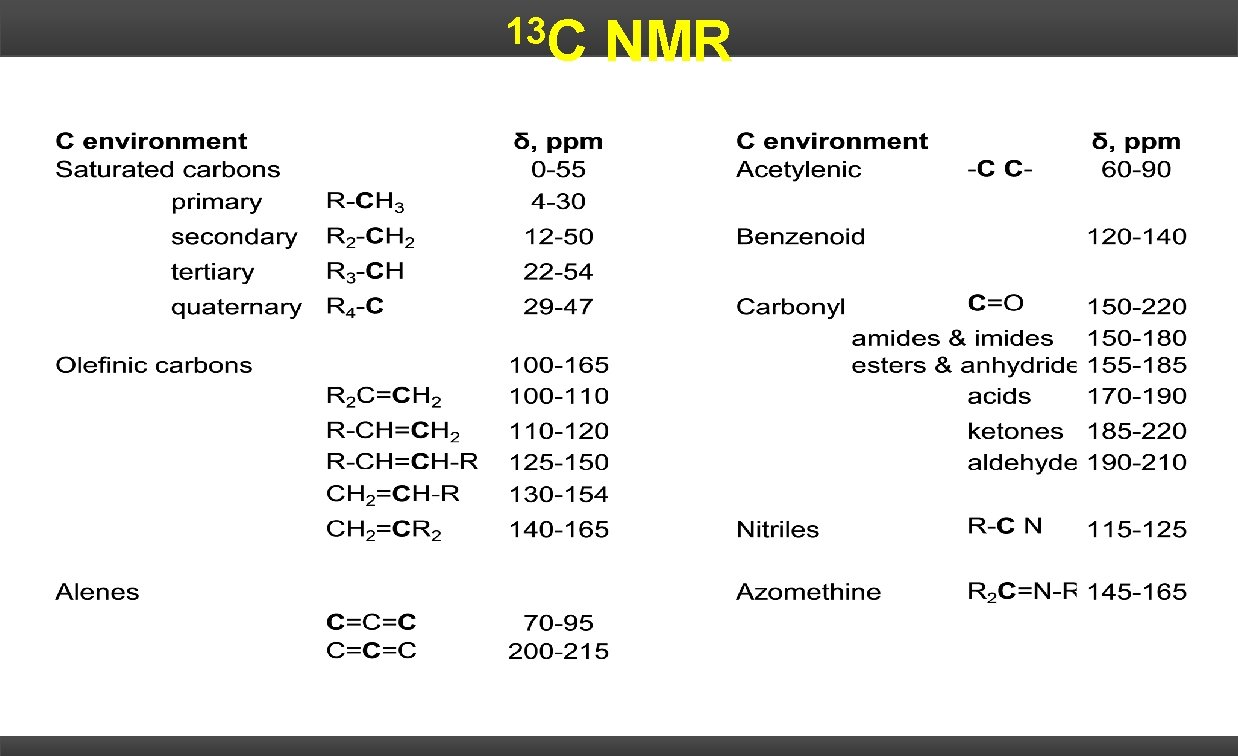
13 C NMR

Chemical Shift - 13 C-NMR Trends • RCH 3 < R 2 CH 2 < R 3 CH • Electronegative atoms cause downfield shift • Pi bonds cause downfield shift • C=O 160 -210 ppm
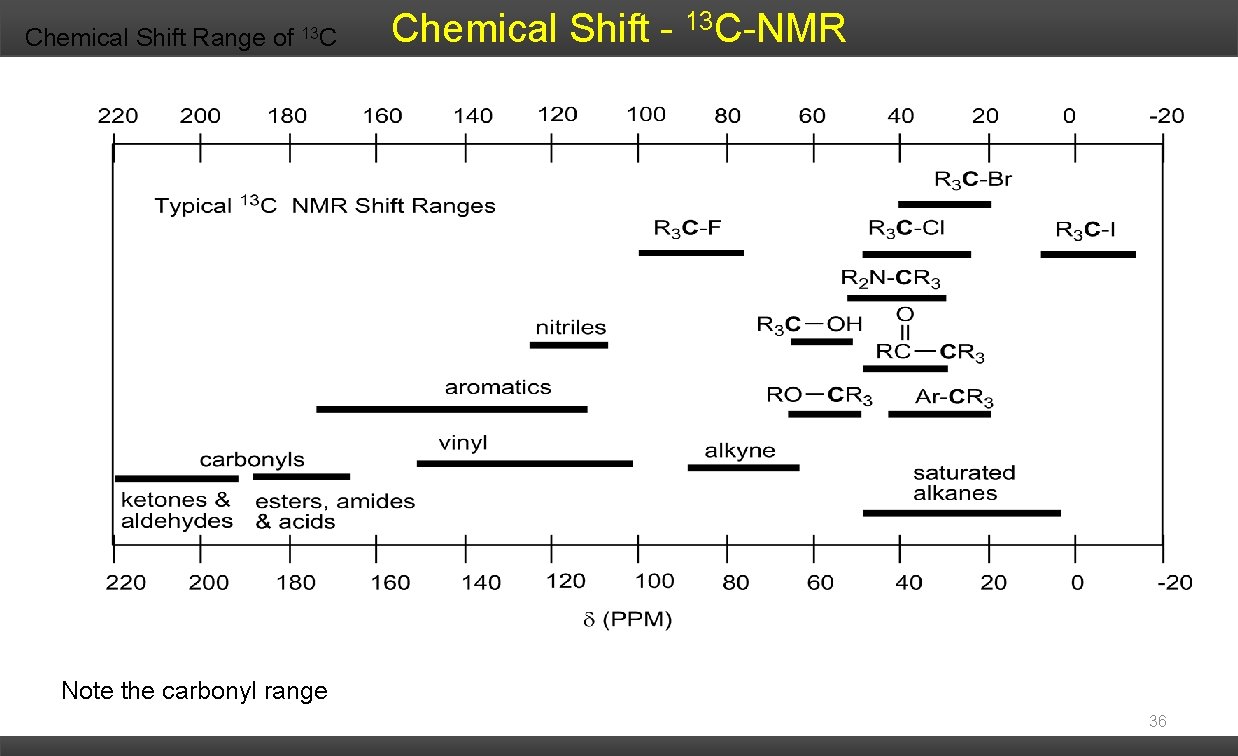
Chemical Shift Range of 13 C Chemical Shift - 13 C-NMR Note the carbonyl range 36

Carbonyl Carbon Chemical Shifts anhydrides nitriles acid chlorides amides esters carboxylic acids aldehydes conj. Ketones ketones 220 210 200 190 180 170 160 150 140 130 120 110
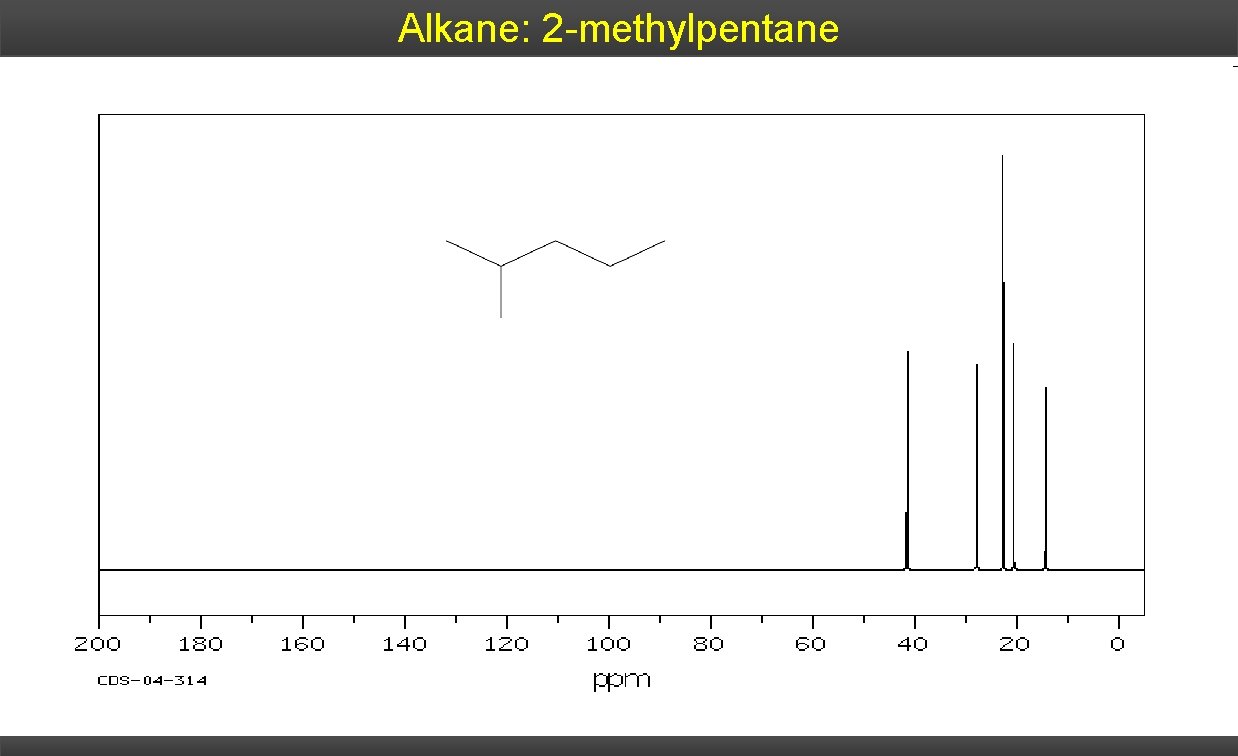
Alkane: 2 -methylpentane
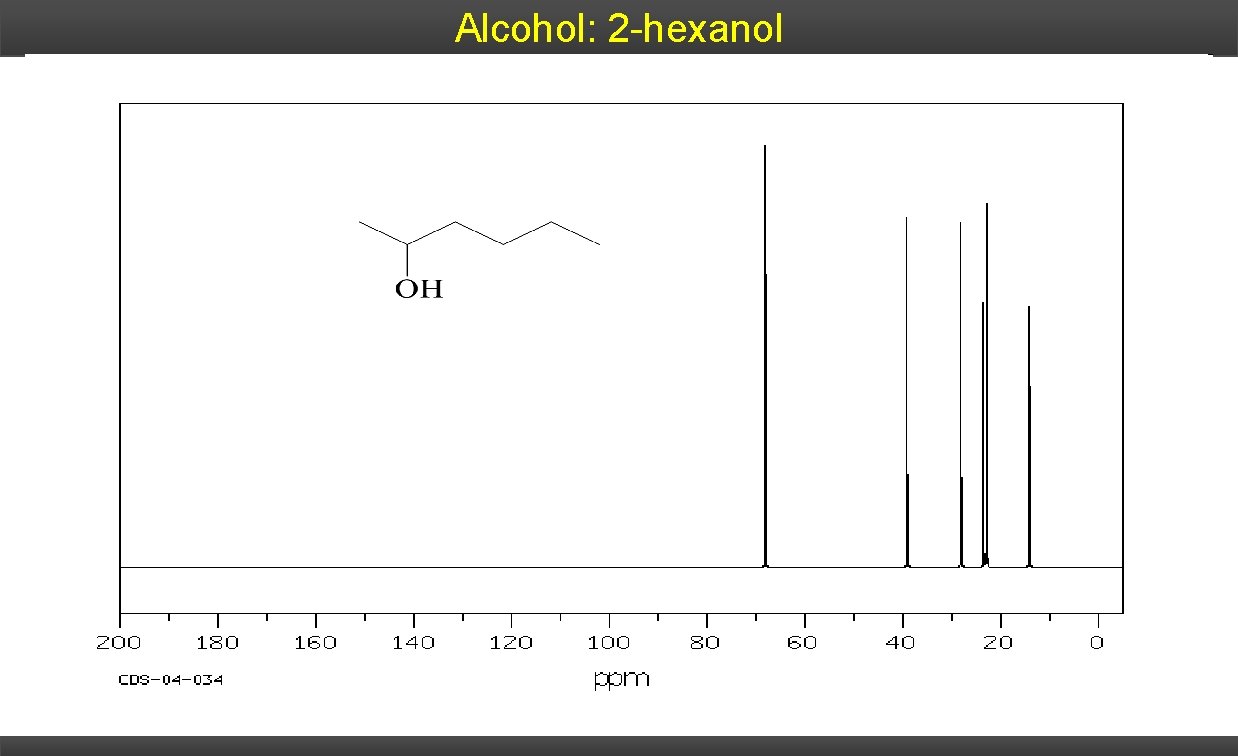
Alcohol: 2 -hexanol
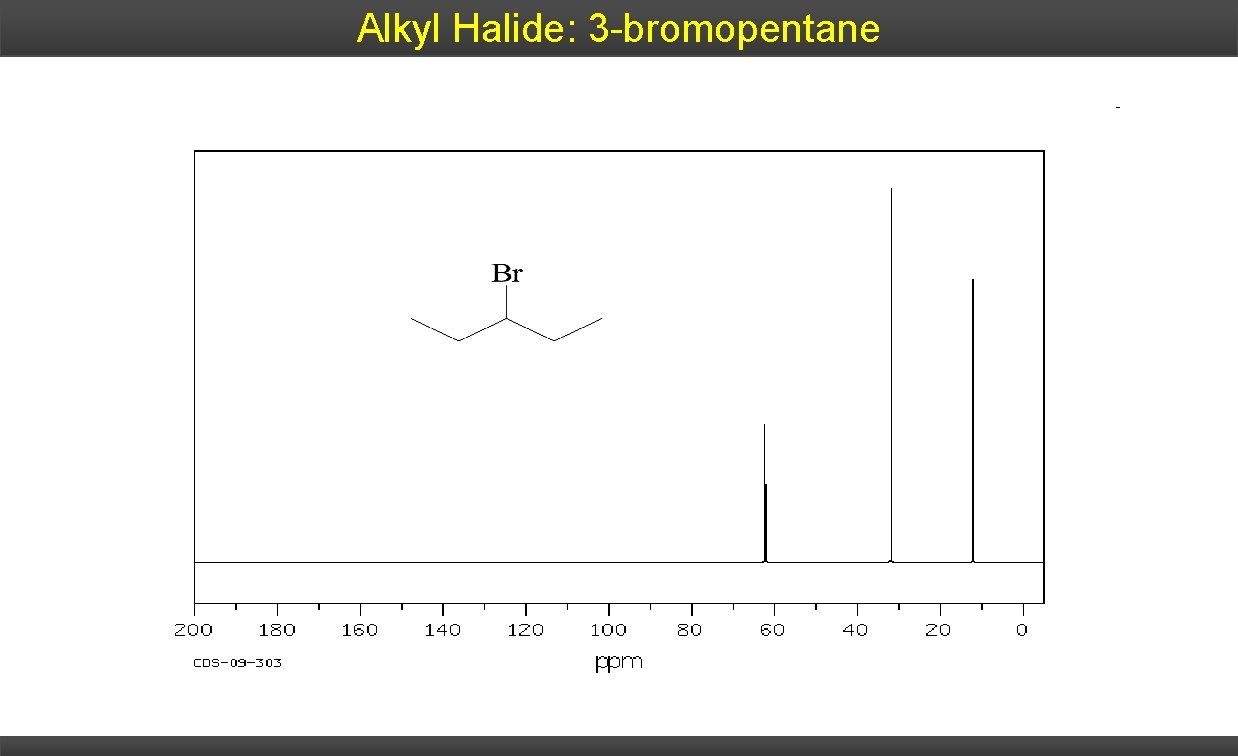
Alkyl Halide: 3 -bromopentane
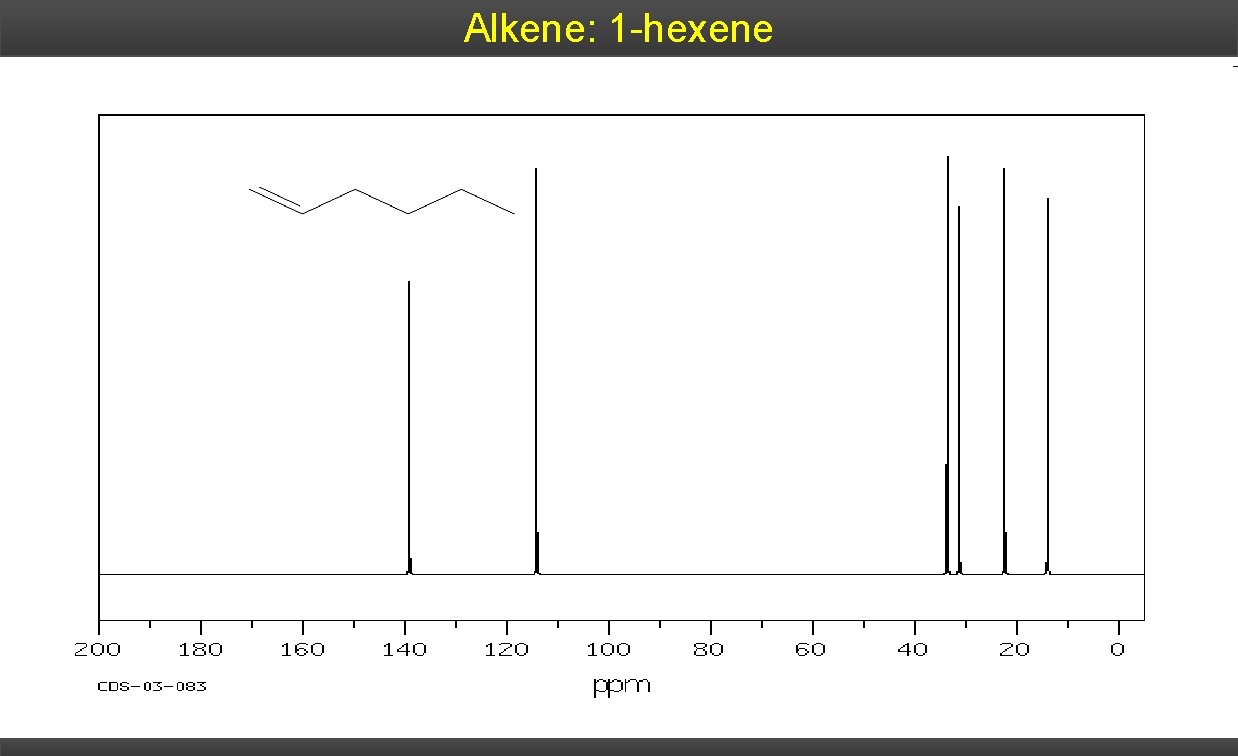
Alkene: 1 -hexene
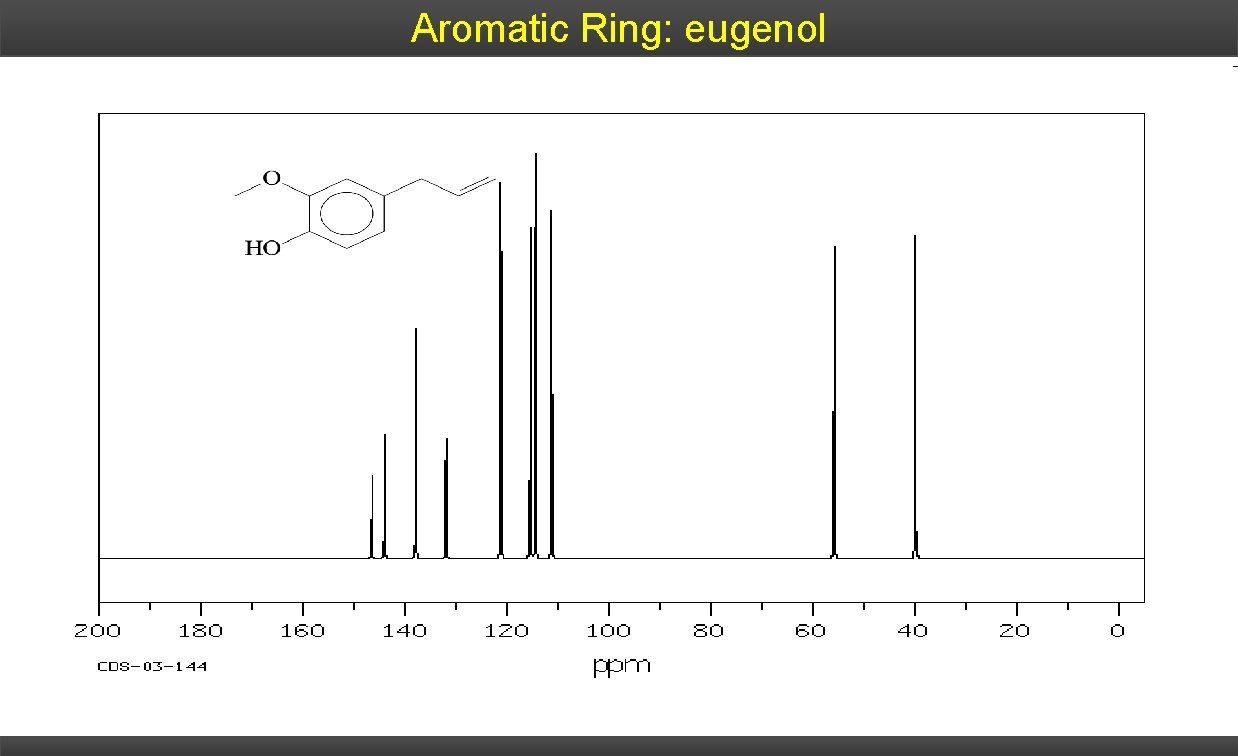
Aromatic Ring: eugenol
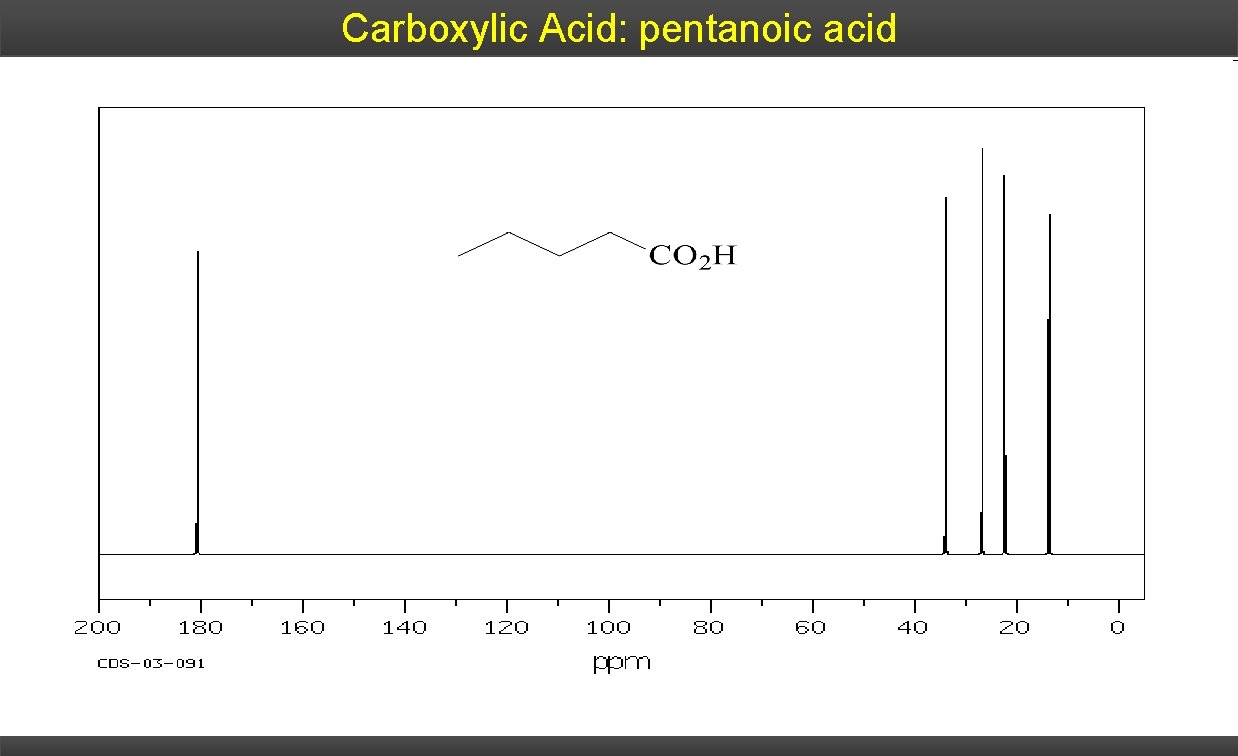
Carboxylic Acid: pentanoic acid
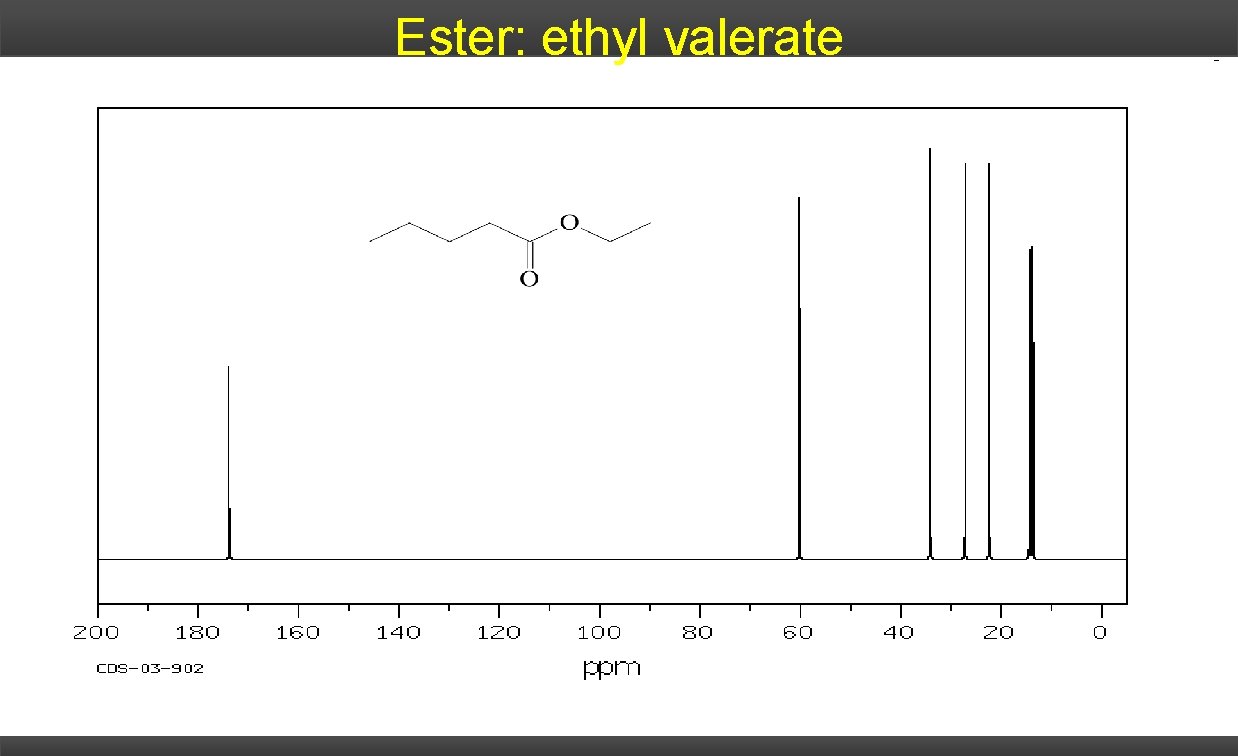
Ester: ethyl valerate
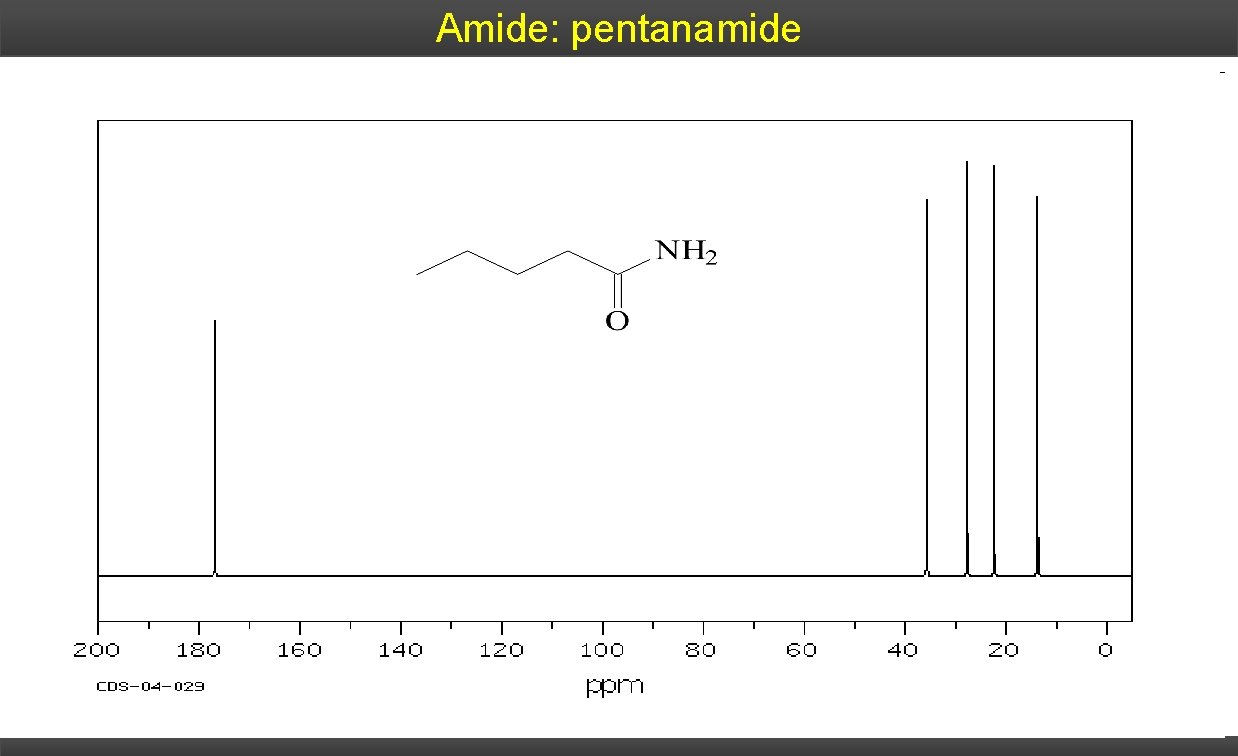
Amide: pentanamide
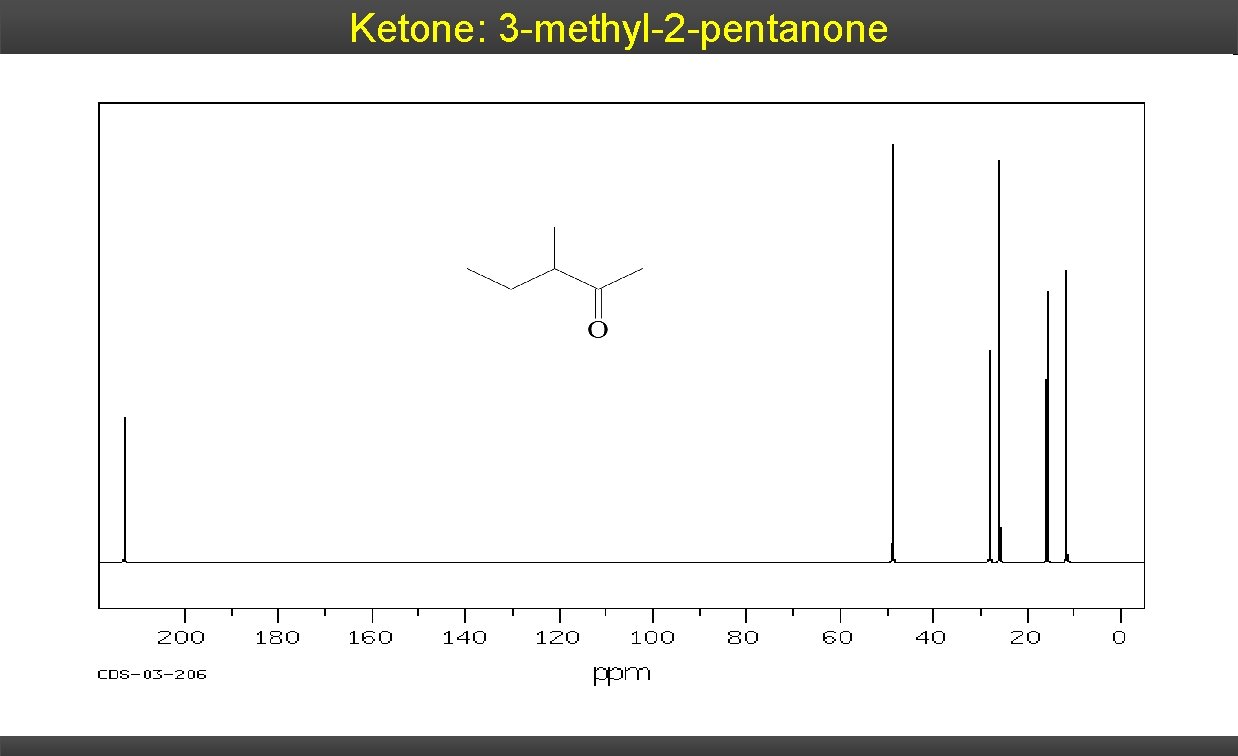
Ketone: 3 -methyl-2 -pentanone
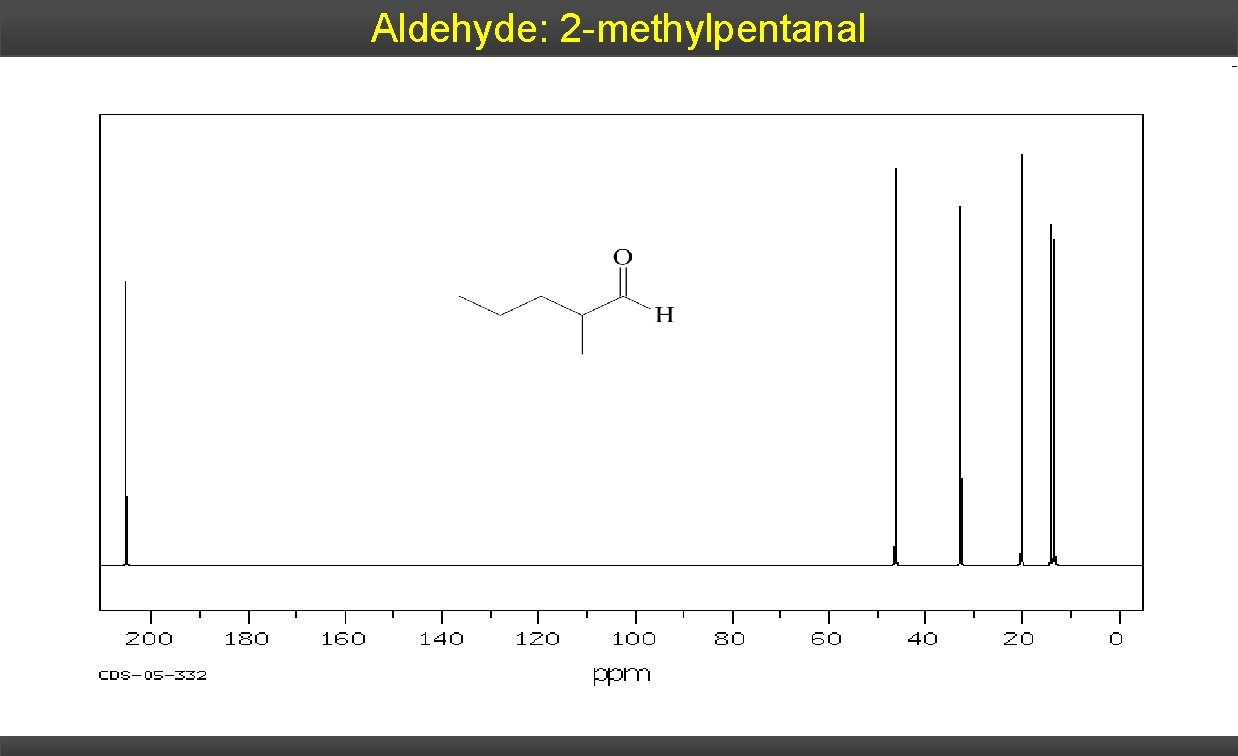
Aldehyde: 2 -methylpentanal
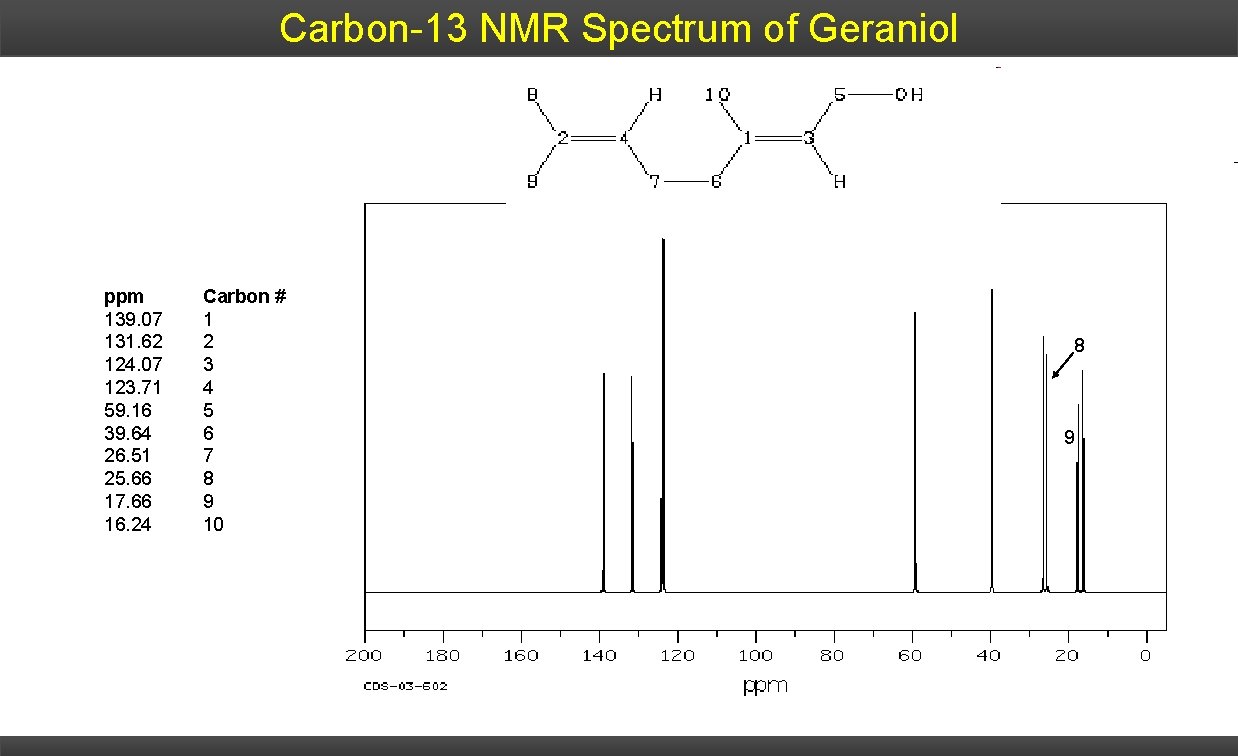
Carbon-13 NMR Spectrum of Geraniol ppm 139. 07 131. 62 124. 07 123. 71 59. 16 39. 64 26. 51 25. 66 17. 66 16. 24 Carbon # 1 2 3 4 5 6 7 8 9 10 8 9

