Chapter 12 Alcohols from Carbonyl Compounds OxidationReduction Organometallic

Chapter 12 Alcohols from Carbonyl Compounds Oxidation-Reduction & Organometallic Compounds Created by Professor William Tam & Dr. Phillis Chang © 2014 by John Wiley & Sons, Inc. All rights reserved.
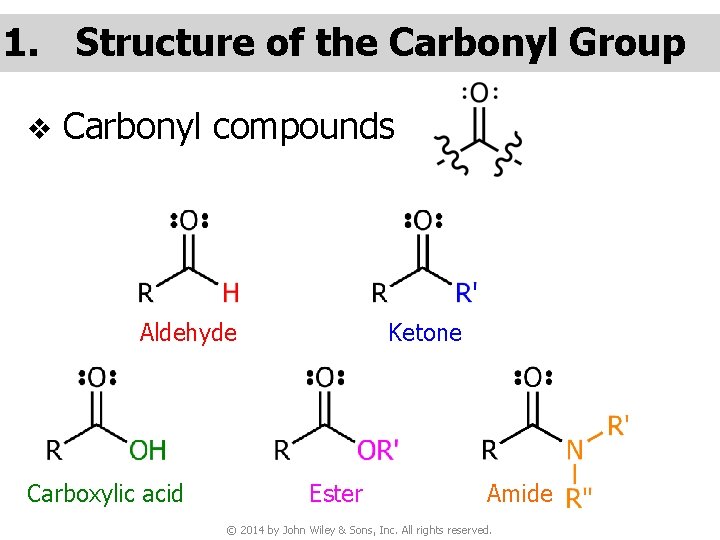
1. Structure of the Carbonyl Group v Carbonyl compounds Aldehyde Carboxylic acid Ketone Ester Amide © 2014 by John Wiley & Sons, Inc. All rights reserved.
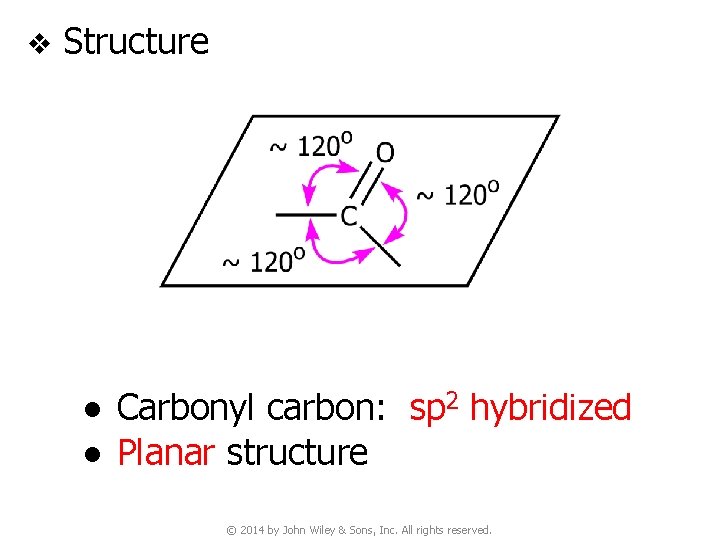
v Structure ● Carbonyl carbon: sp 2 hybridized ● Planar structure © 2014 by John Wiley & Sons, Inc. All rights reserved.
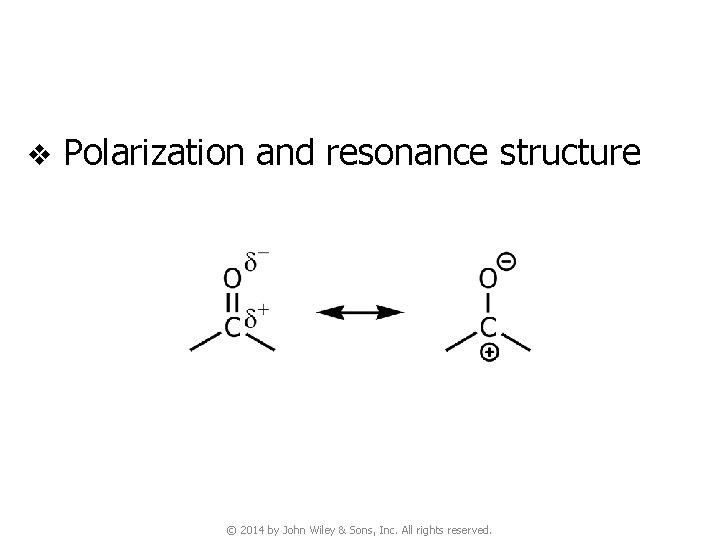
v Polarization and resonance structure © 2014 by John Wiley & Sons, Inc. All rights reserved.

