Chapter 12 Cell Biology of the Immune System

Chapter 12 Cell Biology of the Immune System © 2020 Elsevier Inc. All rights reserved.
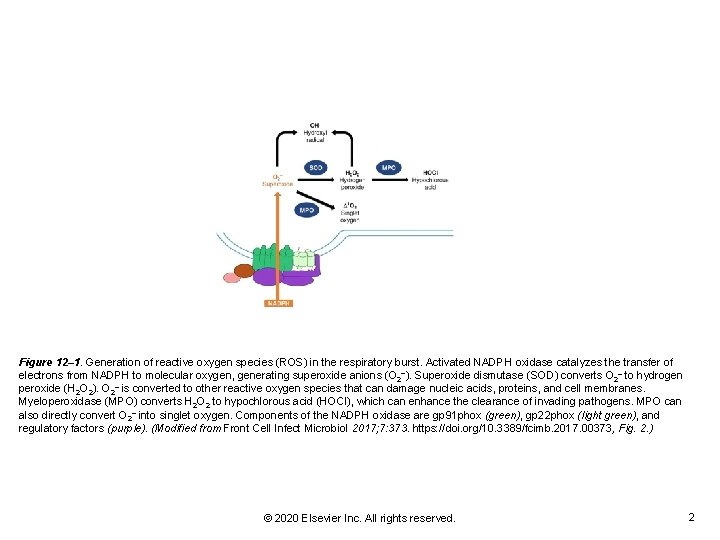
Figure 12– 1. Generation of reactive oxygen species (ROS) in the respiratory burst. Activated NADPH oxidase catalyzes the transfer of electrons from NADPH to molecular oxygen, generating superoxide anions (O 2−). Superoxide dismutase (SOD) converts O 2– to hydrogen peroxide (H 2 O 2). O 2– is converted to other reactive oxygen species that can damage nucleic acids, proteins, and cell membranes. Myeloperoxidase (MPO) converts H 2 O 2 to hypochlorous acid (HOCl), which can enhance the clearance of invading pathogens. MPO can also directly convert O 2– into singlet oxygen. Components of the NADPH oxidase are gp 91 phox (green), gp 22 phox (light green), and regulatory factors (purple). (Modified from Front Cell Infect Microbiol 2017; 7: 373. https: //doi. org/10. 3389/fcimb. 2017. 00373, Fig. 2. ) © 2020 Elsevier Inc. All rights reserved. 2

Figure 12– 2. Scanning electron micrograph of human neutrophil extracellular traps (NETs) with entrapped Salmonella. Bar = 1 μm. (Modified from Brinkmann V, Zychlinsky A. Neutrophil extracellular traps: is immunity the second function of chromatin? J Cell Biol 2012; 198(5): 773– 83. https: //doi. org/10. 1083/jcb. 201203170. ) © 2020 Elsevier Inc. All rights reserved. 3

Figure 12– 3. Leukocyte adhesion and migration. (Modified from Garrod DR. Cell to cell and cell to matrix adhesion. In: Latchman D, ed. Basic Molecular & Cell Biology, 3 rd ed. Oxford, United Kingdom: Blackwell BMJ Books, 1997: 80– 91, by permission. ) © 2020 Elsevier Inc. All rights reserved. 4