Spin-Spin Coupling in 13 C NMR Homonuclear coupling of 13 C-13 C is possible in theory. However, due to the low natural abundance of 13 C, it is rare to find two 13 C’s in the same molecule, let alone adjacent to one another. No need to consider 13 C-13 C coupling except for enrichment studies! Heteronuclear coupling between 13 C and the 1 H atoms attached to them is observed (1 H abundance ~99%). Because the 1 H atoms are directly attached, the coupling constants (1 J)are large, typically 100 -250 Hz. When such spectra are observed, they are referred to as proton coupled spectra (or non-decoupled spectra).

Carbon-13 NMR Spectroscopy Where’s Waldo? 50

Spin-Spin Coupling in 13 C NMR Homonuclear coupling of 13 C-13 C is possible in theory. However, due to the low natural abundance of 13 C, it is rare to find two 13 C’s in the same molecule, let alone adjacent to one another. No need to consider 13 C-13 C coupling except for enrichment studies! Heteronuclear coupling between 13 C and the 1 H atoms attached to them is observed (1 H abundance ~99%). Because the 1 H atoms are directly attached, the coupling constants (1 J)are large, typically 100 -250 Hz. When such spectra are observed, they are referred to as proton coupled spectra (or non-decoupled spectra).
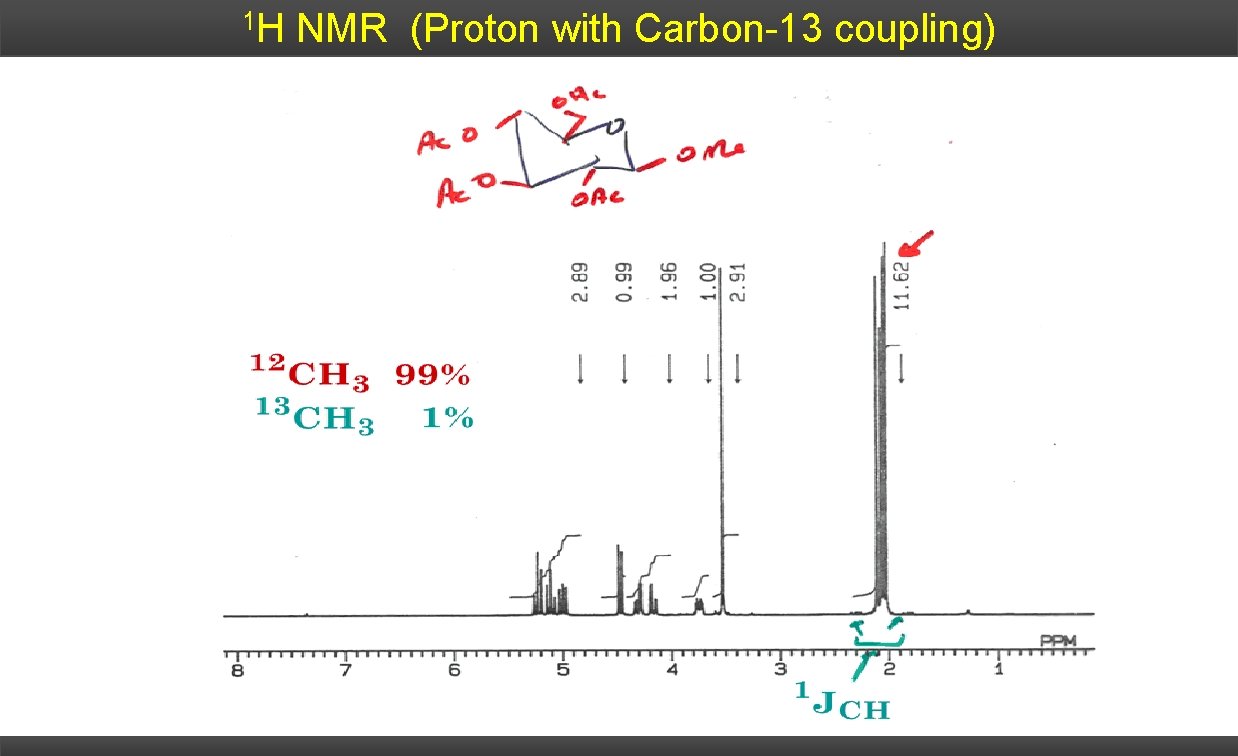
1 H NMR (Proton with Carbon-13 coupling)
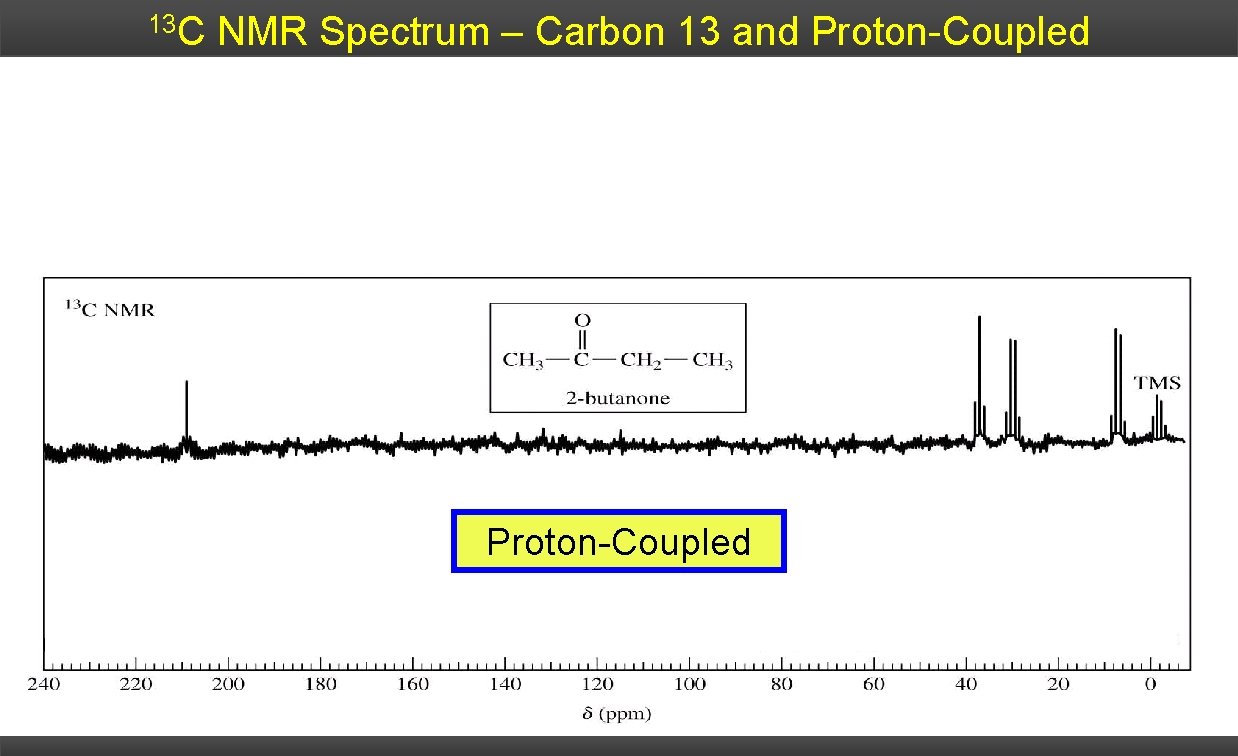
13 C NMR Spectrum – Carbon 13 and Proton-Coupled

1 H – 13 C Splitting The splitting follows the simple N+1 rule: quaternary methine methylene quaternary singlet doublet triplet quartet The multiplet analysis gives useful information, but there are two major limitations: 1) If the 13 C signal is weak (common) the outer peaks of the multiplet may be lost in the noise of the spectrum. 2) Due to the large J-constants, the multiplets quickly begin to overlap and become congested.
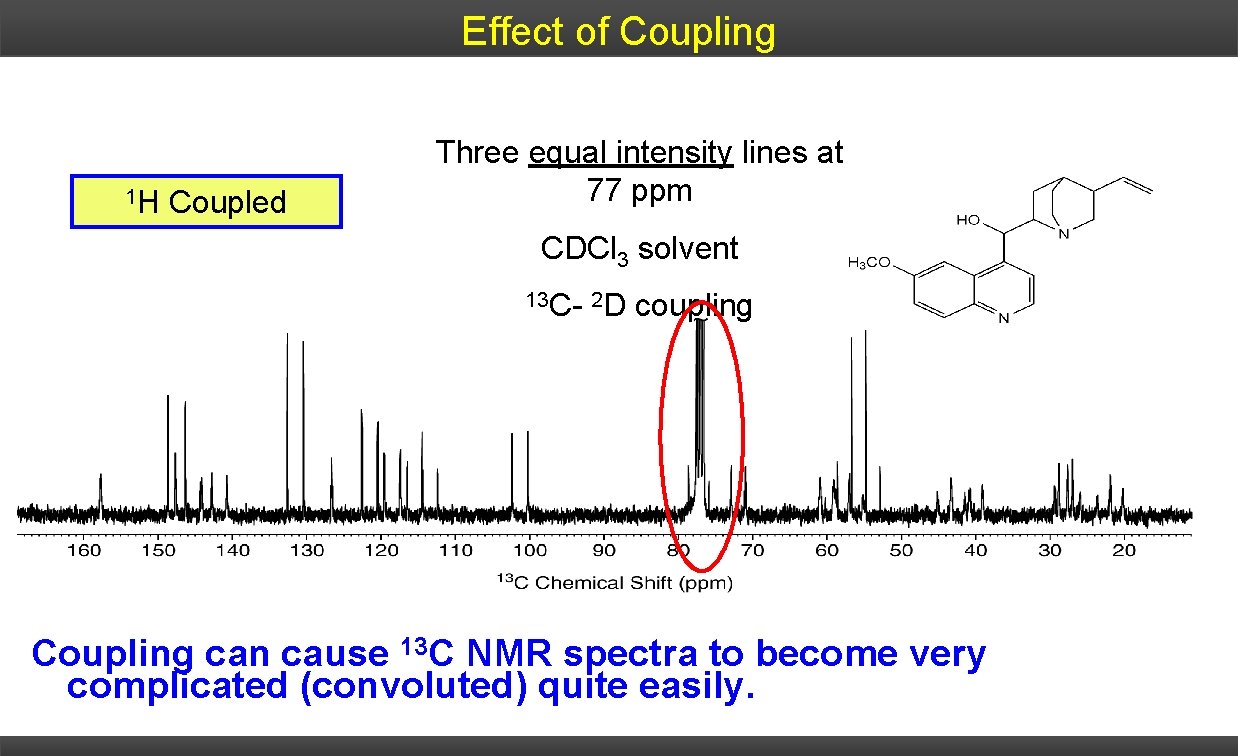
Effect of Coupling 1 H Coupled Three equal intensity lines at 77 ppm CDCl 3 solvent 13 C- 2 D coupling Coupling can cause 13 C NMR spectra to become very complicated (convoluted) quite easily.