1 A. Reactions of Carbonyl Compounds with Nucleophiles v One of the most important reactions of carbonyl compounds is nucleophilic addition to the carbonyl group © 2014 by John Wiley & Sons, Inc. All rights reserved.
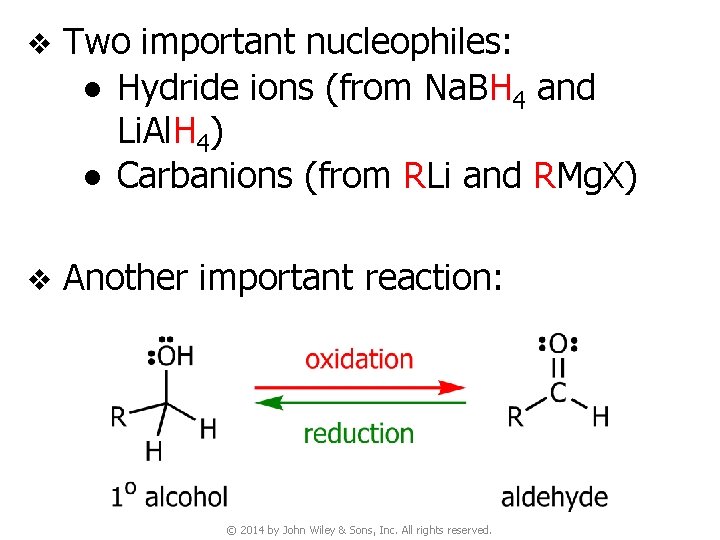
v Two important nucleophiles: ● Hydride ions (from Na. BH 4 and Li. Al. H 4) ● Carbanions (from RLi and RMg. X) v Another important reaction: © 2014 by John Wiley & Sons, Inc. All rights reserved.
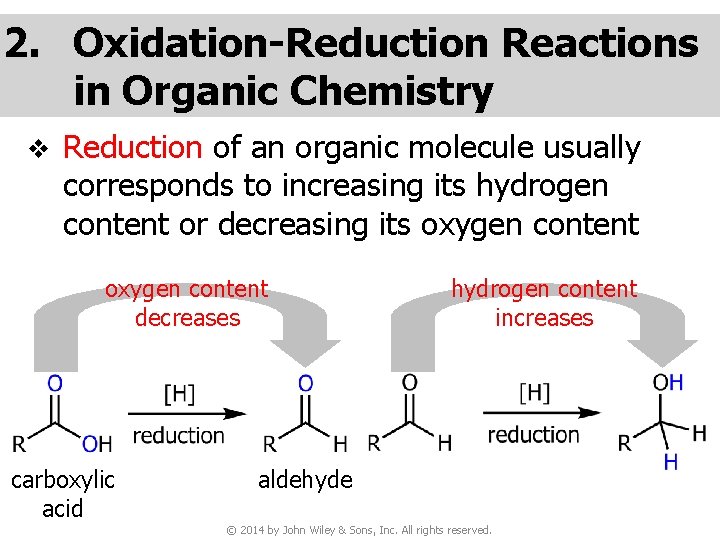
2. Oxidation-Reduction Reactions in Organic Chemistry v Reduction of an organic molecule usually corresponds to increasing its hydrogen content or decreasing its oxygen content decreases carboxylic acid hydrogen content increases aldehyde © 2014 by John Wiley & Sons, Inc. All rights reserved.
