Chapter 12 States of Matter 12 1 Gasses














































- Slides: 55

Chapter 12 States of Matter

12. 1 Gasses • Gasses expand, diffuse, exert pressure, and can be compressed because they are in a low density state consisting of tiny, constantly moving particles

Kinetic Molecular Theory • Describes the behavior of matter in terms of particles in motion – Makes several assumptions about the size, motion, and energy of gas particles

Assumptions of the Kinetic Molecular Theory • Gasses consist of small particles that take up little volume relative to the volume of empty space around them – Gas molecules are very far apart and therefore don’t experience attractive or repulsive forces.

• Gas particles move in constant, random straight lines until they collide with other particles or with the walls of the container – Collisions are elastic -

• The energy of gas particles is determined by the particle’s mass and velocity – KE =

• A gas particle has a mass of 5. 31 x 10 -21 kg and a velocity of 1. 00 x 102 m/s. what is the kinetic energy of the particle?

Explaining the Behavior of Gasses • Kinetic molecular theory helps explain the behavior of gasses – Blowing up a balloon

• Gasses have low densities – Chlorine gas = 0. 00295 g/m. L – Germanium = 5. 323 g/m. L – Why?

• Gasses can be compressed and can expand – Explain

• Gasses diffuse and effuse – Diffusion = gas particles move from an area of high concentration to low concentration • Ex – Effusion = gas particles escape through tiny openings • Ex

• Rates of Effusion – Graham’s Law of Effusion:

• Rates of diffusion – Depends mainly on mass of particles involved – Grahams law allows us to compare rates of diffusion of two gasses

• Ammonia has a molar mass of 17. 0 g/mol, hydrogen chloride has a molar mass of 36. 5 g/mol. What is the ratio of their diffusion rates?

• What is the molar mass of a gas that takes four times as long to effuse as hydrogen?

Gas Pressure • Pressure = force per unit area – Smaller area = more pressure • Air pressure – pressure of the atmosphere – 1 atm – 760 mm Hg – 101. 3 k. Pa

• Dalton’s law of partial pressures: – Total pressure of a mixture of gasses is equal to the sum of the pressures of all of the gasses in the mixture – P 1 + P 2 + P 3…=Ptotal

• A mixture of oxygen, carbon dioxide, and nitrogen has a total pressure of 0. 97 atm. What is the partial pressure of O 2 if the partial pressure of CO 2 is 0. 70 atm and the partial pressure of N 2 is 0. 12 atm?

12. 2 Forces of attraction • Intermolecular forces (dispersion forces, dipole-dipole forces, and hydrogen bonds) determine a substance’s state at a given temperature

Intermolecular forces • Inter- means between or among • Intermolecular forces can hold together identical particles or two different types of particles • Weaker than intramolecular forces (bonds)

Dispersion Forces • Weak forces that result from temporary shifts in the density of electrons in electron clouds

• Exist between all particles – Weak for small particles – Get stronger as the number of electrons involved increases – F 2 – Cl 2 – Br 2 – I 2

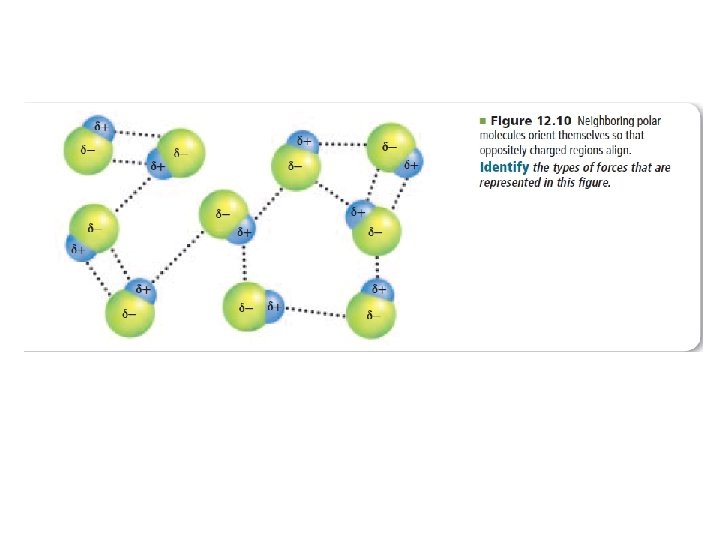
Dipole-dipole forces • Attraction between oppositely charged regions of polar molecules – Polar molecule = • Neighboring polar molecules orient themselves so that oppositely charged regions align


