CHAPTER 12 CHEMICAL BONDING CHEMICAL BOND A force

CHAPTER 12 – CHEMICAL BONDING CHEMICAL BOND – A force that holds two or more atoms together as a unit Individual atoms will naturally bond together to achieve a lower energy state (to be more stable) 3 B-1 (of 42) (9 – 34 -42 + 1 -15) (10 – 16 -26) (11 - 27 -42 + 1 -3)

TYPES OF BONDS 1) METAL ATOMS AND NONMETAL ATOMS Metal atoms easily lose electrons forming positive ions, and nonmetal atoms easily gain electrons forming negative ions IONIC BOND – The electrostatic attraction between positive and negative ions Ionic bonding forms giant crystalline networks containing billions of positive and negative ions that are strongly attracted together Ionic bonding exists between metal and nonmetal ions 3 B-2

Elemental Iron Elemental Oxygen Rust O atoms (molecules) Fe ions and O ions + Fe atoms 3 B-3

2) NONMETAL ATOMS Nonmetal atoms attract each other’s valence electrons, and share the valence electrons between pairs of atoms COVALENT BOND – The electrostatic attraction of shared electrons to the nuclei of bonding nonmetal atoms Covalent bonding forms individual units called molecules, and while the atoms that covalently bond together strongly attract each other, the molecules that are created weakly attract each other Covalent bonding exists between nonmetal atoms 3 B-4

Elemental Carbon Elemental Chlorine Carbon Tetrachloride Cl atoms (molecules) CCl 4 molecules + C atoms 3 B-5

Picture NONPOLAR COVALENT BOND – A bond in which 2 atoms are sharing electrons equally POLAR COVALENT BOND – A bond in which 2 atoms are sharing electrons unequally IONIC BOND – A bond in which two atoms have transferred electrons 3 B-6

ELECTRONEGATIVITY – The attraction of an atom for shared electrons Atom with the highest EN? Atom with the lowest EN? F Fr The difference in the EN’s of 2 atoms tells the type of bond they make EN Difference Bond 0. 0 0. 1 Nonpolar Covalent Polar Covalent Ionic to 1. 6 1. 7 to 3. 3 3 B-7

N-N Bond EN for N = 3. 0 – 3. 0 = 0. 0 Nonpolar Covalent Bond 3 B-8

C-O Bond H-S Bond EN for C = 2. 5, O = 3. 5 EN for H = 2. 1, S = 2. 5 3. 5 – 2. 5 = 1. 0 Polar Covalent Bond 2. 5 – 2. 1 = 0. 4 Polar Covalent Bond Polar covalent bonds have partially positive and partially negative ends C –– O d+ d- C –– O H –– S d+ d- H –– S DIPOLE MOMENT – The amount of separation of the positive and negative charges in a bond DIPOLE MOMENT ARROW – Shows the direction of the dipole moment, pointing toward the negative end of the bond 3 B-9

Na-Cl Bond EN for Na = 0. 9, Cl = 3. 0 – 0. 9 = 2. 1 Ionic Bond Na+ 3 B-10 Cl-

BONDING IN IONIC COMPOUNDS Atoms form ions to obtain a stable, octet electron arrangement Sodium chloride Na + . . . Cl : - . . A sodium chloride crystal is a symmetrical array of sodium and chloride ions in a 1: 1 ratio EMPIRICAL FORMULA – The simplest whole number ratio of atoms of different elements in a compound Empirical Formula: Na. Cl 3 B-11

Magnesium fluoride Mg 2+ . . . F: . . - . . . F: - . . Empirical Formula: Mg. F 2 3 B-12

Potassium nitride K + K + . . N: 3 - . Empirical Formula: K 3 N 3 B-13

SIZES OF ATOMS AND IONS Positive ions are smaller than their neutral atoms and negative ions are bigger than their neutral atoms 3 B-14 Na atom Cl atom Na+ ion Cl- ion