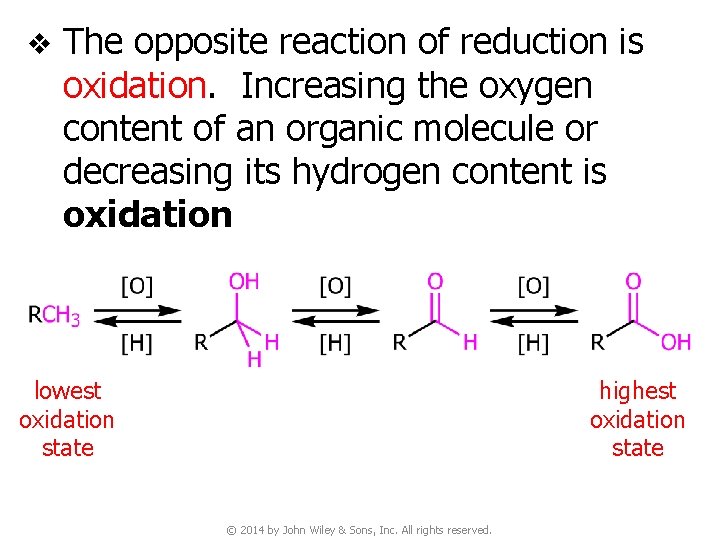
v The opposite reaction of reduction is oxidation. Increasing the oxygen content of an organic molecule or decreasing its hydrogen content is oxidation lowest oxidation state highest oxidation state © 2014 by John Wiley & Sons, Inc. All rights reserved.
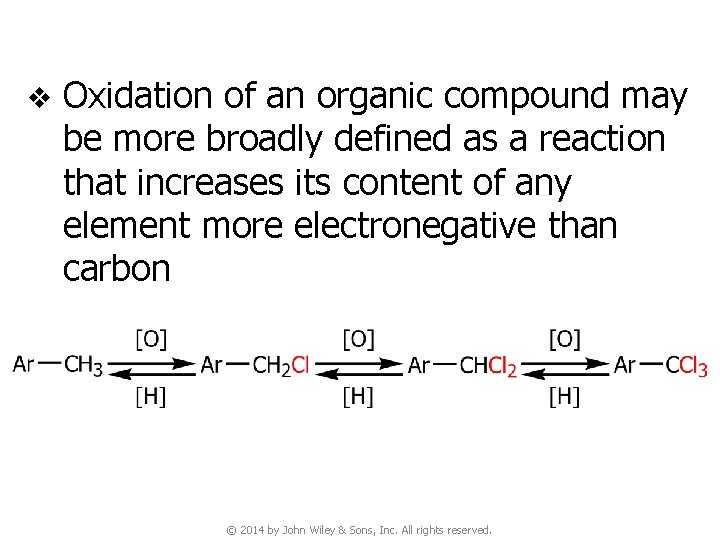
v Oxidation of an organic compound may be more broadly defined as a reaction that increases its content of any element more electronegative than carbon © 2014 by John Wiley & Sons, Inc. All rights reserved.

2 A. Oxidation States in Organic Chemistry v Rules ● For each C–H (or C–M) bond -1 ● For each C–C bond 0 ● For each C–Z bond +1 (where M = electropositive element and is equivalent to H, e. g. Li, K, etc. ; Z = electronegative heteroatom, e. g. OR, SR, PR 2, halogen, etc. ) © 2014 by John Wiley & Sons, Inc. All rights reserved.

v Calculate the oxidation state of each carbon based on the number of bonds it is forming to atoms more (or less) electronegative than carbon © 2014 by John Wiley & Sons, Inc. All rights reserved.
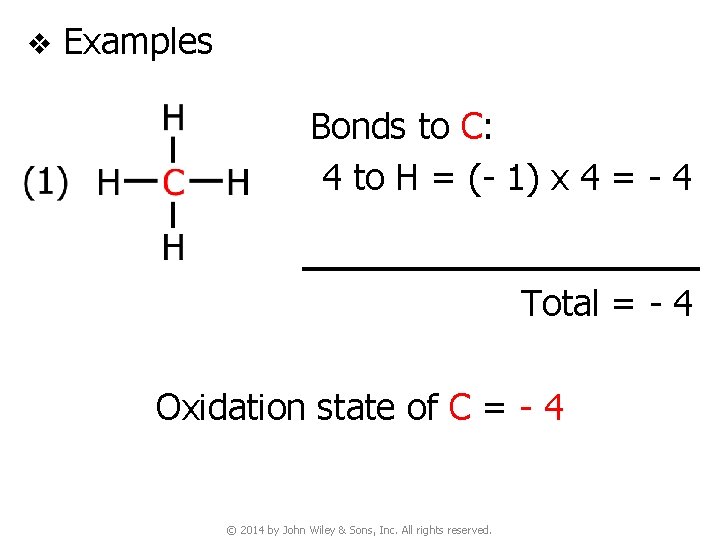
v Examples Bonds to C: 4 to H = (- 1) x 4 = - 4 Total = - 4 Oxidation state of C = - 4 © 2014 by John Wiley & Sons, Inc. All rights reserved.