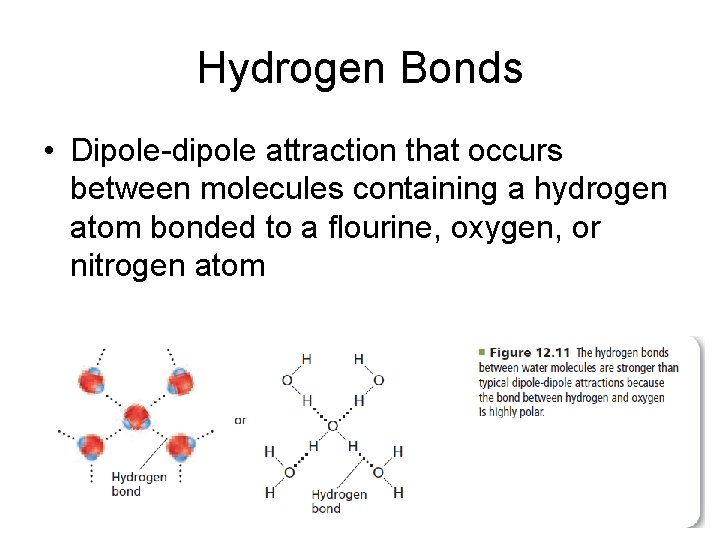
Hydrogen Bonds • Dipole-dipole attraction that occurs between molecules containing a hydrogen atom bonded to a flourine, oxygen, or nitrogen atom

• Explain why water is liquid at room temperature while compounds of similar masses are gasses

12. 3 Liquids and Solids • The particles in solids and liquids have a limited range of motion and are not easily compressed.

Liquids • Kinetic molecular theory also applies to liquids and solids – Must take intermolecular forces into account to apply it

• Density and compression – Much denser than gasses • Due to intermolecular forces holding particles together – Incompressible • Why can you compress a gas but not a liquid?

• Fluidity – both gases and liquids are classified as fluids because they can flow and diffuse – Liquids diffuse more slowly because intermolecular attractions interfere with the flow

• Viscosity - measure of the resistance of a liquid to flow – Attractive forces – stronger intermolecular forces = higher viscosity – Particle size – larger molecules = higher viscosity – Temperature – lower temperature = higher viscosity

• Surface tension – the energy required to increase the surface area of a liquid by a given amount – Caused by intermolecular forces pulling down on the particles on the surface of a liquid which stretches it tight like a drum

• Stronger the attraction between particles in a liquid = greater surface tension • Surfactant – lowers the surface tension of water by disrupting hydrogen bonds between water molecules

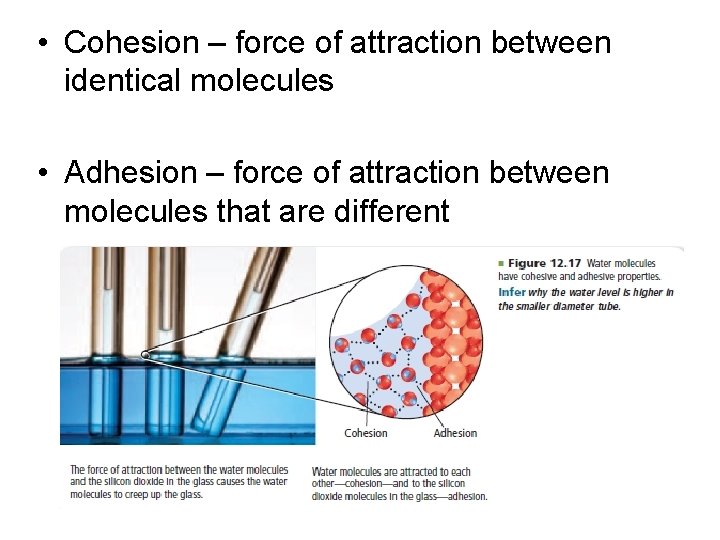
• Cohesion – force of attraction between identical molecules • Adhesion – force of attraction between molecules that are different

Solids • Solid particles have as much kinetic energy as liquids or gasses but much stronger attractive forces between particles – Limit the motion of particles to vibrations

• Density of solids – almost always greater than density of liquids – Exception = water

