Chapter 12 Chemical Bonds Types of Bonds Ionic

Chapter 12 Chemical Bonds

Types of Bonds �Ionic bond: one that holds an ionic cmpd together �Relatively strong �Results in high melting/boiling points �Covalent bond: one that holds a covalent cmpd together �Not as strong �Lower melting/boiling points � 2 types: polar covalent and nonpolar covalent
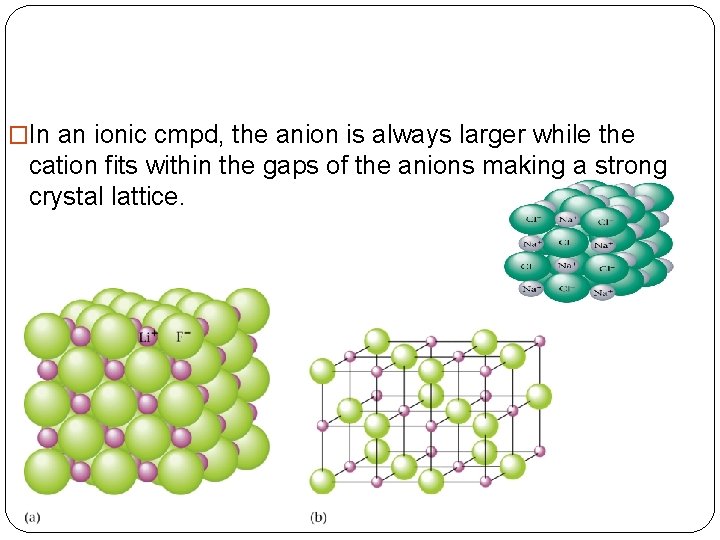
�In an ionic cmpd, the anion is always larger while the cation fits within the gaps of the anions making a strong crystal lattice.
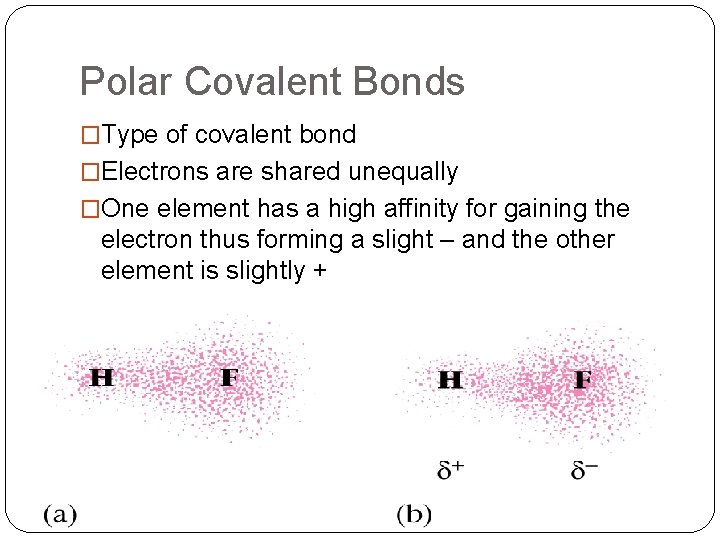
Polar Covalent Bonds �Type of covalent bond �Electrons are shared unequally �One element has a high affinity for gaining the electron thus forming a slight – and the other element is slightly +
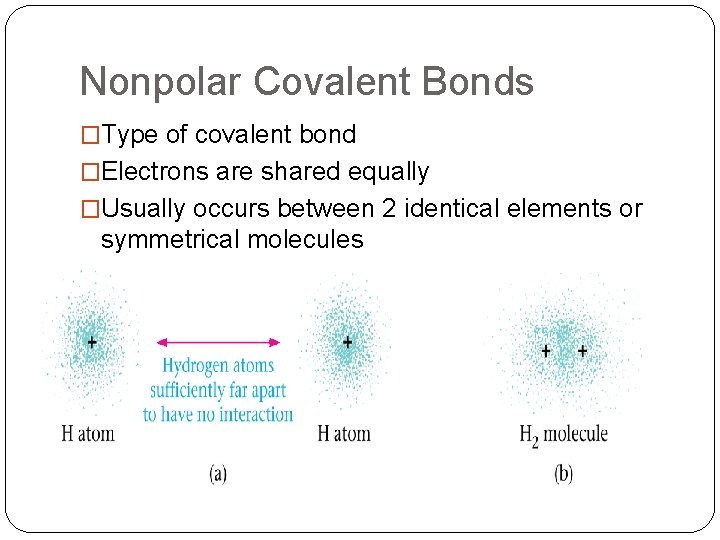
Nonpolar Covalent Bonds �Type of covalent bond �Electrons are shared equally �Usually occurs between 2 identical elements or symmetrical molecules