1 H Decoupling To simplify the 13 C spectrum, and to increase the intensity of the observed signals, a decoupler is used to remove the spin effects of the 1 H nucleus. A second RF generator irradiates at the 1 H resonance frequency causing the saturation – effectively averaging all their spin states to zero 1 H channel- 13 C channel 13 C pulse 13 C FID

Decoupling Proton-decoupled mode, a sample is irradiated with two different radiofrequencies. One to excite all 13 C nuclei, a second to cause all protons in the molecule to undergo rapid transitions between their nuclear spin states. On the time scale of a 13 C-NMR spectrum, each proton is in an average or effectively constant nuclear spin state, with the result that 1 H-13 C spin-spin interactions are not observed and they are decoupled.
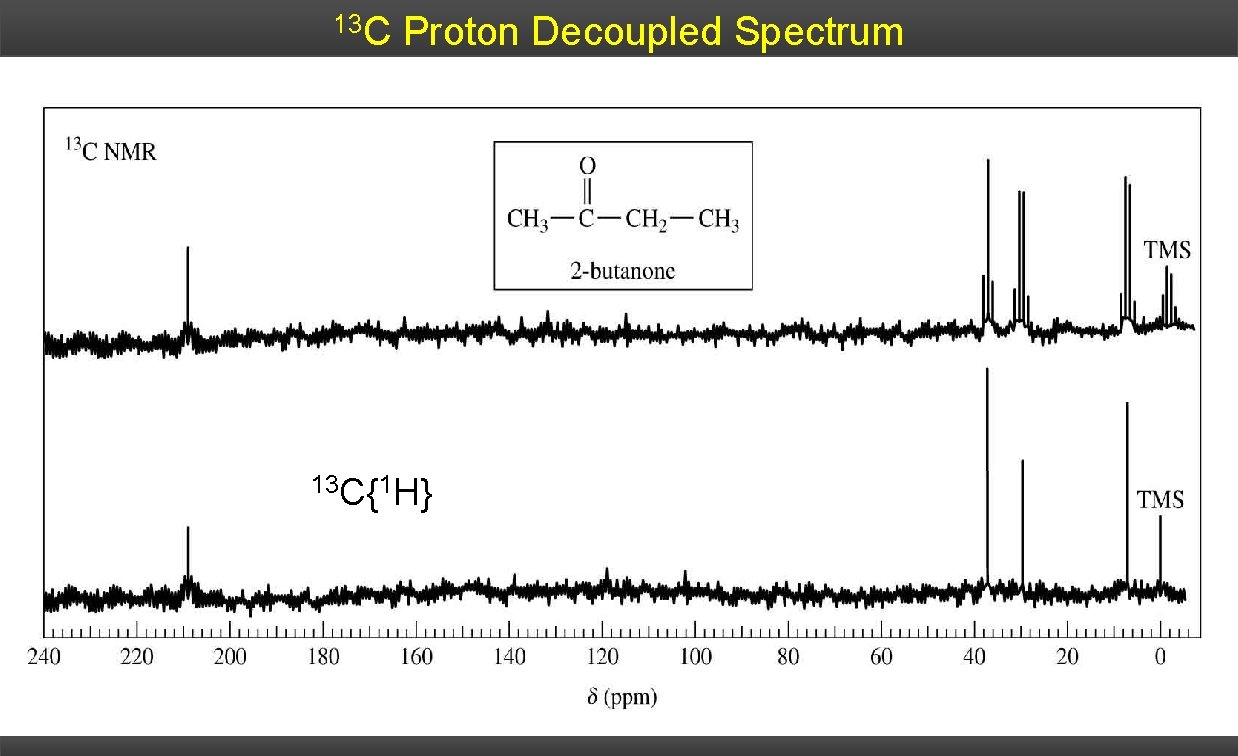
13 C Proton Decoupled Spectrum 13 C{1 H}
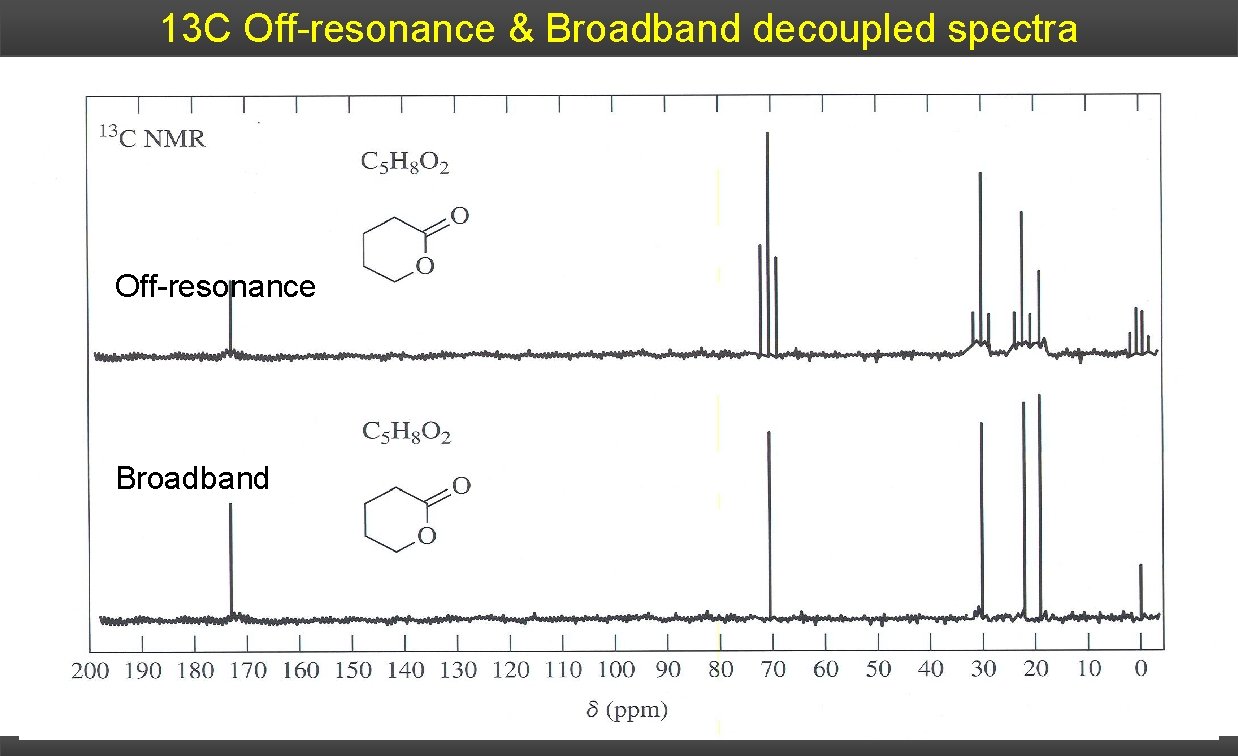
13 C Off-resonance & Broadband decoupled spectra Off-resonance Broadband
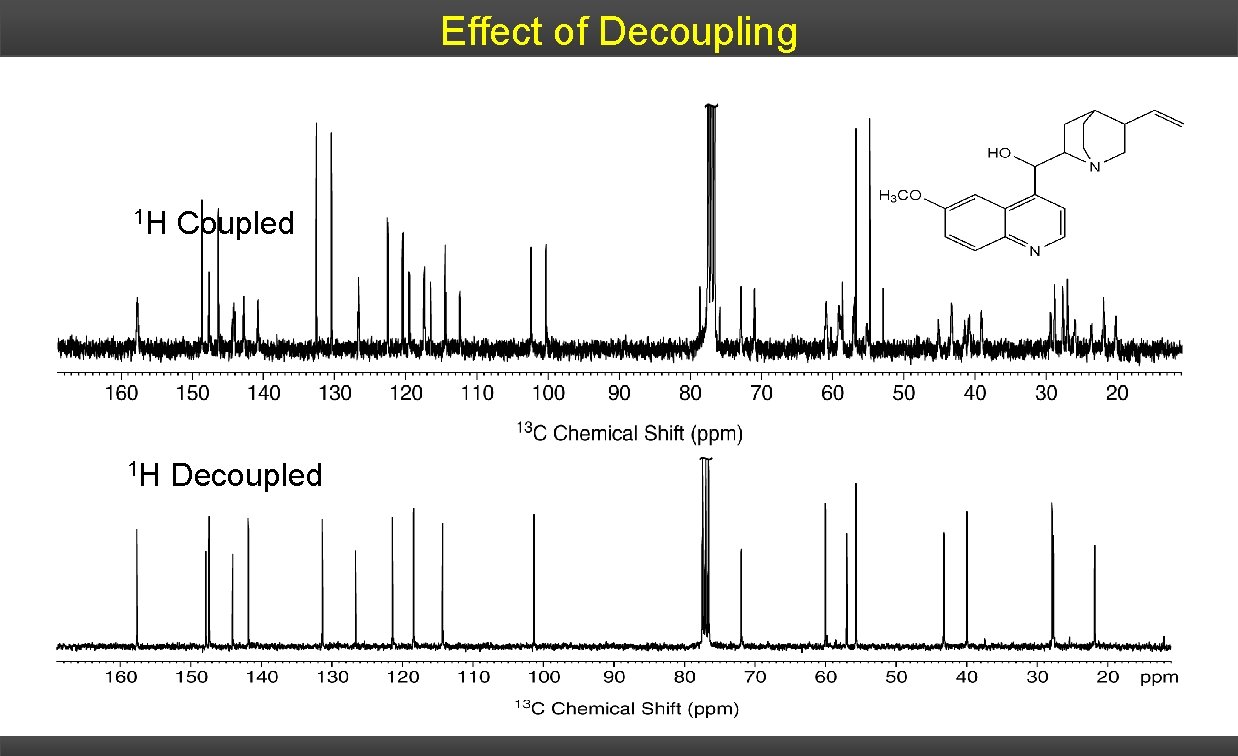
Effect of Decoupling 1 H Coupled 1 H Decoupled
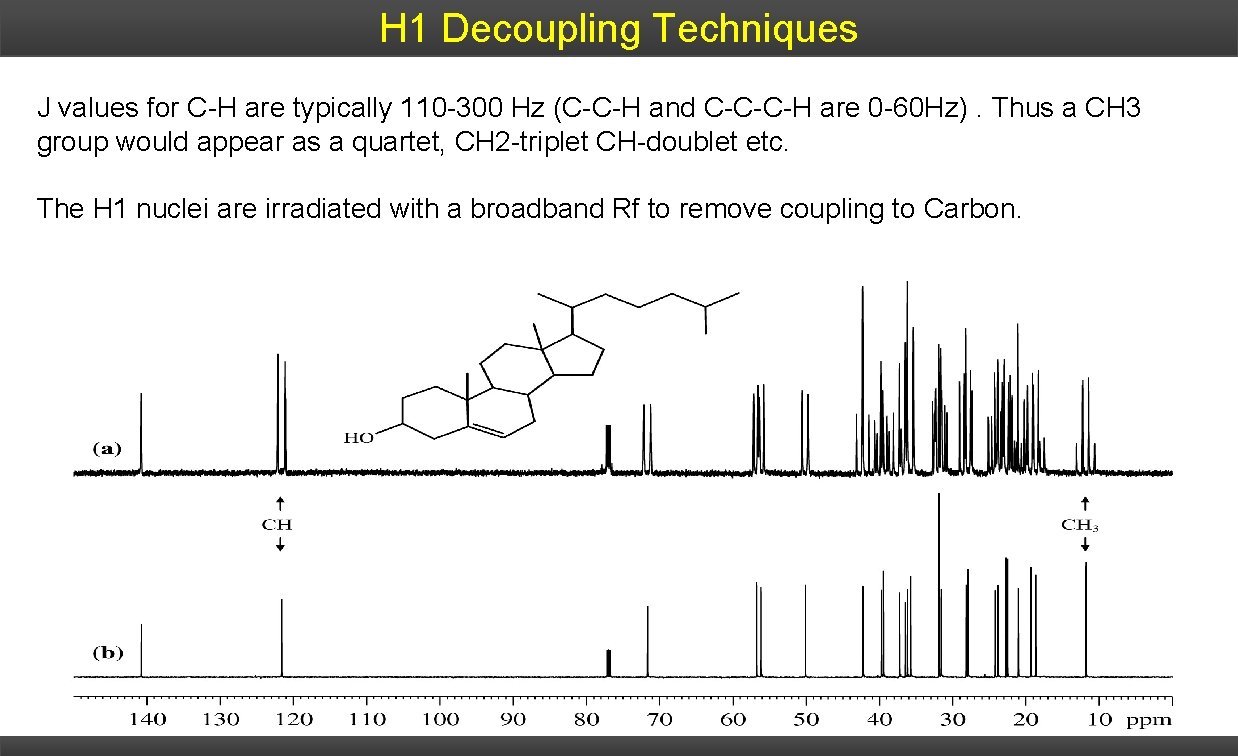
H 1 Decoupling Techniques J values for C-H are typically 110 -300 Hz (C-C-H and C-C-C-H are 0 -60 Hz). Thus a CH 3 group would appear as a quartet, CH 2 -triplet CH-doublet etc. The H 1 nuclei are irradiated with a broadband Rf to remove coupling to Carbon.
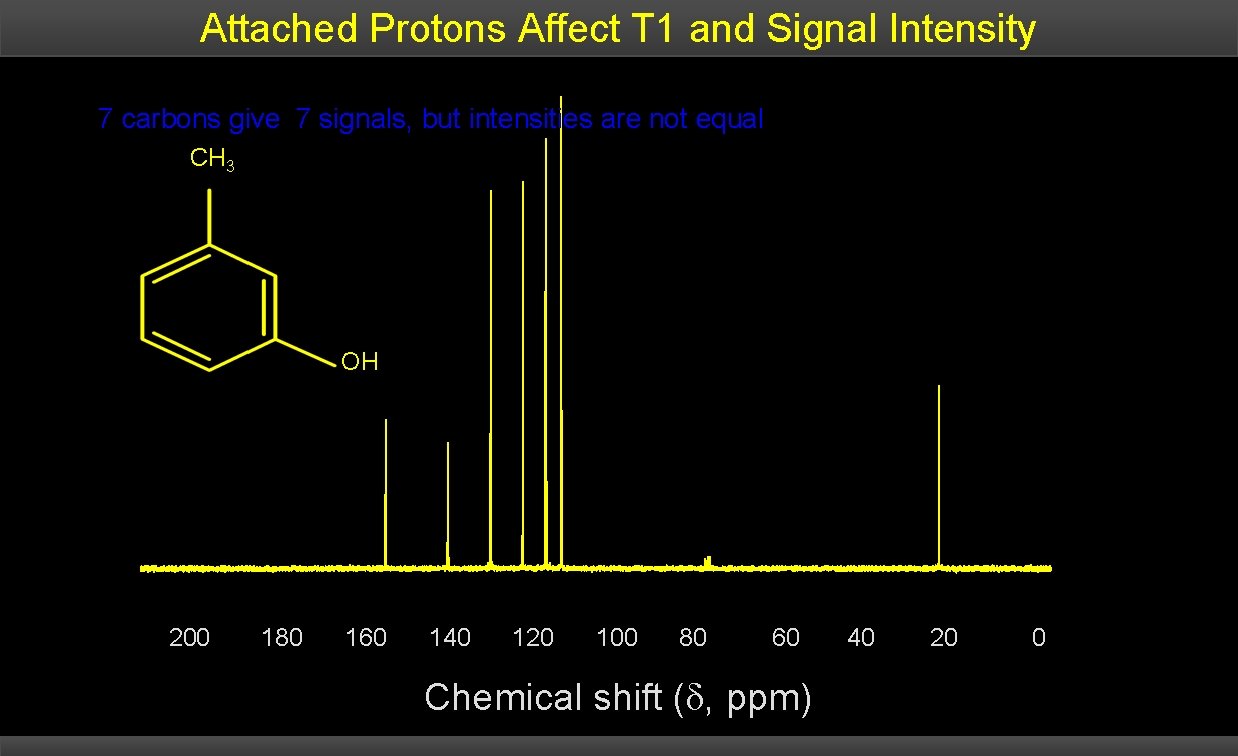
Attached Protons Affect T 1 and Signal Intensity 7 carbons give 7 signals, but intensities are not equal CH 3 OH 200 180 160 140 120 100 80 60 Chemical shift ( , ppm) 40 20 0
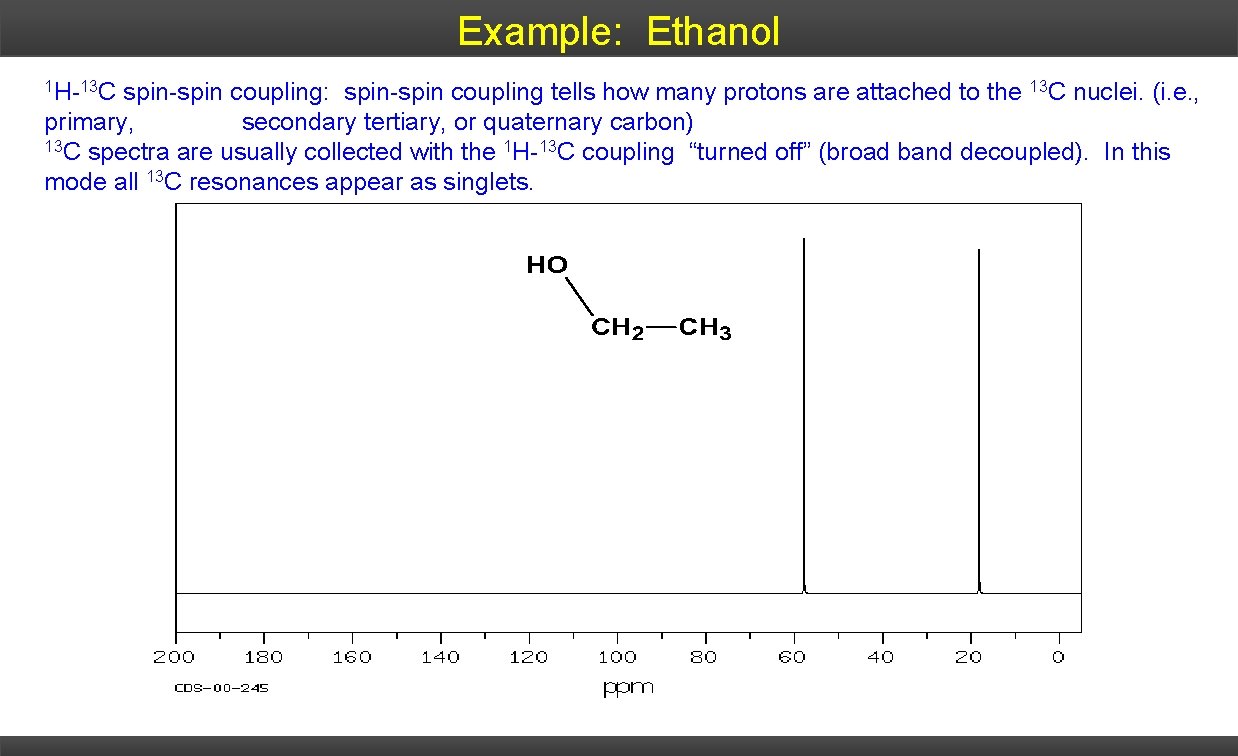
