Chapter 12 Unsaturated Hydrocarbons 12 1 Alkenes and

Chapter 12 Unsaturated Hydrocarbons 12. 1 Alkenes and Alkynes Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 1
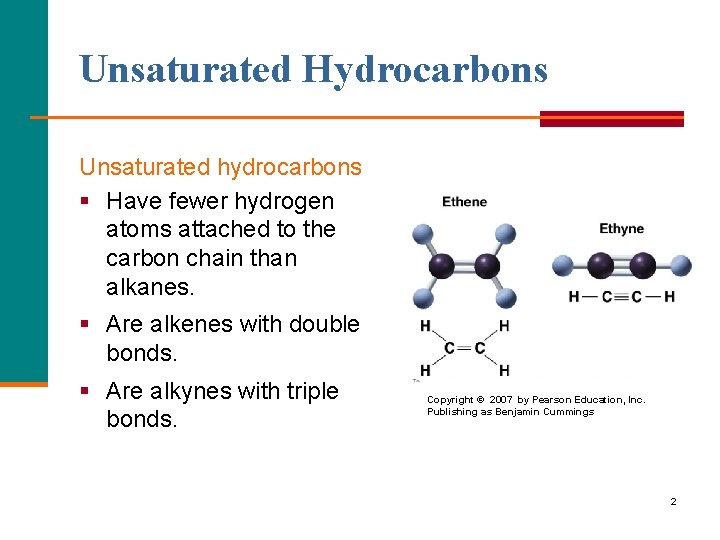
Unsaturated Hydrocarbons Unsaturated hydrocarbons § Have fewer hydrogen atoms attached to the carbon chain than alkanes. § Are alkenes with double bonds. § Are alkynes with triple bonds. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 2
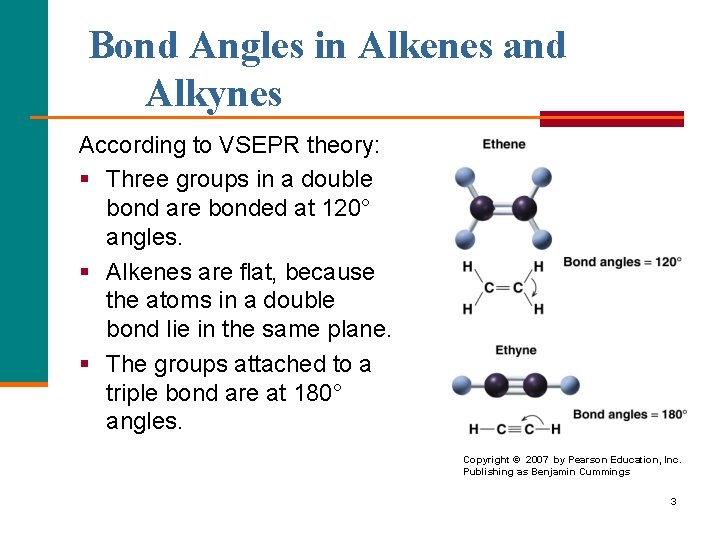
Bond Angles in Alkenes and Alkynes According to VSEPR theory: § Three groups in a double bond are bonded at 120° angles. § Alkenes are flat, because the atoms in a double bond lie in the same plane. § The groups attached to a triple bond are at 180° angles. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 3
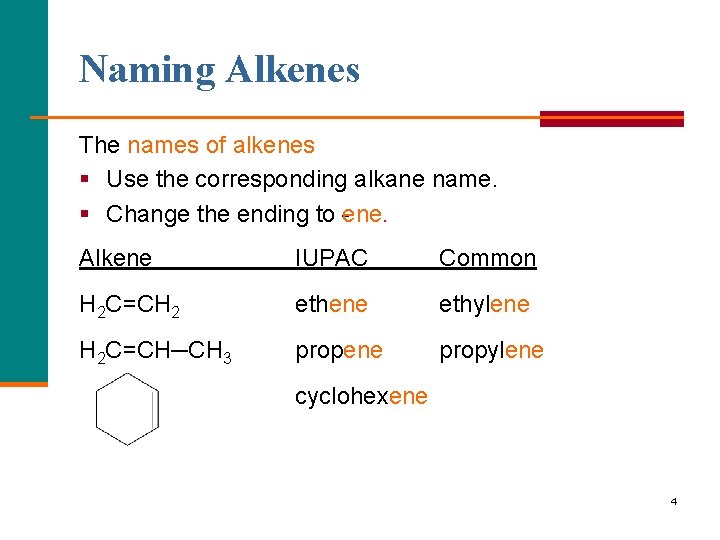
Naming Alkenes The names of alkenes § Use the corresponding alkane name. § Change the ending to ene. Alkene IUPAC Common H 2 C=CH 2 ethene ethylene H 2 C=CH─CH 3 propene propylene cyclohexene 4
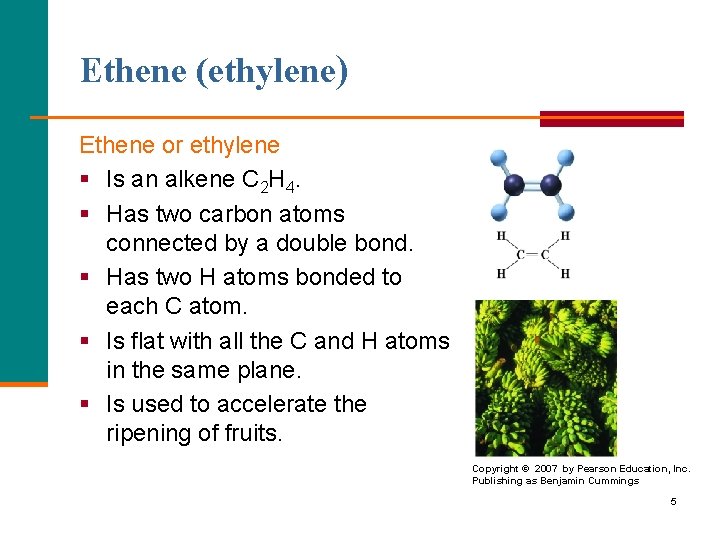
Ethene (ethylene) Ethene or ethylene § Is an alkene C 2 H 4. § Has two carbon atoms connected by a double bond. § Has two H atoms bonded to each C atom. § Is flat with all the C and H atoms in the same plane. § Is used to accelerate the ripening of fruits. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 5
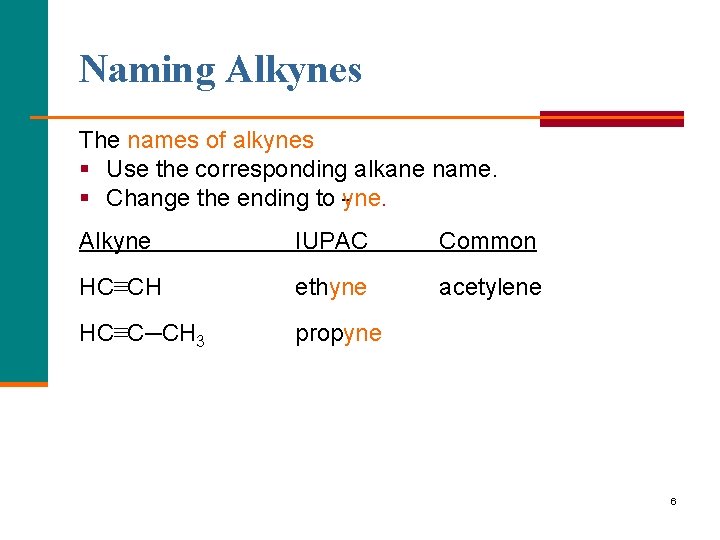
Naming Alkynes The names of alkynes § Use the corresponding alkane name. § Change the ending to yne. Alkyne IUPAC Common HC≡CH ethyne acetylene HC≡C─CH 3 propyne 6

Guide to Naming Alkenes and Alkynes Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 7
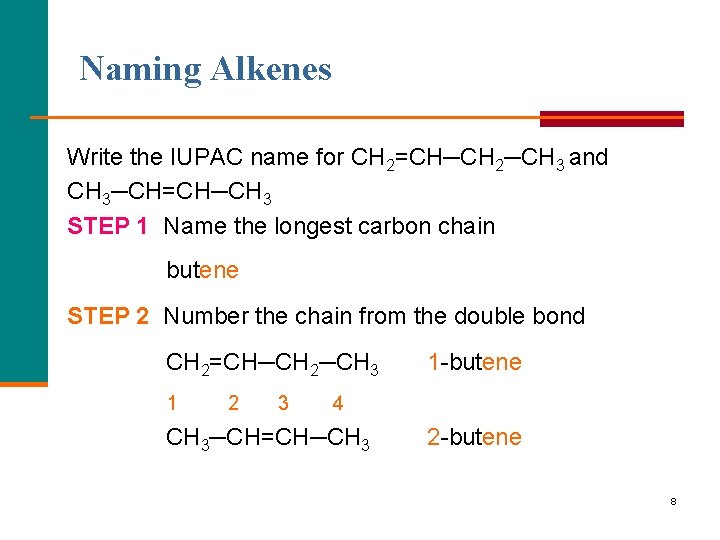
Naming Alkenes Write the IUPAC name for CH 2=CH─CH 2─CH 3 and CH 3─CH=CH─CH 3 STEP 1 Name the longest carbon chain butene STEP 2 Number the chain from the double bond CH 2=CH─CH 2─CH 3 1 2 3 1 butene 4 CH 3─CH=CH─CH 3 2 butene 8