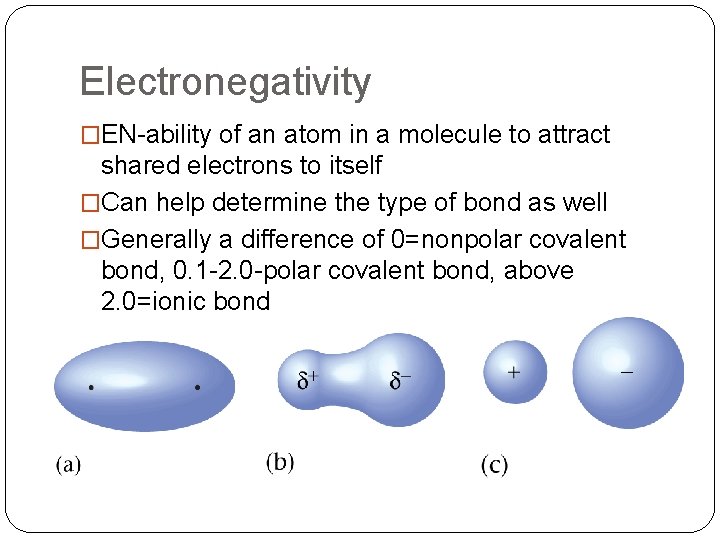
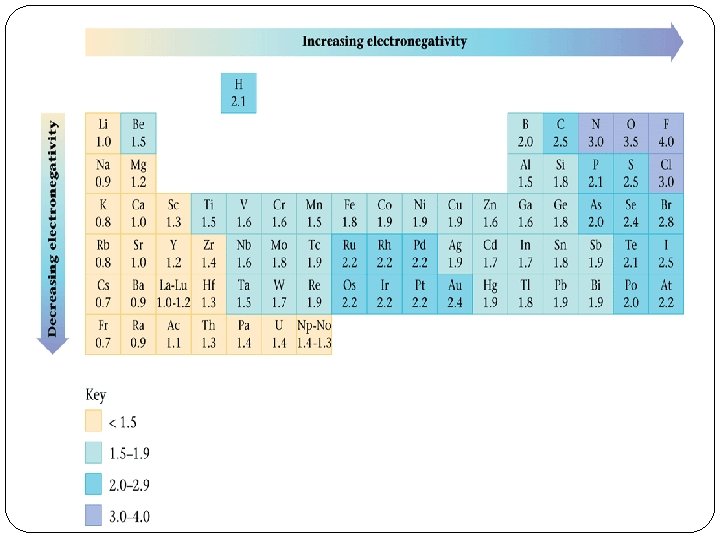
Electronegativity �EN-ability of an atom in a molecule to attract shared electrons to itself �Can help determine the type of bond as well �Generally a difference of 0=nonpolar covalent bond, 0. 1 -2. 0 -polar covalent bond, above 2. 0=ionic bond
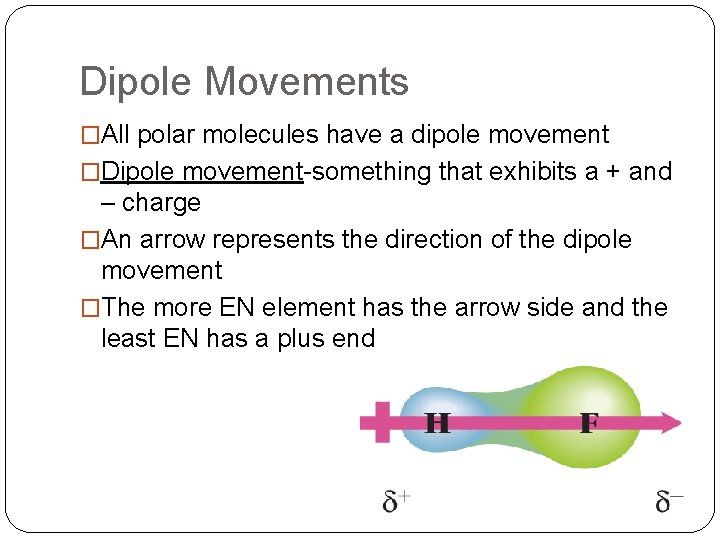
Dipole Movements �All polar molecules have a dipole movement �Dipole movement-something that exhibits a + and – charge �An arrow represents the direction of the dipole movement �The more EN element has the arrow side and the least EN has a plus end
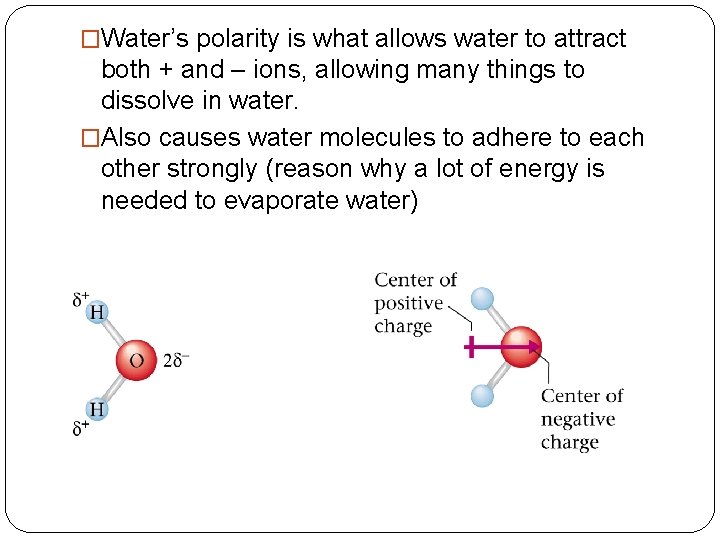
�Water’s polarity is what allows water to attract both + and – ions, allowing many things to dissolve in water. �Also causes water molecules to adhere to each other strongly (reason why a lot of energy is needed to evaporate water)