Figure 12– 4. A diagram depicting the T-cell receptor structure. Each of the TCR α- and β-chain comprises a V and a C domain. The αand β-chains are joined by disulfide bonds. Carbohydrate side chains are indicated (CHO). Positive amino acid residues in the transmembrane region are depicted by +. (Based on Al-Lazikani B, Lesk AM, Chothia C. Canonical structures for the hypervariable regions of T cell alpha-beta receptors. J Mol Biol 2000; 295(4): 979– 95. Fig. 1. ) © 2020 Elsevier Inc. All rights reserved. 5
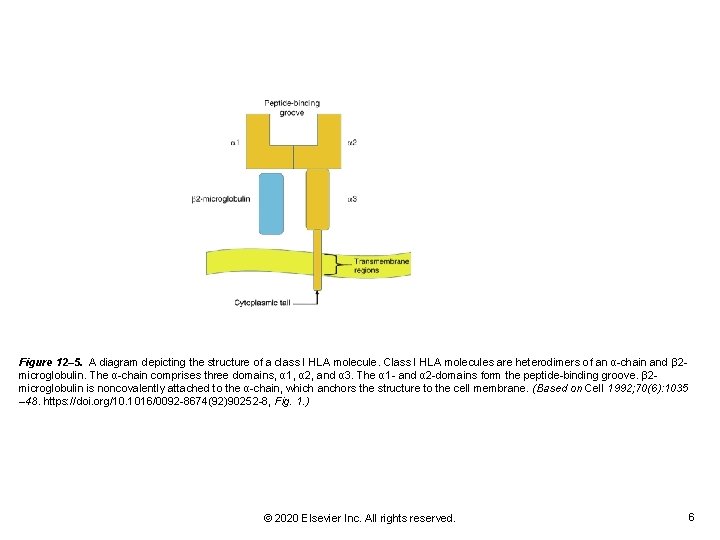
Figure 12– 5. A diagram depicting the structure of a class I HLA molecule. Class I HLA molecules are heterodimers of an α-chain and β 2 microglobulin. The α-chain comprises three domains, α 1, α 2, and α 3. The α 1 - and α 2 -domains form the peptide-binding groove. β 2 microglobulin is noncovalently attached to the α-chain, which anchors the structure to the cell membrane. (Based on Cell 1992; 70(6): 1035 – 48. https: //doi. org/10. 1016/0092 -8674(92)90252 -8, Fig. 1. ) © 2020 Elsevier Inc. All rights reserved. 6
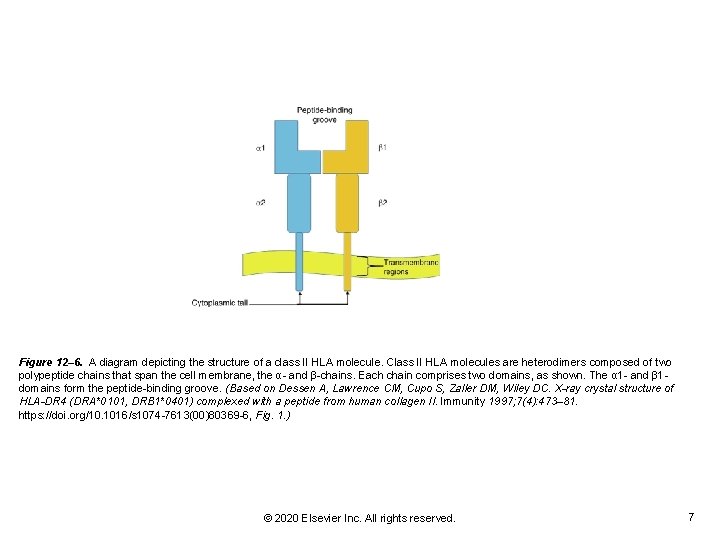
Figure 12– 6. A diagram depicting the structure of a class II HLA molecule. Class II HLA molecules are heterodimers composed of two polypeptide chains that span the cell membrane, the α- and β-chains. Each chain comprises two domains, as shown. The α 1 - and β 1 domains form the peptide-binding groove. (Based on Dessen A, Lawrence CM, Cupo S, Zaller DM, Wiley DC. X-ray crystal structure of HLA-DR 4 (DRA*0101, DRB 1*0401) complexed with a peptide from human collagen II. Immunity 1997; 7(4): 473– 81. https: //doi. org/10. 1016/s 1074 -7613(00)80369 -6, Fig. 1. ) © 2020 Elsevier Inc. All rights reserved. 7
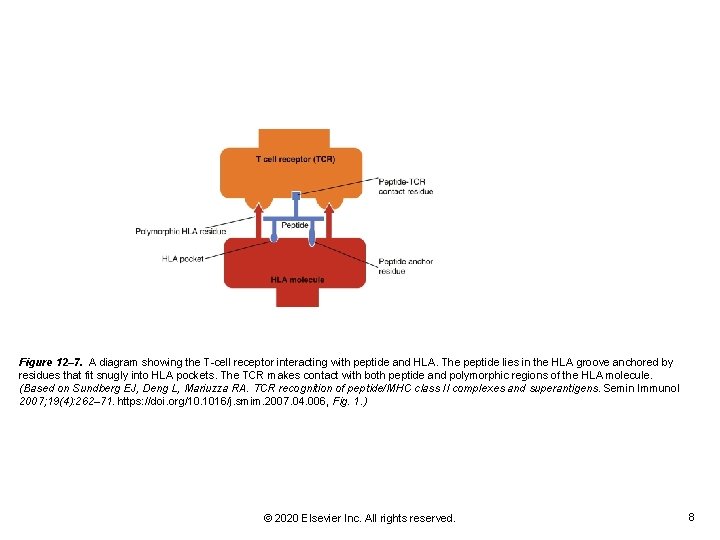
Figure 12– 7. A diagram showing the T-cell receptor interacting with peptide and HLA. The peptide lies in the HLA groove anchored by residues that fit snugly into HLA pockets. The TCR makes contact with both peptide and polymorphic regions of the HLA molecule. (Based on Sundberg EJ, Deng L, Mariuzza RA. TCR recognition of peptide/MHC class II complexes and superantigens. Semin Immunol 2007; 19(4): 262– 71. https: //doi. org/10. 1016/j. smim. 2007. 04. 006, Fig. 1. ) © 2020 Elsevier Inc. All rights reserved. 8

