Chapter 13 Electrochemistry Jacqueline Bennett SUNY Oneonta www

Chapter 13 Electrochemistry Jacqueline Bennett • SUNY Oneonta www. cengage. com/chemistry/brown Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Chapter Objectives • Describe at least three types of corrosion and identify chemical reactions responsible for corrosion • Define oxidation and reduction • Write and balance half-reactions for simple redox processes • Describe the differences between galvanic and electrolytic cells • Use standard reduction potentials to calculate cell potentials under both standard and nonstandard conditions • Use standard reduction potentials to predict the spontaneous direction of a redox reaction 2 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Chapter Objectives (continued) • Calculate the amount of metal plated, the amount of current needed, or the time required for an electrolysis process • Distinguish between primary and secondary batteries • Describe the chemistry of some common battery types, and explain why each type of battery is suitable for a particular application • Describe at least three common techniques for preventing corrosion 3 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Corrosion • Corrosion is the degradation of metals by chemical reactions with the environment • Uniform corrosion occurs evenly over a large portion of the surface area of a metal • Galvanic corrosion occurs when two different metals contact each other in the presence of an appropriate electrolyte • Crevice corrosion occurs in the small gap or crevice seen between two pieces of metal touching each other when the joint is not covered with a coating 4 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Corrosion (continued 1) • Different metals corrode differently • Aluminum has a greater tendency to corrode than iron, but corrosion of aluminum is not problematic compared to iron • Aluminum oxide is a spontaneous corrosion product of aluminum, and it forms a protective layer on the surface of aluminum metal to prevent further corrosion • Iron’s corrosion product is iron oxide that flakes off the surface of iron, exposing fresh iron to corrosion 5 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Corrosion (continued 2) • Corrosion occurs in a variety of forms • The chain shows uniform corrosion • The grill cover shows crevice corrosion where the handle is attached 6

Join in, 1 When two steel parts are held together by steel screws and rust occurs only at the junction, what type of corrosion is most likely occurring? Galvanic Uniform Crevice 7 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 1, Answer • Crevice 8 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Oxidation–Reduction Reactions and Galvanic Cells • Special conditions must be present to promote the reaction between iron and oxygen to form iron(III) oxide • Rust formation is a slow process, so the basics of electrochemistry must be investigated using more easily observed reactions • Reactions involving the transfer of electrons are known as oxidation–reduction or redox reactions • Oxidation is the loss of electrons from some chemical species • Reduction is the gain of electrons by some chemical species 9 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Oxidation–Reduction and Half-Reactions • For an oxidation–reduction reaction to occur, one reactant must be oxidized and one reactant must be reduced • Oxidation cannot occur without reduction • When a copper wire is placed in a colorless solution of silver nitrate, a redox reaction occurs • Observations such as the solution turning blue and the formation of crystals on the wire confirm the occurrence of the reaction 10 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Oxidation–Reduction and Half-Reactions (continued 1) • When a clean copper wire is placed into a colorless solution of silver nitrate, it is quickly apparent that a chemical reaction occurs: Crystals of silver metal form on the copper wire, and the solution turns blue 11

Oxidation–Reduction and Half-Reactions (continued 2) • The solution’s blue color is indicative of Cu 2+ ions in solution • Cu 2+ is formed when a copper atom loses two electrons • The copper metal is oxidized • The crystals formed on the surface of the copper wire are silver metal • Metallic silver is formed when a silver cation gains an electron • The silver in the solution is reduced 12 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Oxidation–Reduction and Half-Reactions (continued 3) • For the reaction between silver cation and copper metal, two half-reactions are written • One for the oxidation of copper and one for the reduction of silver • Neither half-reaction can occur without the other • These half-reactions indicate that Ag+ accepts only one electron, whereas Cu loses two electrons • The electron transfer must be balanced, so the reduction halfreaction is multiplied by 2 13 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Oxidation–Reduction and Half-Reactions (continued 4) • Add the two half-reactions together and the electrons will cancel out, leaving the net ionic equation for the redox reaction 14 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Oxidation–Reduction and Half-Reactions (continued 5) • Reducing agent: The species undergoing oxidation • The Cu was oxidized and is the reducing agent • The Cu facilitated the reduction of Ag+ by losing electrons • Oxidizing agent: The species undergoing reduction • The Ag+ was reduced and is the oxidizing agent • The Ag+ facilitated the oxidation of Cu by gaining electrons 15 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Building a Galvanic Cell • A galvanic cell is any electrochemical cell in which a spontaneous chemical reaction can be used to generate an electric current • The name electrochemistry comes from the observation of electric currents in galvanic cells • To harness electricity from a galvanic cell, each half-reaction is prepared separately in half-cells • Cu metal immersed in Cu 2+ solution is one half-cell • Ag metal immersed in Ag+ solution is the second half-cell 16 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
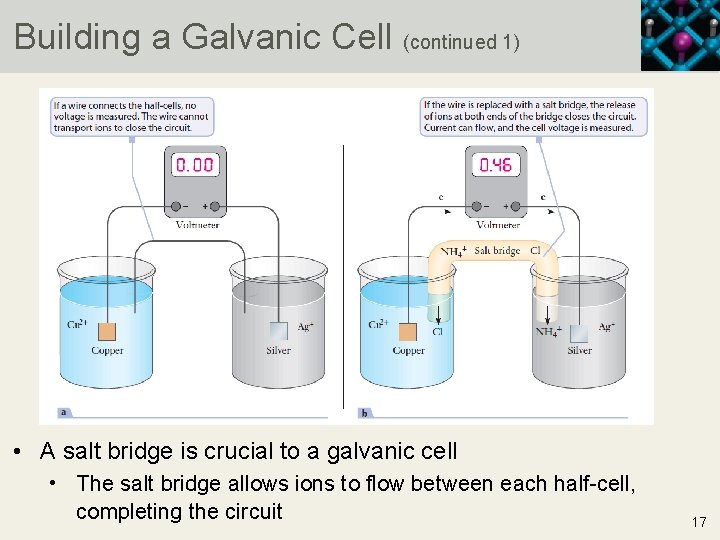
Building a Galvanic Cell (continued 1) • A salt bridge is crucial to a galvanic cell • The salt bridge allows ions to flow between each half-cell, completing the circuit 17

