Chapter 12 Unsaturated Hydrocarbons 12 1 Alkenes and

Chapter 12 Unsaturated Hydrocarbons 12. 1 Alkenes and Alkynes 1
Saturated Hydrocarbons Saturated hydrocarbons • have the maximum number of hydrogen atoms attached to each carbon atom. • are alkanes and cycloalkanes with single C-C bonds. CH 3—CH 2—CH 3 2
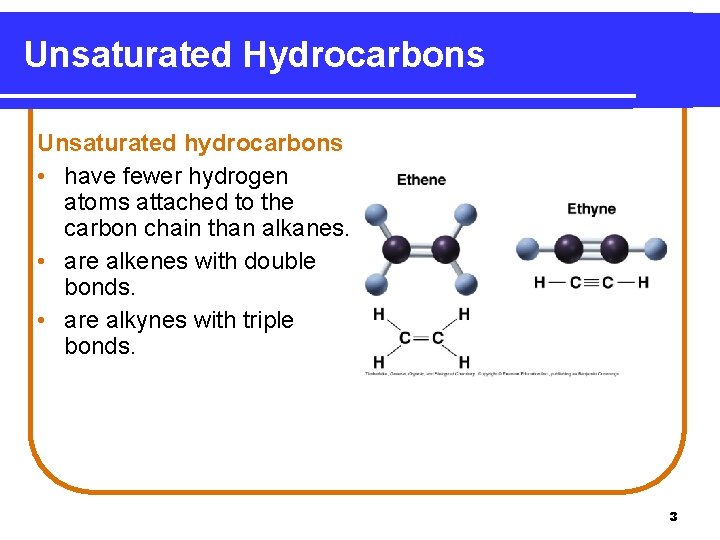
Unsaturated Hydrocarbons Unsaturated hydrocarbons • have fewer hydrogen atoms attached to the carbon chain than alkanes. • are alkenes with double bonds. • are alkynes with triple bonds. 3
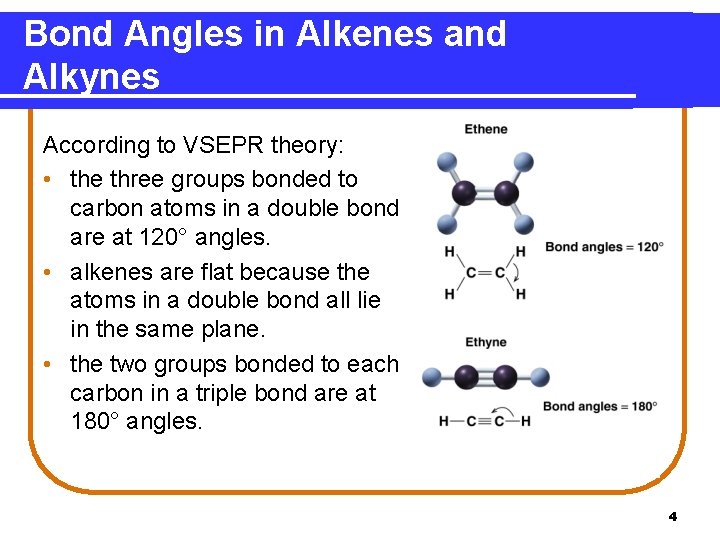
Bond Angles in Alkenes and Alkynes According to VSEPR theory: • the three groups bonded to carbon atoms in a double bond are at 120° angles. • alkenes are flat because the atoms in a double bond all lie in the same plane. • the two groups bonded to each carbon in a triple bond are at 180° angles. 4
