Chapter 12 Unsaturated Hydrocarbons 12 2 CisTrans Isomers

Chapter 12 Unsaturated Hydrocarbons 12. 2 Cis-Trans Isomers 1
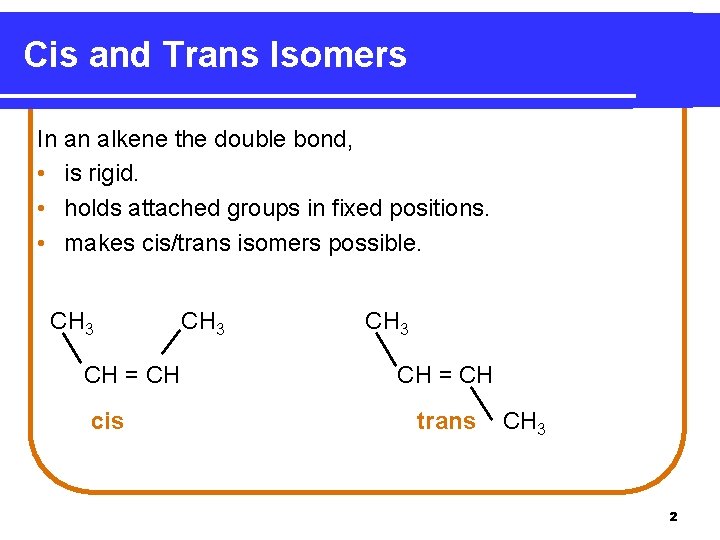
Cis and Trans Isomers In an alkene the double bond, • is rigid. • holds attached groups in fixed positions. • makes cis/trans isomers possible. CH 3 CH = CH cis CH 3 CH = CH trans CH 3 2

Cis-Trans Isomers In cis-trans isomers • there is no rotation around the double bond in alkenes. • groups attached to the double bond are fixed relative to each other. You can make a “double bond” with your fingers with both thumbs on the same side or opposite from each other. 3
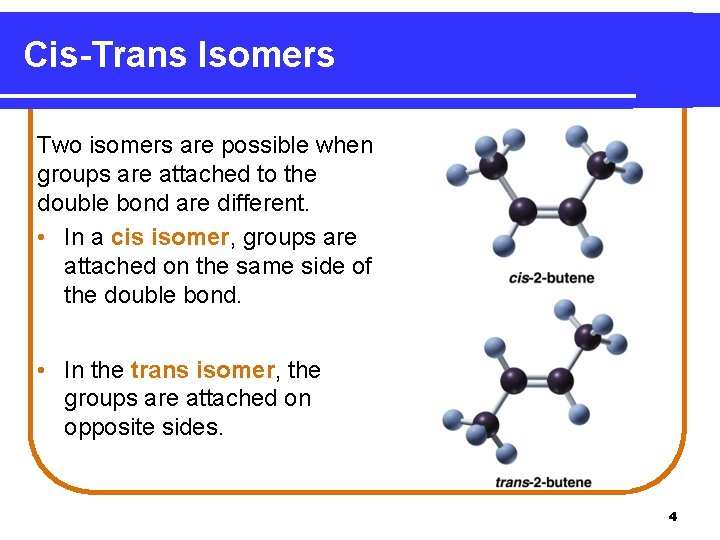
Cis-Trans Isomers Two isomers are possible when groups are attached to the double bond are different. • In a cis isomer, groups are attached on the same side of the double bond. • In the trans isomer, the groups are attached on opposite sides. 4
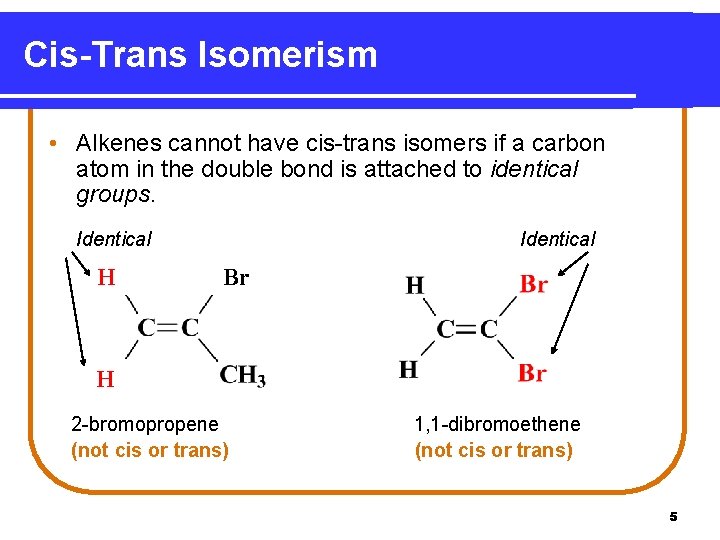