Building a Galvanic Cell (continued 2) • Current flows because of the migration of ions in the solution • To facilitate the flow of electrons between the half-cells, a salt bridge is used • The salt bridge contains a strong electrolyte that allows either cations or anions to migrate into the solution where they are needed to maintain charge neutrality • A metal wire cannot be used as a salt bridge as it cannot transport ions 18 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Building a Galvanic Cell (continued 3) • For a salt bridge composed of NH 4 Cl: • NH 4+ will flow into the Ag+ beaker to offset the removal of Ag+ from the solution • Cl− will flow into the Cu 2+ beaker to offset the production of Cu 2+ in the solution • The circuit is completed by attaching a voltmeter between the Cu and the Ag to measure cell voltage • A voltage potential of 0. 46 V will be measured for the described cell 19 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Terminology for Galvanic Cells • Electrodes are the electrically conducting sites in a galvanic cell at which either oxidation or reduction occurs • The electrode where oxidation occurs is the anode • The electrode where reduction occurs is the cathode • Cell notation: A shorthand notation for representing the specific chemistry of an electrochemical cell • • Cell notation lists the metals and ions involved in the reaction A single vertical line, |, denotes a phase boundary A double vertical line, ||, denotes a salt bridge The anode is written on the left, the cathode on the right 20 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Terminology for Galvanic Cells (continued) • General form of cell notation • For the previous example of copper and silver • The electrolyte concentration is also given • An electrochemical cell is at its standard state when the electrolyte concentrations are 1 M • For half-cells that generate or consume a gas, a partial pressure of 1 atm is required for the standard state 21 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Atomic Perspective on Galvanic Cells • Before half-cells are connected by a salt bridge, a small build up of charge occurs for each half-cell at the interface between the electrode and the electrolyte • At the anode, some oxidation occurs and cations dissolve into solution, leaving a negative charge on the anode • At the cathode, some reduction occurs and cations are removed from solution, leaving a positive charge on the cathode 22 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Atomic Perspective on Galvanic Cells (continued 1) • An equilibrium can be described for each half-cell, the halfreaction equilibrium • Not an oxidation–reduction equilibrium • The build up of charge on the electrode means there is a potential for electrical work • This potential is the cell potential, electromotive force, or EMF • EMF is related to the maximum work that can be obtained from an electrochemical cell • wmax = q. E • q is the charge • E is the cell potential 23 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
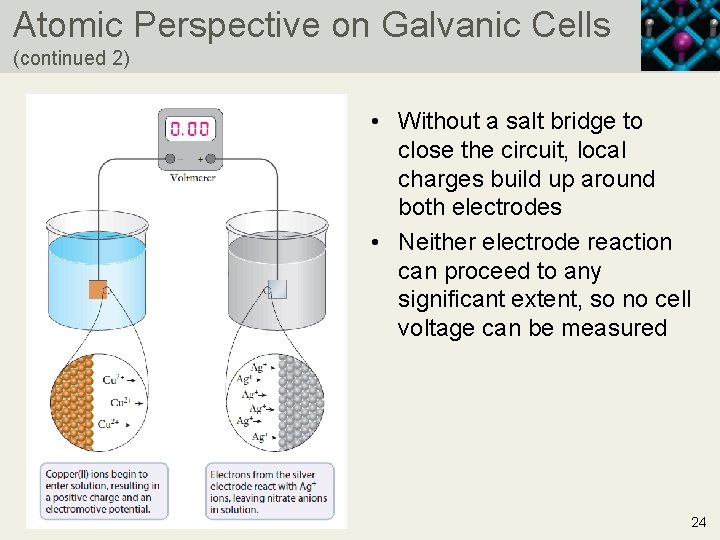
Atomic Perspective on Galvanic Cells (continued 2) • Without a salt bridge to close the circuit, local charges build up around both electrodes • Neither electrode reaction can proceed to any significant extent, so no cell voltage can be measured 24