Example: Ethanol 1 H-13 C spin-spin coupling: spin-spin coupling tells how many protons are attached to the 13 C nuclei. (i. e. , primary, secondary tertiary, or quaternary carbon) 13 C spectra are usually collected with the 1 H-13 C coupling “turned off” (broad band decoupled). In this mode all 13 C resonances appear as singlets.
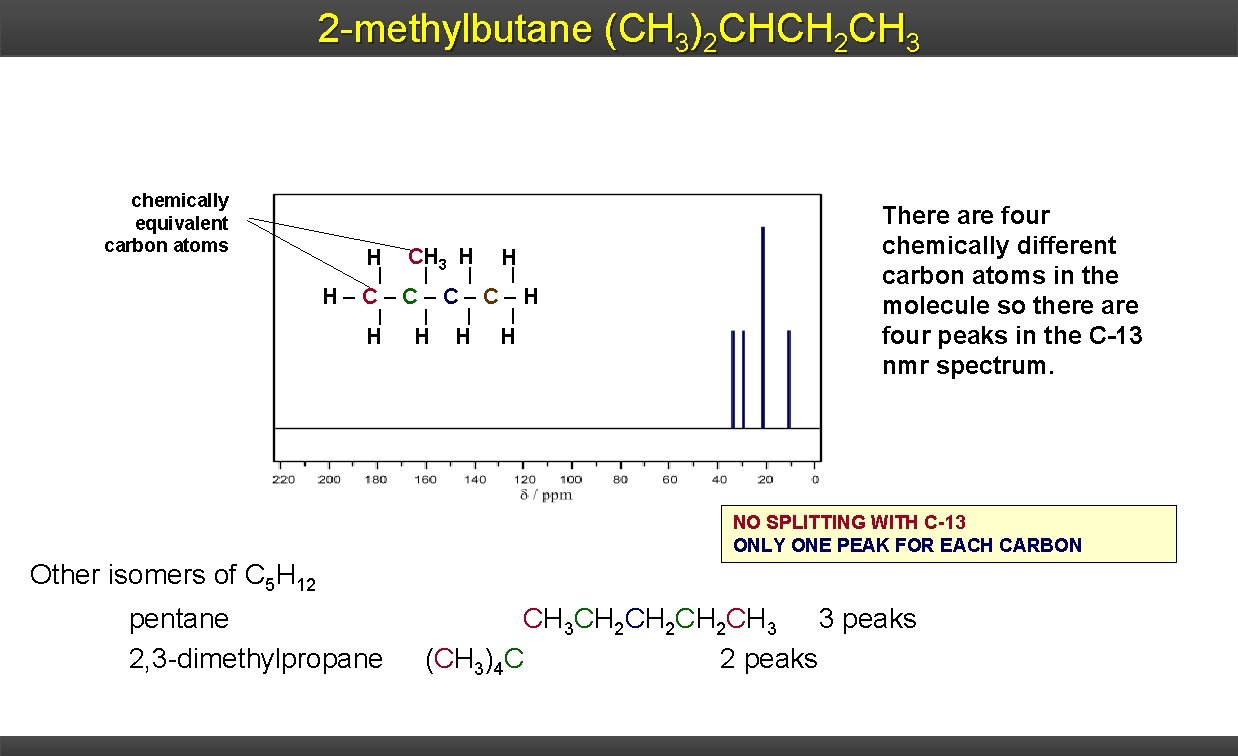
2 -methylbutane (CH 3)2 CHCH 2 CH 3 chemically equivalent carbon atoms H CH 3 H H H C C H H H There are four chemically different carbon atoms in the molecule so there are four peaks in the C-13 nmr spectrum. NO SPLITTING WITH C-13 ONLY ONE PEAK FOR EACH CARBON Other isomers of C 5 H 12 pentane 2, 3 -dimethylpropane CH 3 CH 2 CH 2 CH 3 3 peaks (CH 3)4 C 2 peaks
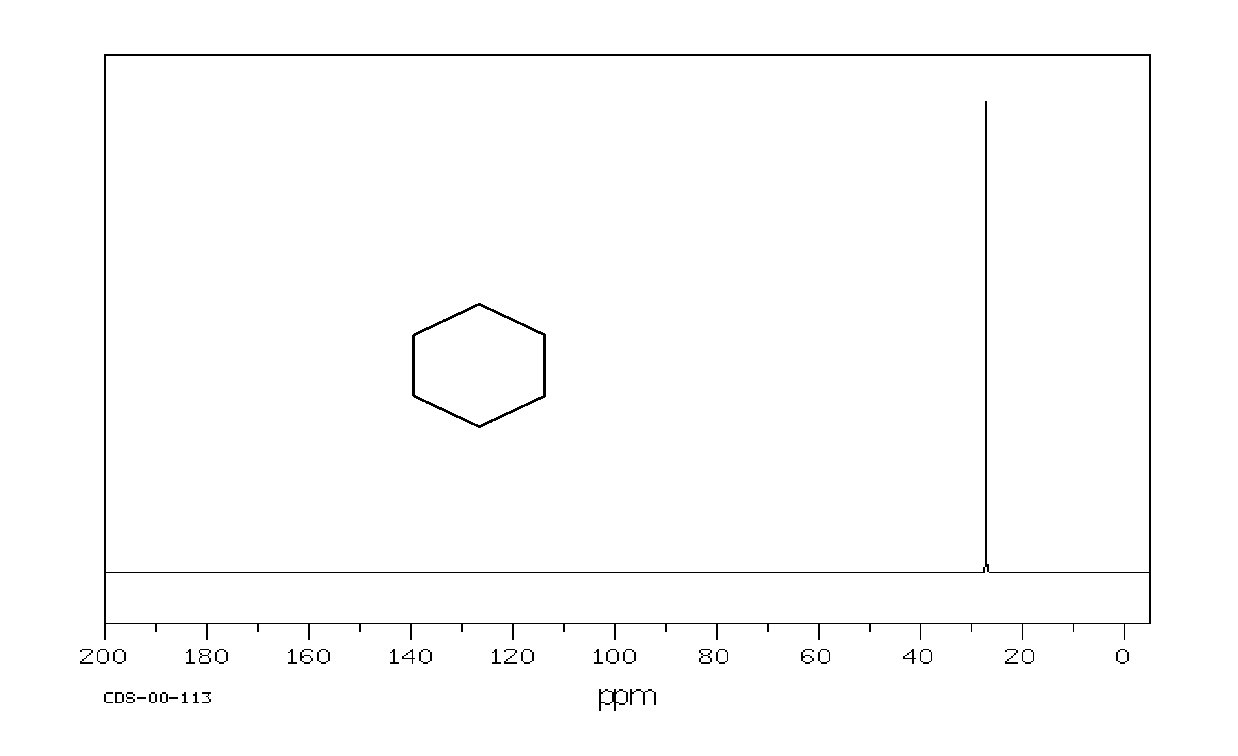
Example: cyclohexane
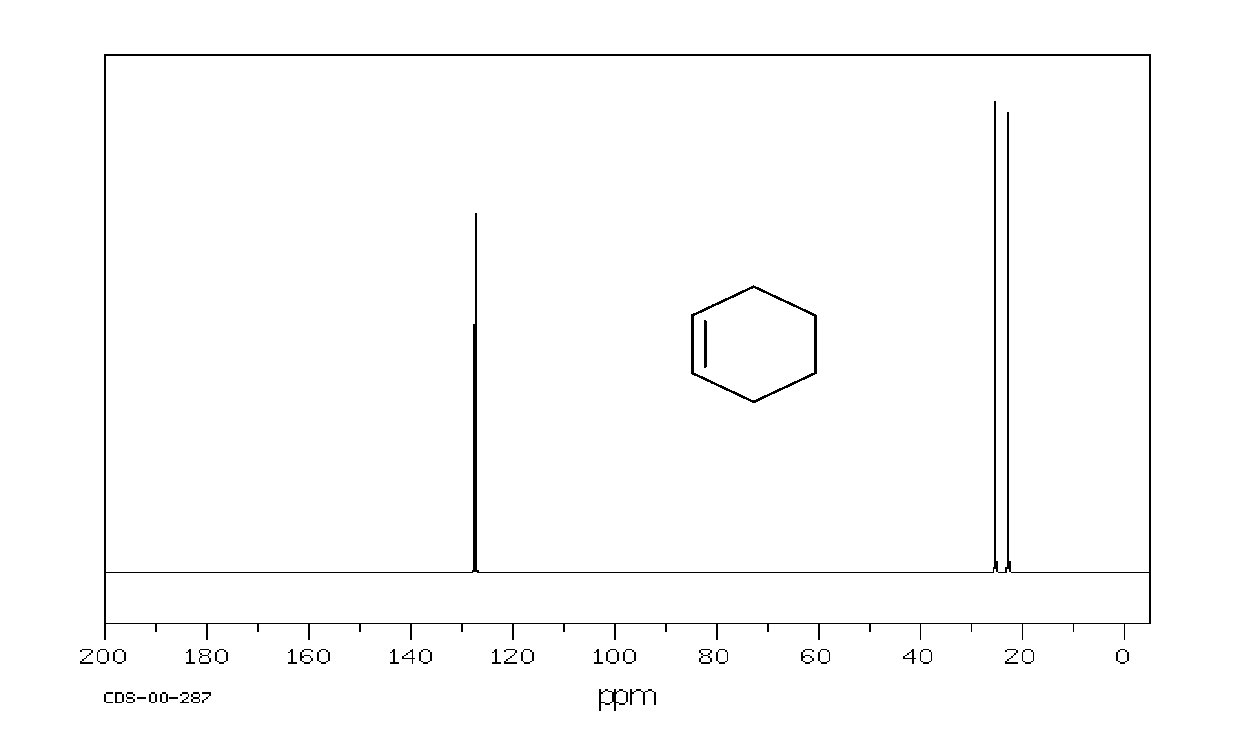
Example: cyclohexene
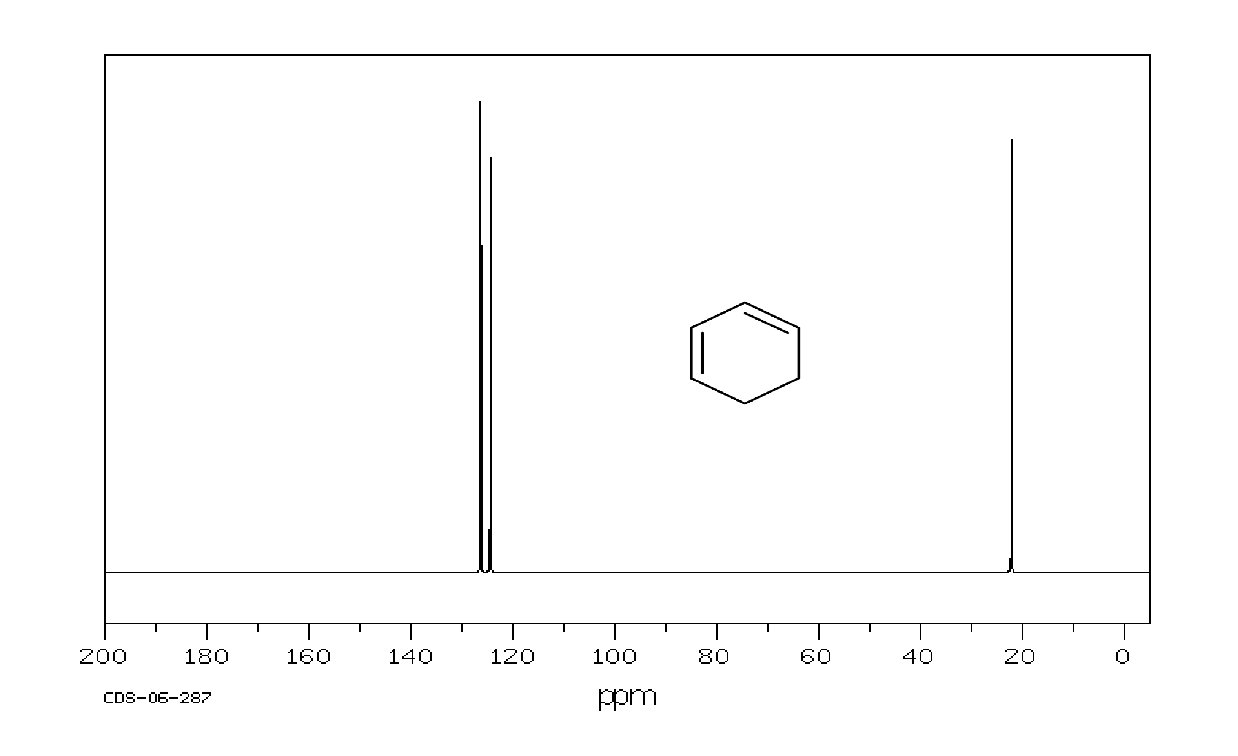
Example: 1, 3 -cyclohexadiene
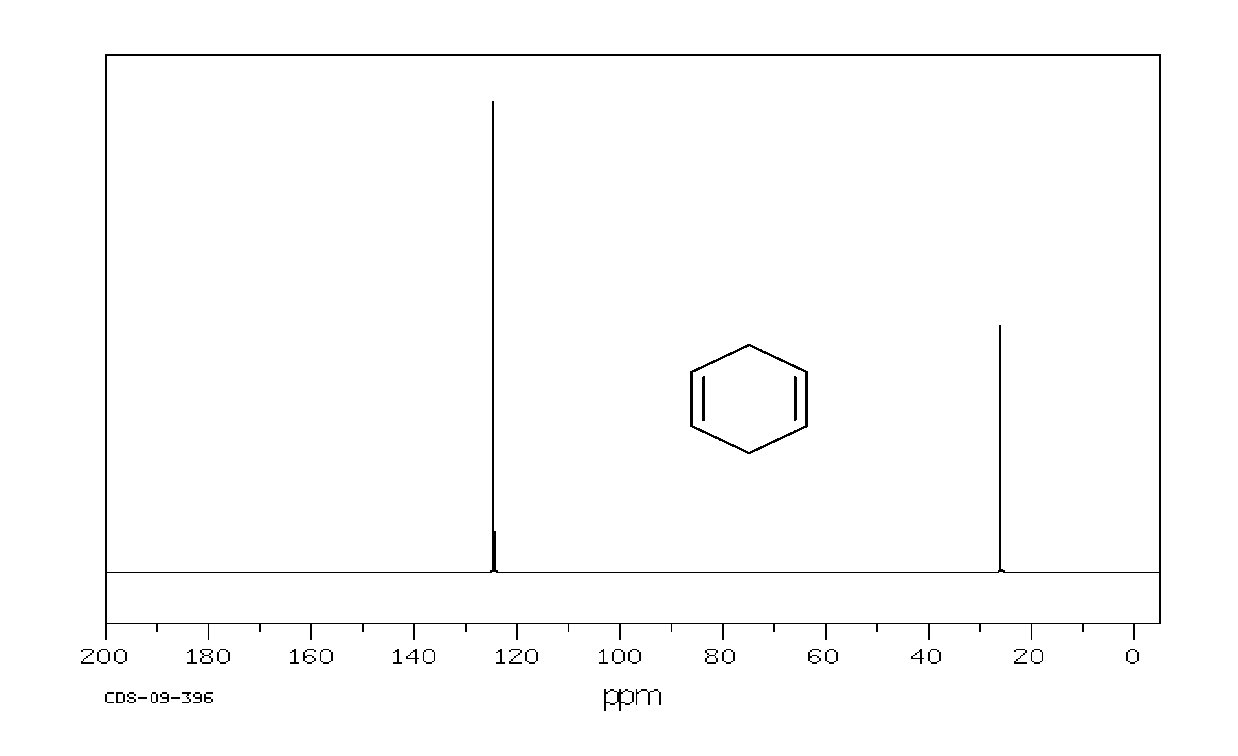
Example: 1, 4 -cyclohexadiene
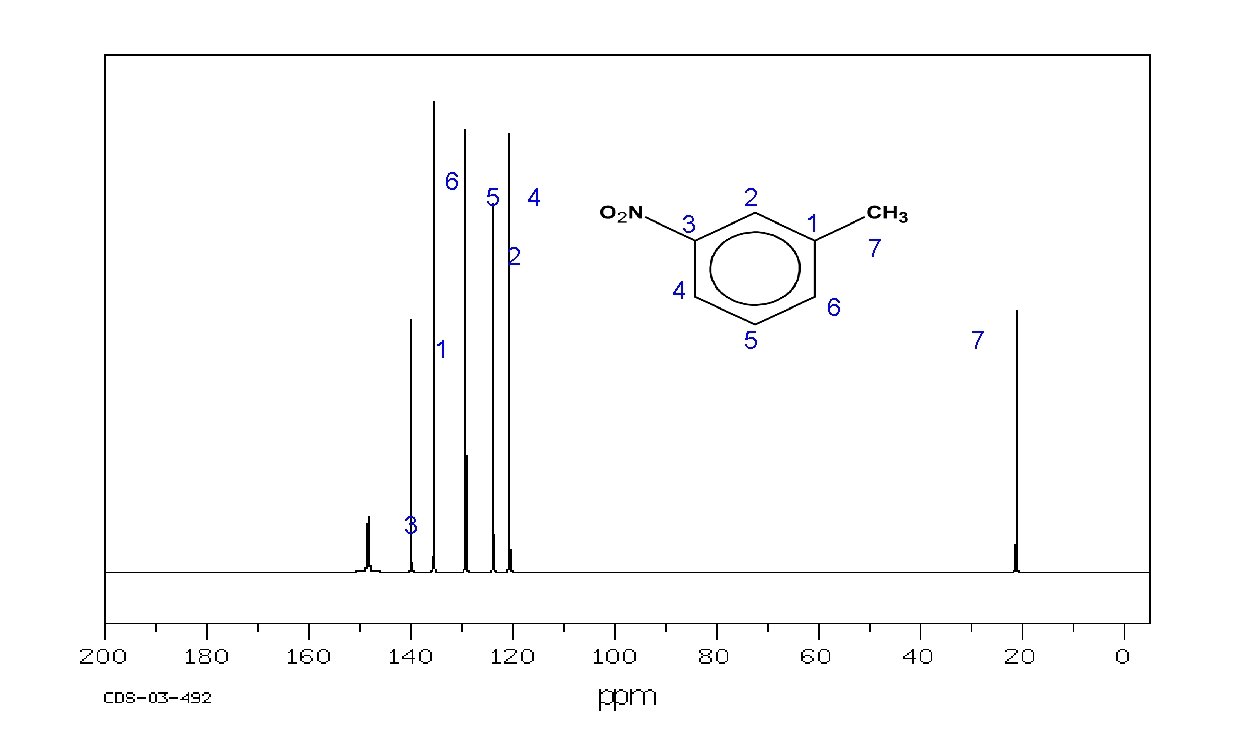
Example: m-nitrotoluene 6 5 4 3 2 1 7 2 4 1 3 6 5 7
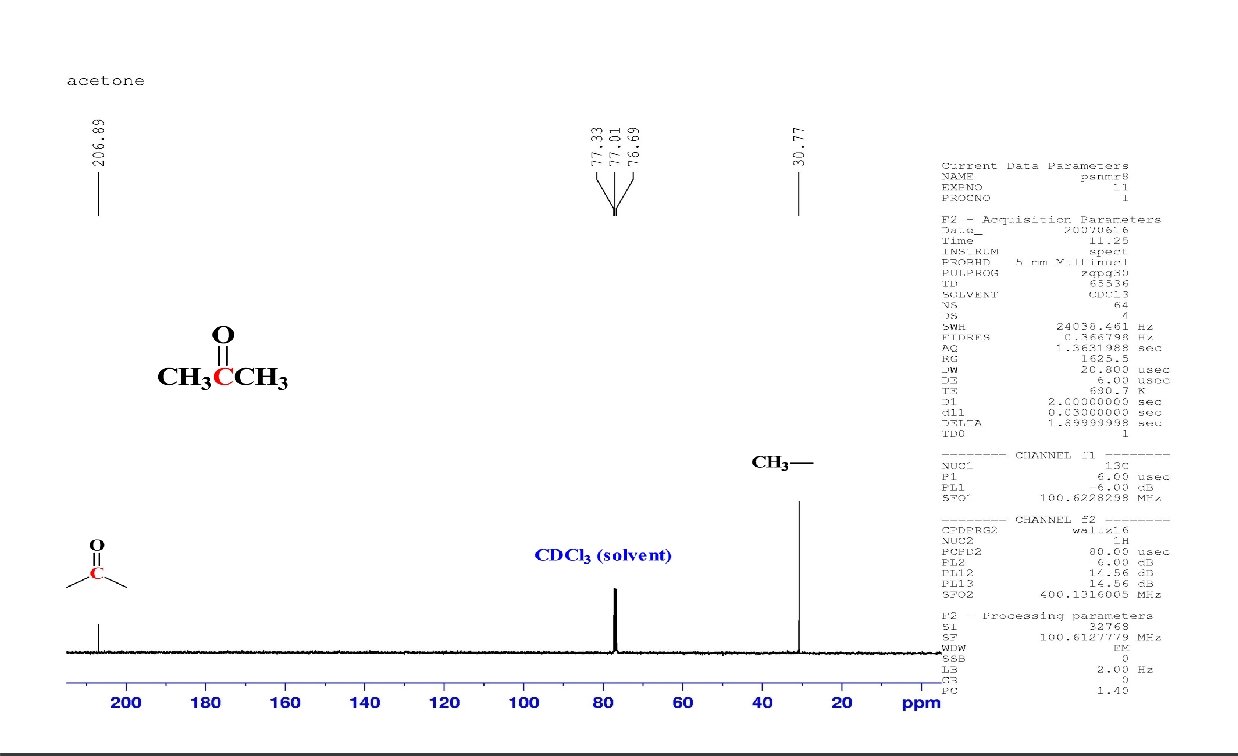
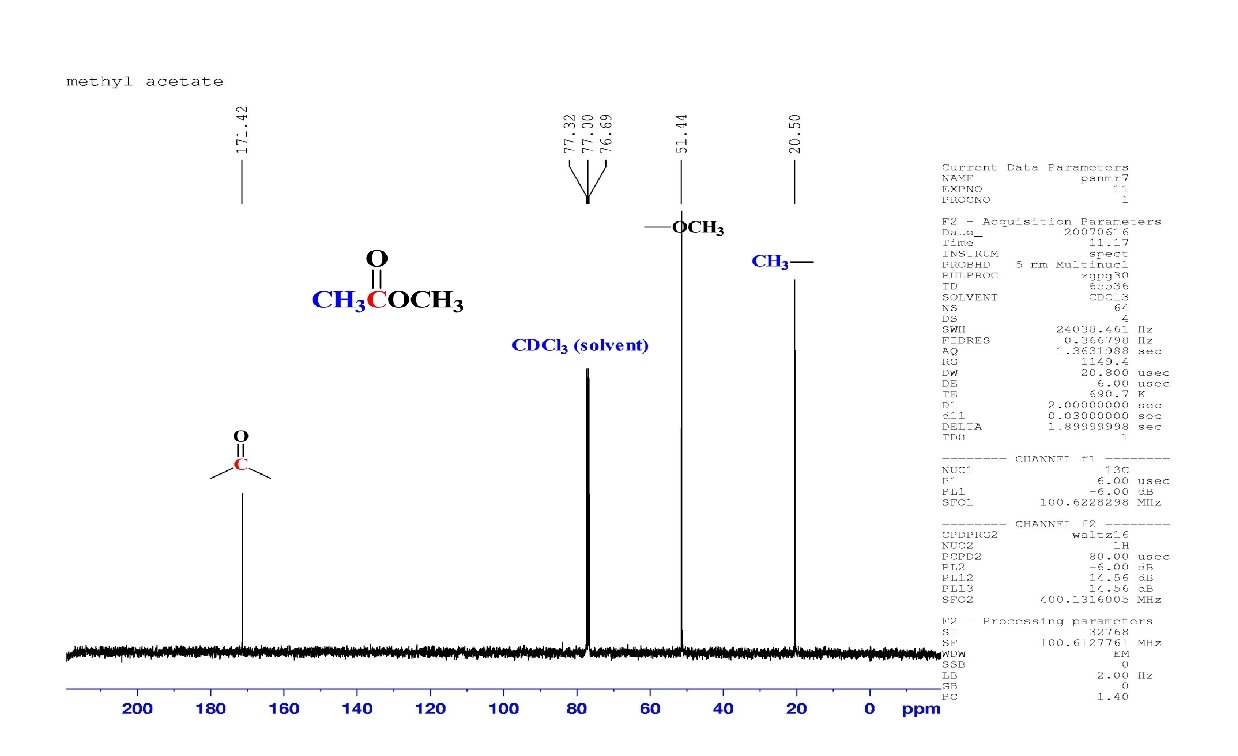
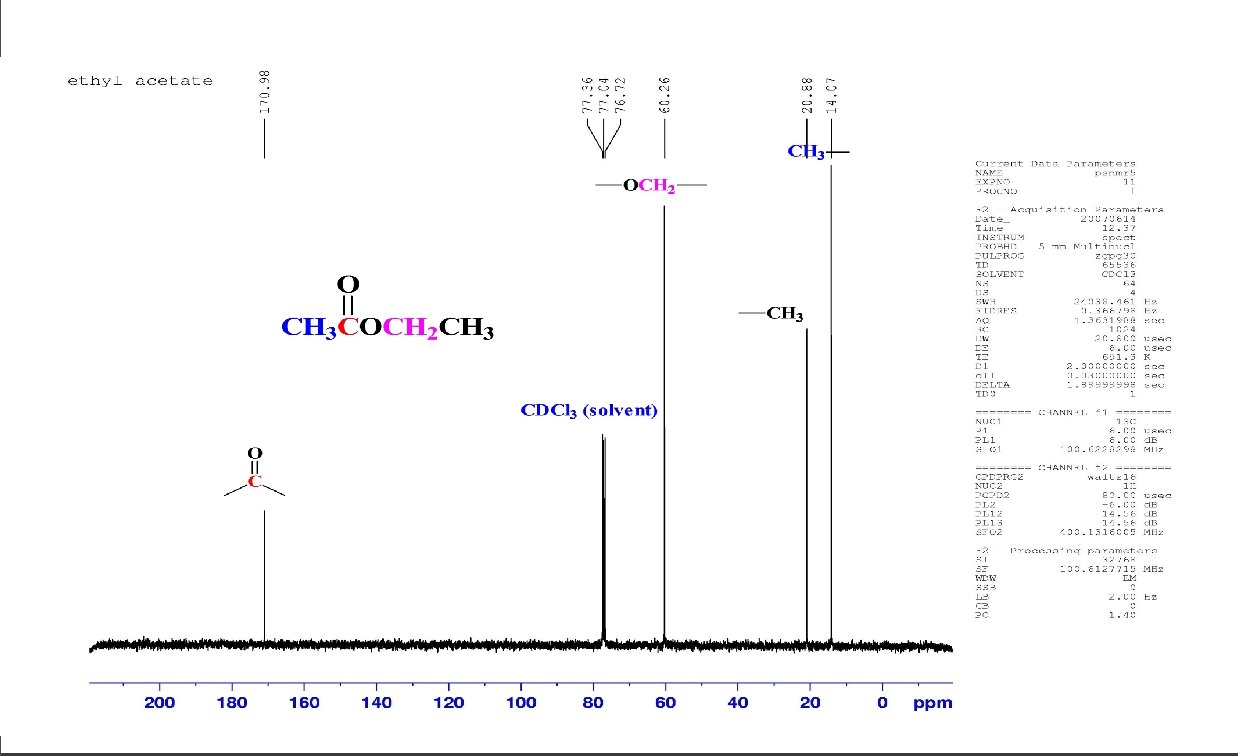
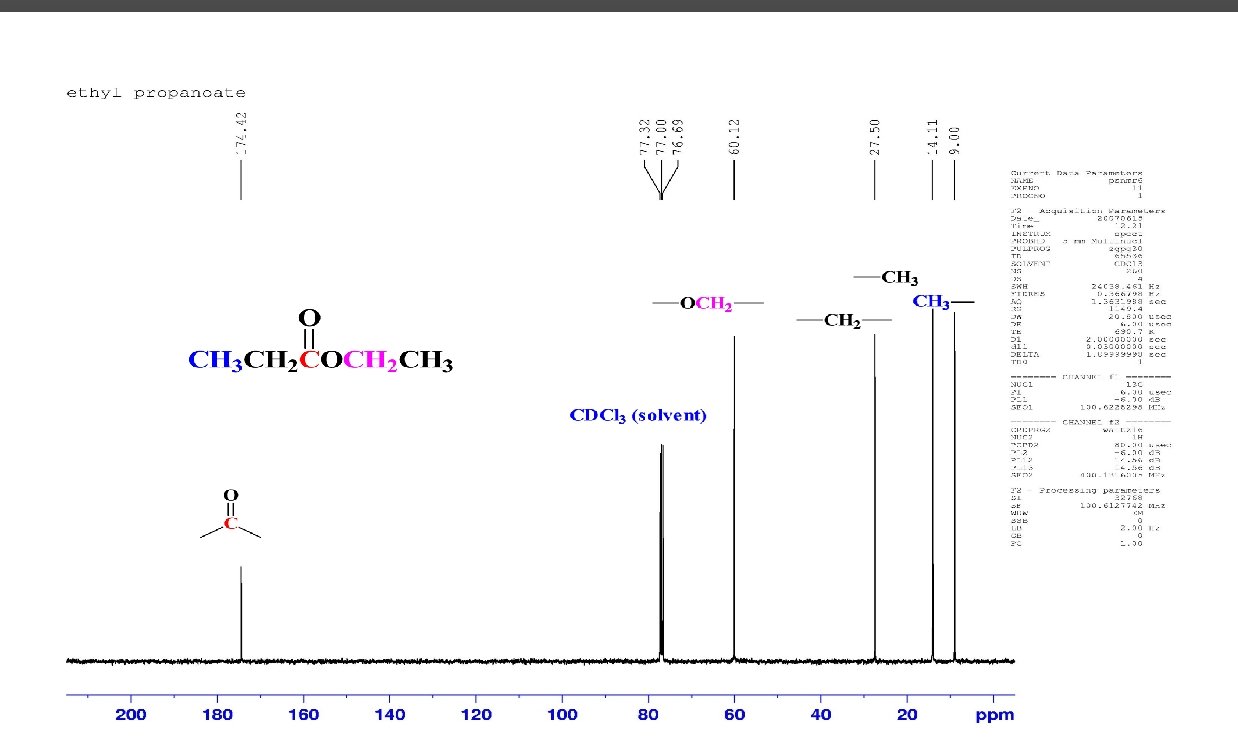
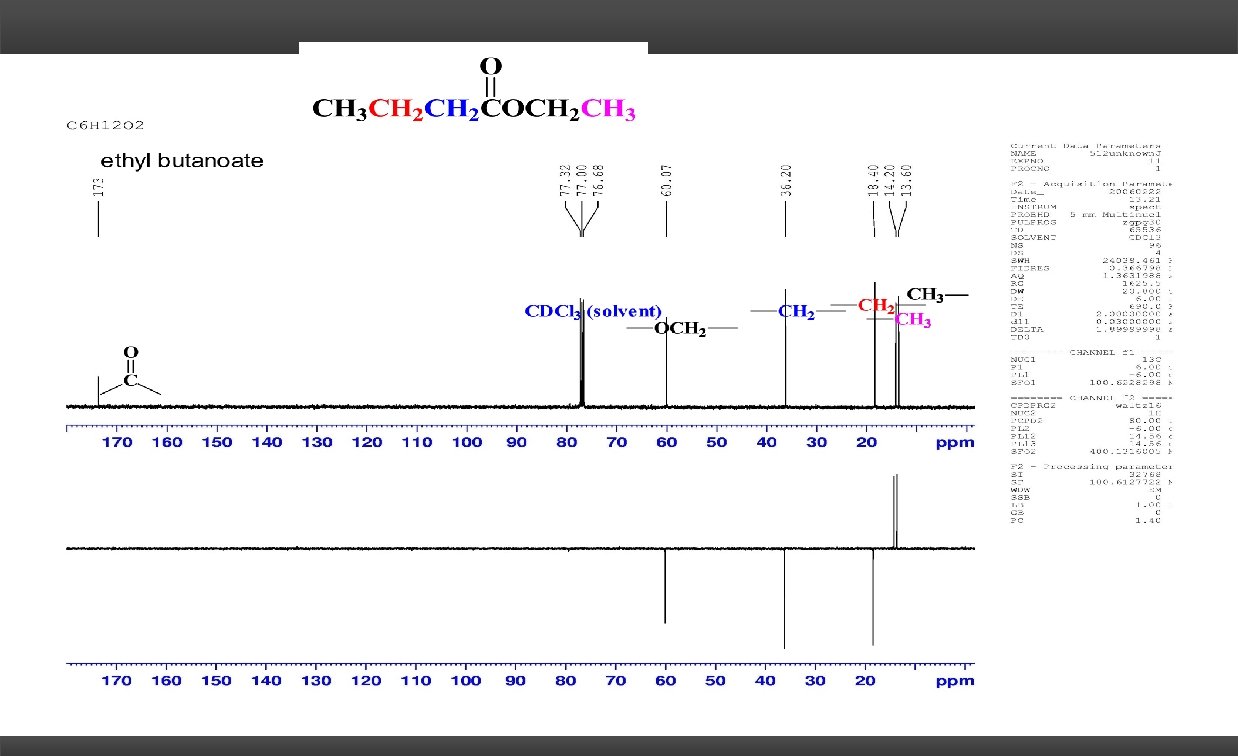
C 6 H 12 O 2
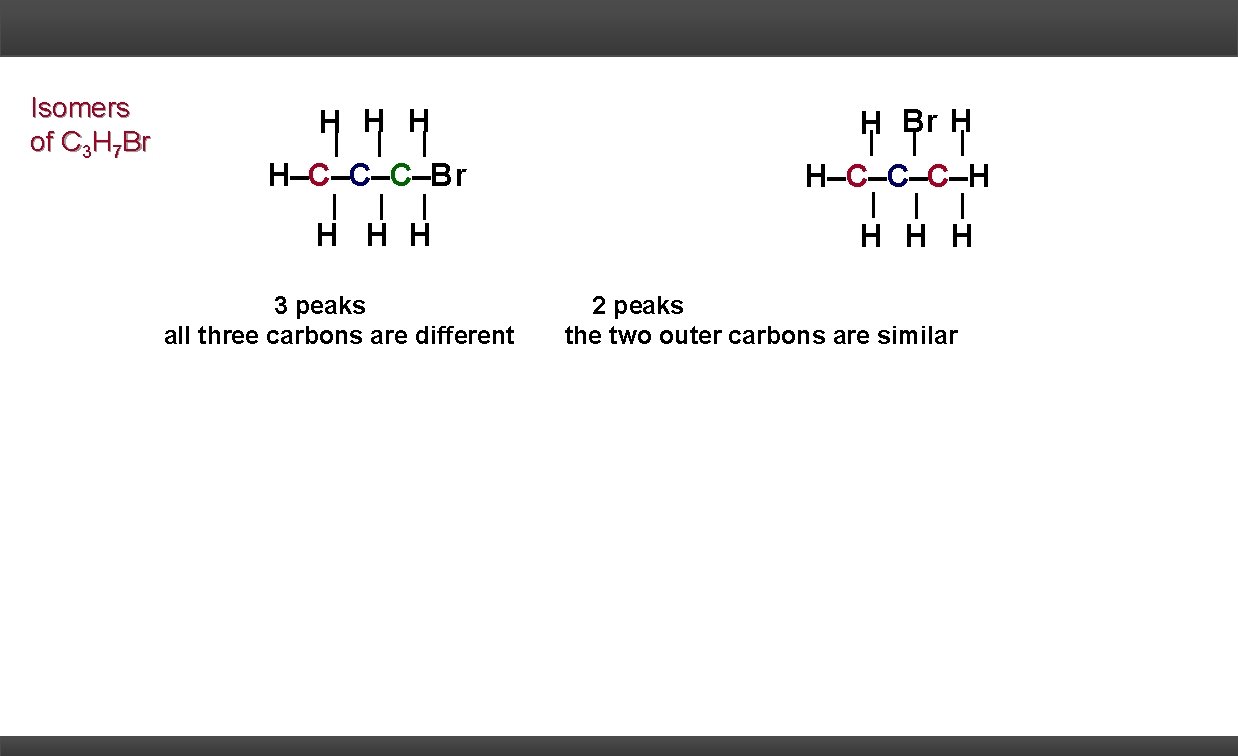
Isomers of C 3 H 7 Br H H C C C Br H H H 3 peaks all three carbons are different H Br H H C C C H H 2 peaks the two outer carbons are similar
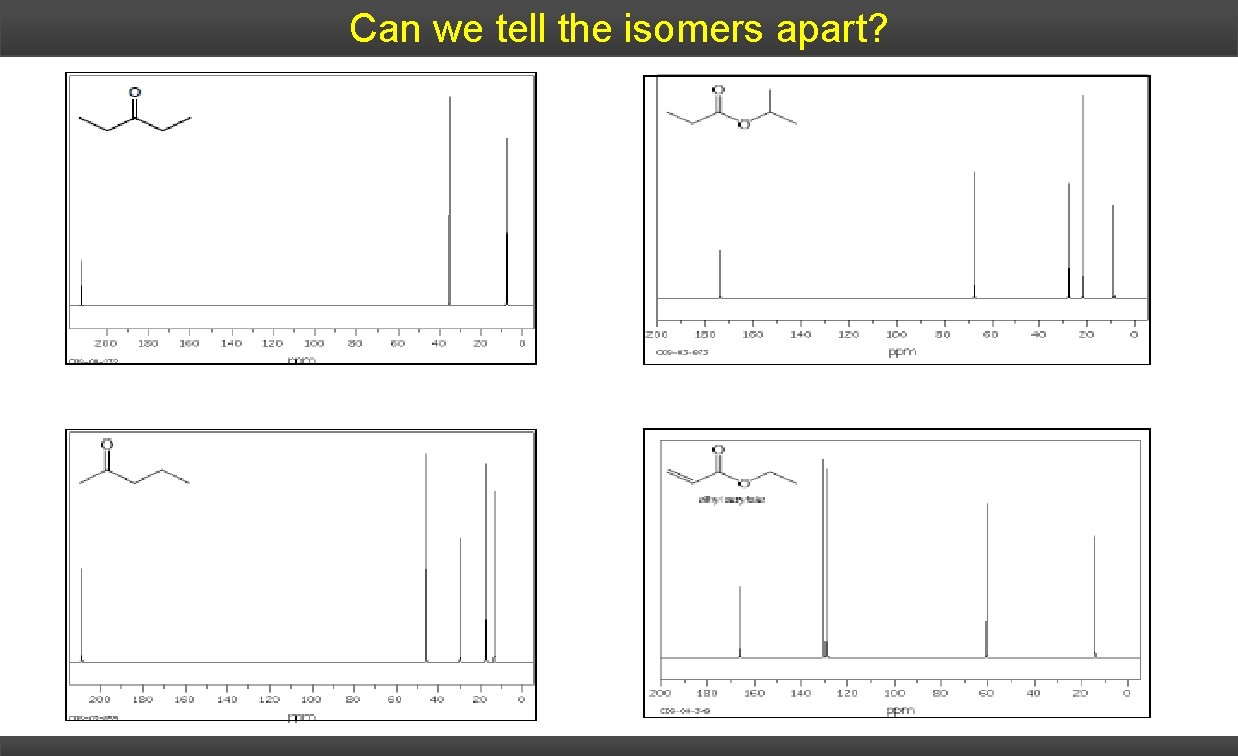
Can we tell the isomers apart?
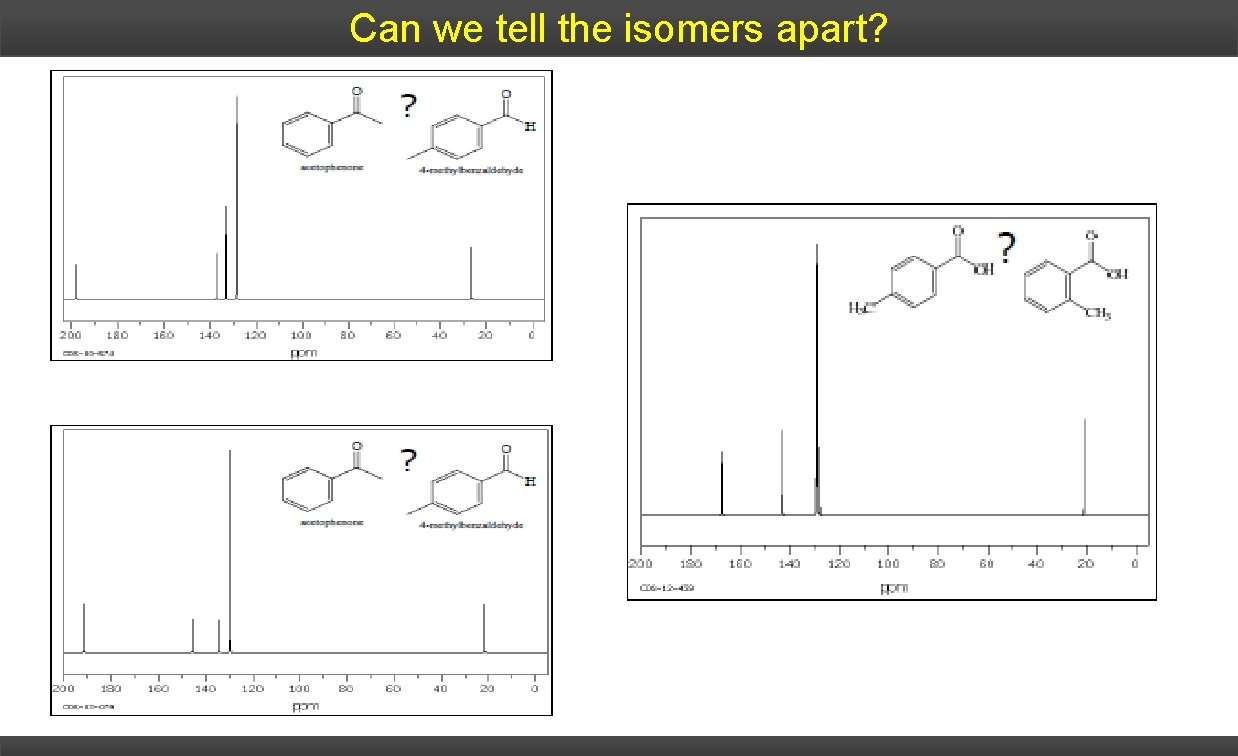
Can we tell the isomers apart?