Naming Alkenes The names of alkenes • use the corresponding alkane name. • change the ending to –ene. Alkene IUPAC Common H 2 C=CH 2 ethene ethylene H 2 C=CH─CH 3 propene propylene cyclohexene 5

Ethene (Ethylene) Ethene or ethylene • is an alkene with the formula C 2 H 4. • has two carbon atoms connected by a double bond. • has two H atoms bonded to each C atom. • is flat with all the C and H atoms in the same plane. • is used to accelerate the ripening of fruits. 6
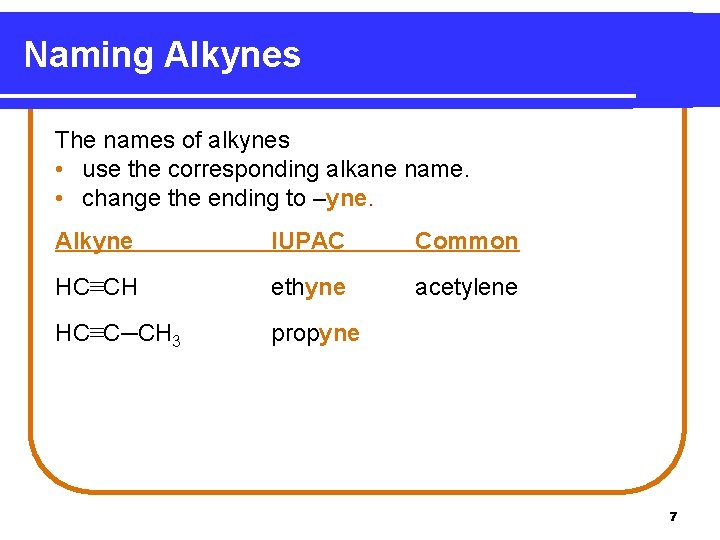
Naming Alkynes The names of alkynes • use the corresponding alkane name. • change the ending to –yne. Alkyne IUPAC Common HC≡CH ethyne acetylene HC≡C─CH 3 propyne 7
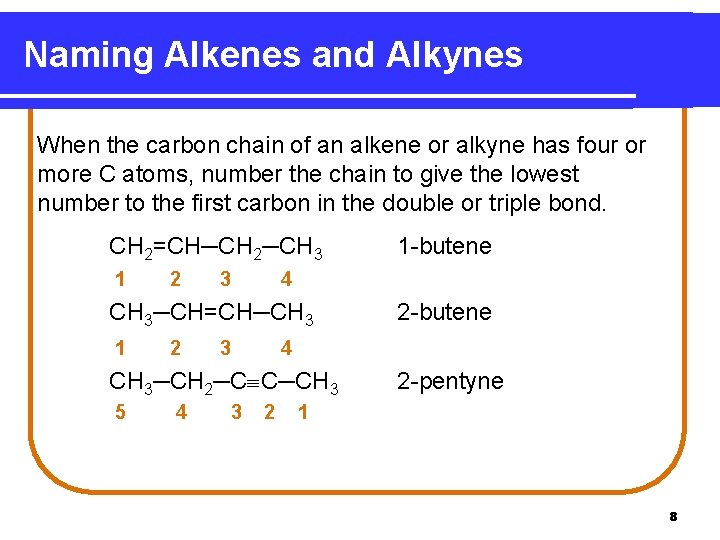
Naming Alkenes and Alkynes When the carbon chain of an alkene or alkyne has four or more C atoms, number the chain to give the lowest number to the first carbon in the double or triple bond. CH 2=CH─CH 2─CH 3 1 2 3 4 CH 3─CH=CH─CH 3 1 2 3 4 3 2 -butene 4 CH 3─CH 2─C C─CH 3 5 1 -butene 2 2 -pentyne 1 8