Sizes of atoms or ions are determined by 1) The more energy levels an atom or ion has the larger it will be 2) With the same number of energy levels, the more protons an atom or ion has the smaller it will be Electrons Energy Levels Protons Big to Small Li F 3 2 3 3 rd 9 2 9 4 th Na 11 Cl Li+ F 10 17 2 1 Na+ Cl- 10 18 2 3 3 4 th 9 ISOELECTRONIC – Ions or atoms with 11 the 17 same number of electrons 11 2 nd 1 st 2 nd 3 rd 3 B-15 1 st

BONDING IN COVALENT MOLECULES Atoms share electrons to obtain a stable, octet (or duet) arrangements Water (H 2 O) H H . . . O: . . . H–O: H ← LONE PAIR ← BONDING PAIR LEWIS STRUCTURE – A diagram using electron dot notation to show the valence electrons are arranged among bonded atoms 3 B-16

To draw a proper Lewis Structure: 1 – Add up the valence e-s for all of the atoms in the molecule or ion 2 – Draw a skeletal structure by using pairs of electrons to make bonds 3 – Complete octets (or duets for H) for all atoms, outer atoms first, using the remaining valence e-s 4 – If octets are not produced, make the atoms that have octets share more e pairs with atoms that do not have octets 3 B-17

Sulfur dichloride, SCl 2 6 + 7 = 20 valence e-s Cl 3 B-18 S Cl

Phosphorus tribromide, PBr 3 5 + 7 + 7 = 26 valence e-s Br P Br 3 B-19 Br

Ammonia, NH 3 5 + 1 + 1 = 8 valence e-s H N H 3 B-20 H

Methane, CH 4 4 + 1 + 1 = 8 valence e-s H H C H 3 B-21 H

Fluorine, F 2 7 + 7 = 14 valence e-s F F SINGLE BOND – One shared pair of e-s between two atoms 3 B-22

Oxygen, O 2 6 + 6 = 12 valence e-s O O DOUBLE BOND – Two shared pairs of e-s between two atoms 3 B-23

Nitrogen, N 2 5 + 5 = 10 valence e-s N N TRIPLE BOND – Three shared pairs of e-s between two atoms 3 B-24

Hydrogen cyanide, HCN Carbon disulfide, CS 2 1 + 4 + 5 = 10 valence e-s 4 + 6 = 16 valence e-s H 3 B-25 C N S C S

Sulfate, SO 42 - Ammonium, NH 4+ 6 + 4(6) + 2 = 32 valence e-s 5 + 4(1) - 1 = 8 valence e-s 2 - O O S O 3 B-26 O H H N H + H

Ozone, O 3 6 + 6 = 18 valence e-s O O O or O O O ↔ O O O RESONANCE – When more than one Lewis structure can be drawn for a molecule or ion RESONANCE STRUCTURES – The Lewis structures that can be drawn for the molecule or ion The real ozone molecule is an average of its resonance structures 3 B-27

O O O ↔ O O 2 “ 1½” bonds 3 B-28 O O

MOLECULAR SHAPE VSEPR THEORY (Valence Shell Electron Pair Repulsion) – All atoms and lone pairs attached to a central atom will spread out as far as possible to minimize repulsion A Lewis structure must be drawn to use the VSEPR Theory 3 B-29