Comparing Names of Alkanes, Alkenes, and Alkynes TABLE 12. 1 Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 9
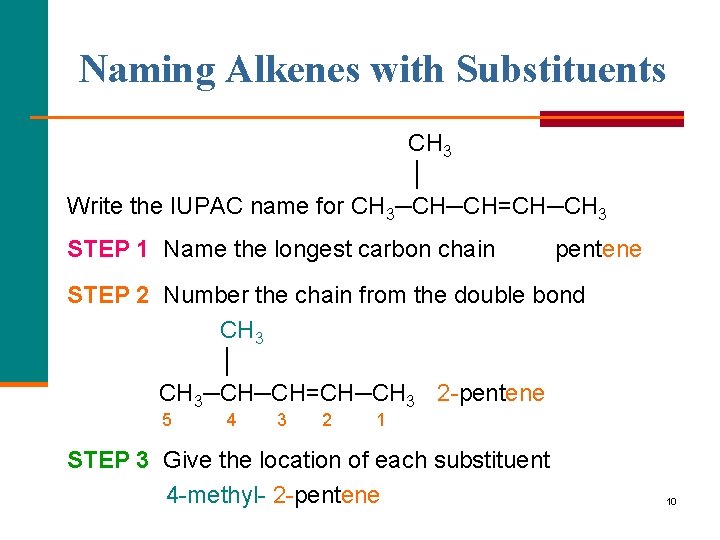
Naming Alkenes with Substituents CH 3 │ Write the IUPAC name for CH 3─CH─CH=CH─CH 3 STEP 1 Name the longest carbon chain pentene STEP 2 Number the chain from the double bond CH 3 │ CH 3─CH─CH=CH─CH 3 2 pentene 5 4 3 2 1 STEP 3 Give the location of each substituent 4 methyl 2 pentene 10
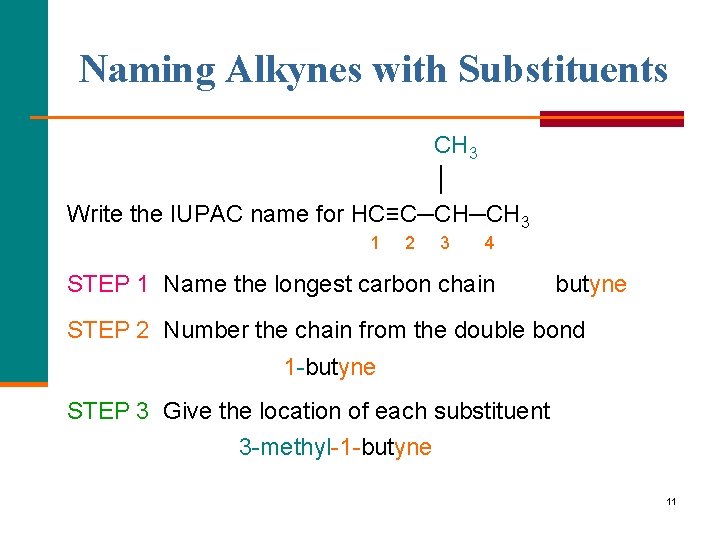
Naming Alkynes with Substituents CH 3 │ Write the IUPAC name for HC≡C─CH─CH 3 1 2 3 4 STEP 1 Name the longest carbon chain butyne STEP 2 Number the chain from the double bond 1 butyne STEP 3 Give the location of each substituent 3 methyl 1 butyne 11
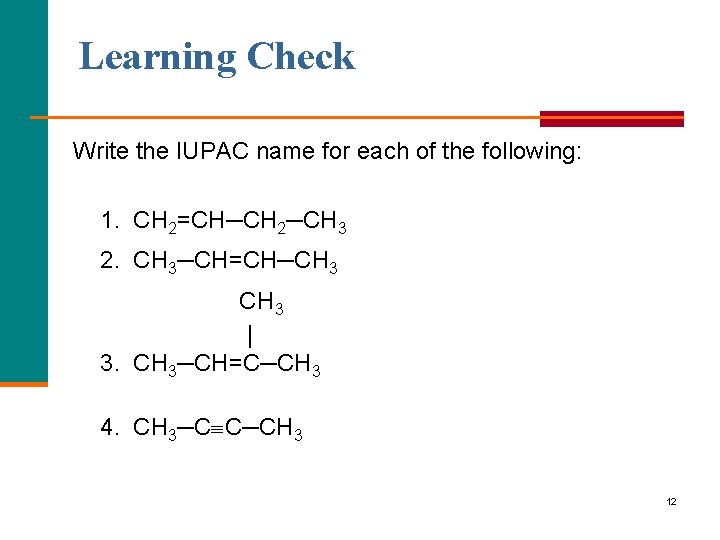
Learning Check Write the IUPAC name for each of the following: 1. CH 2=CH─CH 2─CH 3 2. CH 3─CH=CH─CH 3 | 3. CH 3─CH=C─CH 3 4. CH 3─C C─CH 3 12
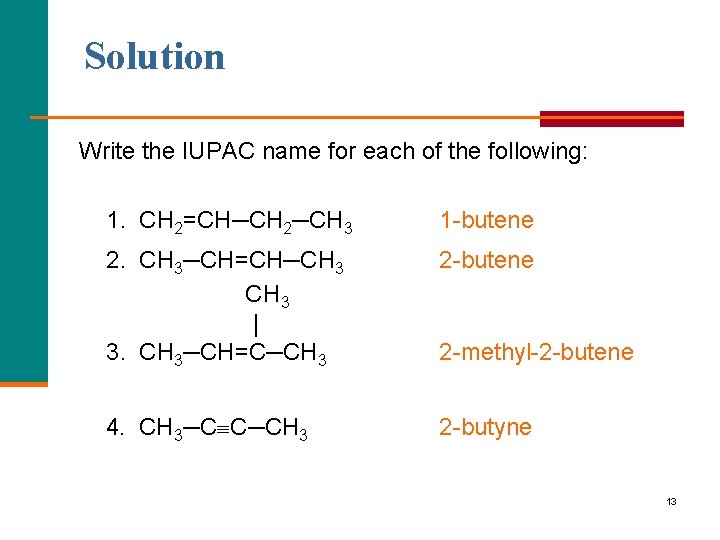
Solution Write the IUPAC name for each of the following: 1. CH 2=CH─CH 2─CH 3 1 butene 2. CH 3─CH=CH─CH 3 | 3. CH 3─CH=C─CH 3 2 butene 4. CH 3─C C─CH 3 2 butyne 2 methyl 2 butene 13

Learning Check Write the structural formula for each of the following: A. 2 pentyne B. 3 methyl 2 pentene 14
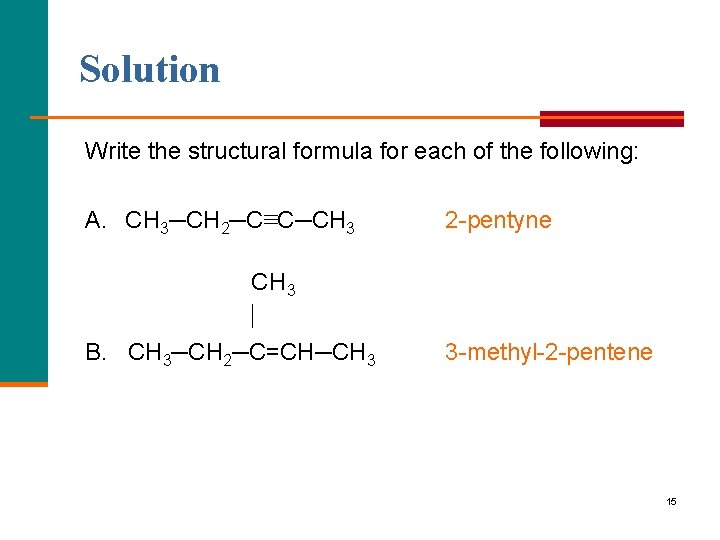
Solution Write the structural formula for each of the following: A. CH 3─CH 2─C≡C─CH 3 2 pentyne CH 3 B. CH 3─CH 2─C=CH─CH 3 3 methyl 2 pentene 15

Chapter 12 Unsaturated Hydrocarbons 12. 2 Cis-Trans Isomers Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 16
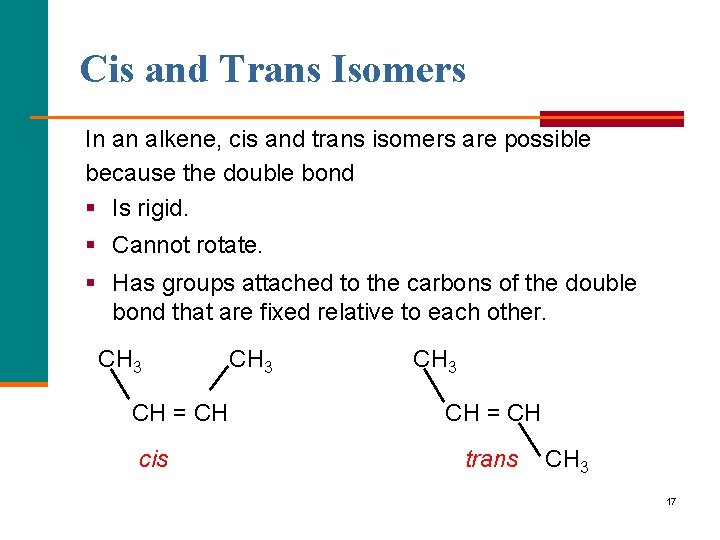
Cis and Trans Isomers In an alkene, cis and trans isomers are possible because the double bond § Is rigid. § Cannot rotate. § Has groups attached to the carbons of the double bond that are fixed relative to each other. CH 3 CH = CH cis CH 3 CH = CH trans CH 3 17