Figure 12– 8. Genomic organization of immunoglobulin genes. The structure of germline Ig genes is depicted for the heavy (A) and light chain (B and C) genes. The boxes represent V, D, J, and C DNA segments. Boxes labeled L represent leader sequences. An example is shown of a V(D)J recombination event leading to the formation of the VH 2 -D 3 -J 5 -Cμ coding sequence, with the deletion of intervening DNA, which codes for an Ig. M heavy chain (μ). (Data from Burmester G-R. Color Atlas of Immunology. New York: Thieme, 2003. ) © 2020 Elsevier Inc. All rights reserved. 9
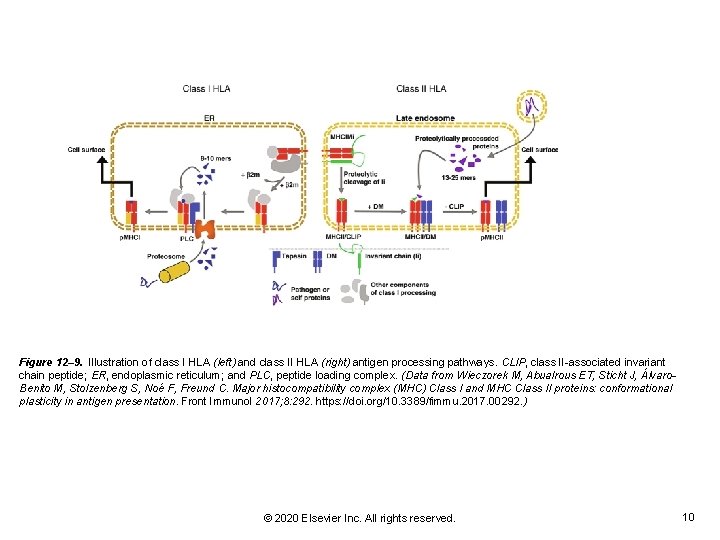
Figure 12– 9. Illustration of class I HLA (left) and class II HLA (right) antigen processing pathways. CLIP, class II-associated invariant chain peptide; ER, endoplasmic reticulum; and PLC, peptide loading complex. (Data from Wieczorek M, Abualrous ET, Sticht J, Álvaro. Benito M, Stolzenberg S, Noé F, Freund C. Major histocompatibility complex (MHC) Class I and MHC Class II proteins: conformational plasticity in antigen presentation. Front Immunol 2017; 8: 292. https: //doi. org/10. 3389/fimmu. 2017. 00292. ) © 2020 Elsevier Inc. All rights reserved. 10
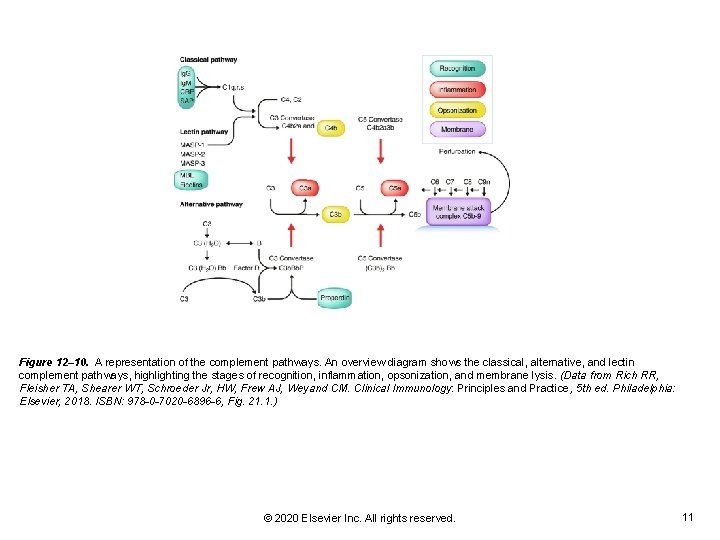
Figure 12– 10. A representation of the complement pathways. An overview diagram shows the classical, alternative, and lectin complement pathways, highlighting the stages of recognition, inflammation, opsonization, and membrane lysis. (Data from Rich RR, Fleisher TA, Shearer WT, Schroeder Jr, HW, Frew AJ, Weyand CM. Clinical Immunology: Principles and Practice, 5 th ed. Philadelphia: Elsevier, 2018. ISBN: 978 -0 -7020 -6896 -6, Fig. 21. 1. ) © 2020 Elsevier Inc. All rights reserved. 11
- Slides: 11