H H C H H STERIC NUMBER (SN) – The sum of the bonded atoms and lone pairs on a central atom The steric number of carbon is 4 (SN = 4): 4 bonded atoms and no lone pairs H Tetrahedral Bond angle is 109. 5° 3 B-30 H C H H
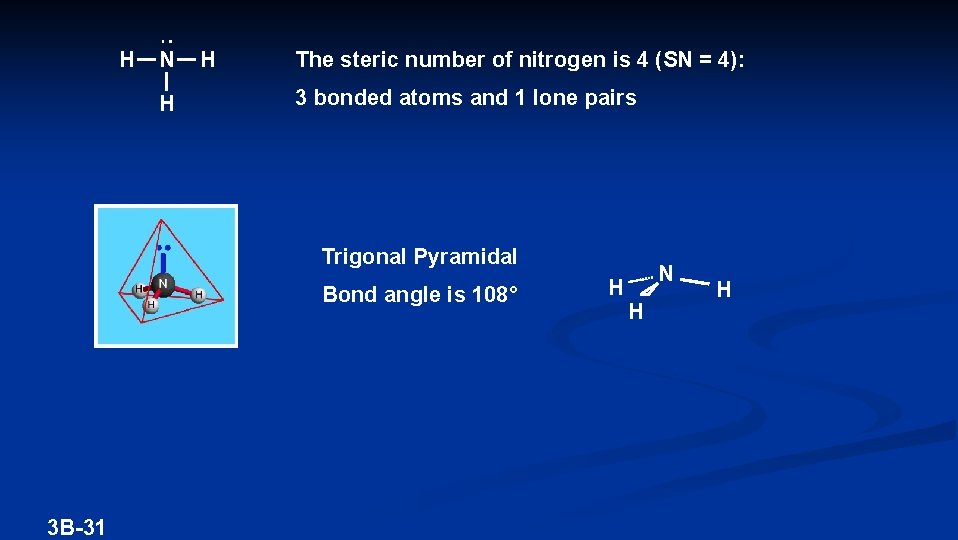
H N H H The steric number of nitrogen is 4 (SN = 4): 3 bonded atoms and 1 lone pairs Trigonal Pyramidal Bond angle is 108° 3 B-31 H N H H
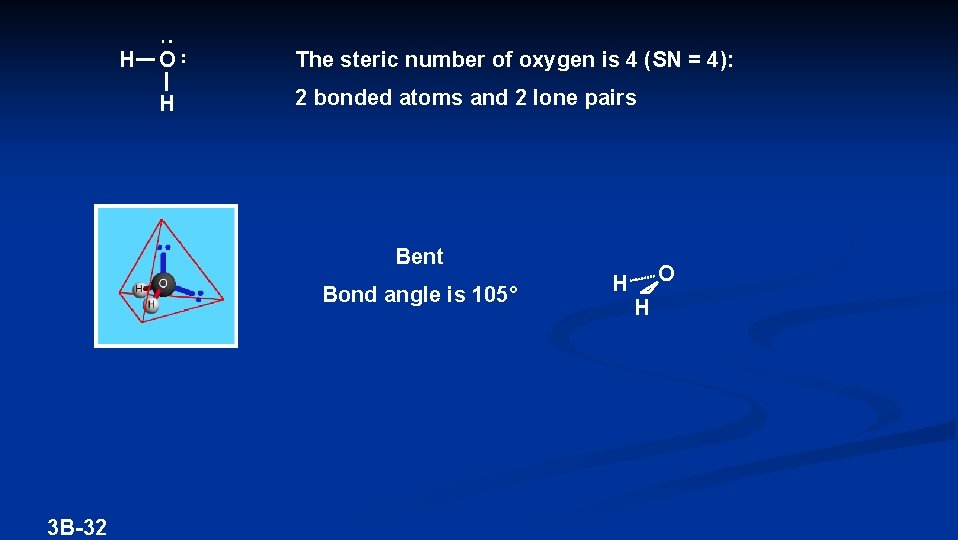
H O The steric number of oxygen is 4 (SN = 4): H 2 bonded atoms and 2 lone pairs Bent Bond angle is 105° 3 B-32 H O H
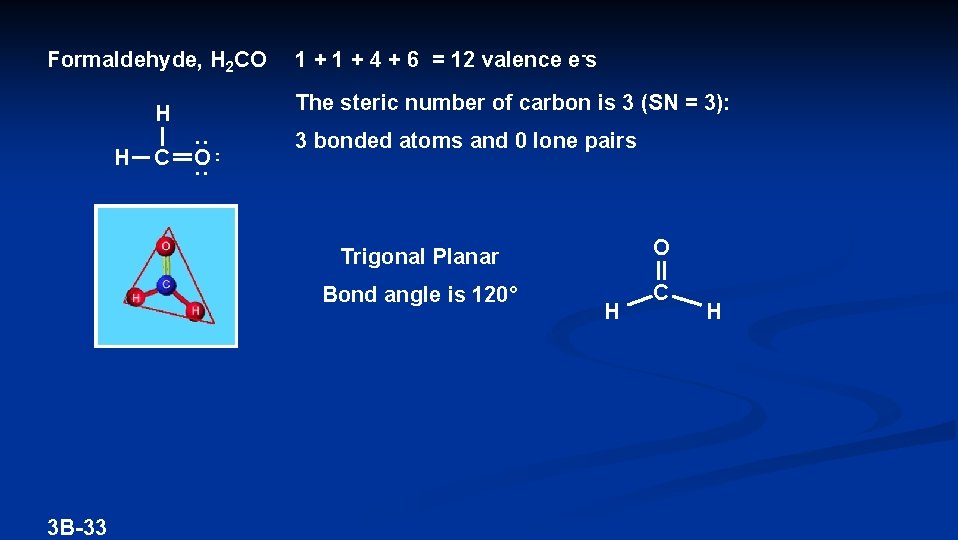