Cis-Trans Isomers Cis trans isomers § Can be modeled by making a “double bond” with your fingers with both thumbs on the same side or opposite from each other. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 18
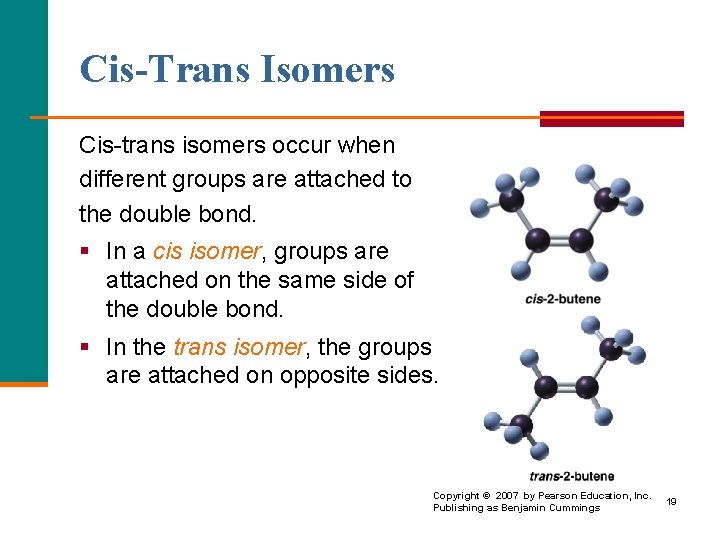
Cis-Trans Isomers Cis trans isomers occur when different groups are attached to the double bond. § In a cis isomer, groups are attached on the same side of the double bond. § In the trans isomer, the groups are attached on opposite sides. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 19
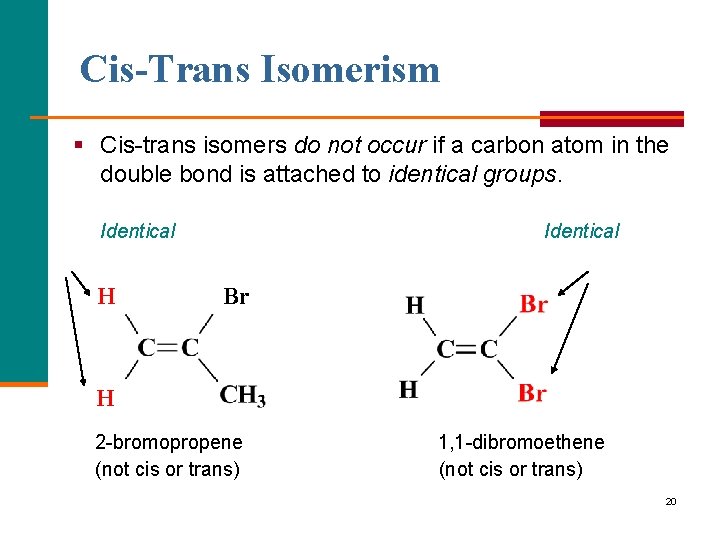
Cis-Trans Isomerism § Cis trans isomers do not occur if a carbon atom in the double bond is attached to identical groups. Identical H Identical Br H H 2 bromopropene (not cis or trans) 1, 1 dibromoethene (not cis or trans) 20
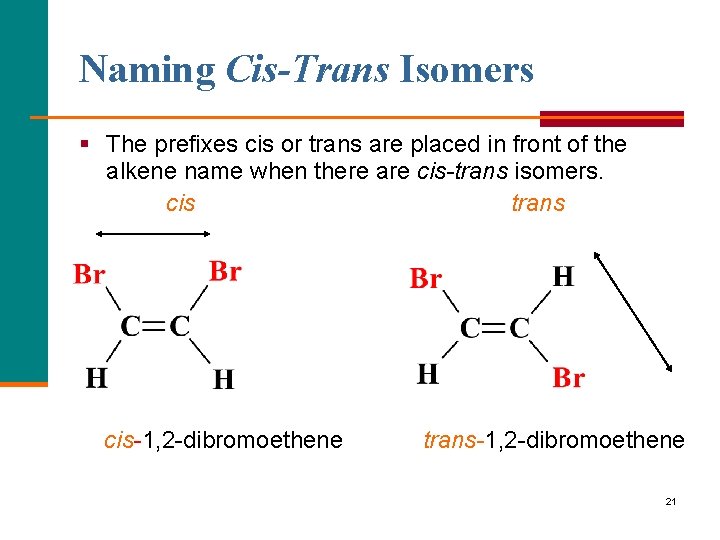
Naming Cis-Trans Isomers § The prefixes cis or trans are placed in front of the alkene name when there are cis-trans isomers. cis trans cis 1, 2 dibromoethene trans-1, 2 dibromoethene 21
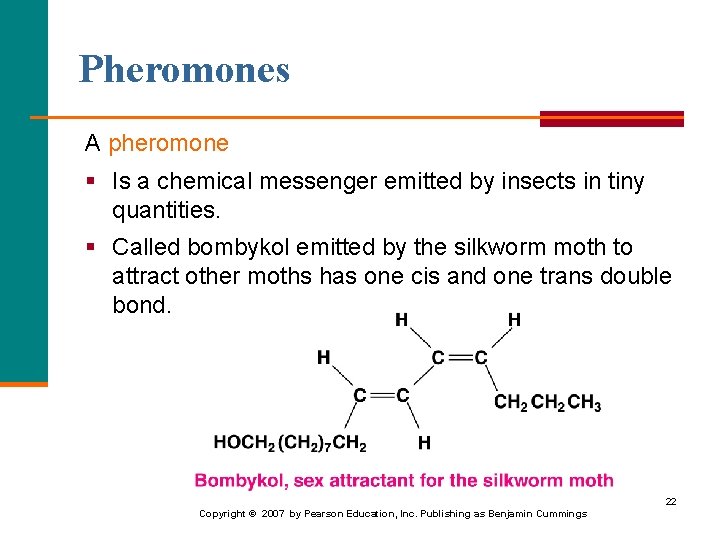
Pheromones A pheromone § Is a chemical messenger emitted by insects in tiny quantities. § Called bombykol emitted by the silkworm moth to attract other moths has one cis and one trans double bond. 22 Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings
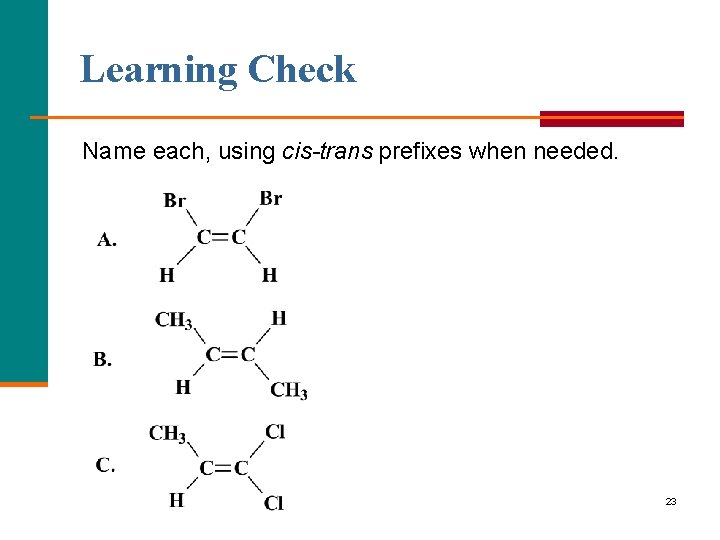
Learning Check Name each, using cis-trans prefixes when needed. 23
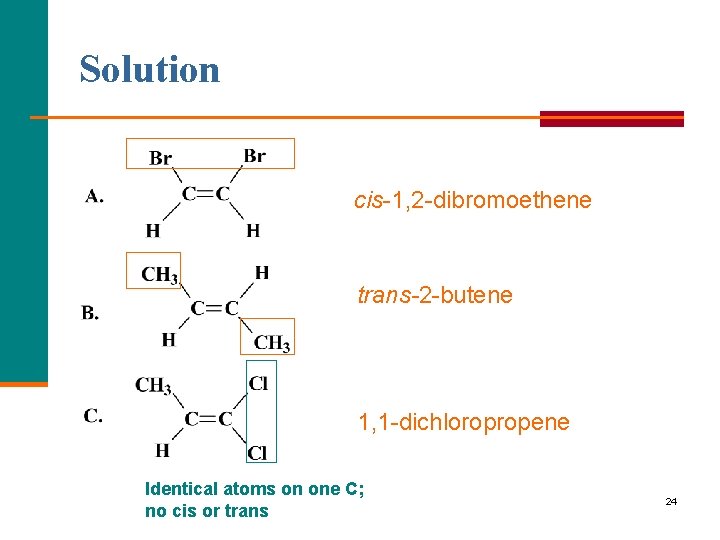
Solution cis 1, 2 dibromoethene trans-2 butene 1, 1 dichloropropene Identical atoms on one C; no cis or trans 24

Chapter 12 Unsaturated Hydrocarbons 12. 3 Addition Reactions Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 25