Examples �Page 404 Ex. 12. 1 �Page 406 # 4 and 6

Lewis Structures �A representation that shows how valence electrons bond. �Hydrogen is the only element that follows the duet rule-needs 2 electrons to be stable �Other elements follow the octet rule-need 8 electrons to be stable �Bonding electrons-those involved in bonding �Lone pair electrons-those not involved in bonding

Examples �H 2 O �PH 3 �CCl 4

Multiple Bonds �Single bond-sharing of 2 electrons �Double bond-sharing of 4 electrons �Triple bond-sharing of 6 electrons �Ex: HCN

Resonance & Formal Charge �Some elements exhibit multiple Lewis structures like carbon dioxide. See if you can draw all 3.

�Resonance structures-when more than 1 valid Lewis Structure can be drawn �Use arrows to show resonance �How do we know which is right? �Want the formal charge on all elements to be as close to zero as possible �# of valence – nonbonding – (bonding / 2)

�Some resonance structures will not require formal charge as they are mirror images of each other �Ex: ozone(O 3), SO 3 �What is the formal charge on each element in the above?

Polyatomic Ions �Add/subtract electrons as needed �Put the structure in brackets with the charge outside �Check for resonance �Ex: NO 2 -1, CO 3 -2

Exceptions to the rule �Boron is happy with 6 electrons �Anything to the right and below phosphorus can technically have expanded octets. We will work mostly with phosphorus and sulfur. �Ex: BF 3, SF 6, PCl 5

Molecular Structure � 3 D arrangement of the valence electrons (both bonded and non) �VSEPR theory-valence shell electron pair repulsion theory �The structure around a given atom is determined by minimizing repulsions between electrons pairs. �AKA: electrons spread out as far away from each other as possible

Electron pair geometry vs. molecular geometry �Electron pair includes all electrons including bonded and non �Molecular refers only to the bonding electrons