Edited C 13 NMRs APT= Attached Proton Test “APT” DEPT= Distortionless Enhancement by Polarization Transfer
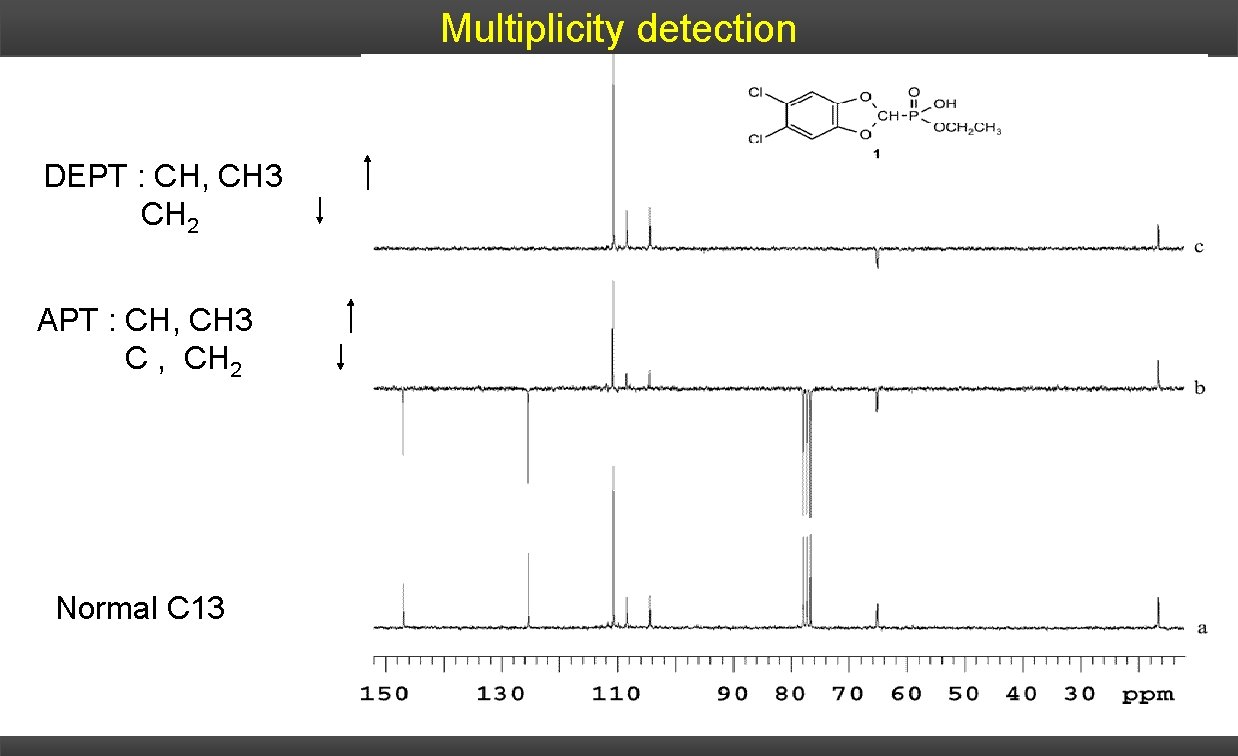
Multiplicity detection DEPT : CH, CH 3 CH 2 APT : CH, CH 3 C , CH 2 Normal C 13

Attached Proton Test “APT” Acquiring a 13 C after 1/J seconds: methine and methyl C’s produce negative peaks (odd number of attached H’s) methylene and quaternary C’s produce positive peaks (even number of attached H’s) Remember 1 JCH is essentially the same for all tetrahedral carbons Thus acquiring the C signal after a pre-determined time can give positive peaks, negative peaks, or even no peaks at all, depending on how many H’s are attached. APT has been superseded by DEPT Distortionless Enhancement by Polarization Transfer Complex pulse sequence allowing selective reception of signals from different C types: -C, -CH 2, CH 3 DEPT spectra (Distortionless Enhancement by Polarization Transfer) a modern allows you to determine the number of attached hydrogens. 13 C NMR spectra that

Measuring a 13 C NMR spectrum involves 1 H In DEPT, a second transmitter irradiates during the sequence, which affects the appearance of the 13 C spectrum. some 13 C signals stay the same 13 some C signals disappear 13 some C signals are inverted

13 C NMR - DEPT Distortionless enhancement by polarization transfer (DEPT) spectra permit identification of CH 3, CH 2, and CH carbon atoms. o 1, o 2 , and o 3 DEPT 45 shows carbons. DEPT 90 shows only 3 o carbons. DEPT 135 shows 1 o and 3 o carbons as positive peaks and 2 o carbons as negative peaks.