Formaldehyde, H 2 CO The steric number of carbon is 3 (SN = 3): H H 3 B-33 C 1 + 4 + 6 = 12 valence e-s O 3 bonded atoms and 0 lone pairs Trigonal Planar O Bond angle is 120° C H H
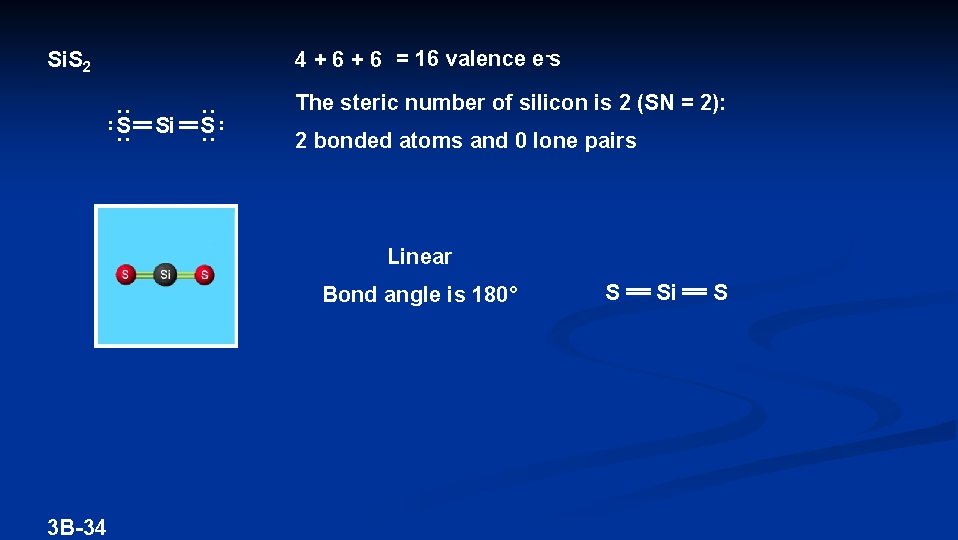
4 + 6 = 16 valence e-s Si. S 2 S Si S The steric number of silicon is 2 (SN = 2): 2 bonded atoms and 0 lone pairs Linear Bond angle is 180° 3 B-34 S Si S
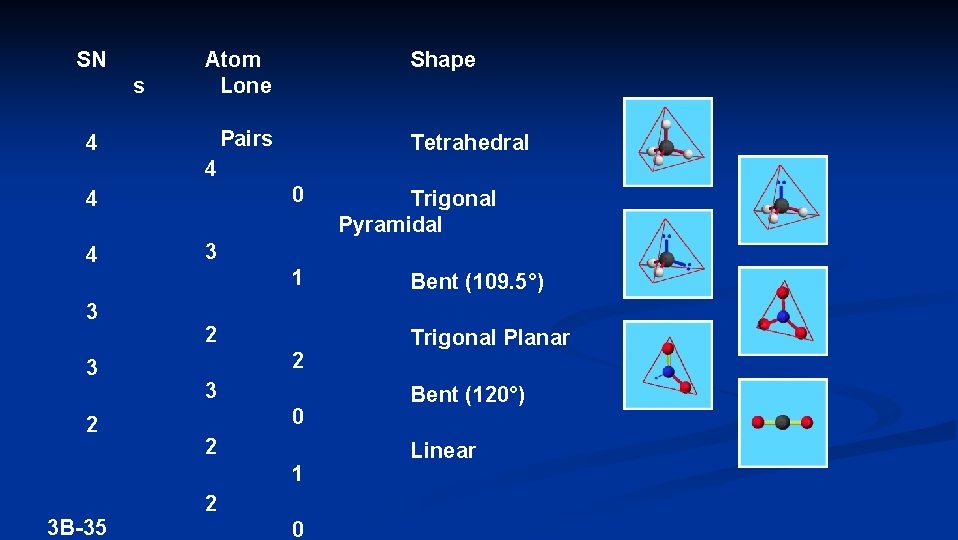
SN s Atom Lone Shape Pairs 4 Tetrahedral 4 0 4 4 3 3 2 3 1 2 2 3 0 2 1 3 B-35 Trigonal Pyramidal 2 0 Bent (109. 5°) Trigonal Planar Bent (120°) Linear

