Chapter 12 Lecture Basic Chemistry Fourth Edition Chapter

Chapter 12 Lecture Basic Chemistry Fourth Edition Chapter 12 Solutions 12. 6 Properties of Solutions Learning Goal Identify a mixture as a solution, a colloid, or a suspension. Describe how the number of particles in a solution affects the freezing point, boiling point, and osmotic pressure. © 2014 Pearson Education, Inc.

Solution and Solute Particles The solute particles in a solution • play an important role in determining the properties of that solution • are dispersed uniformly through the solution and cannot be visibly distinguished from the solvent • are so small that they go through filters and semipermeable membranes © 2014 Pearson Education, Inc.

Colloids are homogeneous mixtures that do not settle out. The solute particles in a colloidal suspension or colloid are • much larger than the particles in a solution • large molecules such as proteins, or groups of molecules or ions • small enough to pass through filters but too large to go through a semipermeable membrane © 2014 Pearson Education, Inc.
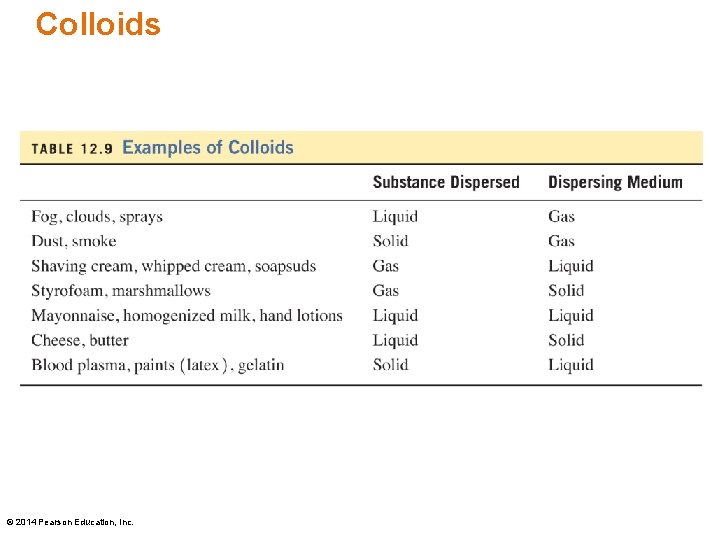
Colloids © 2014 Pearson Education, Inc.

Suspensions • are heterogeneous, nonuniform mixtures • contain very large particles that are trapped by filters and do not pass through semipermeable membranes • contain particles that settle out of solution and must be stirred to stay suspended Examples: muddy water, liquid penicillin © 2014 Pearson Education, Inc.
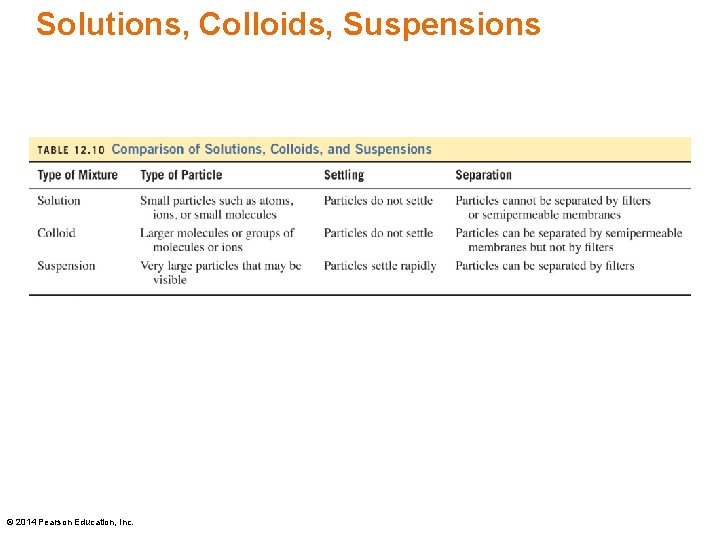
Solutions, Colloids, Suspensions © 2014 Pearson Education, Inc.

Solutions, Colloids, Suspensions © 2014 Pearson Education, Inc.

Colligative Properties Colligative properties of solutions depend on the number of solute particles in the solution. • Adding a solute to water lowers the freezing point of the solvent. • Adding a solute to water elevates the boiling point of the solution. © 2014 Pearson Education, Inc.

Freezing Point Lowering Ethylene glycol (antifreeze) HO—CH 2—OH • is an organic compound with two –OH groups, that make it very soluble in water • is added to water in a car radiator to lower its freezing point and raise its boiling point • prevents the water in a car radiator from freezing in cold weather and boiling in hot weather © 2014 Pearson Education, Inc.

Ethylene Glycol A 50 -50% by mass mixture of ethylene glycol and water freezes at about − 30 o. F and does not boil until it reaches 225 o. F. Ethylene glycol dissolves as molecules. © 2014 Pearson Education, Inc.