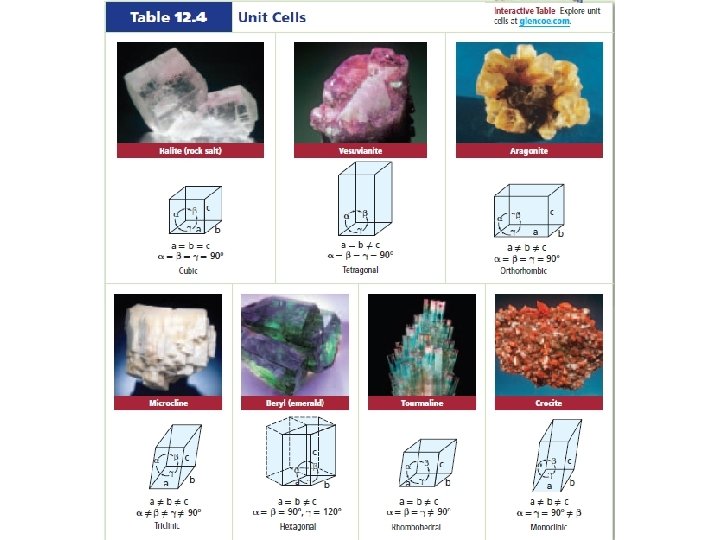
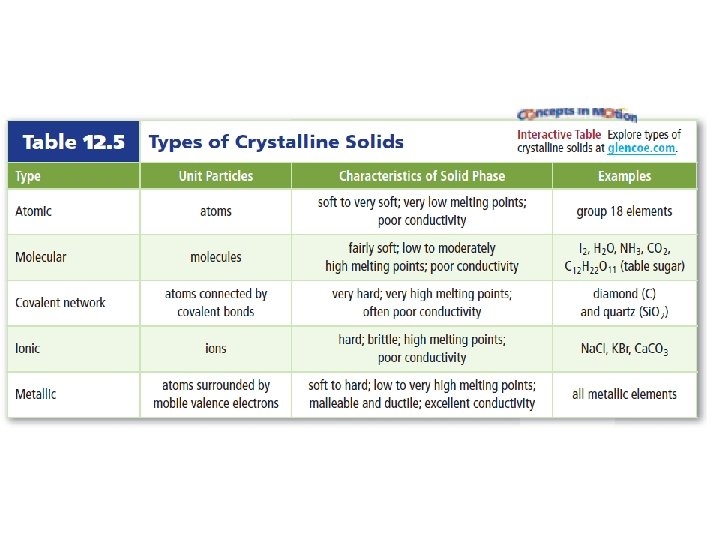
• Crystalline solids – solid whose atoms, ions, or molecules are arranged in an orderly, geometric structure – Unit cell = smallest arrangement of atoms in a crystalline solid that has the same shape as the whole crystal


• Categories of crystalline solids – Classified based on the types of particles they contain and how they are bonded together

• Molecular solids – Molecules are held together by dispersion forces, dipole-dipole forces or hydrogen bonds – Most are not solid at room temperature – Poor conductors

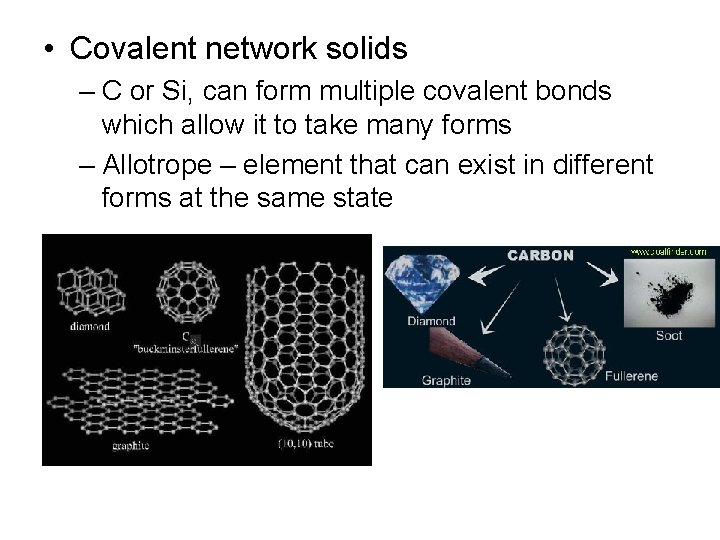
• Covalent network solids – C or Si, can form multiple covalent bonds which allow it to take many forms – Allotrope – element that can exist in different forms at the same state

• Ionic solids – Made of cation + anion – Each ion is surrounded by ions of the opposite charge – High melting point – Brittle

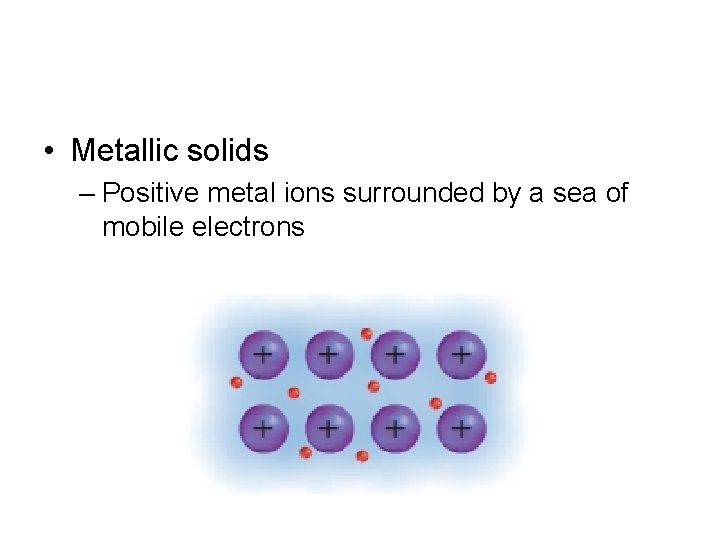
• Metallic solids – Positive metal ions surrounded by a sea of mobile electrons