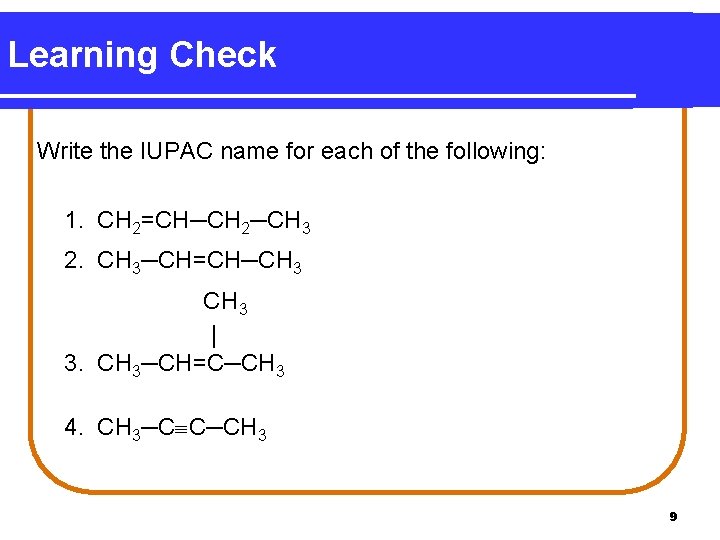
Learning Check Write the IUPAC name for each of the following: 1. CH 2=CH─CH 2─CH 3 2. CH 3─CH=CH─CH 3 | 3. CH 3─CH=C─CH 3 4. CH 3─C C─CH 3 9
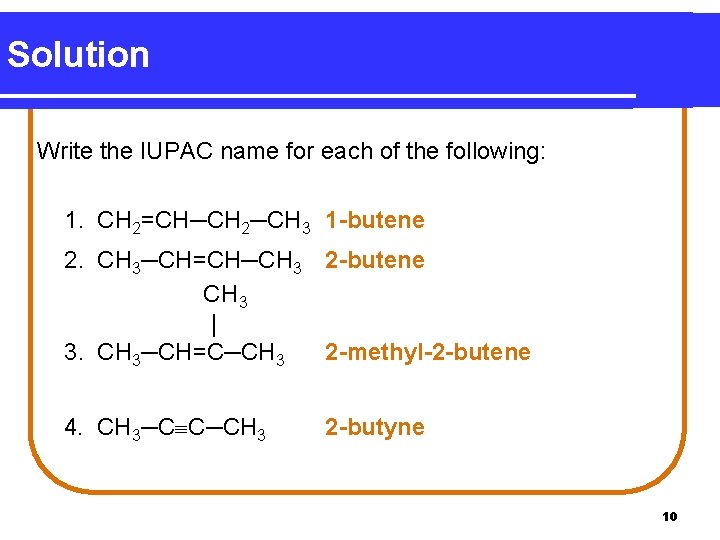
Solution Write the IUPAC name for each of the following: 1. CH 2=CH─CH 2─CH 3 1 -butene 2. CH 3─CH=CH─CH 3 2 -butene CH 3 | 3. CH 3─CH=C─CH 3 2 -methyl-2 -butene 4. CH 3─C C─CH 3 2 -butyne 10
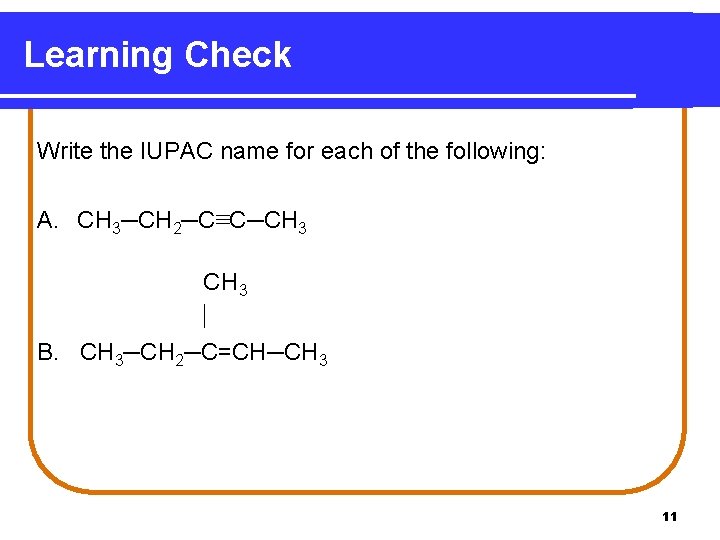
Learning Check Write the IUPAC name for each of the following: A. CH 3─CH 2─C≡C─CH 3 B. CH 3─CH 2─C=CH─CH 3 11
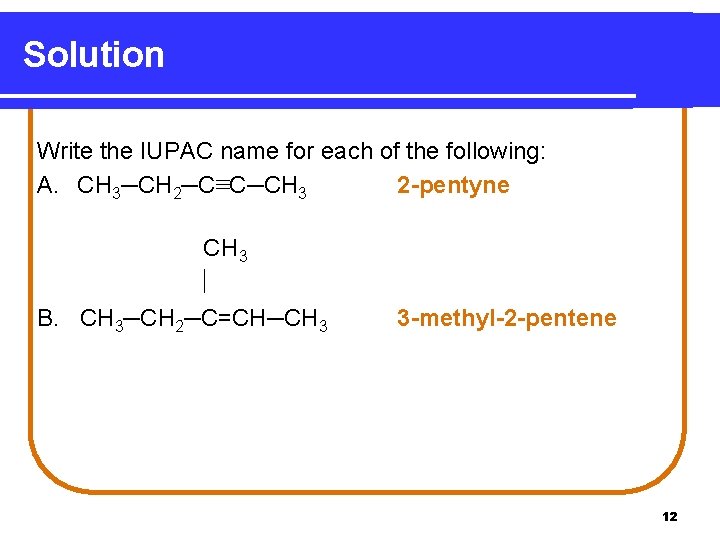
Solution Write the IUPAC name for each of the following: A. CH 3─CH 2─C≡C─CH 3 2 -pentyne CH 3 B. CH 3─CH 2─C=CH─CH 3 3 -methyl-2 -pentene 12
- Slides: 12