Particles in Solution Solutes that are • nonelectrolytes dissolve as molecules 1 mole of C 2 H 6 O 2(l) = 1 mole of C 2 H 6 O 2(aq) • strong electrolytes dissolve as ions 1 mole of Na. Cl(s) = 2 moles of particles, Na+(aq) and Cl–(aq) © 2014 Pearson Education, Inc.

Molality (m) Molality is a unit of concentration: Molality is used to calculate freezing point lowering or boiling point elevation. © 2014 Pearson Education, Inc.
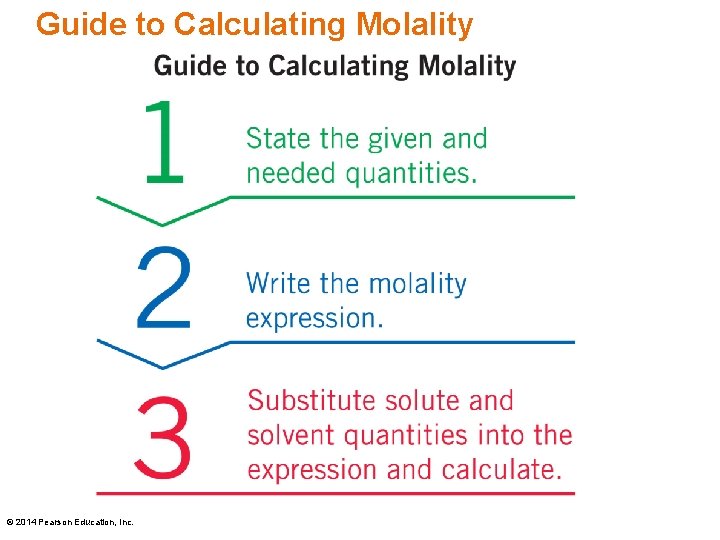
Guide to Calculating Molality © 2014 Pearson Education, Inc.

Learning Check What is the molality of particles in a solution of 125 g of Mg. Cl 2, a strong electrolyte, dissolved in 0. 500 kg of water? © 2014 Pearson Education, Inc.

Solution What is the molality of particles in a solution of 125 g of Mg. Cl 2, a strong electrolyte, dissolved in 0. 500 kg of water? Step 1 State the given and needed quantities. © 2014 Pearson Education, Inc.

Solution What is the molality of particles in a solution of 125 g of Mg. Cl 2, a strong electrolyte, dissolved in 0. 500 kg of water? Step 2 Write the molality expression. © 2014 Pearson Education, Inc.

Solution What is the molality of particles in a solution of 125 g of Mg. Cl 2, a strong electrolyte, dissolved in 0. 500 kg of water? Step 3 Substitute the solute and solvent quantities into the expression and calculate. © 2014 Pearson Education, Inc.

Solution What is the molality of particles in a solution of 125 g of Mg. Cl 2, a strong electrolyte, dissolved in 0. 500 kg of water? Step 3 Substitute the solute and solvent quantities into the expression and calculate. © 2014 Pearson Education, Inc.

Solution What is the molality of particles in a solution of 125 g of Mg. Cl 2, a strong electrolyte, dissolved in 0. 500 kg of water? Step 3 Substitute the solute and solvent quantities into the expression and calculate. © 2014 Pearson Education, Inc.

Molality, Colligative Properties Freezing point lowering (ΔTf) is determined from the molality (m) of the particles in the solution and the freezing point constant, Kf. To determine the new solution freezing point The freezing point constant (Kf) for water is 1. 86 o C/m. © 2014 Pearson Education, Inc.

Molality, Colligative Properties For nonelectrolyte solutions such as glucose with 1 mole of particles per one mole of solute, For electrolyte solutions, such as Na. Cl with 2 moles of particles per mole of solute, Na. Cl(s) Na+(aq) + Cl−(aq) © 2014 Pearson Education, Inc.

Molality, Colligative Properties Boiling point elevation (ΔTb) is determined from the molality (m) of the particles in the solution and the boiling point constant, Kb. To determine the new boiling point The boiling point constant (Kb) for water is 0. 52 o. C/m. © 2014 Pearson Education, Inc.