MOLECULAR POLARITY A BOND is polar if it has a positive end a negative end A MOLECULE is polar if it has a positive end a negative end To determine if a molecule is polar or nonpolar: 1) Draw the correct Lewis structure 2) Draw its correct shape 3) Use EN’s to determine if the BONDS in the molecule are polar or nonpolar 4) For the polar bonds, label the positive and negative ends with δ+ and δ 5) If a line can be drawn separating all δ+’s from all δ-’s, the molecule is polar, if not its nonpolar 3 B-36

. . H O: H δ+ H H δ+ 3 B-37 δO δ- EN’s: O = 3. 5, H = 2. 1 3. 5 – 2. 1 = 1. 4 the O-H BONDS are polar All of the δ+’s can be separated from all of the δ-’s, the H 2 O MOLECULE is polar

H N H H δδ- N δ- δ+ H H δ+ 3 B-38 EN’s: N = 3. 0, H = 2. 1 H 3. 0 – 2. 1 = 0. 9 the N-H BONDS are polar δ+ All of the δ+’s can be separated from all of the δ-’s, the NH 3 MOLECULE is polar

Carbon tetrafluoride, CF 4 4 + 4(7) = 32 valence e-s F F C F F δF δ+ δ+δ+ C δ- F δ+ F δF δ- 3 B-39 EN’s: C = 2. 5, F = 4. 0 – 2. 5 = 1. 5 the C-F BONDS are polar All of the δ+’s cannot be separated from all of the δ-’s, the CF 4 MOLECULE is nonpolar

REVIEW FOR TEST Electromagnetic Radiation, Photons Ground State, Excited State Orbital Energy Levels (Sublevels and Number of Orbitals They Contain) Sublevels (Shape, Number, and Starting Energy Level) Orbital Notation Electron Configuration Notation Electron Dot Notation 2 B-40

REVIEW FOR TEST Valence Electrons Octet Electron Pair Periodic Trends in Metal, Nonmetal Activity Atomic Radii Ionization Energy (and Write Equation) Electron Affinity (and Write Equation) 2 B-41

REVIEW FOR TEST Ionic Bonds, Covalent Bonds Electronegativity and its Periodic Trend Bond Polarity from Electronegativities Ion Sizes Lewis Structures for Ionic Compounds and Covalent Compounds Resonance Molecular Shapes Molecular Polarity 2 B-42
- Slides: 42