Galvanic Corrosion and Uniform Corrosion • Metals in contact with a solution establish an oxidation halfreaction equilibrium • If the solution contains a substance that can undergo reduction, a redox reaction may occur • For two metals in contact, such as a tin-plated steel can, there is the possibility of galvanic corrosion • A scratch in the tin exposes the underlying steel (mostly iron) to air and water, resulting in rapid corrosion • The half-reaction equilibrium for the tin facilitates the process by which iron is oxidized • This is an example of galvanic corrosion 25 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
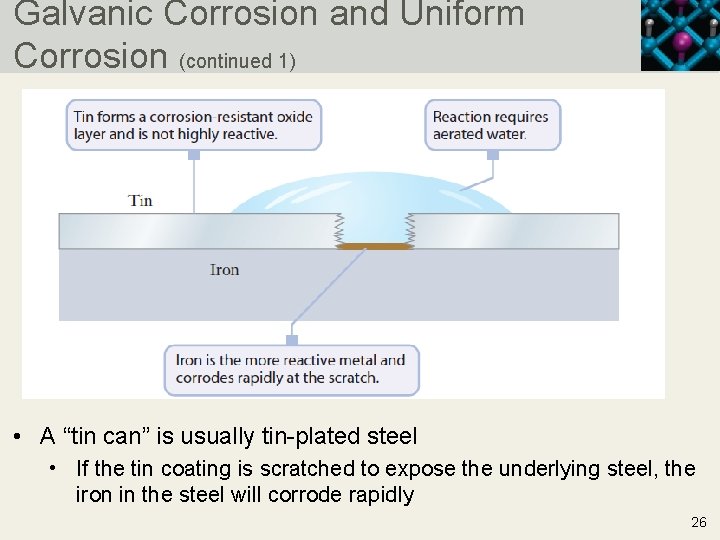
Galvanic Corrosion and Uniform Corrosion (continued 1) • A “tin can” is usually tin-plated steel • If the tin coating is scratched to expose the underlying steel, the iron in the steel will corrode rapidly 26

Galvanic Corrosion and Uniform Corrosion (continued 2) • Metal not in contact with another metal can also corrode • A nonmetal is involved in the second half-cell • For the corrosion of iron, iron is the first half-cell and oxygen dissolved in water is the second half-cell • The electrode for the oxygen half-cell is the iron itself • Dissolved salts facilitate the corrosion reaction • This is an example of uniform corrosion 27 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Galvanic Corrosion and Uniform Corrosion (continued 3) • Iron is oxidized, and oxygen from the air is reduced • Water is needed for ion mobility between the anodic and cathodic regions, and the presence of ionic salts speeds up the reaction considerably 28 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 2 When a metal is in water, some of the metal is lost in the form of metal ions in the water. What part of a reaction has occurred? • Reduction • Oxidation • Disproportionation 29 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 2, Answer • Oxidation 30 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 3 In a classroom demonstration of the reaction between copper metal and silver ions, silver plates out on the copper wire. Which is the oxidizing agent in this reaction? • • Silver metal Silver ions Copper metal Copper ions 31 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 3, Answer • Silver ions 32 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 4 What actually flows through a salt bridge in a galvanic cell? • • Nothing Electrons Salt Water 33 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 4, Answer • Nothing 34 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 5 In a galvanic cell, where does oxidation occur? • • In the electrolyte At the anode At the cathode In the salt bridge 35 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 5, Answer • At the anode 36 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 6 Why are there some steel shipwrecks at the bottom of the ocean that have no signs of uniform corrosion? • Mg ions from salt water inhibit corrosion • There is not enough oxygen dissolved in the water • There are no other metals other than steel present 37 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 6, Answer • There is not enough oxygen dissolved in the water 38 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 7 Based on Table 13. 1, which metallic solids could be used galvanically to convert hydrogen gas to hydrogen ions? • • It cannot be done because H 2 has 0 V Zinc and iron Copper and silver Any metal will do this because H 2 has 0 V 39 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 7, Answer • Copper and silver 40 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 8 How does galvanizing steel reduce corrosion? • The iron ions that would form react with the zinc • The zinc ions in water prevent iron ions from dissolving to form rust • Zinc is more easily oxidized than iron 41 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 8, Answer • Zinc is more easily oxidized than iron 42 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Cell Potentials • The relative corrosivities of various plated steels can be expressed as cell potential • A voltmeter measures the size of the electrical potential and also its polarity • Polarity denotes the locations of the negative charge or negative pole and the positive charge or positive pole • An electric potential has a fixed polarity and voltage • Reversing the poles of a battery with respect to a voltmeter changes the sign on the measured voltage • However, it does not influence the electrochemical reaction in the battery 43 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Measuring Cell Potential • When a voltmeter is connected to the copper or silver cell described in earlier slides, a potential of 0. 462 V is measured • When the copper half-cell is connected to a half-cell that reduces iron(III) to iron(II), a cell potential of 0. 434 V is measured • Connecting the iron(III) or iron(II) half-cell to the silver half-cell results in a cell potential of 0. 028 • For the three cell potentials measured, the fact that 0. 462 V = 0. 434 V + 0. 028 V suggests two things • The behavior of cell potentials is akin to that of state functions • If a specific standard electrode is chosen to which all other electrodes can be compared, a practical system for determining cell potential can be devised 44 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
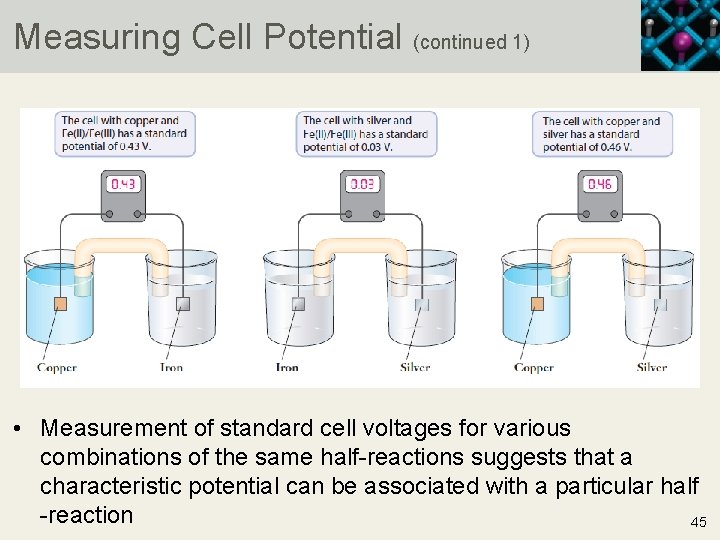
Measuring Cell Potential (continued 1) • Measurement of standard cell voltages for various combinations of the same half-reactions suggests that a characteristic potential can be associated with a particular half -reaction 45