�http: //intro. chem. okstate. edu/1314 f 00/lecture/chapter 1 0/vsepr. html �Main types/Subtypes with bond angles �Linear- 180 �Trigonal Planar -120 �Bent- under 118 �Tetrahedral- 109. 5 �Trigonal pyramidal- 107 �Bent- 105 �Trigonal bipyramidal- 90, 120, 180 �Octahedral- 90 �Square Planar- 90
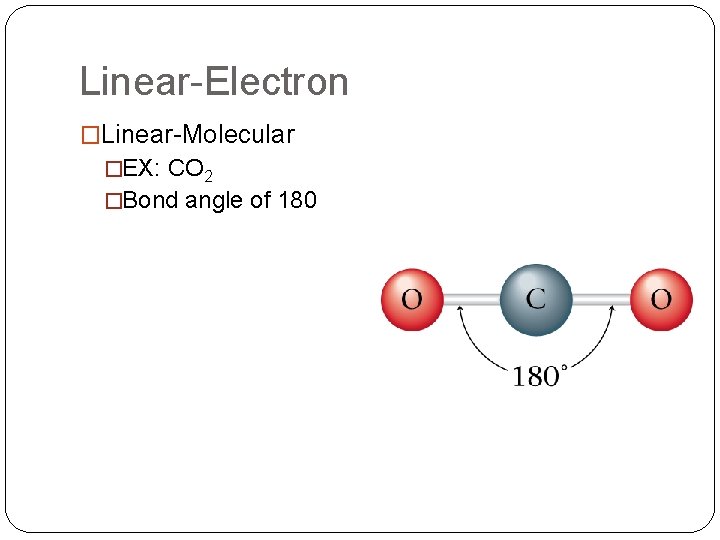
Linear-Electron �Linear-Molecular �EX: CO 2 �Bond angle of 180
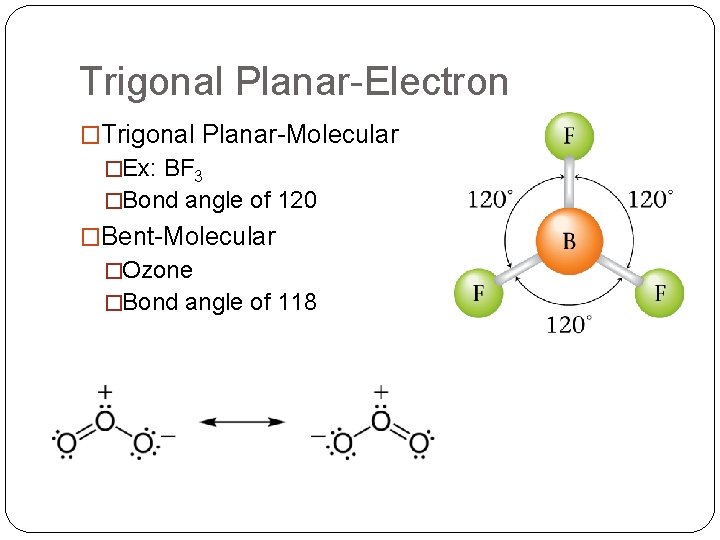
Trigonal Planar-Electron �Trigonal Planar-Molecular �Ex: BF 3 �Bond angle of 120 �Bent-Molecular �Ozone �Bond angle of 118
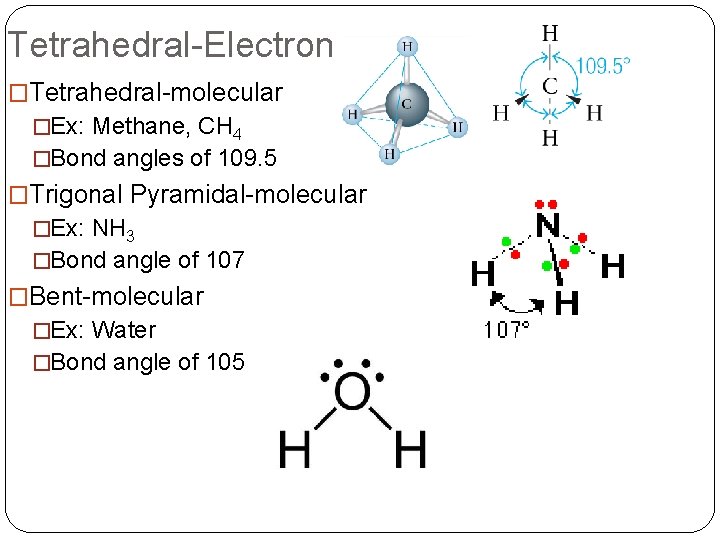