Using DEPT to Count Hydrogens Attached to 13 C
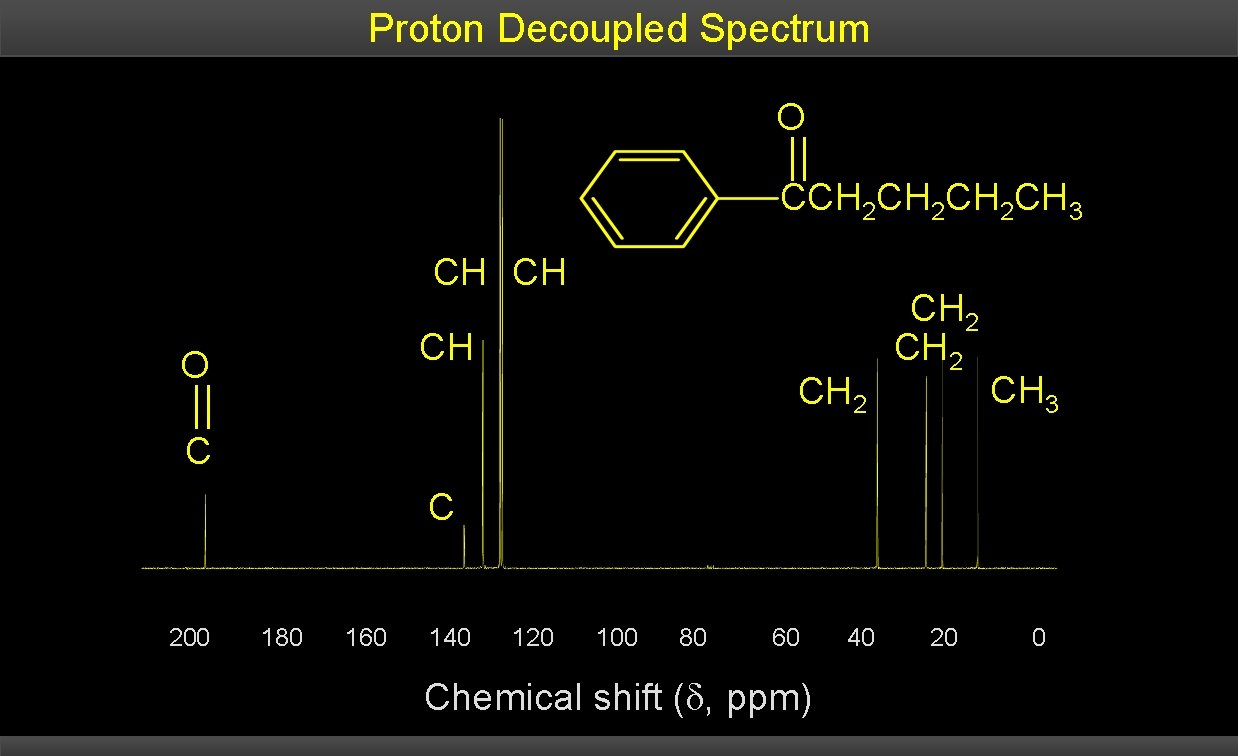
Proton Decoupled Spectrum O CCH 2 CH 2 CH 3 CH CH CH O CH 2 CH 3 C C 200 180 160 140 120 100 80 60 Chemical shift ( , ppm) 40 20 0
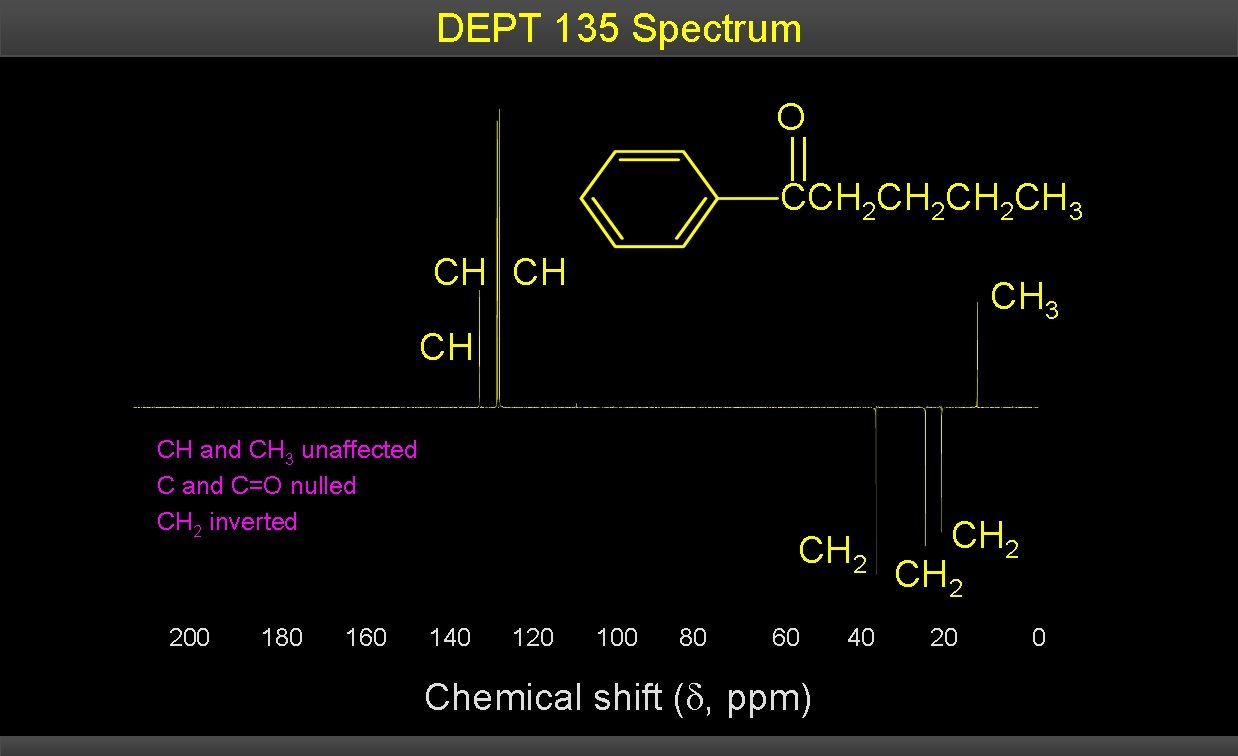
DEPT 135 Spectrum O CCH 2 CH 2 CH 3 CH CH and CH 3 unaffected C and C=O nulled CH 2 inverted 200 180 160 CH 2 140 120 100 80 60 Chemical shift ( , ppm) 40 CH 2 20 0
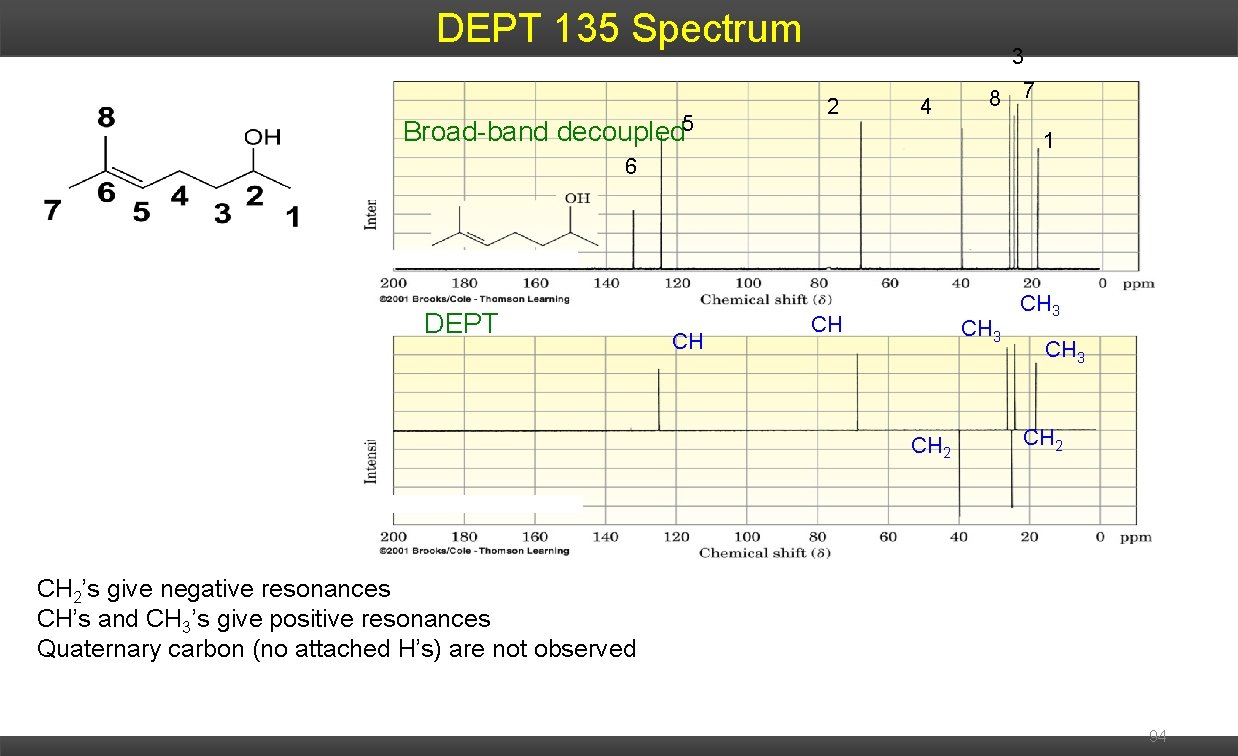
DEPT 135 Spectrum Broad-band decoupled 5 3 2 4 8 7 1 6 DEPT CH CH CH 3 CH 2’s give negative resonances CH’s and CH 3’s give positive resonances Quaternary carbon (no attached H’s) are not observed 94
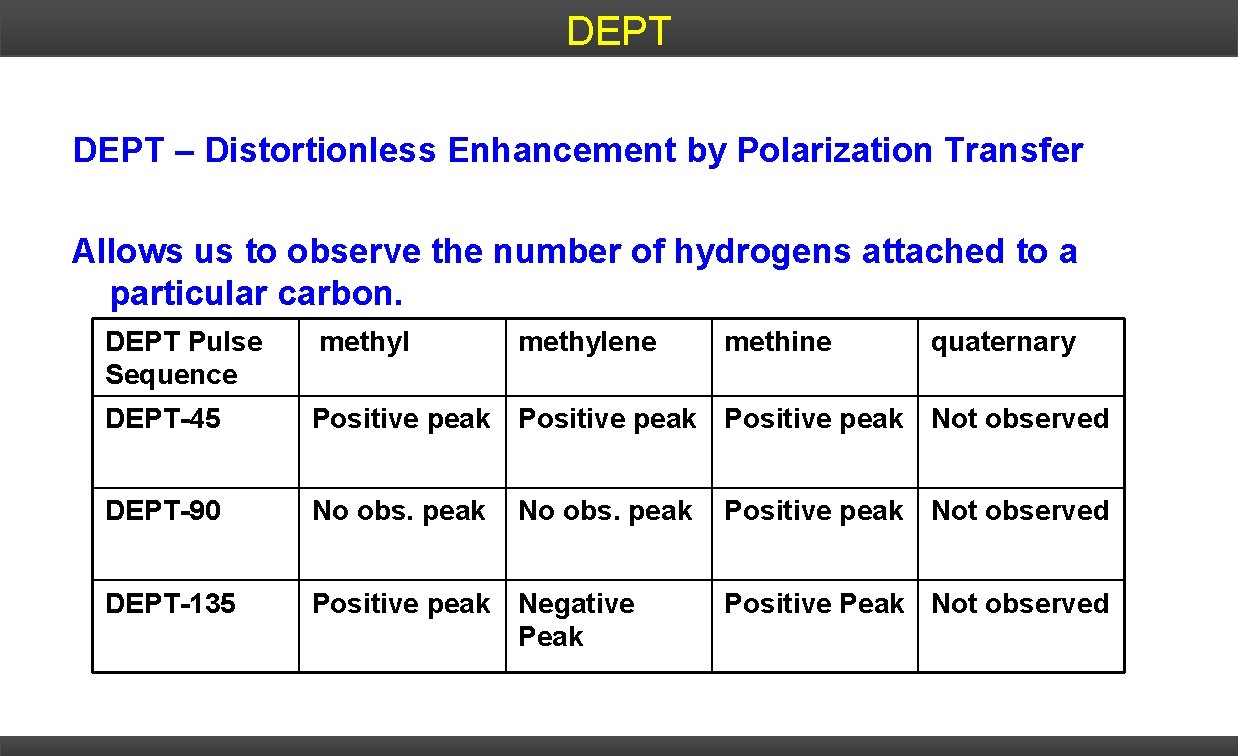
DEPT – Distortionless Enhancement by Polarization Transfer Allows us to observe the number of hydrogens attached to a particular carbon. DEPT Pulse Sequence methylene DEPT-45 Positive peak Not observed DEPT-90 No obs. peak DEPT-135 Positive peak Negative Peak No obs. peak methine quaternary Positive peak Not observed Positive Peak Not observed
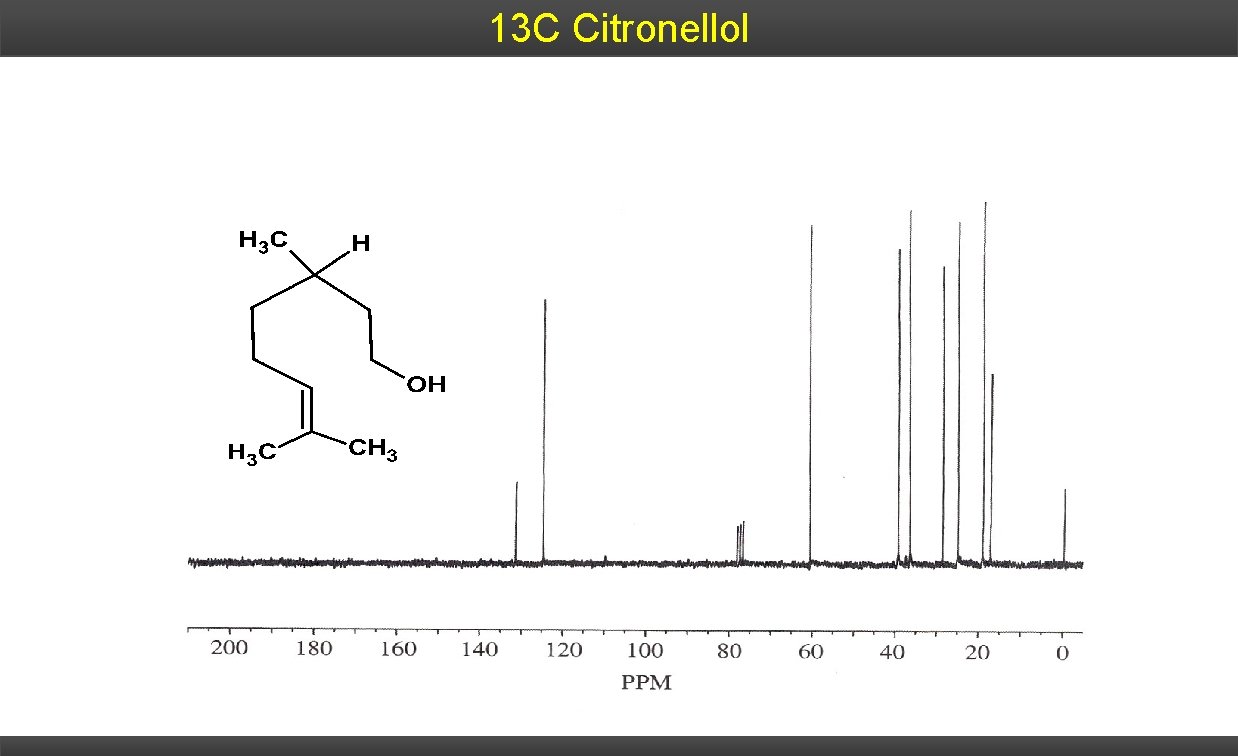
13 C Citronellol
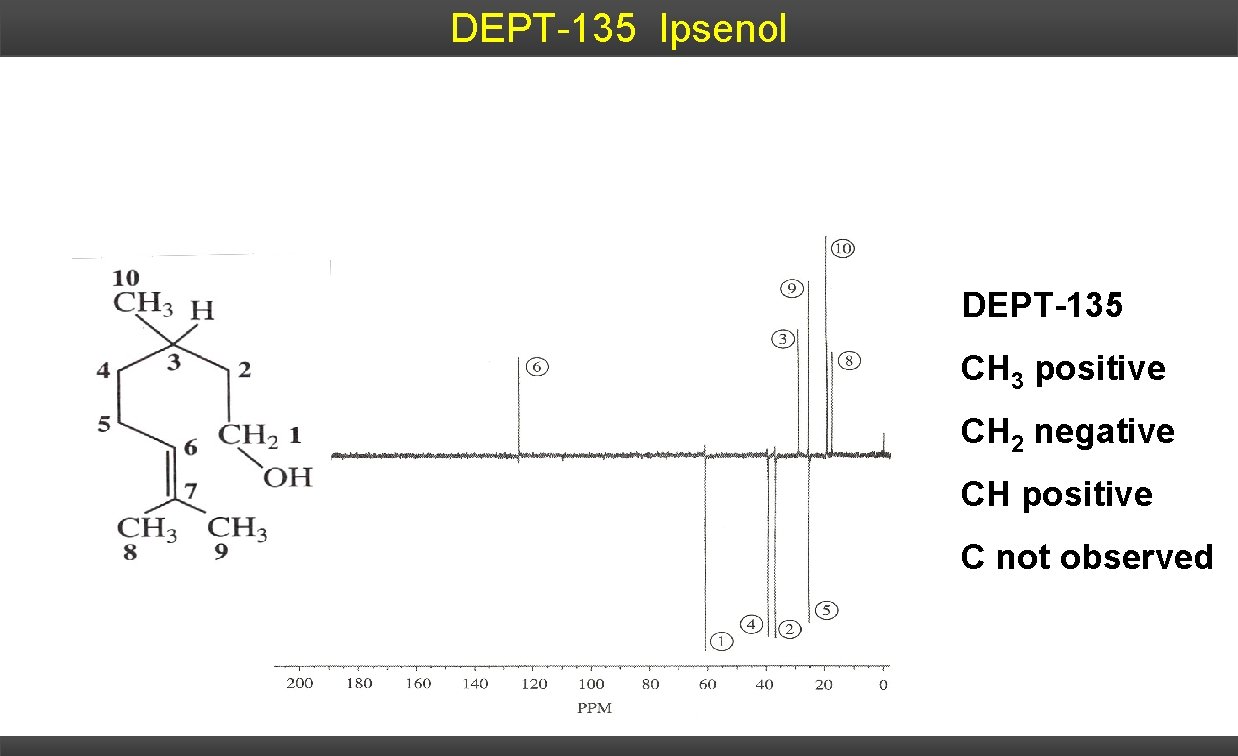
DEPT-135 Ipsenol DEPT-135 CH 3 positive CH 2 negative CH positive C not observed
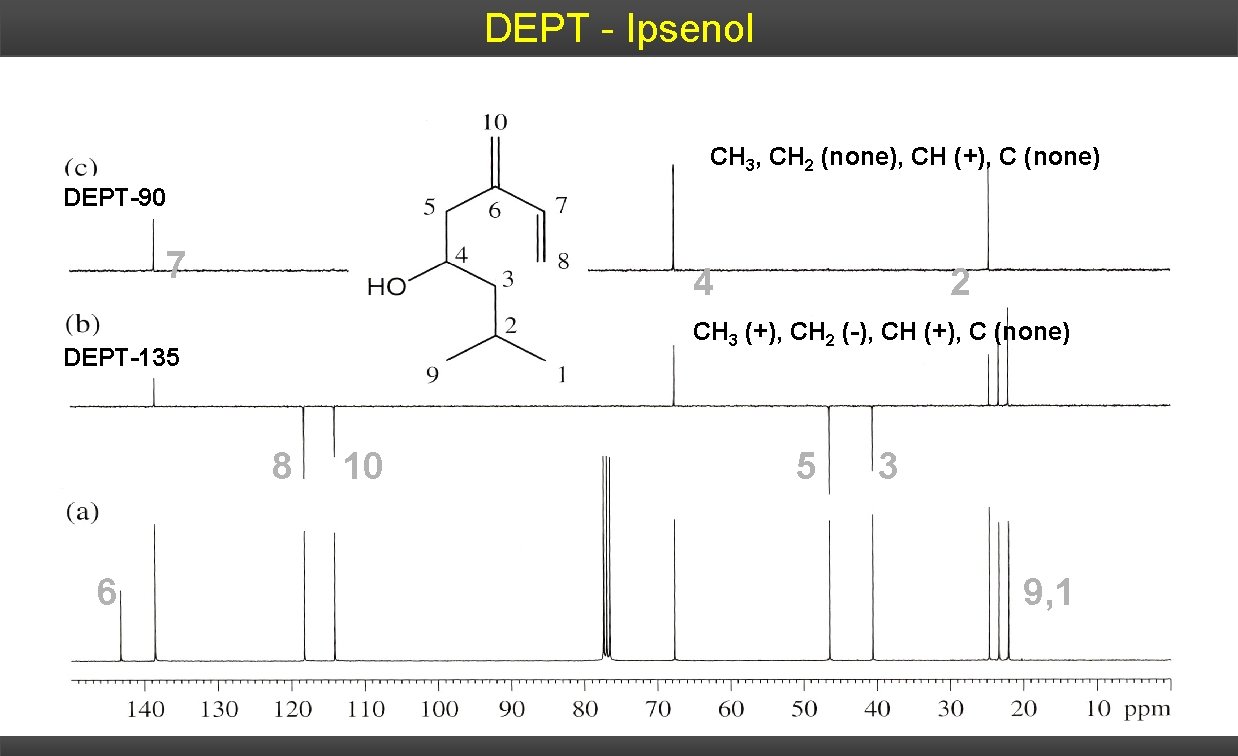
DEPT - Ipsenol CH 3, CH 2 (none), CH (+), C (none) DEPT-90 7 4 CH 3 (+), CH 2 (-), CH (+), C (none) DEPT-135 8 6 2 10 5 3 9, 1
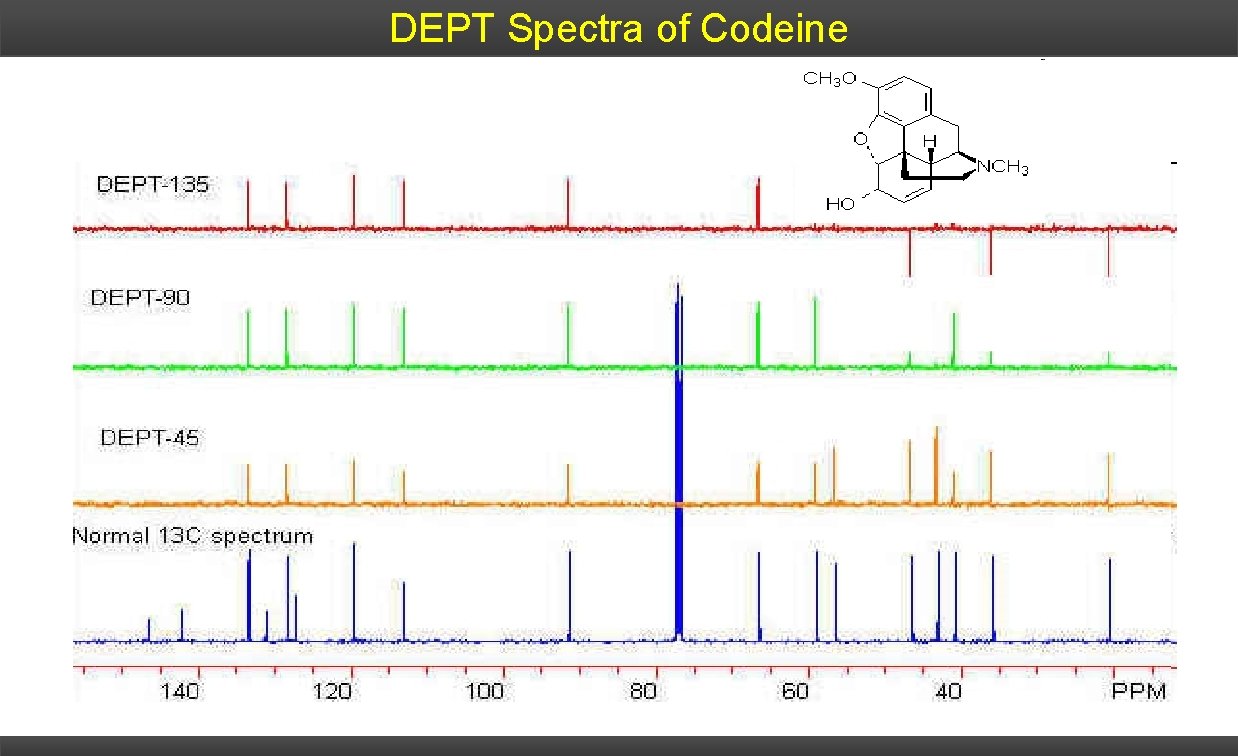
DEPT Spectra of Codeine
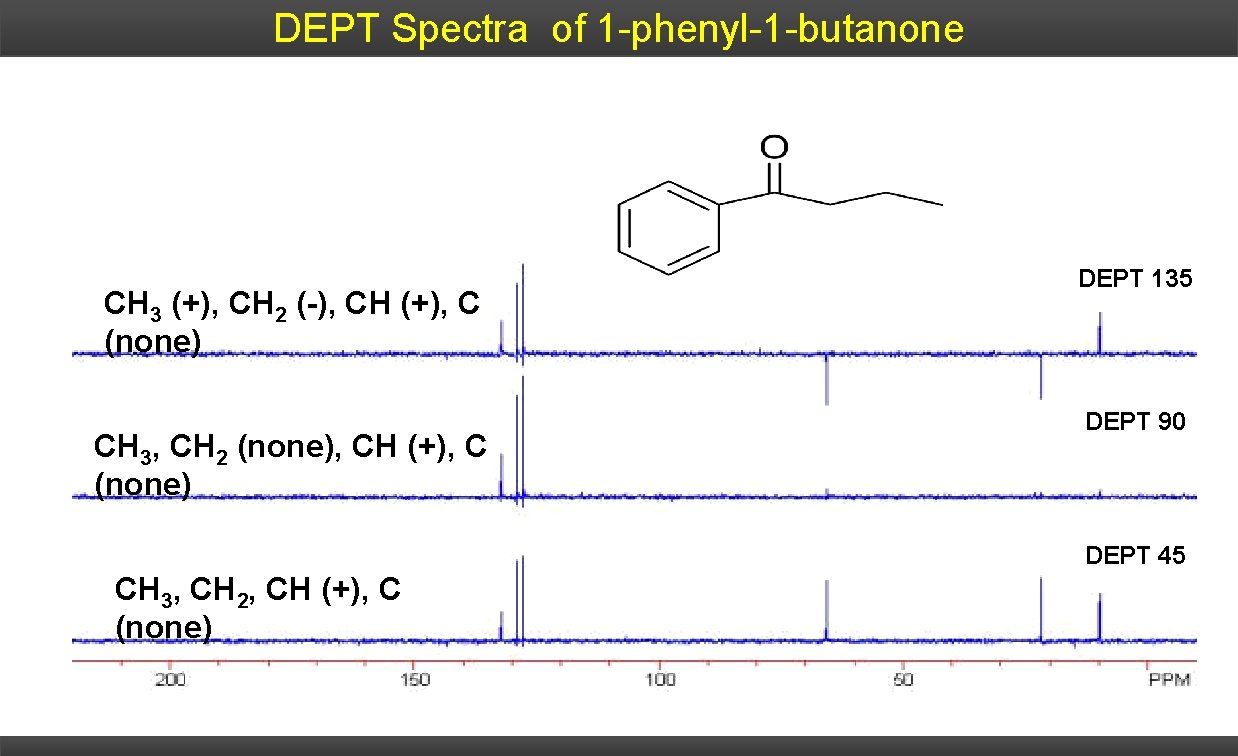
DEPT Spectra of 1 -phenyl-1 -butanone CH 3 (+), CH 2 (-), CH (+), C (none) CH 3, CH 2 (none), CH (+), C (none) DEPT 135 DEPT 90 DEPT 45 CH 3, CH 2, CH (+), C (none)
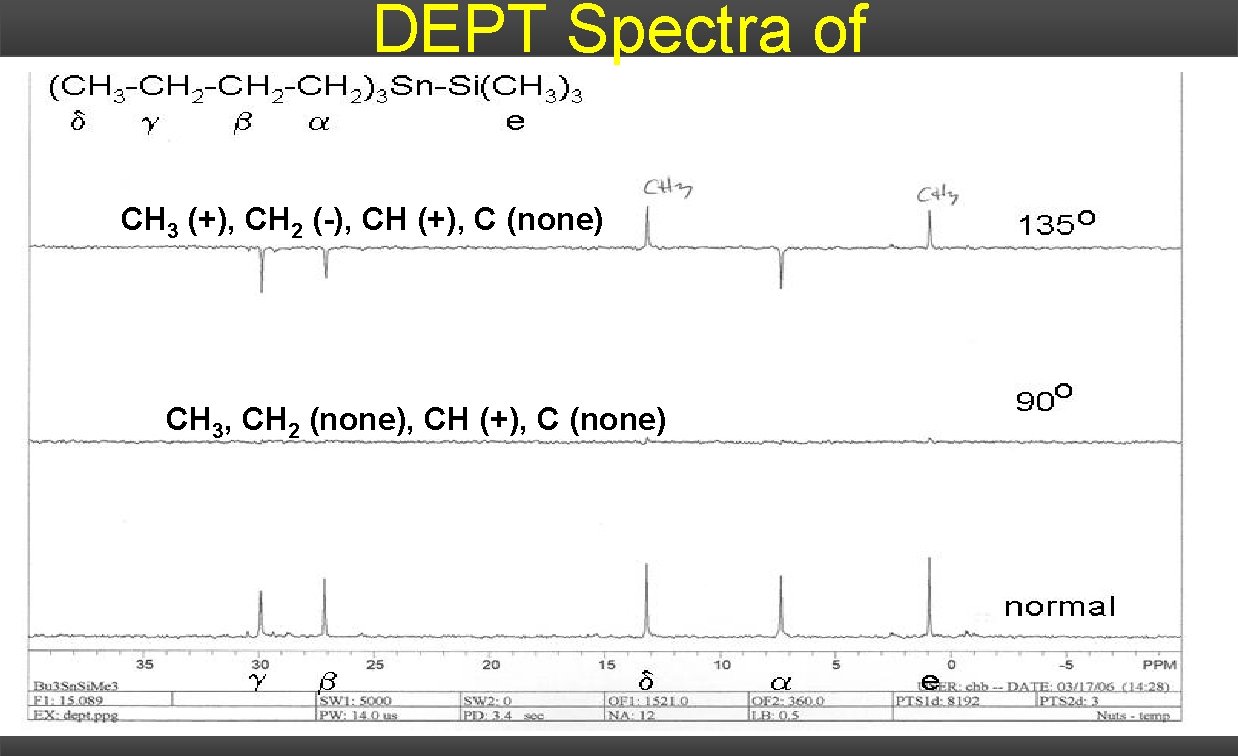
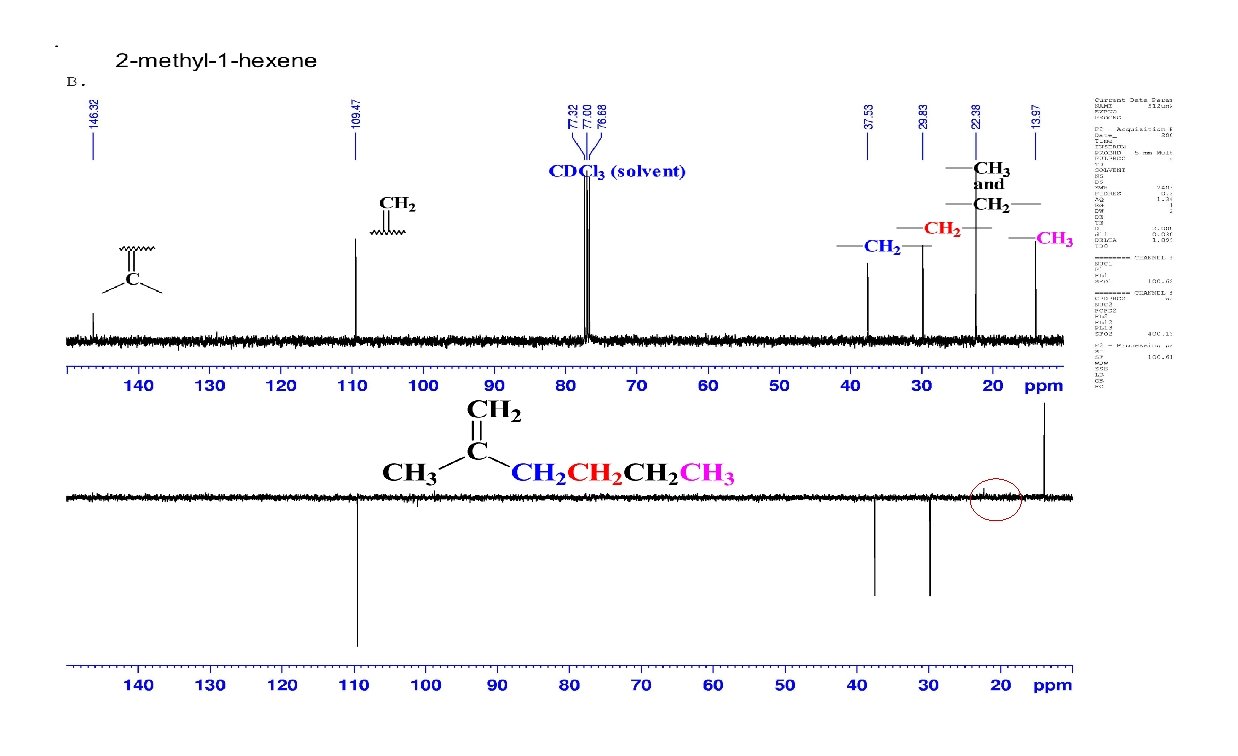
DEPT Spectra of CH 3 (+), CH 2 (-), CH (+), C (none) CH 3, CH 2 (none), CH (+), C (none)
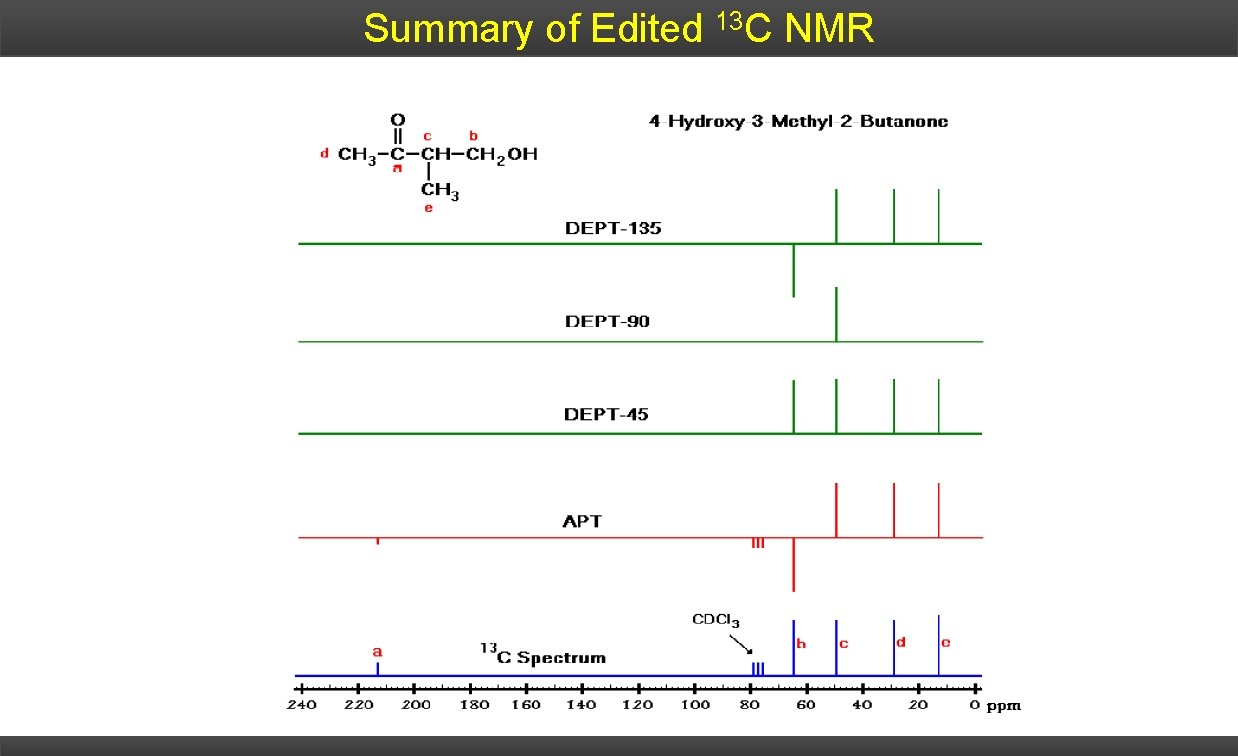
Summary of Edited 13 C NMR
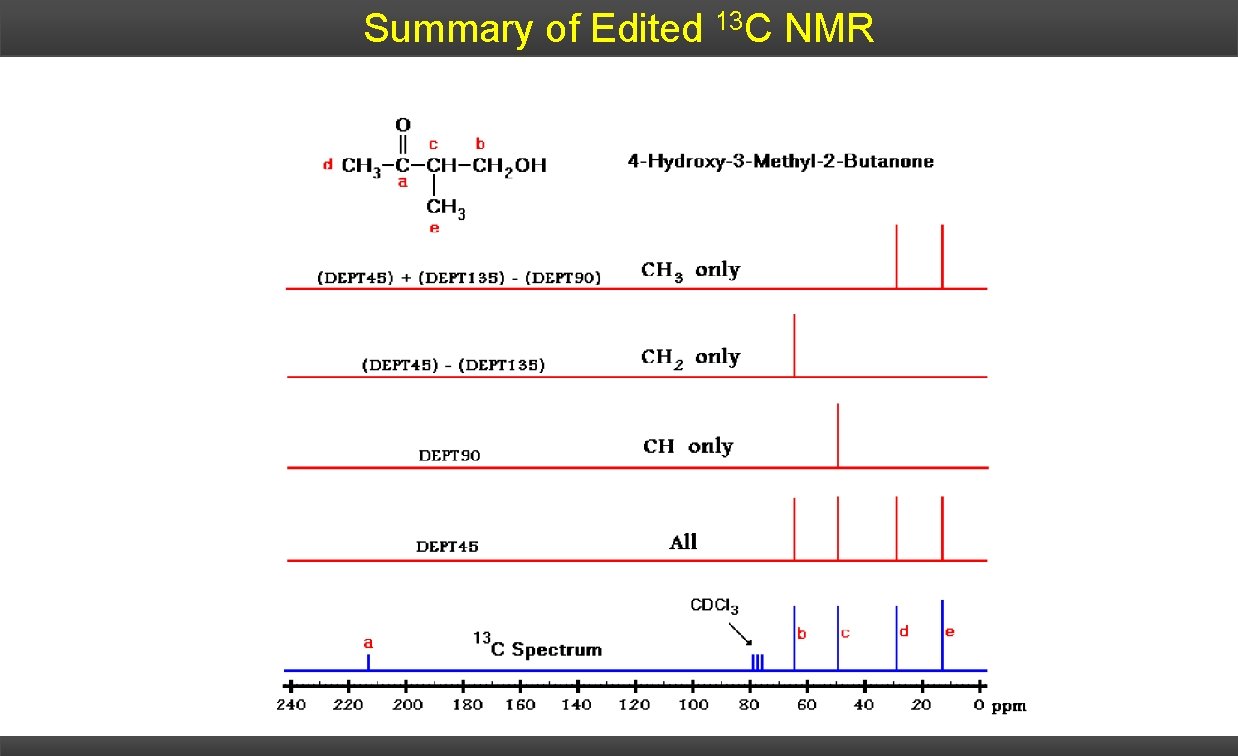
Summary of Edited 13 C NMR