Measuring Cell Potential (continued 2) • The standard hydrogen electrode or SHE is the choice for the standard component in cell potential measurements • The cell is constructed using a platinum wire or foil as the electrode • The electrode is immersed in a 1 M HCl solution, and H 2 gas is bubbled over the electrode at a pressure of 1 atm • The SHE has been chosen as the reference point for the scale of standard reduction potentials and assigned a potential of exactly zero volts 46 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Measuring Cell Potential (continued 3) • For the standard hydrogen electrode • The half-reaction for the SHE is • The half-cell notation is Pt(s) | H 2(g, 1 atm) | H+(1 M) • The half-cell is assigned a potential of exactly zero volts • The cell potential is attributed to the other half-reaction 47 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Measuring Cell Potential (continued 4) • For some galvanic cells, the SHE acts as the anode, and for other galvanic cells, the SHE acts as the cathode • The anode is the site of oxidation, releasing electrons and creating a negatively charged electrode • If the anode is connected to the positive terminal of the voltmeter, a negative potential is measured • The cathode is the site of reduction, consuming electrons and creating a positively charged electrode • If the cathode is connected to the positive terminal of the voltmeter, a positive potential is measured 48 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Measuring Cell Potential (continued 5) • When measuring standard potentials, the SHE is always connected to the positive terminal • The sign of the measured potential tells us the direction of the redox reaction • When the potential is negative, the SHE is the anode and H 2 is oxidized to H+(aq) • When the potential is positive, the SHE is the cathode and H+(aq) is reduced to H 2 49 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
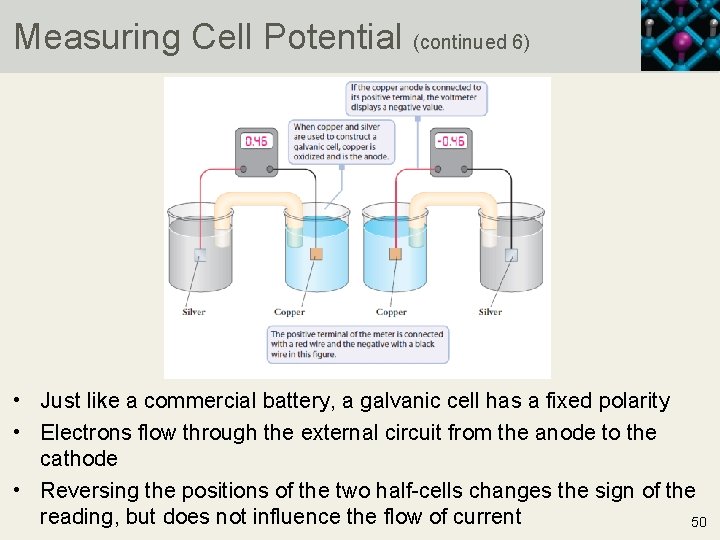
Measuring Cell Potential (continued 6) • Just like a commercial battery, a galvanic cell has a fixed polarity • Electrons flow through the external circuit from the anode to the cathode • Reversing the positions of the two half-cells changes the sign of the reading, but does not influence the flow of current 50

Standard Reduction Potentials • To compare the oxidation–reduction trends of species used in electrochemistry, all half-cell potentials are written as reductions • A table of standard reduction potentials lists the potential of any half-reaction when connected to a SHE • All electrodes must be constructed to meet the conditions of standard states • All materials are in thermodynamic standard states • Solutions have a concentration of 1 M • Gases have a pressure of 1 atm 51 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Standard Reduction Potentials (continued 1) • Standard reduction potentials for several half-reactions involved in the cells discussed in the text 52