Addition Reactions In addition reactions, § Reactants add to the carbon atoms in double or triple bonds. § A double or triple bond is easily broken, which makes them very reactive. TABLE 12. 2 Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 26
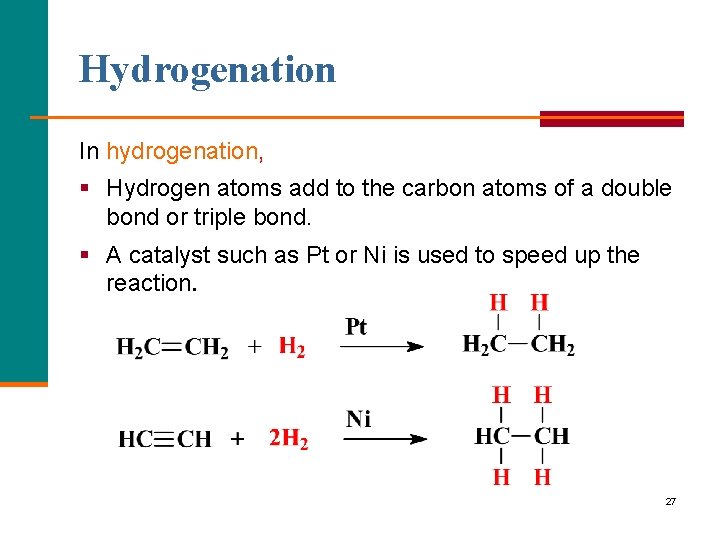
Hydrogenation In hydrogenation, § Hydrogen atoms add to the carbon atoms of a double bond or triple bond. § A catalyst such as Pt or Ni is used to speed up the reaction. 27

Hydrogenation of Oils Adding H 2 to double bonds in vegetable oils produces § Compounds with higher melting points. § Solids at room temperature such as margarine, soft margarine, Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings and shortening. 28

Learning Check Write an equation for the hydrogenation of 1 butene using a platinum catalyst. 29
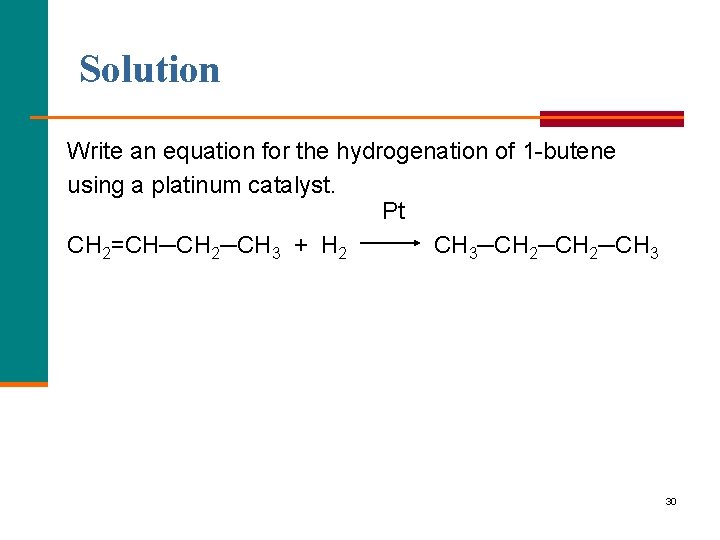
Solution Write an equation for the hydrogenation of 1 butene using a platinum catalyst. Pt CH 2=CH─CH 2─CH 3 + H 2 CH 3─CH 2─CH 3 30

Trans Fats In vegetable oils, the unsaturated fats usually contain cis double bonds. § During hydrogenation, some cis double bonds are converted to trans double bonds (more stable) causing a change in the fatty acid structure § If a label states “partially” or “fully hydrogenated”, the fats contain trans fatty acids. 31

Learning Check (T) True or (F) False A. ____ There are more unsaturated fats in vegetable oils. B. ____ Hydrogenation converts some cis-double bonds to trans- double bonds. C. ____ Animal fats have more unsaturated fats. 32

Solution (1) True or (2) False A. T There are more unsaturated fats in vegetable oils. B. T Hydrogenation of oils converts some cis-double bonds to trans- double bonds. C. F Animal fats have more unsaturated fats. 33
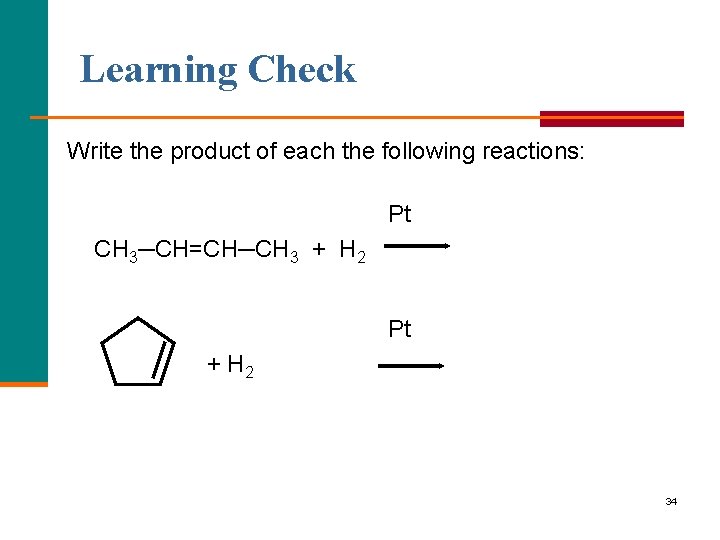
Learning Check Write the product of each the following reactions: Pt CH 3─CH=CH─CH 3 + H 2 Pt + H 2 34
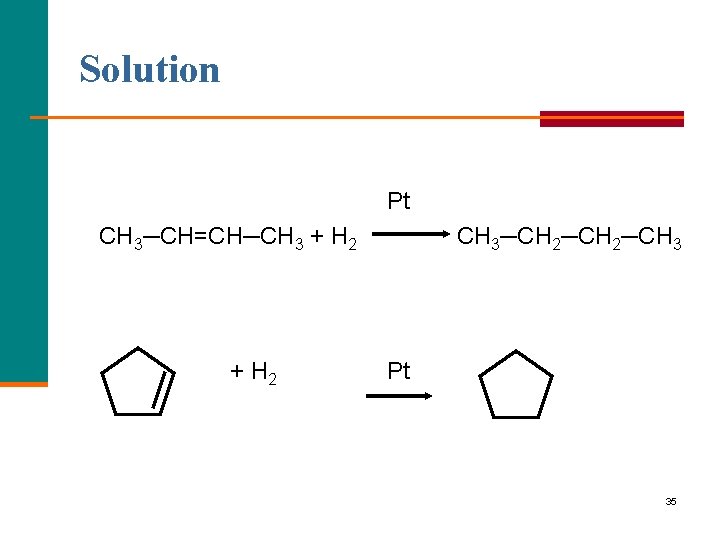
Solution Pt CH 3─CH=CH─CH 3 + H 2 CH 3─CH 2─CH 3 Pt 35
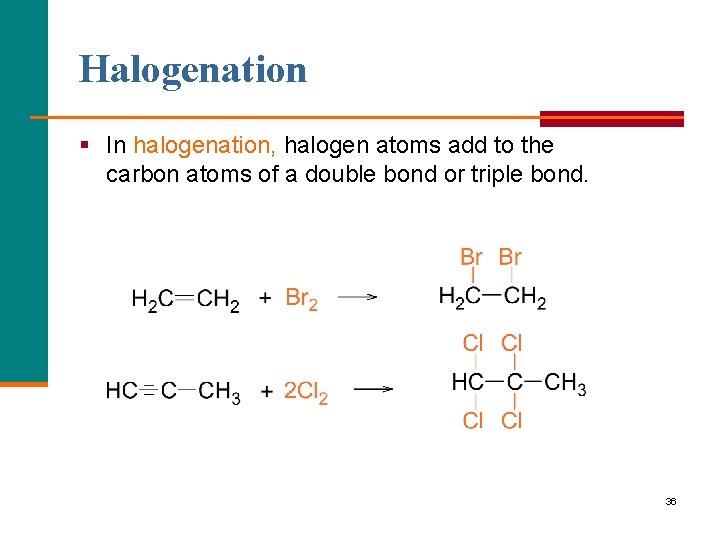
Halogenation § In halogenation, halogen atoms add to the carbon atoms of a double bond or triple bond. 36
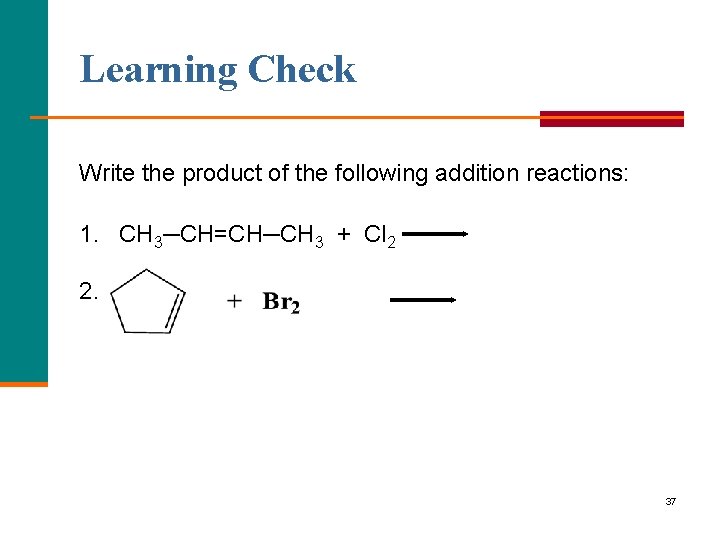
Learning Check Write the product of the following addition reactions: 1. CH 3─CH=CH─CH 3 + Cl 2 2. 37
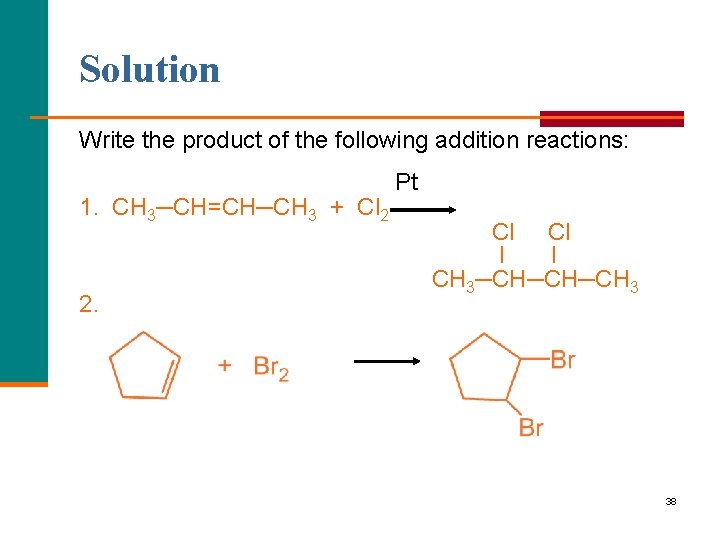
Solution Write the product of the following addition reactions: 1. CH 3─CH=CH─CH 3 + Cl 2 2. Pt Cl Cl l l CH 3─CH─CH─CH 3 38