Summary Number of signals indicates the number of types of carbon in the sample. (Is symmetry present? ) Chemical shifts show what types of carbons are in the sample. Quaternary/ipso carbons will be smaller than carbons with protons attached. DEPT differentiates between primary, secondary, and tertiary carbons.
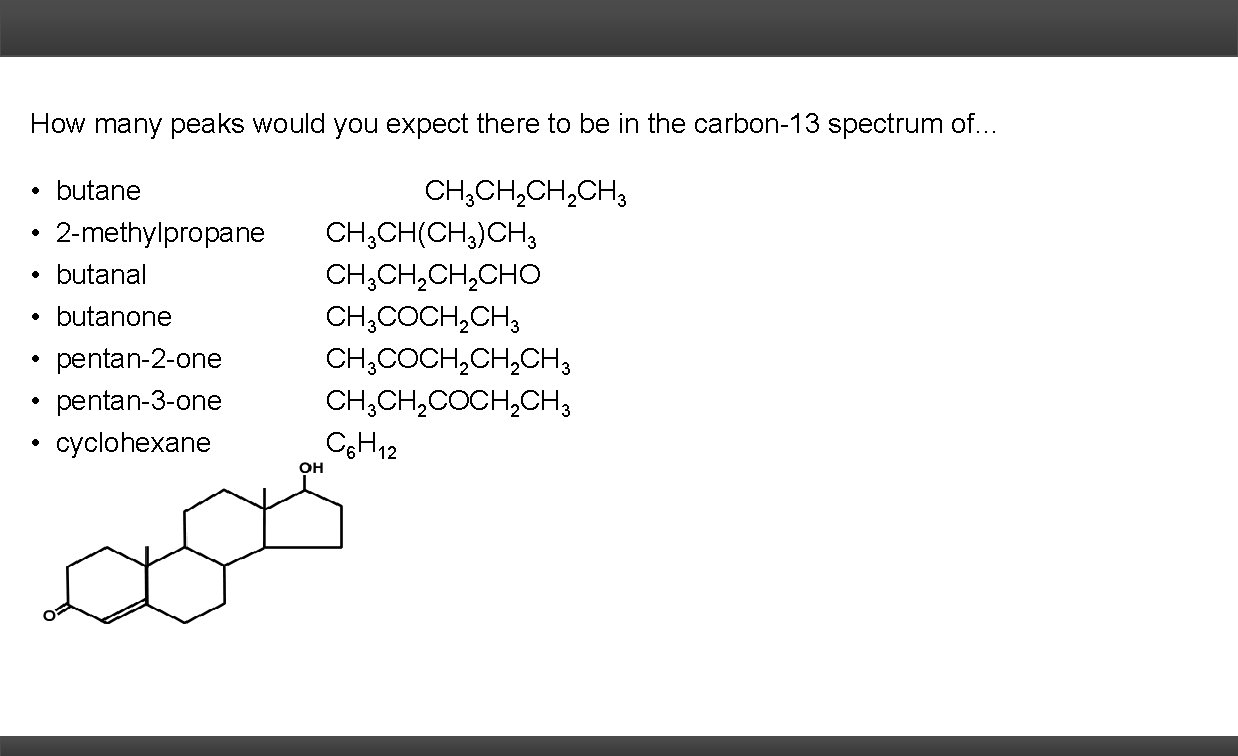
How many peaks would you expect there to be in the carbon-13 spectrum of… • • butane 2 -methylpropane butanal butanone pentan-2 -one pentan-3 -one cyclohexane CH 3 CH 2 CH 3 CH(CH 3)CH 3 CH 2 CHO CH 3 COCH 2 CH 3 CH 3 CH 2 COCH 2 CH 3 C 6 H 12

How many peaks would you expect there to be in the carbon-13 spectrum of… • • butane 2 -methylpropane butanal butanone pentan-2 -one pentan-3 -one cyclohexane CH 3 CH 2 CH 3 CH(CH 3)CH 3 CH 2 CHO CH 3 COCH 2 CH 3 CH 3 CH 2 COCH 2 CH 3 C 6 H 12 1 2 2 4 4 5 3 19
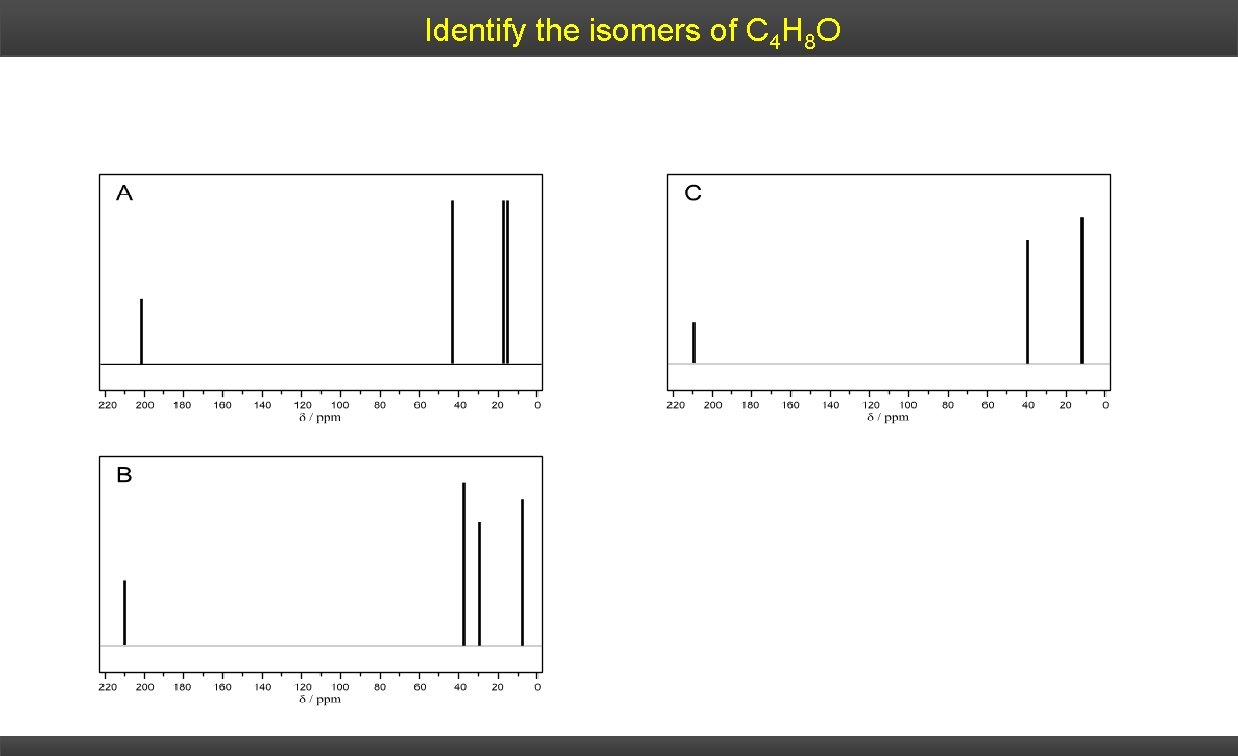
Identify the isomers of C 4 H 8 O
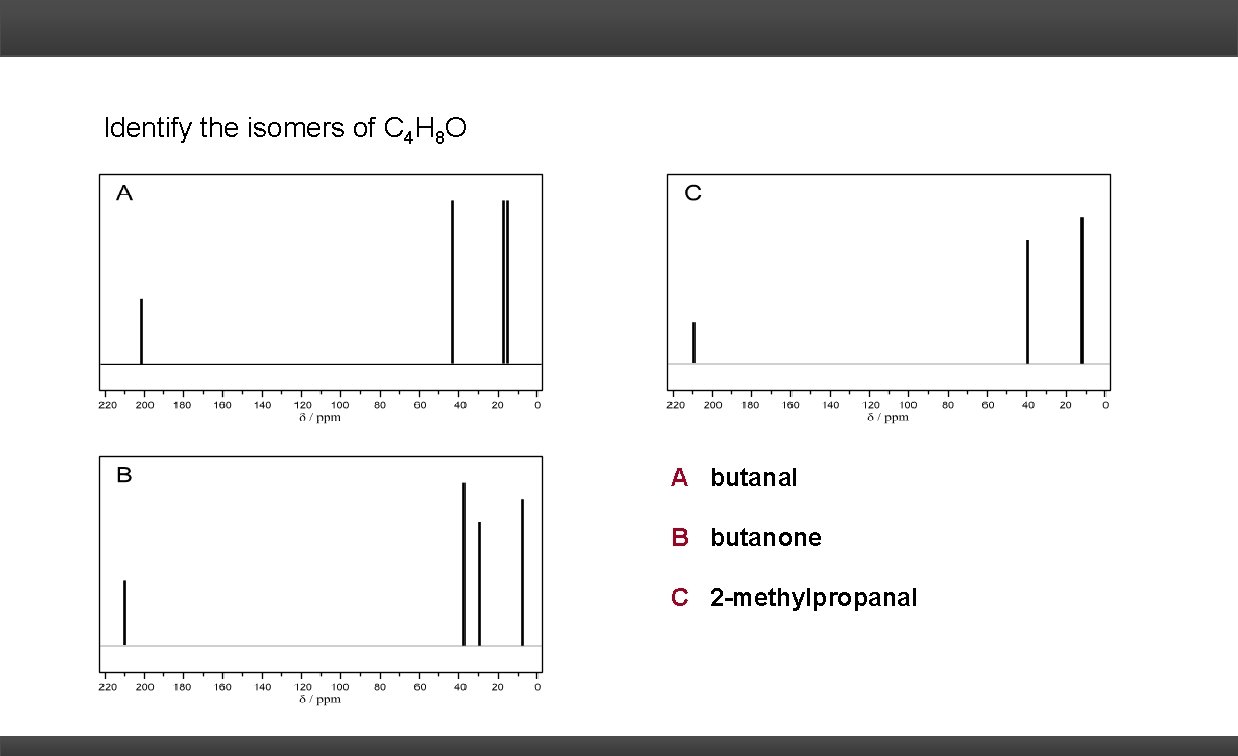
Identify the isomers of C 4 H 8 O A butanal B butanone C 2 -methylpropanal

13 C NMR-Important points • The 13 C nucleus is present in only 1. 08% natural abundance. Therefore, acquisition of a spectrum usually takes much longer than in 1 H NMR. • The magnetogyric ratio of the 13 C nucleus is about 1/4 that of the 1 H nucleus. Therefore, the resonance frequency in 13 C NMR is much lower than in 1 H NMR. (75 MHz for 13 C as opposed to 300 MHz for 1 H in a 7. 04 Tesla field). • At these lower frequencies, the excess population of nuclei in the lower spin state is reduced, which, in turn, reduces the sensitivity of NMR detection. • Unlike 1 H NMR, the area of a peak is not proportional to the number of carbons giving rise to the signal. Therefore, integrations are usually not done. • Each unique carbon in a molecule gives rise to a 13 C NMR signal. Therefore, if there are fewer signals in the spectrum than carbon atoms in the compound, the molecule must possess symmetry. • When running a spectrum, the protons are usually decoupled from their respective carbons to give a singlet for each carbon atom. This is called a proton-decoupled spectrum.

2 D NMR: COSY AND HETCOR

2 D NMR Terminology 1 D NMR = 1 frequency axis 2 D NMR = 2 frequency axes COSY = Correlated Spectroscopy H-1 H COSY provides connectivity information by allowing one to identify spin-coupled protons. 1 x, y-coordinates of cross peaks are spin-coupled protons
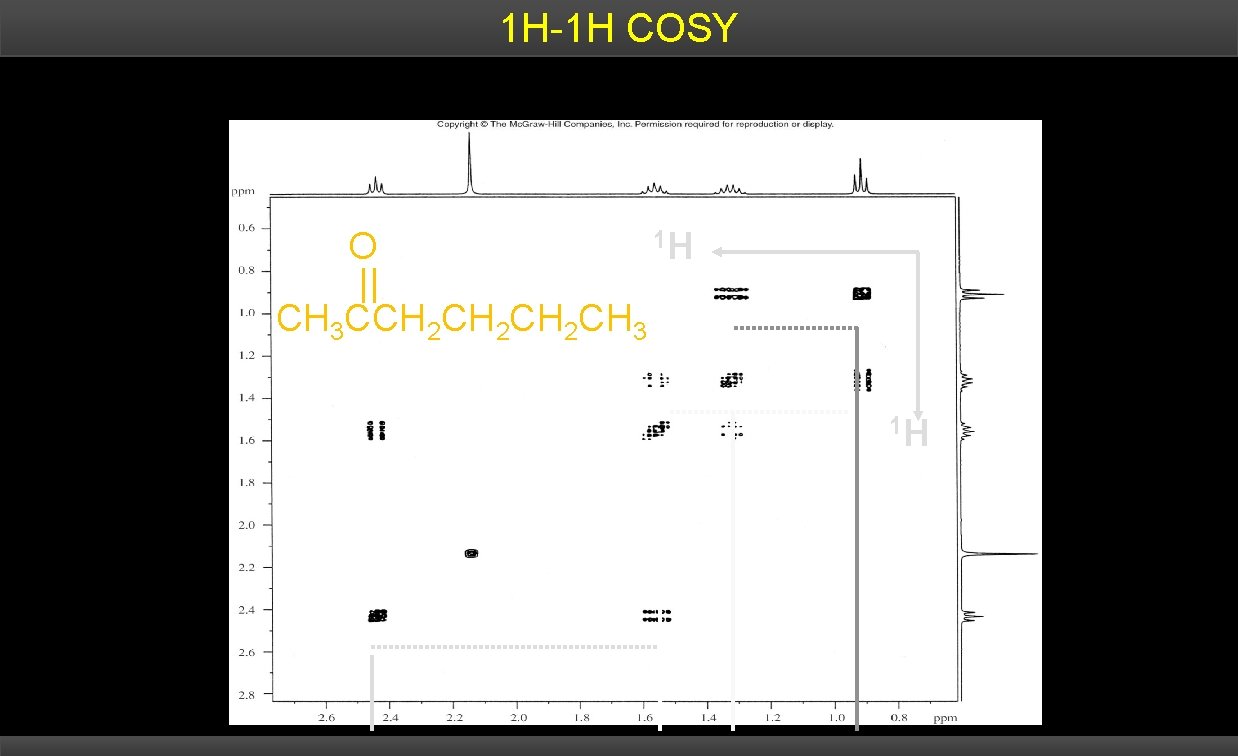
1 H-1 H COSY O 1 H CH 3 CCH 2 CH 2 CH 3 1 H

HETCOR H and 13 C spectra plotted separately on two frequency axes 1 Coordinates of cross peak connect signal of carbon to protons that are bonded to it.
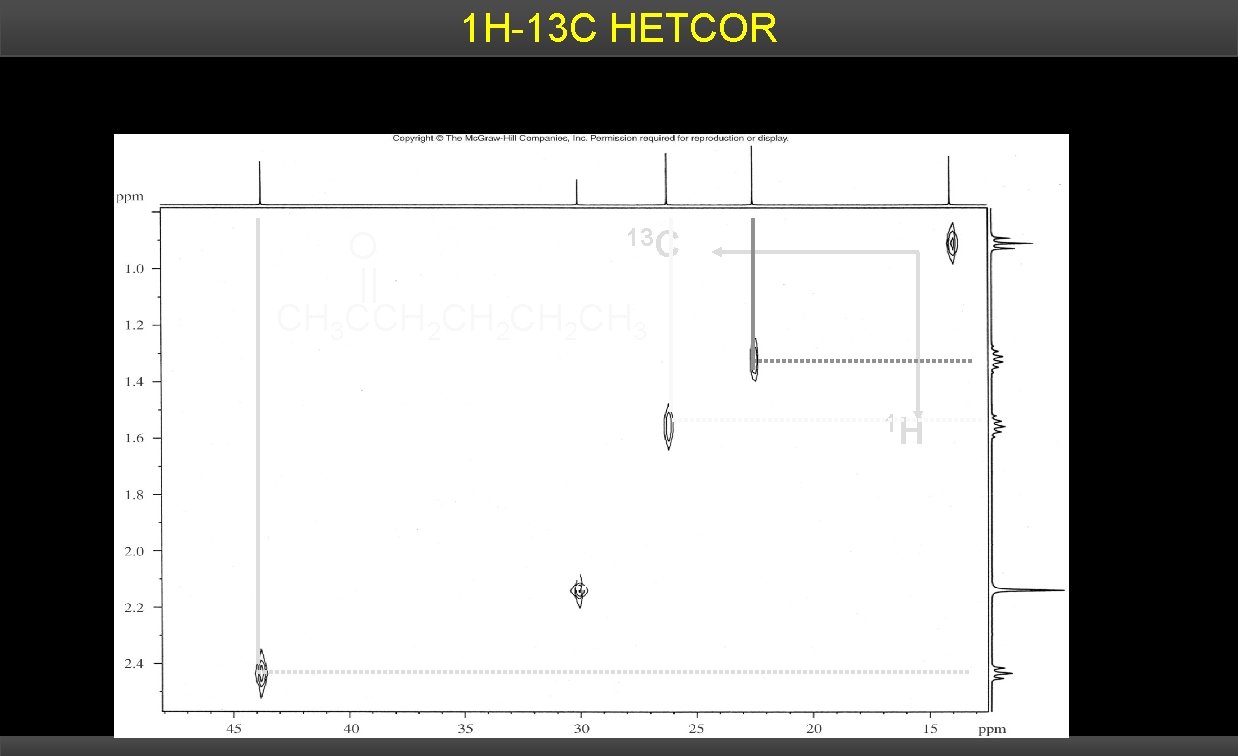
1 H-13 C HETCOR O 13 C CH 3 CCH 2 CH 2 CH 3 1 H