Cis-Trans Isomerism • Alkenes cannot have cis-trans isomers if a carbon atom in the double bond is attached to identical groups. Identical H Identical Br H H 2 -bromopropene (not cis or trans) 1, 1 -dibromoethene (not cis or trans) 5
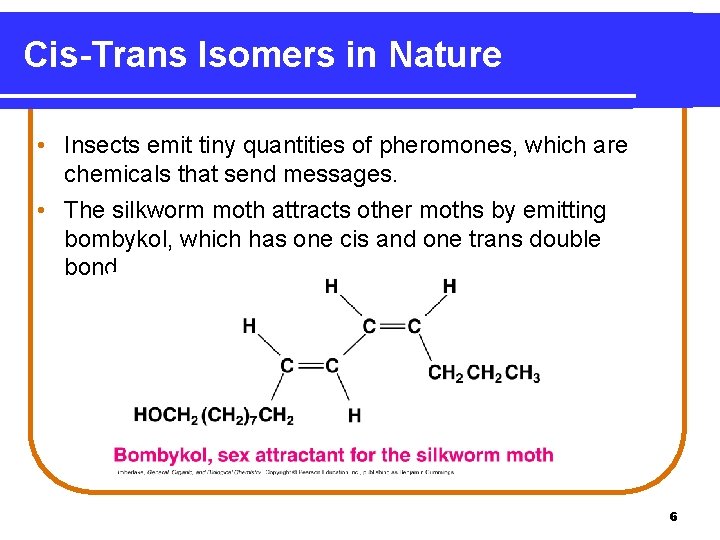
Cis-Trans Isomers in Nature • Insects emit tiny quantities of pheromones, which are chemicals that send messages. • The silkworm moth attracts other moths by emitting bombykol, which has one cis and one trans double bond. 6
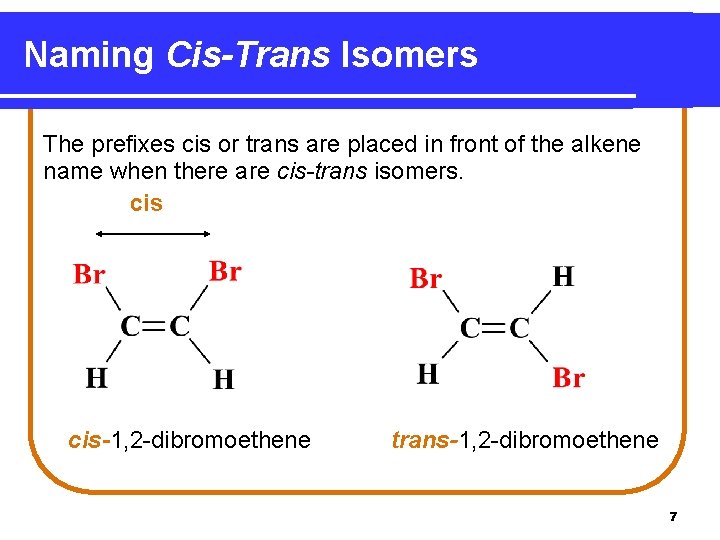
Naming Cis-Trans Isomers The prefixes cis or trans are placed in front of the alkene name when there are cis-trans isomers. cis-1, 2 -dibromoethene trans-1, 2 -dibromoethene 7
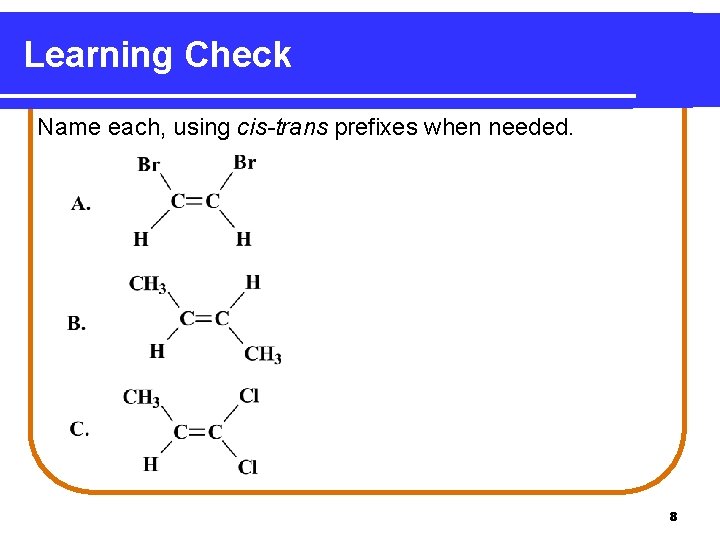
Learning Check Name each, using cis-trans prefixes when needed. 8
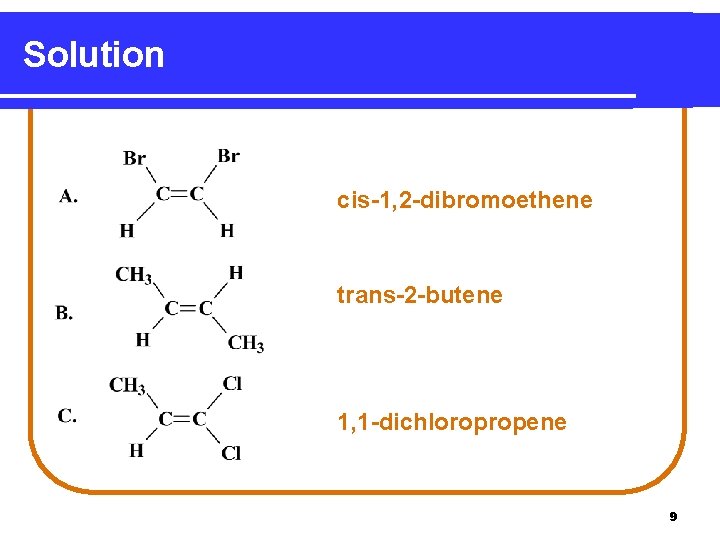
Solution cis-1, 2 -dibromoethene trans-2 -butene 1, 1 -dichloropropene 9
- Slides: 9