Molality, Colligative Properties For nonelectrolyte solutions such as glucose with 1 mole of particles per one mole of solute, For electrolyte solutions, such as Na. Cl with 2 moles of particles per mole of solute, Na. Cl(s) Na+(aq) + Cl−(aq) © 2014 Pearson Education, Inc.
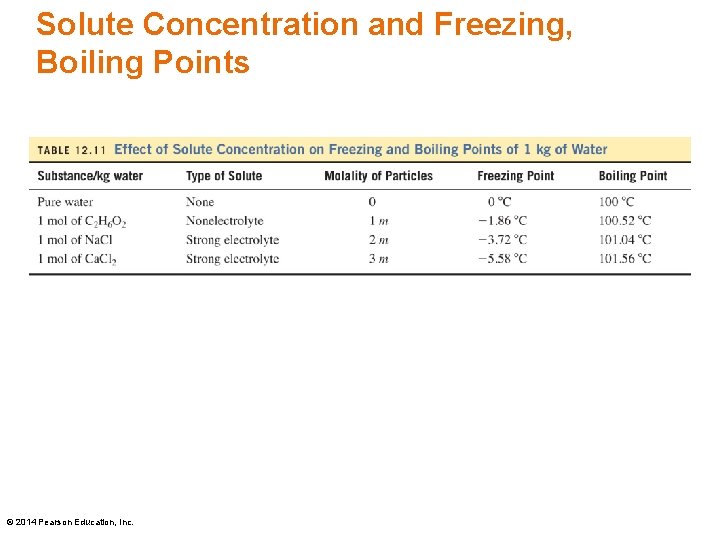
Solute Concentration and Freezing, Boiling Points © 2014 Pearson Education, Inc.
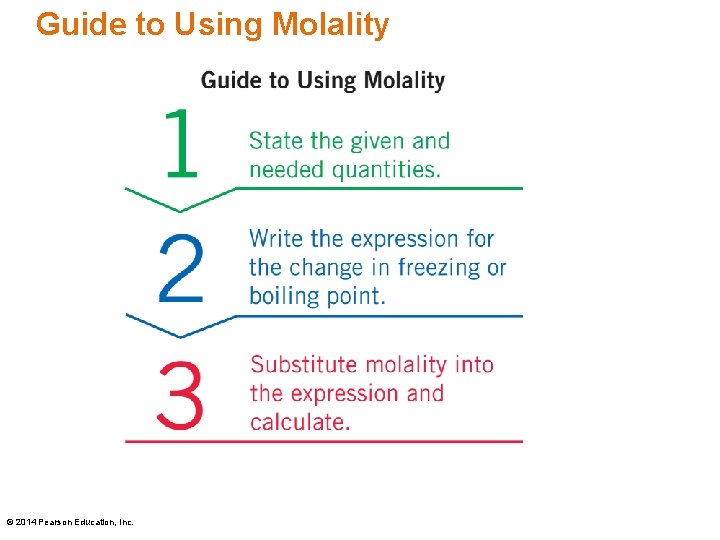
Guide to Using Molality © 2014 Pearson Education, Inc.

Learning Check In the northeastern United States during freezing temperatures, Ca. Cl 2 is spread on icy highways to melt the ice. Calculate the freezing point lowering and freezing point of a solution containing 225 g of Ca. Cl 2 in 500. g of water. © 2014 Pearson Education, Inc.

Solution Calculate the freezing point lowering and freezing point of a solution containing 225 g of Ca. Cl 2 in 500. g of water. Step 1 State the given and needed quantities. © 2014 Pearson Education, Inc.

Solution Calculate the freezing point lowering and freezing point of a solution containing 225 g of Ca. Cl 2 in 500. g of water. Step 2 Write the expression for the change in freezing or boiling point. © 2014 Pearson Education, Inc.

Solution Calculate the freezing point lowering and freezing point of a solution containing 225 g of Ca. Cl 2 in 500. g of water. Step 3 Substitute the molality into the expression and calculate. © 2014 Pearson Education, Inc.

Solution Calculate the freezing point lowering and freezing point of a solution containing 225 g of Ca. Cl 2 in 500. g of water. Step 3 Substitute the molality into the expression and calculate. Calculating the molality of the Ca. Cl 2 solution: moles of particles © 2014 Pearson Education, Inc.

Solution Calculate the freezing point lowering and freezing point of a solution containing 225 g of Ca. Cl 2 in 500. g of water. Step 3 Substitute the molality into the expression and calculate. © 2014 Pearson Education, Inc.

Solution Calculate the freezing point lowering and freezing point of a solution containing 225 g of Ca. Cl 2 in 500. g of water. Step 3 Substitute the molality into the expression and calculate. © 2014 Pearson Education, Inc.

Osmosis In osmosis, • water (solvent flows from the lower solute concentration into the higher solute concentration • the level of the solution with the higher solute concentration rises • the concentrations of the two solutions become equal with time © 2014 Pearson Education, Inc.