Standard Reduction Potentials (continued 2) • Although the half-reactions are listed as reductions in the table, one half-reaction in any electrochemical cell must be an oxidation and, therefore, reversed from what appears in the table • The cell potential sign must be changed when writing the halfreaction as an oxidation • Some half-reactions have positive potentials, whereas others have negative potentials 53 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Standard Reduction Potentials (continued 3) • All potentials are measured with a SHE connected to the positive terminal • If the voltage is positive, the SHE is the anode or the oxidation site • A positive standard reduction potential means the half-reaction proceeds as a reduction reaction • If the voltage is negative, the SHE is the cathode or the reduction site • A negative standard reduction potential means the half-reaction proceeds as an oxidation reaction 54 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Standard Reduction Potentials (continued 4) • The tendency for the chemicals involved in a half-reaction to be an oxidation or reduction depends on the value of the reduction potential • A large, positive value for the standard reduction potential implies the substance is reduced readily and is a good oxidizing agent • A large, negative value for the standard reduction potential implies the substance is oxidized readily and is a good reducing agent 55 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Standard Reduction Potentials (continued 5) • For a galvanic cell, the half-reaction with the more positive reduction potential will be the cathode • The half-reaction with the more negative reduction potential will be the anode • The standard reduction potential for any pair of half-reactions, E°cell, is calculated using the standard reduction potentials for the cathode and anode • E°red is the standard reduction potential for the cathode • E°ox is the standard reduction potential for the anode 56 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
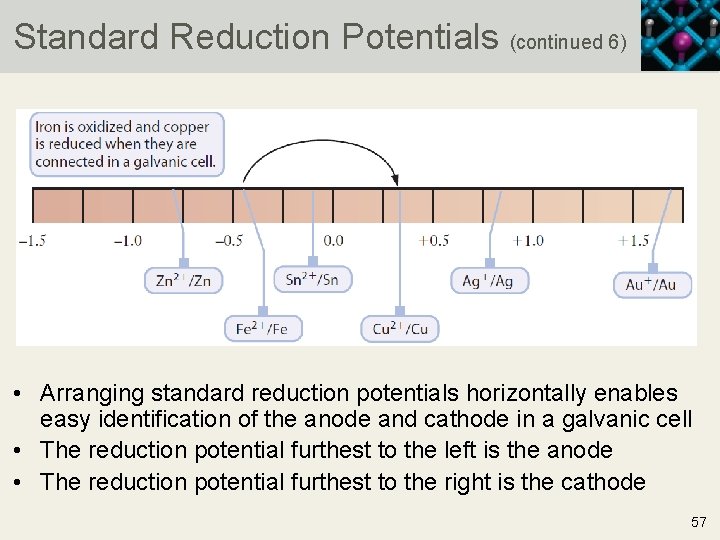
Standard Reduction Potentials (continued 6) • Arranging standard reduction potentials horizontally enables easy identification of the anode and cathode in a galvanic cell • The reduction potential furthest to the left is the anode • The reduction potential furthest to the right is the cathode 57