Testing for Unsaturation Br 2 § When bromine (Br 2) is added to an alkane, the red color of bromine persists. § When bromine (Br 2) is added to an alkene or alkyne, the red color of bromine disappears immediately. Br 2 39 Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 15
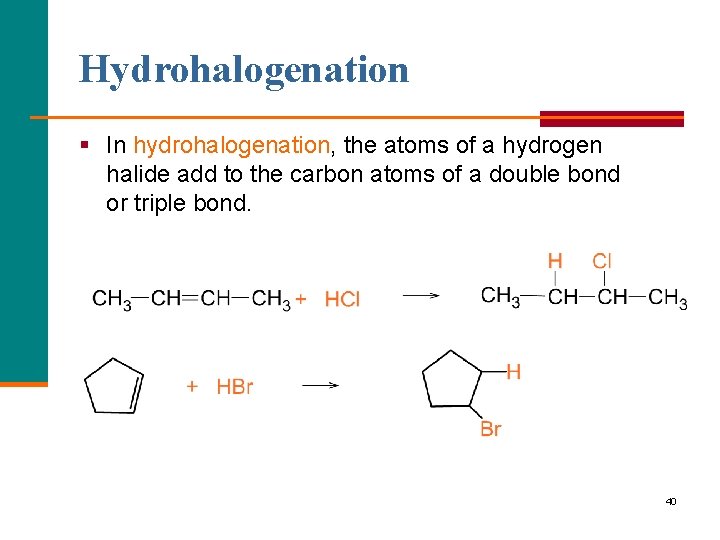
Hydrohalogenation § In hydrohalogenation, the atoms of a hydrogen halide add to the carbon atoms of a double bond or triple bond. 40
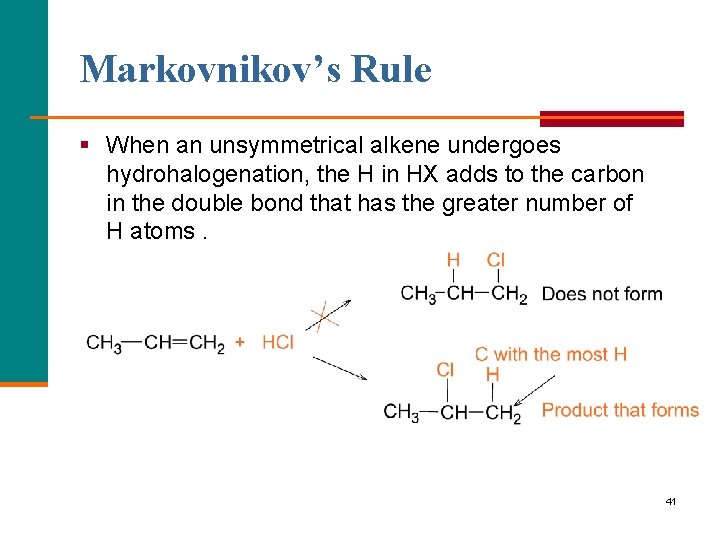
Markovnikov’s Rule § When an unsymmetrical alkene undergoes hydrohalogenation, the H in HX adds to the carbon in the double bond that has the greater number of H atoms. 41

Hydration In the addition reaction called hydration § An acid H+ catalyst is required. § Water (HOH) adds to a double bond. § An H atom bonds to one C in the double bond. § An OH bonds to the other C. H OH H+ │ │ CH 3─CH=CH─CH 3 + H─OH CH 3─CH─CH─CH 3 42

Hydration When hydration occurs with a double bond that has an unequal number of H atoms, § The H atom bonds to the C in the double bond with the most H. § The OH bonds to the C in the double bond with the fewest H atoms. OH H H+ │ │ CH 3─CH=CH 2 + H─OH CH 3─CH─CH 2 (1 H) (2 H) 43
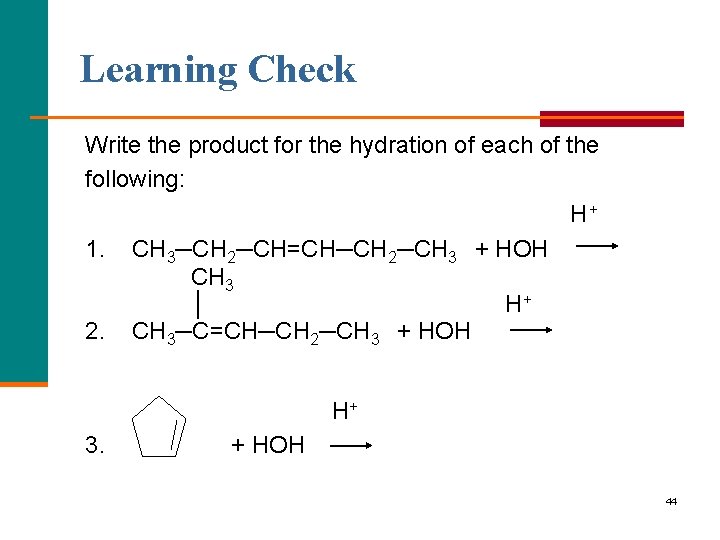
Learning Check Write the product for the hydration of each of the following: H+ 1. CH 3─CH 2─CH=CH─CH 2─CH 3 + HOH CH 3 │ H+ 2. CH 3─C=CH─CH 2─CH 3 + HOH H+ 3. + HOH 44
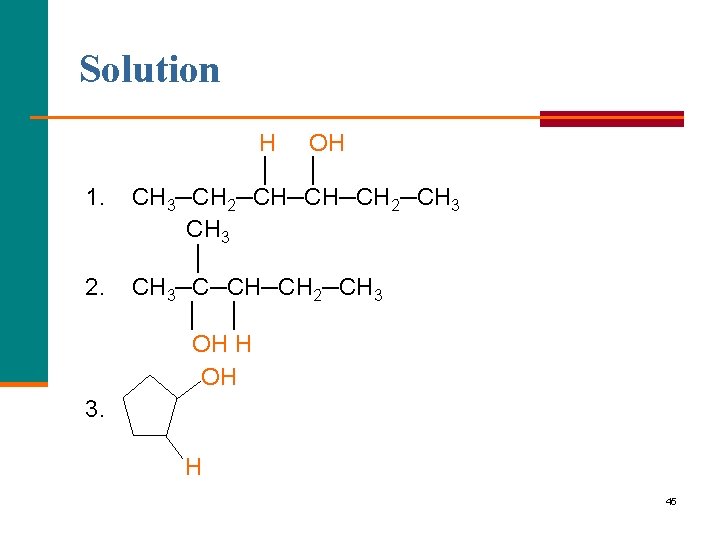
Solution 1. 2. H OH │ │ CH 3─CH 2─CH─CH─CH 2─CH 3 │ CH 3─C─CH─CH 2─CH 3 │ │ OH H OH 3. H 45
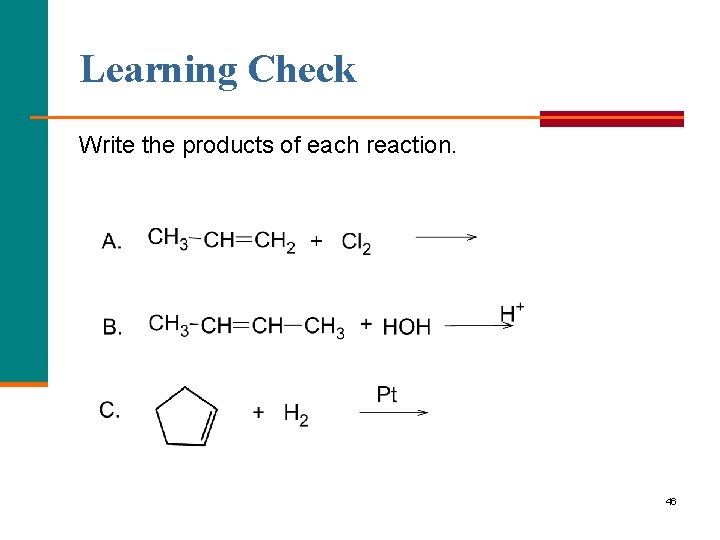
Learning Check Write the products of each reaction. 46
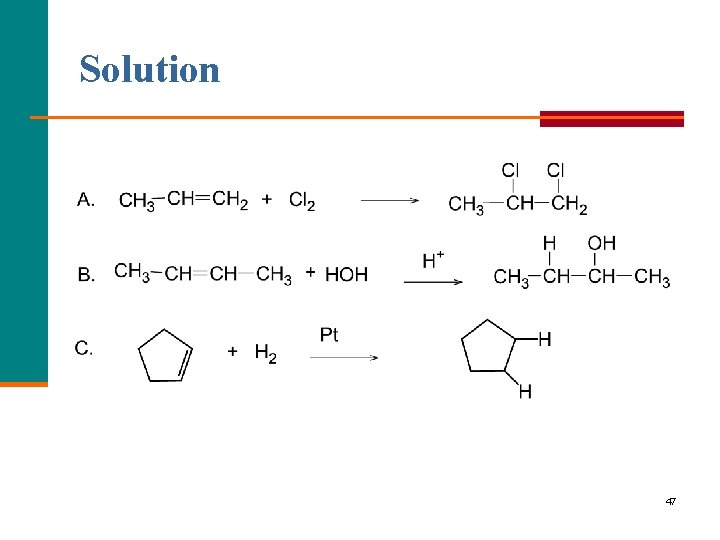
Solution 47