Amorphous solids • Particles are not arranged in a regular, repeating pattern • Does not contain crystals • Forms when molten material cools too quickly for crystals to form – Glass – Rubber – Some plastics

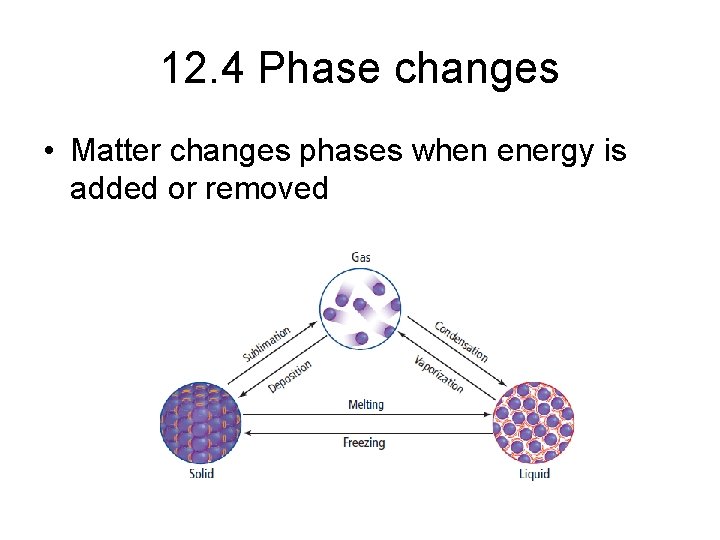
12. 4 Phase changes • Matter changes phases when energy is added or removed

Phase changes that require energy • Melting – Heat flows from an object at a higher temperature to an object at a lower temperature – Ice absorbs heat which does not raise temperature but is used to break hydrogen bonds – When hydrogen bonds are broken molecules can move further apart into the liquid phase

• Melting point – temperature in which forces holding a solid together are broken and it becomes a liquid

• Vaporization – process by which liquid changes to vapor – Vapor – gaseous state of a substance that is normally liquid at room temperature – Evaporation – when vaporization occurs only at the surface of a liquid – Vapor pressure – the pressure exerted by a vapor over a liquid

– Boiling – temperature at which the vapor pressure of a liquid equals the atmospheric pressure – Energy being input causes molecules to move around more and vaporize

• Sublimation – changing from solid to gas without becoming a liquid – Dry ice – Moth balls – Solid air fresheners

Phase changes that release energy • Freezing – Heat flows out of warmer object into cooler object – Molecules slow down & become less likely to flow past one another – Intermolecular forces cause the molecules to become fixed into set positions – Freezing point – temperature in which a liquid becomes a solid

• Condensation – process by which a gas or vapor becomes a liquid • Deposition – substance changes from gas or vapor to solid without first becoming a liquid – frost

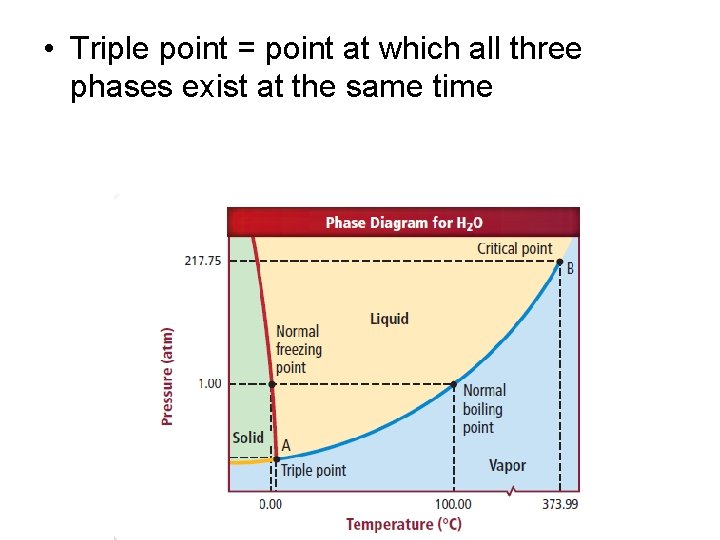
Phase Diagrams • Temperature and pressure both effect the phase of a substance – Have opposite effects • Phase diagram – graph of pressure vs temperature that shows which phase a substance will be in under different conditions.

• Triple point = point at which all three phases exist at the same time