Example Problem 13. 1 • Using standard reduction potentials, identify the anode and the cathode and determine the cell potential for a galvanic cell composed of copper and iron • Assume standard conditions • Confirm that the potential of the following galvanic cell is 0. 462 V 58 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Nonstandard Conditions • Cell potentials under nonstandard conditions are calculated using the Nernst equation • Q is the reaction quotient, F is the Faraday constant, and n is the number of electrons transferred in the reaction • F = 96, 485 J V− 1 mol− 1 or 96, 485 C mol− 1 59 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Example Problem 13. 2 • Assume that you have a cell that has an iron(II) concentration of 0. 015 M and an H+ concentration of 1. 0 × 10− 3 M. The cell temperature is 38°C, and the pressure of hydrogen gas is maintained at 0. 04 atm. What would the cell potential be under these conditions? 60 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 9 In a galvanic cell where iron is oxidized and copper is reduced, the standard potential is 0. 78 V. How does the voltage change if the concentration of copper ions is 0. 5 M? • It increases • It decreases • There is no change because solid copper is formed 61 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 9, Answer • It decreases 62 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Cell Potentials and Free Energy • Corrosion is a spontaneous process and has a negative Gibbs free energy change • The Gibbs free energy change for an electrochemical reaction can be calculated from the standard reduction potential • n is the number of electrons transferred, and F is Faraday’s constant • A galvanic cell always has a positive cell potential, so all three terms on the right are positive • The minus sign is required because a galvanic cell spontaneously generates electrical work, and thus, must have a negative ΔG value 63 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Example Problem 13. 3 • Suppose that we wish to study the possible galvanic corrosion between zinc and chromium, so we set up the following cell: • What is the chemical reaction that takes place, and what is the standard free energy change for that reaction? 64 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Equilibrium Constants • Cell potential can be used to calculate the equilibrium constant for an electrochemical reaction • n is the number of electrons transferred, R is the universal gas law constant, and F is Faraday’s constant 65 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Equilibrium Constants (continued 1) • The relationship between the cell potential and the equilibrium constant can be re-written in terms of the common (base 10) log • The equation can be simplified for a reaction carried out at standard temperature, 25°C (298 K) 66 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
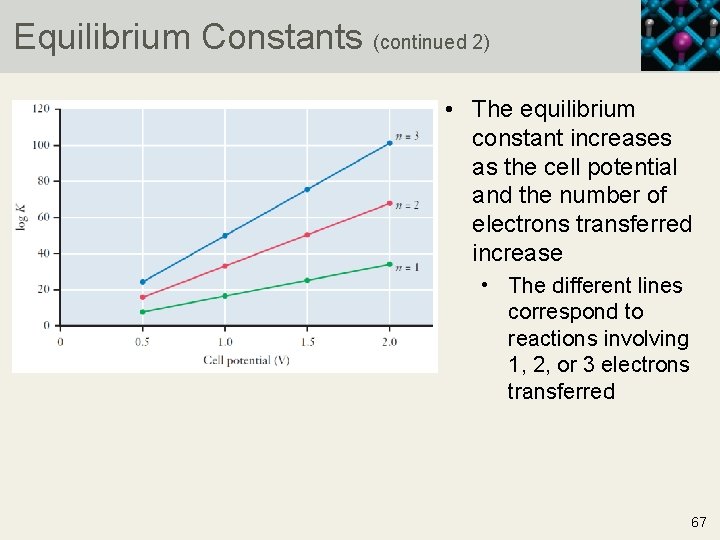
Equilibrium Constants (continued 2) • The equilibrium constant increases as the cell potential and the number of electrons transferred increase • The different lines correspond to reactions involving 1, 2, or 3 electrons transferred 67

Join in, 10 For galvanic cells, what is always true about the equilibrium constant? • • It is positive It is greater than one It is greater than the cell potential All these are true 68 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 10, Answer • All these are true 69 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Batteries • A battery is a cell or a series of cells that generates an electrical current • Batteries are the means by which we harness the electrical work of a galvanic cell and use it productively 70 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Primary Cells • Single-use batteries that cannot be recharged are primary cells, or primary batteries • The most prevalent type of primary cell is the alkaline battery • An alkaline battery has a zinc electrode at which oxidation occurs • The cathode is derived from manganese(IV) oxide 71 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Primary Cells (continued 1) • The net reaction representing the chemistry of an alkaline dry cell battery is shown above • The alkaline battery is termed a dry cell because the KOH electrolyte is in the form of a paste or gel 72 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Primary Cells (continued 2) • Lithium batteries are small and long-lasting • They are used for medical devices like pacemakers • Lithium metal is the anode • Manganese(IV) oxide is the cathode as in the alkaline battery, but in this case, the Mn. O 2 reacts with lithium ions • The lithium battery provides a stable current and electrical potential for long periods 73 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Primary Cells (continued 3) • Zinc-air batteries are also primary cells • Zinc is the anode • Oxygen reacts at the cathode • In a zinc-air battery, one of the reactants is oxygen from the surrounding air • As a result, these batteries can offer a very attractive energy density 74 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Secondary Cells • Rechargeable batteries are secondary cells or secondary batteries • Nickel-metal-hydride batteries are an example of secondary cells • The anode reaction is • The complex cathode reaction can be represented as 75 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Secondary Cells (continued 1) • Nickel-metal-hydride batteries have become popular as rechargeable cells 76