Chapter 12 Unsaturated Hydrocarbons 12. 4 Polymers of Alkenes Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 48

Polymers are § Large, long chain molecules. § Found in nature, including cellulose in plants, starches in food, proteins and DNA in the body. § Also synthetic such as polyethylene and polystyrene, Teflon, and nylon. § Made up of small repeating units called monomers. § Made by reaction of small alkenes. 49
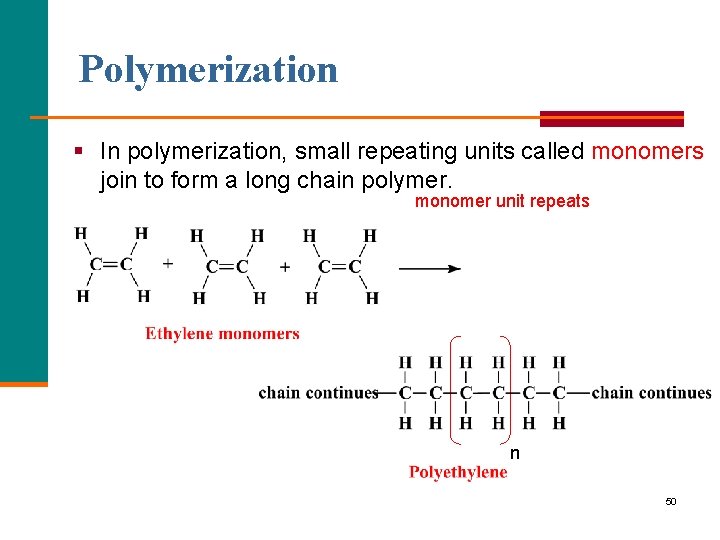
Polymerization § In polymerization, small repeating units called monomers join to form a long chain polymer. monomer unit repeats n 50

Common Synthetic Polymers Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 51

Polymers from Addition Reactions TABLE 12. 3 Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 52
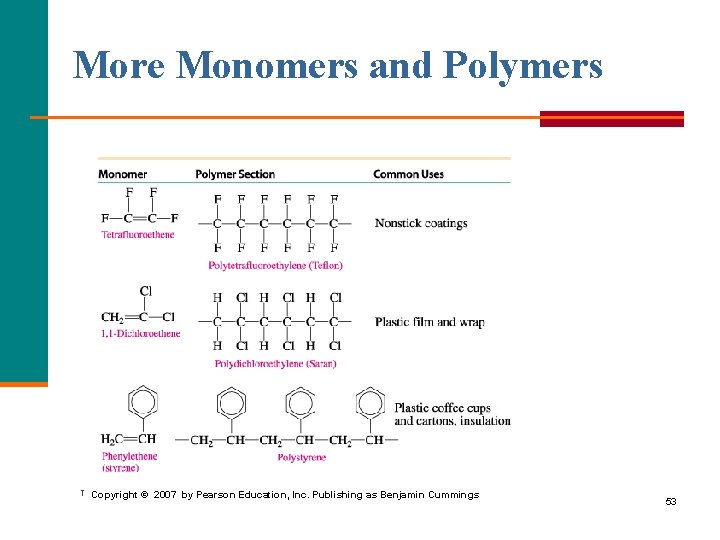
More Monomers and Polymers Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 53

Learning Check What is the starting monomer for polyethylene? 54
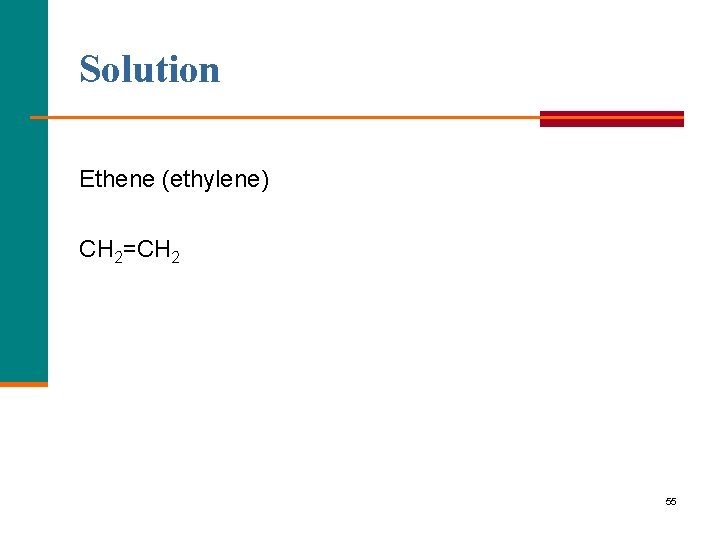
Solution Ethene (ethylene) CH 2=CH 2 55

Learning Check Name the monomer used to make Teflon and write a portion of a Teflon polymer using four monomers. 56
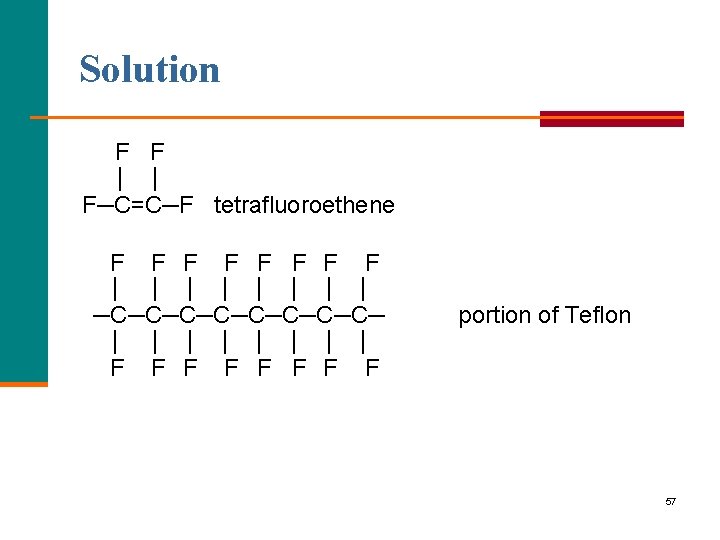
Solution F F │ │ F─C=C─F tetrafluoroethene F F F F │ │ │ │ F │ ─C─C─C─C─ │ │ │ F F F │ │ F F portion of Teflon │ F 57

Recycling Plastics Recycling is simplified by using codes found on plastic items. 1 2 3 4 5 6 PETE Polyethyleneterephtalate HDPE High density polyethylene PV Polyvinyl chloride LDPE Low density polyethylene PP Polypropylene PS Polystyrene Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 58

Learning Check What types of plastic are indicated by the following codes? 3 PV A. B. C. 5 PP 6 PS 59

Solution What types of plastic are indicated by the following codes? A. B. C. . 3 PV 5 PP 6 PS Polyvinyl chloride Polypropylene Polystyrene 60