Tetrahedral-Electron �Tetrahedral-molecular �Ex: Methane, CH 4 �Bond angles of 109. 5 �Trigonal Pyramidal-molecular �Ex: NH 3 �Bond angle of 107 �Bent-molecular �Ex: Water �Bond angle of 105
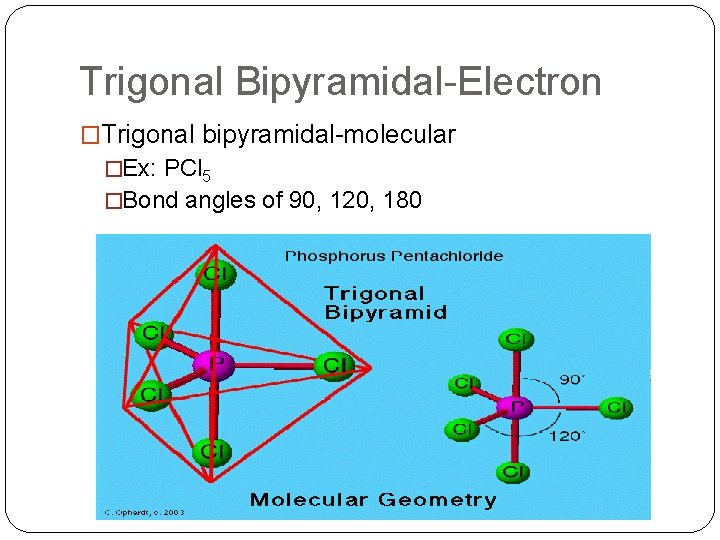
Trigonal Bipyramidal-Electron �Trigonal bipyramidal-molecular �Ex: PCl 5 �Bond angles of 90, 120, 180
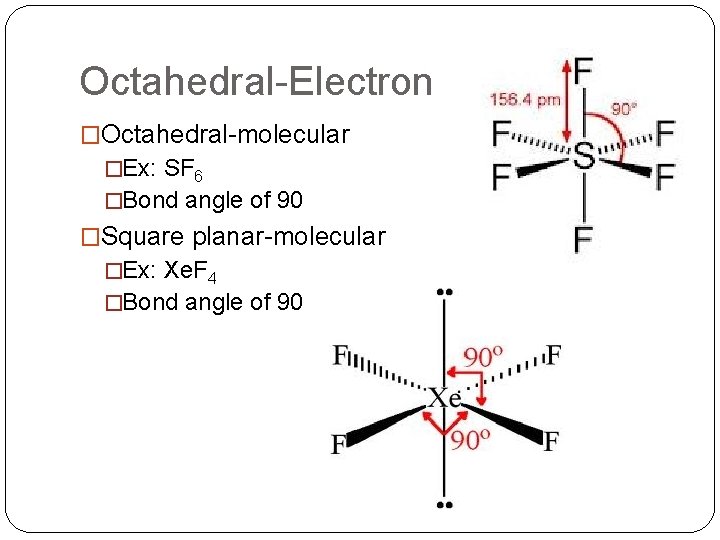
Octahedral-Electron �Octahedral-molecular �Ex: SF 6 �Bond angle of 90 �Square planar-molecular �Ex: Xe. F 4 �Bond angle of 90
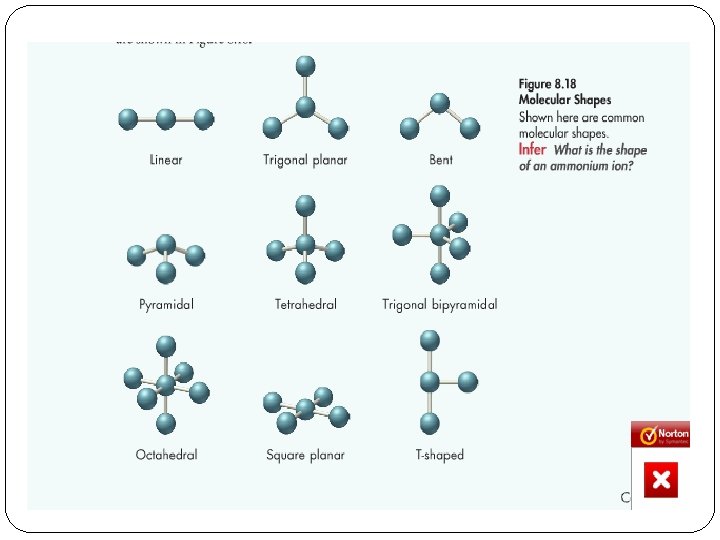

In-Class Practice �Provide bond angles for all �Page 429 honors book Ex. 12. 6 �Others: nitrite ion, sulfur trioxide, phosphorus pentafluoride, Si. F 6 -2
- Slides: 28