Solving Combined Spectra Problems: Mass Spectra: Molecular Formula Nitrogen Rule # of nitrogen atoms in the molecule M+1 peak # of carbons Degrees of Unsaturation: # of rings and/or -bonds Infrared Spectra: Functional Groups C=O O-H C=C N-H C C CO-OH C N 1 H NMR: Chemical Shift ( ) chemical environment of the H's Integration # of H's giving rise to the resonance Spin-Spin Coupling (multiplicity) # of non-equivalent H's on the adjacent carbons (vicinal coupling). 13 C NMR: # of resonances symmetry of carbon framework Type of Carbonyl Each piece of evidence gives a fragment (puzzle piece) of the structure. Piece the puzzle together to give a proposed structure. The proposed structure should be consistent with all the evidence. 116
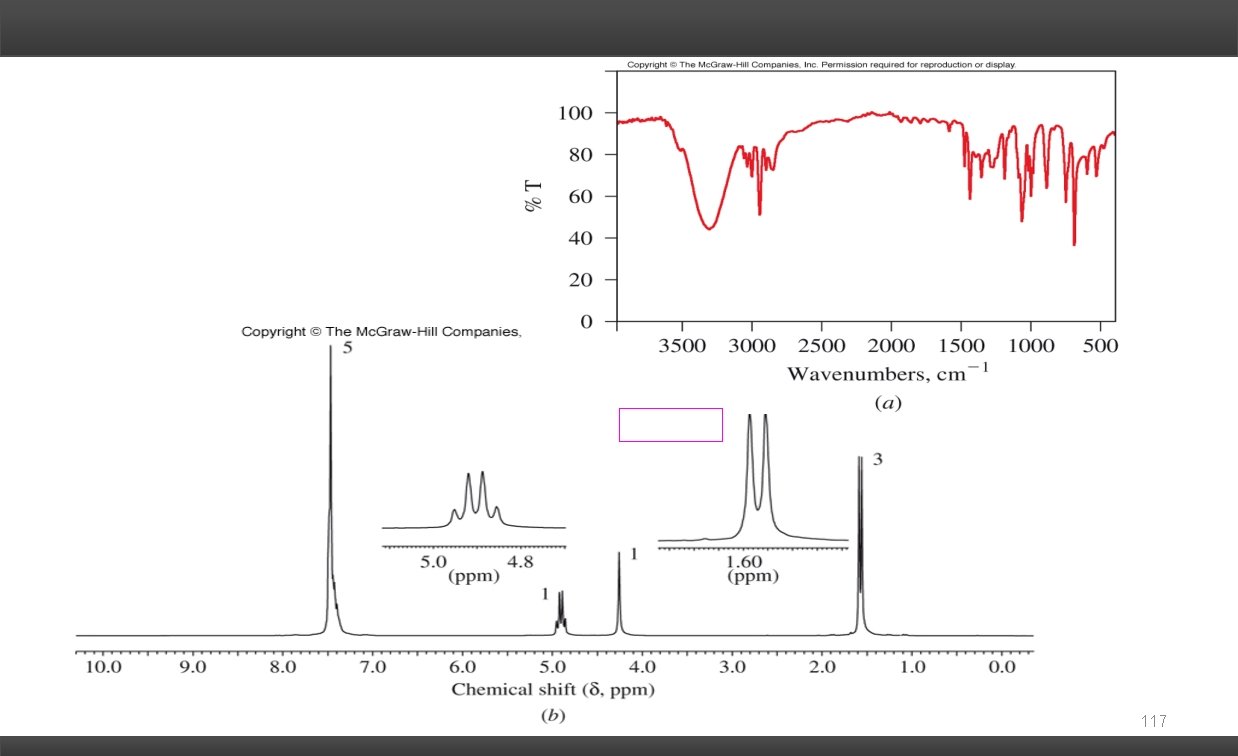
117
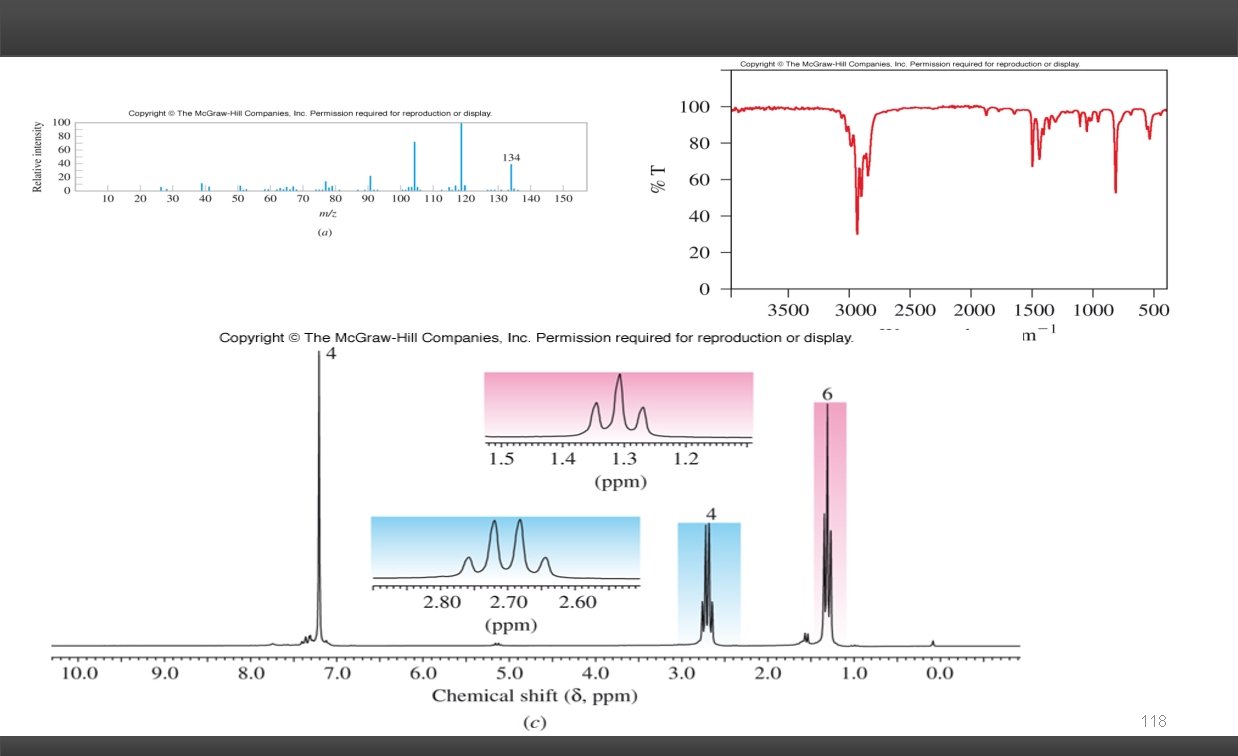
118
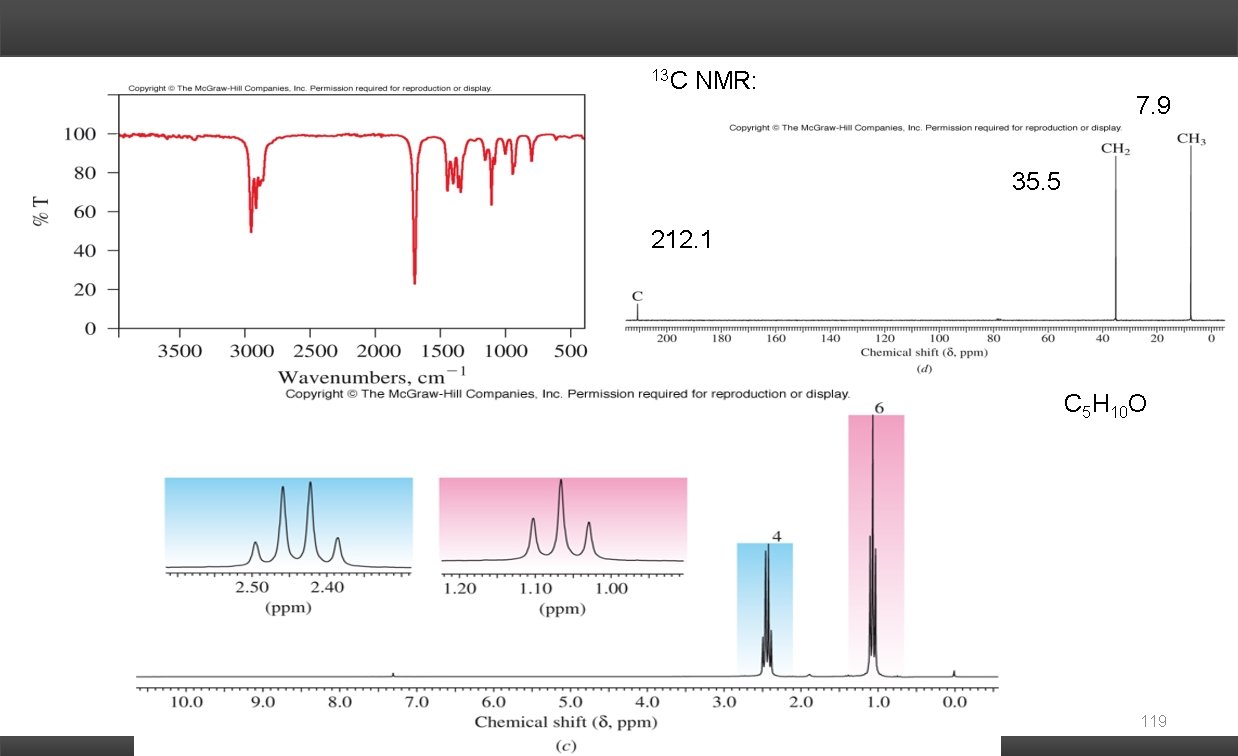
13 C NMR: 7. 9 35. 5 212. 1 C 5 H 10 O 119
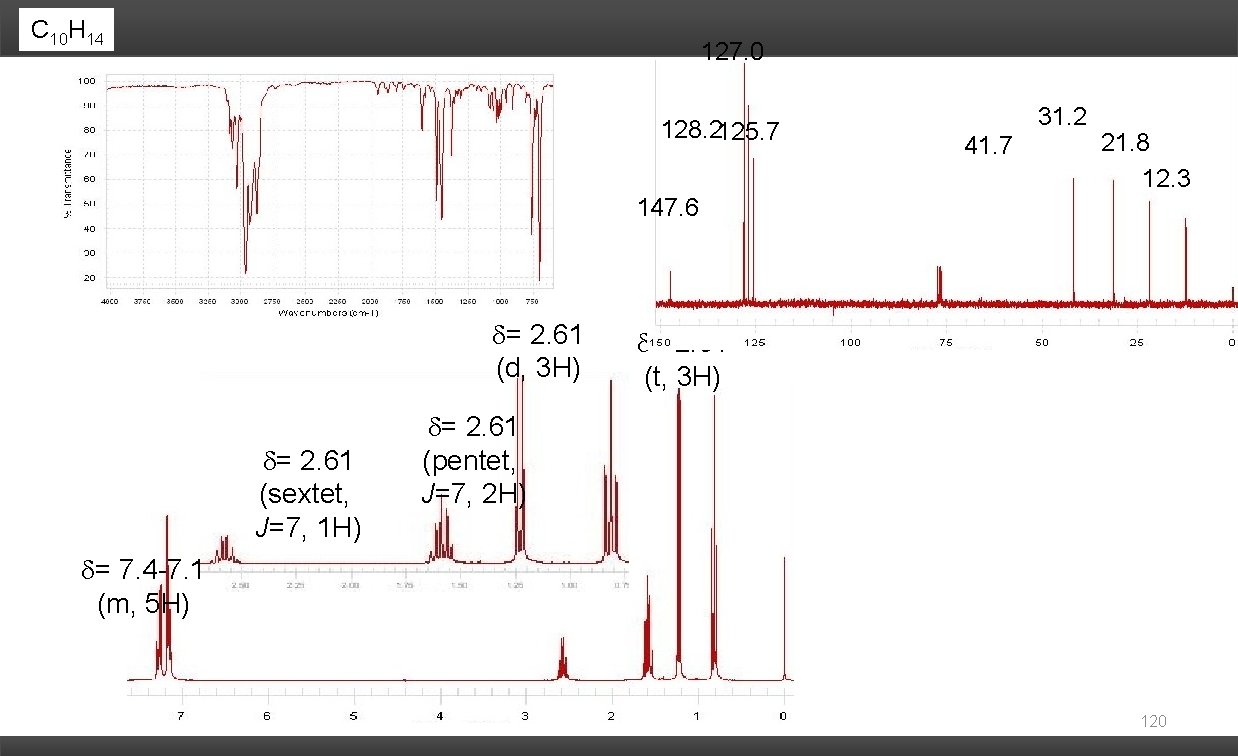
C 10 H 14 127. 0 128. 2125. 7 31. 2 41. 7 21. 8 12. 3 147. 6 = 2. 61 (d, 3 H) = 2. 61 (sextet, J=7, 1 H) = 2. 61 (t, 3 H) = 2. 61 (pentet, J=7, 2 H) = 7. 4 -7. 1 (m, 5 H) 120
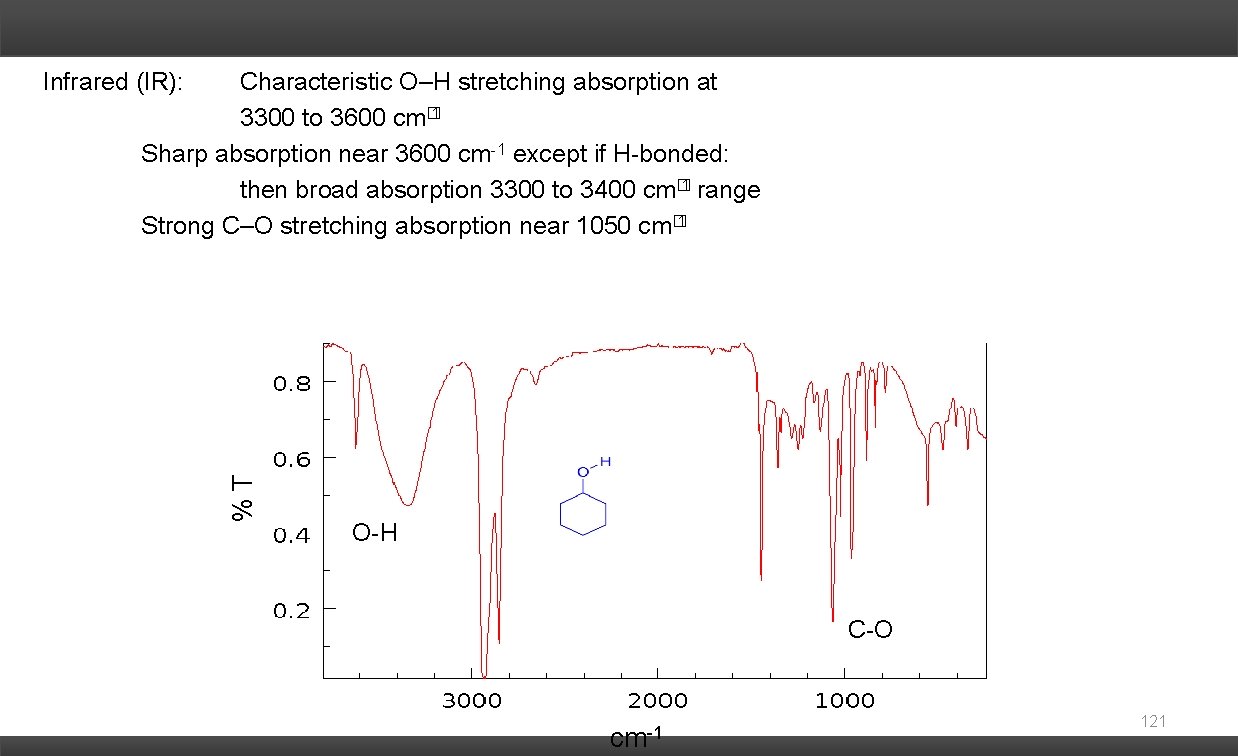
Characteristic O–H stretching absorption at 3300 to 3600 cm� 1 Sharp absorption near 3600 cm-1 except if H-bonded: then broad absorption 3300 to 3400 cm� 1 range Strong C–O stretching absorption near 1050 cm� 1 %T Infrared (IR): O-H C-O cm-1 121
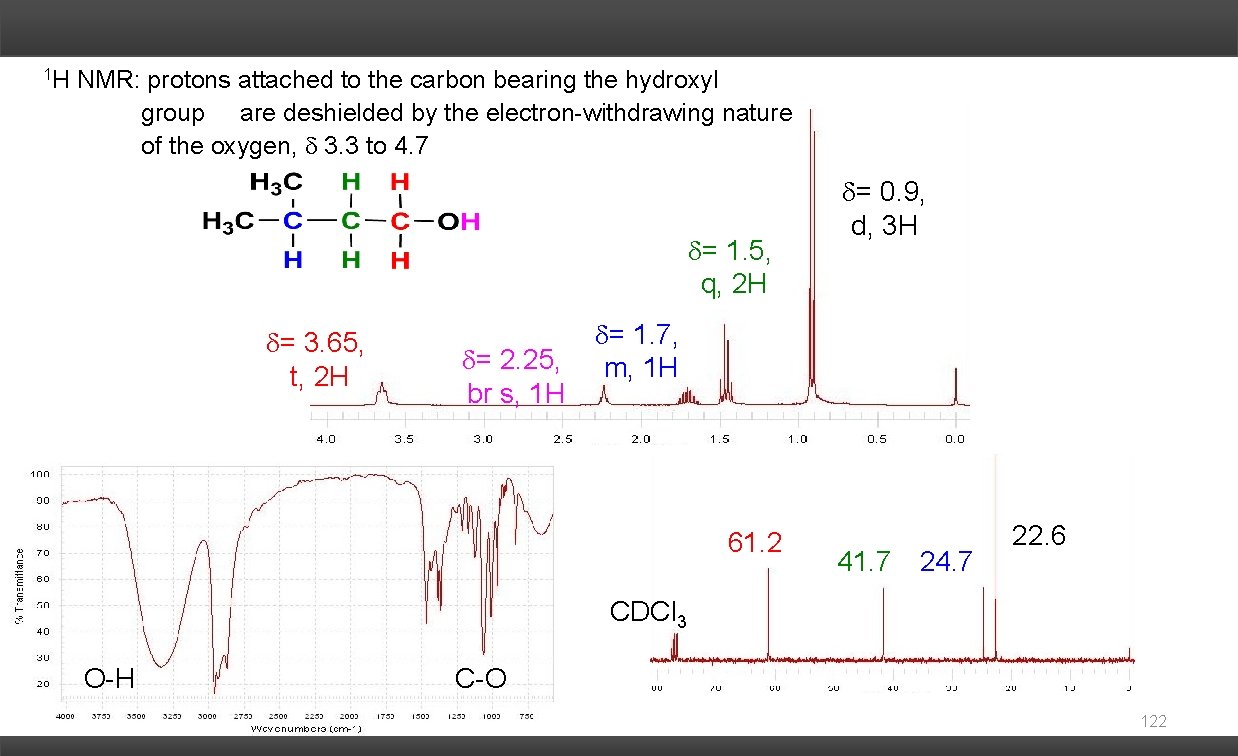
1 H NMR: protons attached to the carbon bearing the hydroxyl group are deshielded by the electron-withdrawing nature of the oxygen, 3. 3 to 4. 7 = 1. 5, q, 2 H = 3. 65, t, 2 H = 2. 25, br s, 1 H = 0. 9, d, 3 H = 1. 7, m, 1 H 61. 2 41. 7 24. 7 22. 6 CDCl 3 O-H C-O 122

Usually no spin-spin coupling between the O–H proton and neighboring protons on carbon due to exchange reaction The chemical shift of the -OH proton occurs over a large range (2. 0 - 5. 5 ppm). It chemical shift is dependent upon the sample concentration and temperature. This proton is often observed as a broad singlet (br s). Exchangable protons are often not to be observed at all. 123
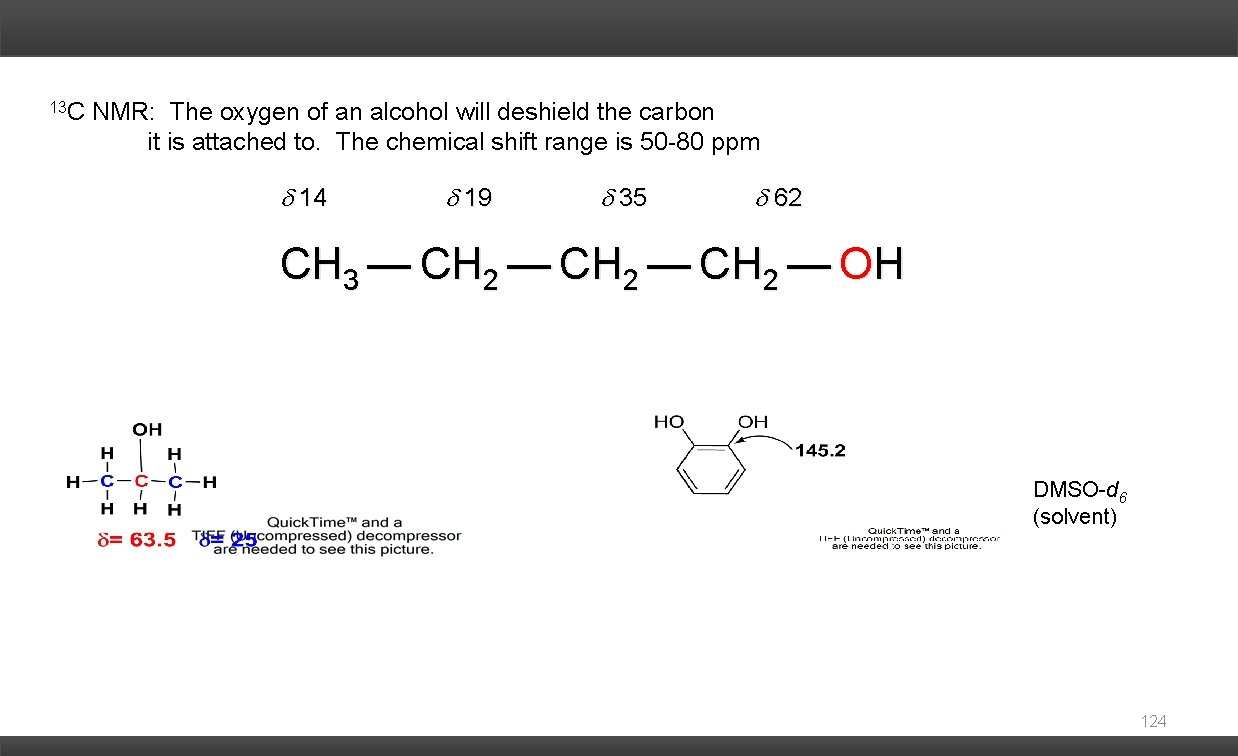
13 C NMR: The oxygen of an alcohol will deshield the carbon it is attached to. The chemical shift range is 50 -80 ppm 14 19 35 62 CH 3 — CH 2 — OH DMSO-d 6 (solvent) 124
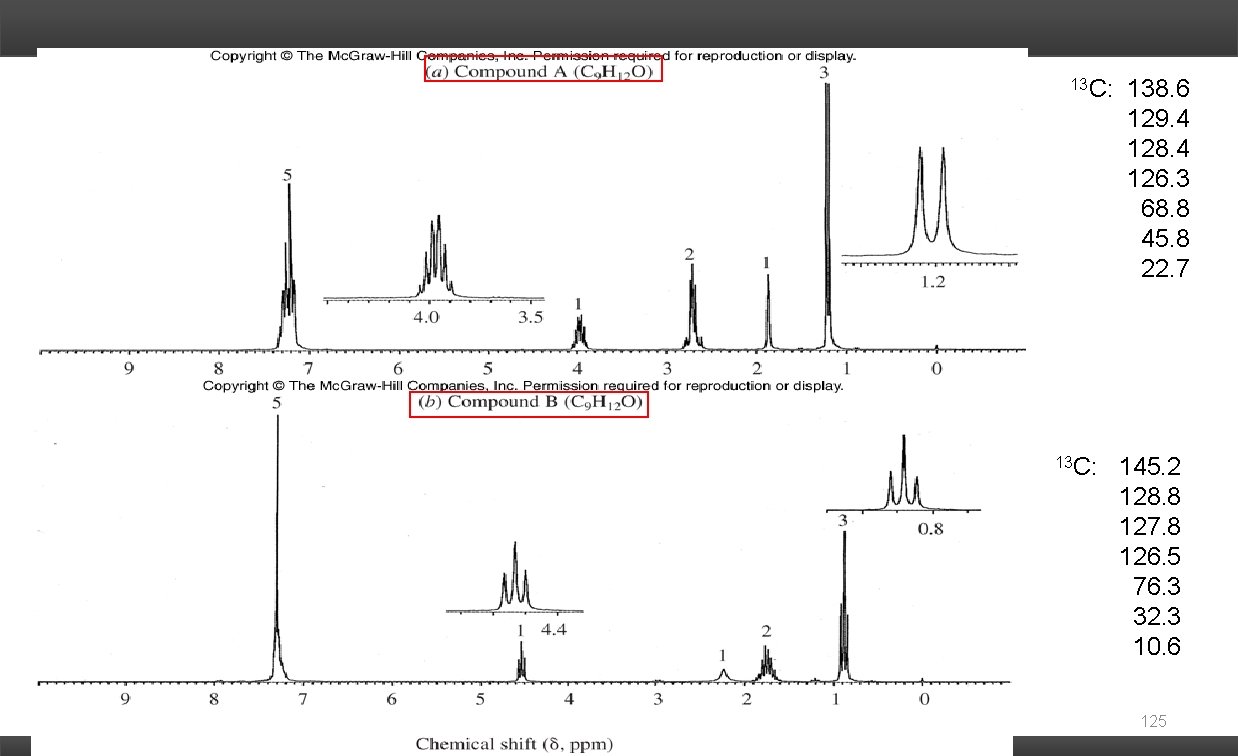
13 C: 138. 6 129. 4 128. 4 126. 3 68. 8 45. 8 22. 7 145. 2 128. 8 127. 8 126. 5 76. 3 32. 3 10. 6 125
Magnetic Resonance Imaging (MRI) MRI uses the principles of nuclear magnetic resonance to image tissue MRI normally uses the magnetic resonance of protons on water and very sophisticated computer methods to obtain images. Other nuclei within the tissue can also be used (31 P) or a imaging (contrast) agent can be administered 126
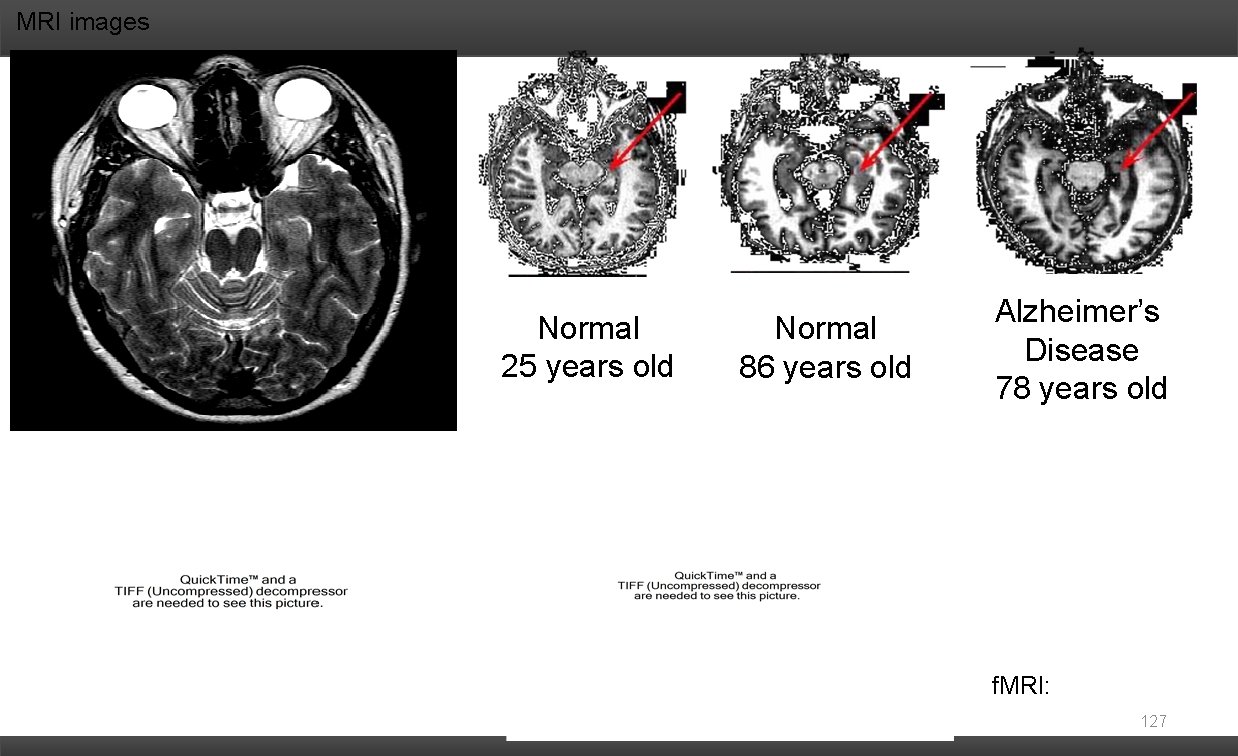
MRI images Normal 25 years old Normal 86 years old Alzheimer’s Disease 78 years old f. MRI: 127
- Slides: 127