Chapter 12 Unsaturated Hydrocarbons 12. 5 Aromatic Compounds Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 61
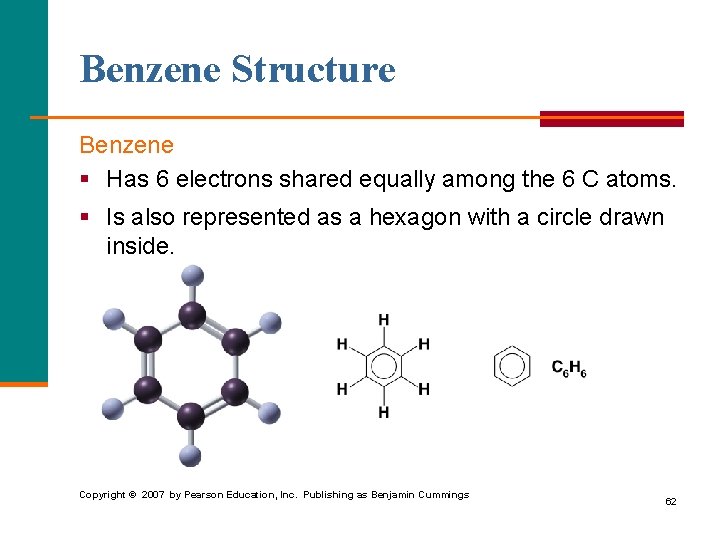
Benzene Structure Benzene § Has 6 electrons shared equally among the 6 C atoms. § Is also represented as a hexagon with a circle drawn inside. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 62
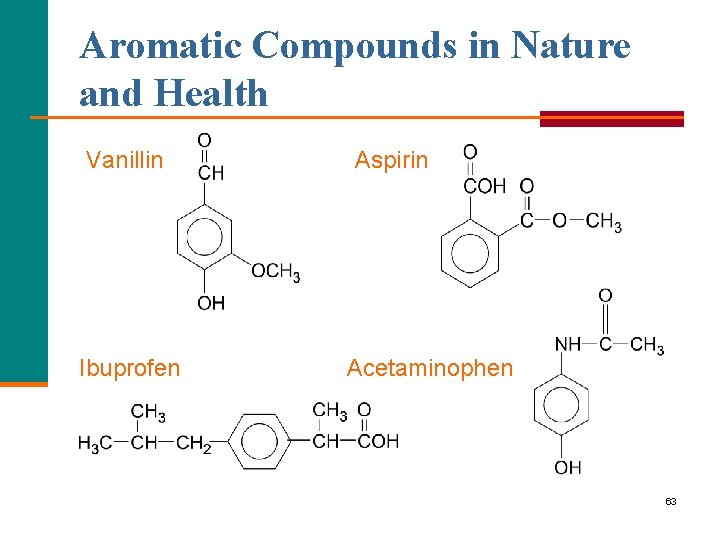
Aromatic Compounds in Nature and Health Vanillin Ibuprofen Aspirin Acetaminophen 63

Naming Aromatic Compounds Aromatic compounds are named § With benzene as the parent chain. § With one side group named in front of benzene. methylbenzene chlorobenzene 64
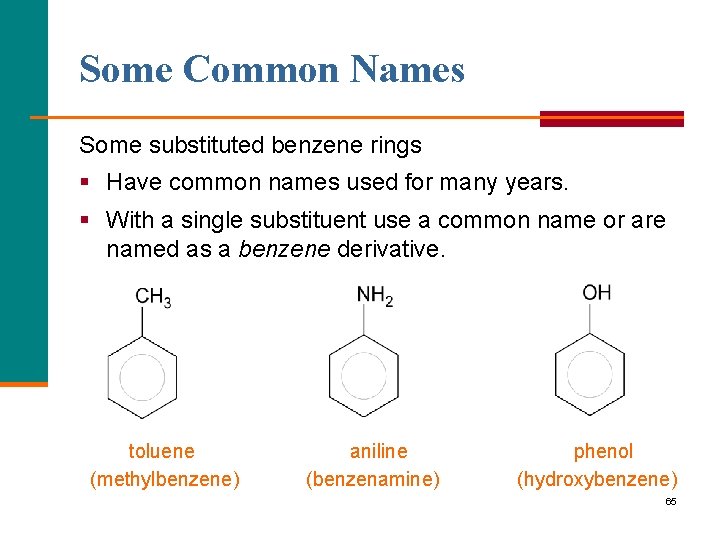
Some Common Names Some substituted benzene rings § Have common names used for many years. § With a single substituent use a common name or are named as a benzene derivative. toluene (methylbenzene) aniline (benzenamine) phenol (hydroxybenzene) 65

Aromatic Compounds with Two Groups Two naming systems are used when two groups are attached to a benzene ring. § Number the ring to give the lowest numbers to the side groups. § Use prefixes to show the arrangement: ortho(o ) for 1, 2 meta(m ) for 1, 3 para(p ) for 1, 4 66
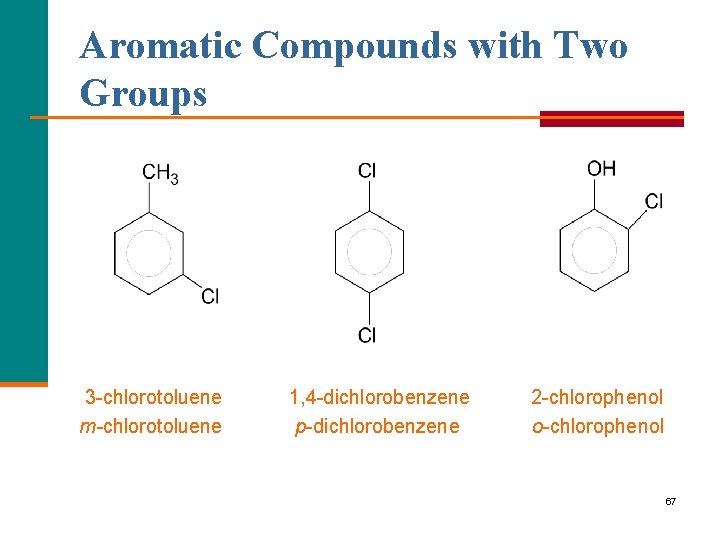
Aromatic Compounds with Two Groups 3 chlorotoluene m-chlorotoluene 1, 4 dichlorobenzene p dichlorobenzene 2 chlorophenol o chlorophenol 67
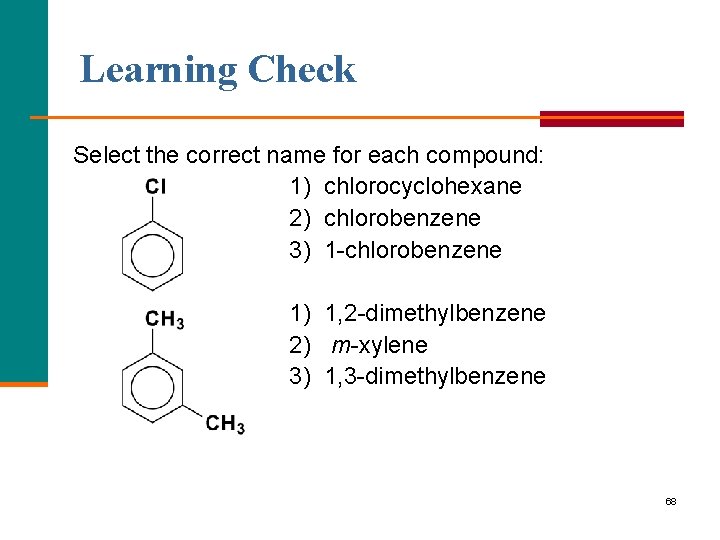
Learning Check Select the correct name for each compound: 1) chlorocyclohexane 2) chlorobenzene 3) 1 chlorobenzene 1) 1, 2 -dimethylbenzene 2) m xylene 3) 1, 3 dimethylbenzene 68
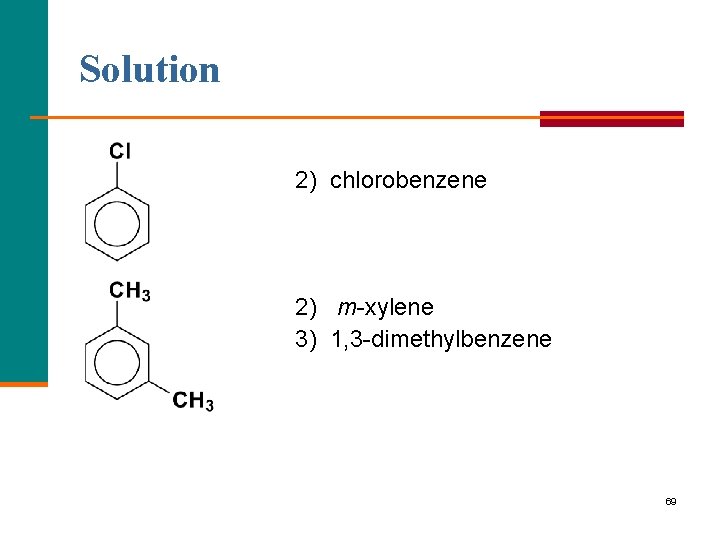
Solution 2) chlorobenzene 2) m xylene 3) 1, 3 dimethylbenzene 69