Secondary Cells (continued 2) • The lead-acid storage battery found in cars is a secondary cell • The anode for a lead-acid battery is lead metal • The cathode for a lead-acid battery is lead oxide • The lead-acid storage battery consists of Pb anodes alternating with Pb. O 2 cathodes, all immersed in sulfuric acid 77 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
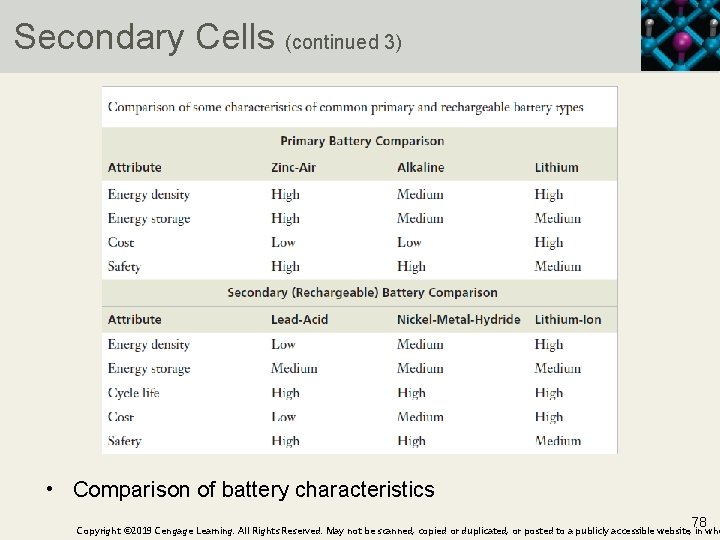
Secondary Cells (continued 3) • Comparison of battery characteristics 78 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Fuel Cells • A fuel cell is a voltaic cell in which the reactants can be supplied continuously and the products of the cell reaction are continuously removed • Most common type is based on the reaction of hydrogen and oxygen to produce water • Oxygen is reduced at the cathode • Hydrogen is oxidized at the anode 79 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Limitations of Batteries • Corrosion is a major cause for the loss of performance in batteries • Protective plating of materials used in batteries is an attempt to limit the performance-diminishing effects of corrosion on batteries 80 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 11 Which battery is NOT a secondary cell? • • Nickel-cadmium Nickel-metal-hydride Lead-acid Zinc-air 81 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 11, Answer • Zinc-air 82 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Electrolysis • Electrolysis is the process of passing an electric current through an ionic solution or molten salt to produce a chemical reaction • Electrolytic cells are divided into two categories based on the nature of the electrodes used • Passive electrolysis: The electrodes are chemically inert materials that simply provide a path for electrons • Active electrolysis: The electrodes are part of the electrolytic reaction 83 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Electrolysis and Polarity • Electrolysis changes the polarity of the electrodes in a system • For reduction, electrons are forced to the cathode • The cathode becomes the negative electrode • For oxidation, electrons are pulled from the anode • The anode becomes the positive electrode • In electrolysis, an external source of current drives a redox reaction that would otherwise not be spontaneous • The flow of ions through the solution completes the circuit 84 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Passive Electrolysis in Refining Aluminum • Electrolysis provides the means to overcome the nonspontaneous reaction of separating aluminum from its oxide • The Hall–Heroult refining process uses carbon electrodes as inert sites for passive electrolysis • The Hall–Heroult process involves the electrolytic refining of aluminum from Al 2 O 3 to produce aluminum metal and oxygen gas 85 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Active Electrolysis and Electroplating • The process of depositing a thin coat of metal on another metal by using electrolysis is called electroplating • In some cases, the thin coating is cosmetic or used to provide some vital functionality for the coated piece, such as corrosion resistance or desirable conducting properties • Silver is plated onto electrical devices because silver is a good conductor and resistant to corrosion • The solution from which silver is plated contains CN−(aq) ions, which form a complex with Ag+ • The need for uniform coatings makes this step of complex formation an important step 86 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Active Electrolysis and Electroplating (continued 1) • The object being electroplated is the cathode • Anode • Cathode • Opposite reactions at the anode and cathode are common for electroplating operations • Silver is transferred from the anode to the cathode, coating the cathode in a thin layer of silver • The zero cell potential is not critical since an external current drives electrolysis 87 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
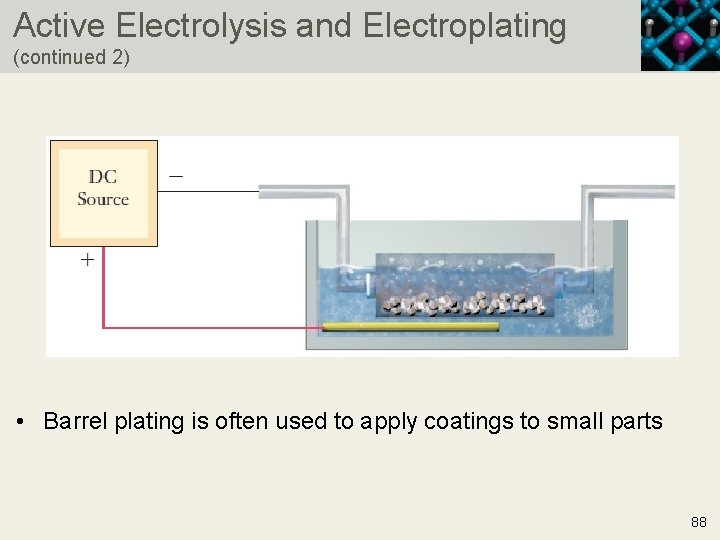
Active Electrolysis and Electroplating (continued 2) • Barrel plating is often used to apply coatings to small parts 88