Osmotic Pressure Osmotic pressure • is produced by the solute particles dissolved in a solution • is the pressure that prevents the flow of additional water into the more concentrated solution • increases as the number of dissolved particles in the solution increases © 2014 Pearson Education, Inc.
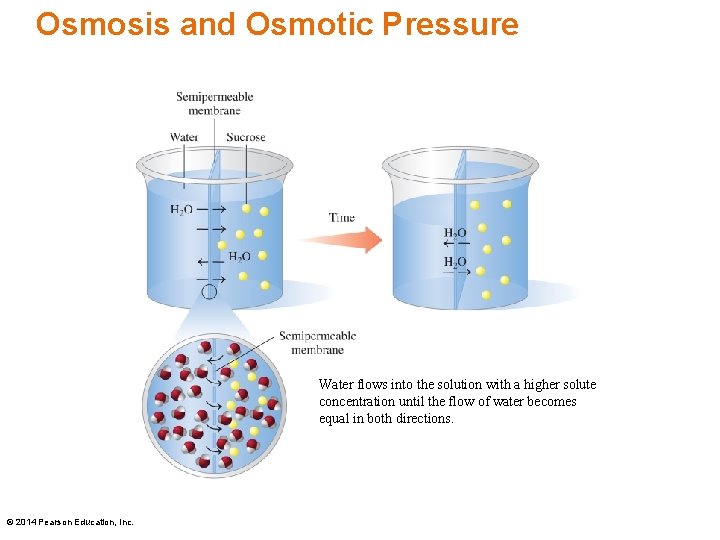
Osmosis and Osmotic Pressure Water flows into the solution with a higher solute concentration until the flow of water becomes equal in both directions. © 2014 Pearson Education, Inc.

Learning Check A semipermeable membrane separates a 10% starch solution (A) from a 5% starch solution (B). If starch is a colloid, fill in the blanks in the statements below. 1. Solution ____ has the greater osmotic pressure. 2. Water initially flows from ___ into ___. 3. The level of solution ____ will be lower. © 2014 Pearson Education, Inc.

Solution A semipermeable membrane separates a 10% starch solution (A) from a 5% starch solution (B). If starch is a colloid, fill in the blanks in the statements below. 1. Solution (A) has the greater osmotic pressure. 2. Water initially flows from (B) into (A). 3. The level of solution (B) will be lower. © 2014 Pearson Education, Inc.
Isotonic Solutions Solutes in body solutions such as blood, tissue fluids, lymph, and plasma exert osmotic pressure on semipermeable cell membranes. Most intravenous solutions used in hospitals are isotonic solutions, which exert the same osmotic pressure as body fluids such as blood. A 0. 9% Na. Cl solution is isotonic with the solute concentration of the blood cells of the body. © 2014 Pearson Education, Inc.

Hypotonic Solutions When a red blood cell is placed in a hypotonic solution, which has a lower solute concentration, water flows into the cell by osmosis. The increase in fluid causes the cell to swell and possibly burst—a process called hemolysis. © 2014 Pearson Education, Inc.

Hypotonic Solutions When a red blood cell is placed in a hypertonic solution, which has a higher solute concentration, water flows out of the cell by osmosis. The decrease in fluid causes the cell to shrink and possibly burst—a process called crenation. © 2014 Pearson Education, Inc.
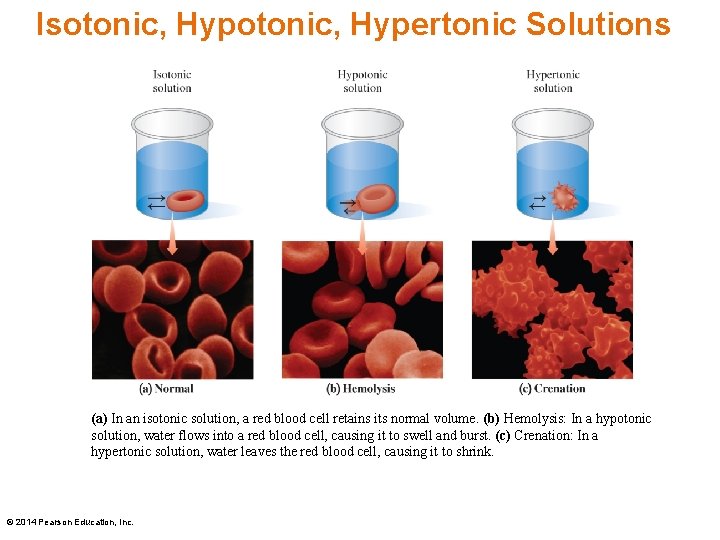
Isotonic, Hypertonic Solutions (a) In an isotonic solution, a red blood cell retains its normal volume. (b) Hemolysis: In a hypotonic solution, water flows into a red blood cell, causing it to swell and burst. (c) Crenation: In a hypertonic solution, water leaves the red blood cell, causing it to shrink. © 2014 Pearson Education, Inc.
Isotonic, Hypertonic Solutions © 2014 Pearson Education, Inc.
- Slides: 42