Learning Check Write the structural formulas for each of the following: A. 1, 3 dichlorobenzene B. o-chlorotoluene 70
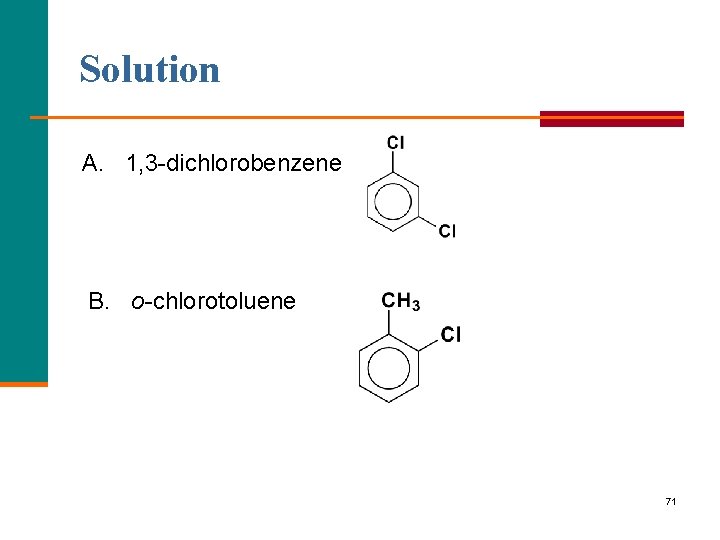
Solution A. 1, 3 dichlorobenzene B. o chlorotoluene 71
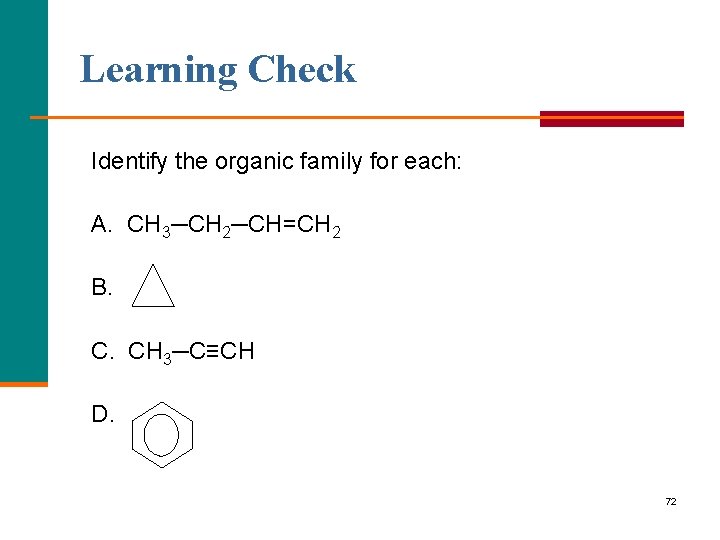
Learning Check Identify the organic family for each: A. CH 3─CH 2─CH=CH 2 B. C. CH 3─C≡CH D. 72
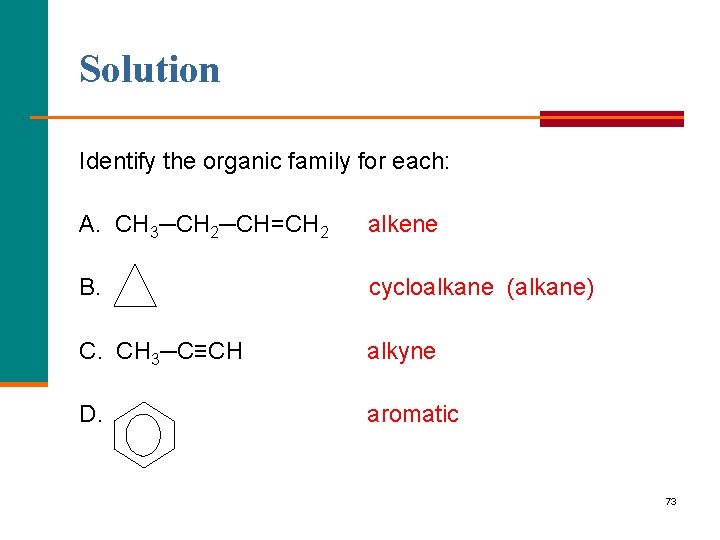
Solution Identify the organic family for each: A. CH 3─CH 2─CH=CH 2 alkene B. cycloalkane (alkane) C. CH 3─C≡CH alkyne D. aromatic 73

Chapter 12 Unsaturated Hydrocarbons 12. 6 Properties of Aromatic Compounds Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings 74

Properties of Aromatic Compounds Aromatic compounds § Have a stable aromatic bonding system. § Are resistant to many reactions. § Undergo substitution reactions, which retain the stability of the aromatic bonding system. 75

Substitution Reactions In a substitution reaction, a hydrogen atom on a benzene ring is replaced by an atom or group of atoms. Type of substitution H on benzene replaced by § Halogenation chlorine or bromine atom § Nitration nitro group (—NO 2) § Sulfonation —SO 3 H group 76
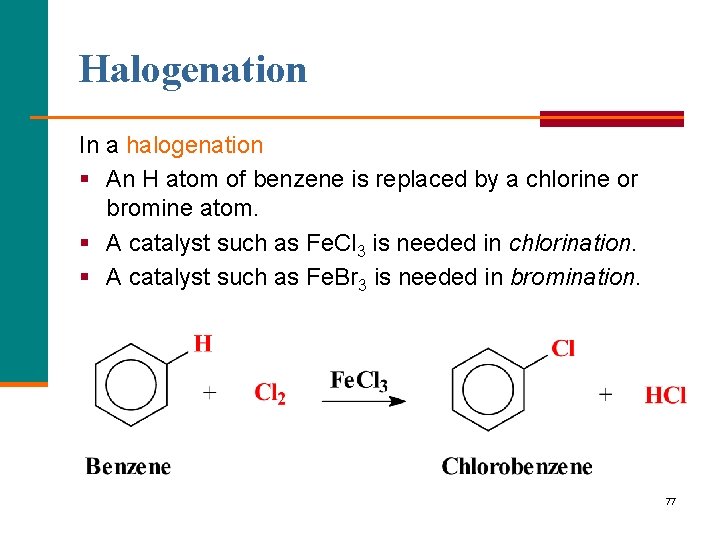
Halogenation In a halogenation § An H atom of benzene is replaced by a chlorine or bromine atom. § A catalyst such as Fe. Cl 3 is needed in chlorination. § A catalyst such as Fe. Br 3 is needed in bromination. 77
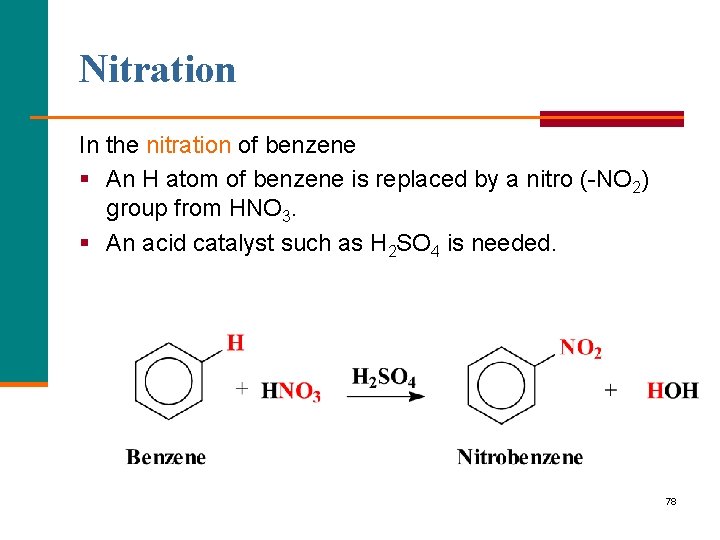
Nitration In the nitration of benzene § An H atom of benzene is replaced by a nitro ( NO 2) group from HNO 3. § An acid catalyst such as H 2 SO 4 is needed. 78
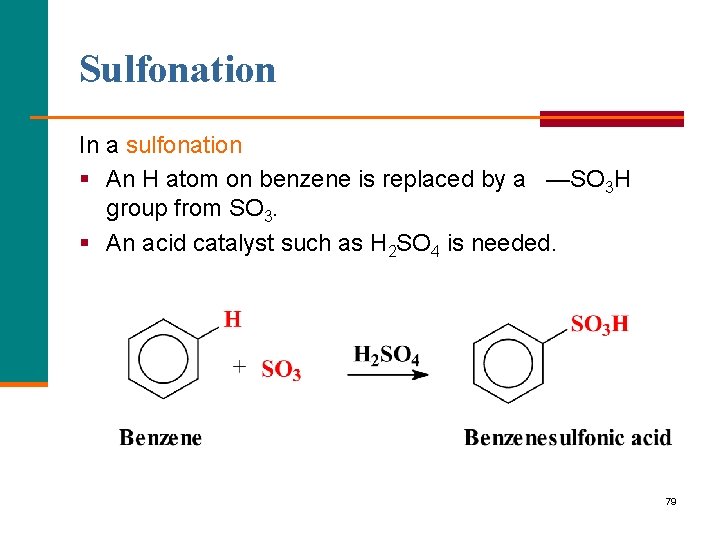
Sulfonation In a sulfonation § An H atom on benzene is replaced by a —SO 3 H group from SO 3. § An acid catalyst such as H 2 SO 4 is needed. 79

Learning Check Write the equation for the bromination of benzene including catalyst. 80
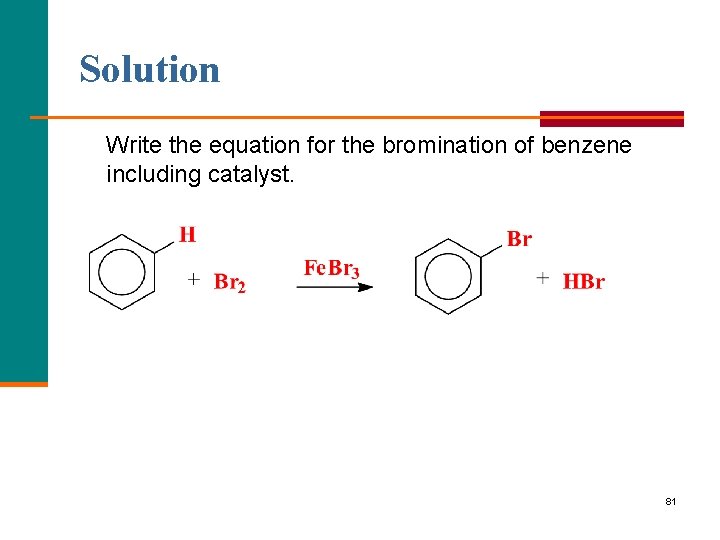
Solution Write the equation for the bromination of benzene including catalyst. 81
- Slides: 81