Electrolysis and Stoichiometry • For electroplating, it can be vitally important to use carefully controlled amounts of materials • Controlling the flow of electrons, or current, in an electroplating operation provides a method to accurately limit the amount of material deposited • Electroplating is often used to prevent galvanic corrosion in an electrical apparatus in places where different metals come into contact with one another 89 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Current and Charge • When current is measured in an electric circuit, the observation is the flow of charge for a period of time • The unit of current, the ampere or A, is defined as one coulomb per second: 1 A = 1 C s− 1 • If a known current flows through a circuit for a known time, the charge can be easily calculated 90 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Current and Charge (continued) • Using Faraday’s constant, F = 96, 485 C mol− 1 and the calculated charge, the number of moles of electrons that pass through the circuit can be calculated • If the number of electrons required to reduce each metal cation is known, the number of moles of material plated can be calculated • Electricity use is often measured in terms of power • The SI unit for power is the watt: 1 watt = 1 J s− 1 • Electrical utilities normally determine consumption in kilowatthours, k. Wh: 1 k. Wh = 3. 60 × 106 J 91 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Example Problem 13. 4 • In a process called flash electroplating, a current of 2. 50 x 103 A passes through an electrolytic cell for 5. 00 minutes. How many moles of electrons are driven through the cell? 92 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Example Problem 13. 5 • Suppose that a batch of parts is plated with copper in an electrolytic bath running at 0. 15 V and 15. 0 A for exactly 2 hours. What is the energy cost of this process if the electric utility charges the company $0. 0500 per k. Wh? 93 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Calculations Using Masses of Substances in Electrolysis • A knowledge of current, how long the current flows, stoichiometry, and the number of electrons required to reduce a metal cation are used to answer the following questions: • How much material is plated given a specific current for an allotted time or electrical energy expenditure? • How long must a given current pass through the cell to yield a desired mass of plated material? 94 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Example Problem 13. 6 • An electrolysis cell that deposits gold (from Au+(aq)) operates for 15. 0 minutes at a current of 2. 30 A. What mass of gold is deposited? 95 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Example Problem 13. 7 • Suppose that you have a part that requires tin coating. You have calculated that you need to deposit 3. 60 g of tin to achieve an adequate coating. If your electrolysis cell (using Sn 2+) runs at 2. 00 A, how long must you operate the cell to obtain the desired coating? 96 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 12 When the electrode is part of the electrolytic process, what type of electrolysis is occurring? • Galvanic • Active • Passive 97 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 12, Answer • Active 98 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 13 If you need to plate 15. 7 mg of gold from a Au+ solution, how long would you need to run the electrolysis at 0. 050 A? • • 1. 6 sec 7. 7 sec 154 sec 7700 sec 99 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 13, Answer • 154 sec 100 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Batteries in Engineering Design • Lithium-ion batteries have recently come under scrutiny because of a high-profile fire during the first commercial flight of the Boeing Dreamliner 787 • More recently, Samsung was forced to cancel production of its Galaxy Note 7 and recall millions of the phones after a series of incidents in which batteries caught fire 101 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Batteries in Engineering Design (continued 1) • What factors led to the Dreamliner’s Problems? • The Dreamliner requires higher levels of electrical power compared to conventional aircrafts due in large part to the replacement of the traditional hydraulic system for controlling the flight of the plane with an all-electronic alternative • Increased fuel efficiency was a major goal, so it was important to minimize weight • Lithium-ion batteries have high energy capacity and low weight 102 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
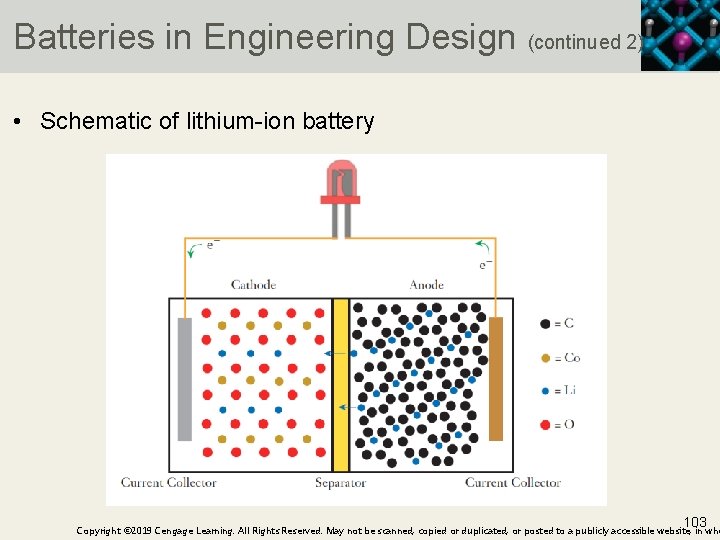
Batteries in Engineering Design (continued 2) • Schematic of lithium-ion battery 103 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Batteries in Engineering Design (continued 3) • The anode is a form of graphite into which lithium atoms have been incorporated, or “intercalated” • The cathode is cobalt oxide that also has lithium incorporated into it • Because the electrodes both consist of solids with lithium embedded in them, it is difficult to write a simple chemical equation for the cell reaction • The chemistry is summarized reasonably by the following reactions: 104 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Batteries in Engineering Design (continued 4) • Two reasons explain why the energy density for a battery based on these half-reactions will be high • Li+/Li has one of the largest standard reduction potentials • Both lithium and carbon are relatively light • High temperatures cause lithium-ion batteries to degrade fairly quickly, so engineering designs that use them generally must account for this 105

Join in, 14 What chemical strategy do rust inhibitors in paint often employ? • Passivation of the iron oxide layer to prevent further corrosion • Sacrificial oxidation of the inhibitors themselves • Raising the oxidation potential of the iron before rust occurs 106 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who

Join in, 14, Answer • Passivation of the iron oxide layer to prevent further corrosion 107 Copyright © 2019 Cengage Learning. All Rights Reserved. May not be scanned, copied or duplicated, or posted to a publicly accessible website, in who
- Slides: 107