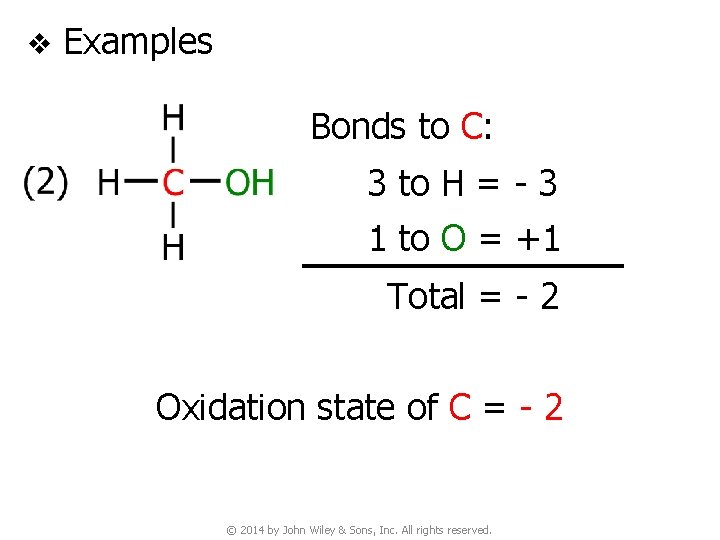
v Examples Bonds to C: 3 to H = - 3 1 to O = +1 Total = - 2 Oxidation state of C = - 2 © 2014 by John Wiley & Sons, Inc. All rights reserved.
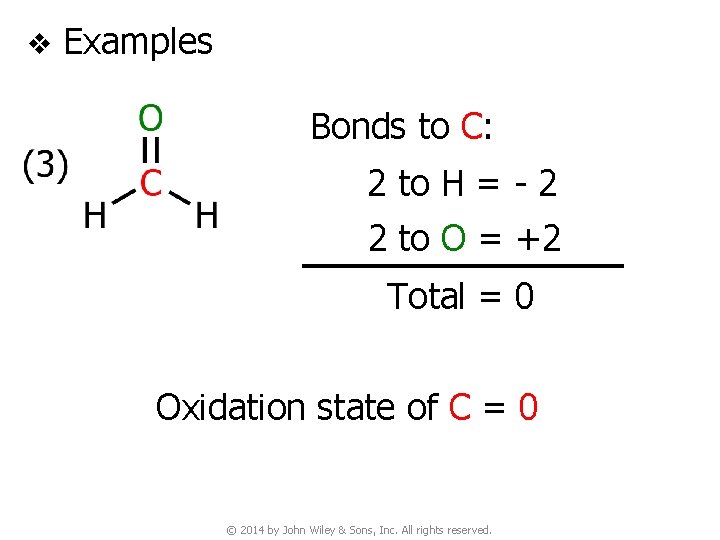
v Examples Bonds to C: 2 to H = - 2 2 to O = +2 Total = 0 Oxidation state of C = 0 © 2014 by John Wiley & Sons, Inc. All rights reserved.
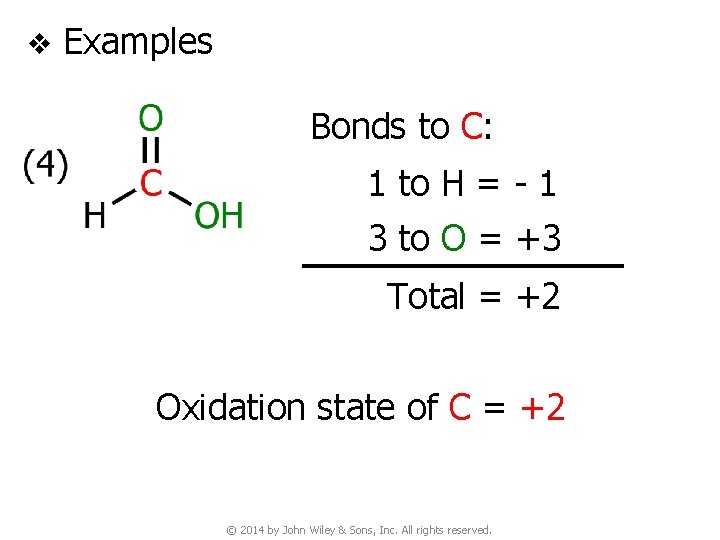
v Examples Bonds to C: 1 to H = - 1 3 to O = +3 Total = +2 Oxidation state of C = +2 © 2014 by John Wiley & Sons, Inc. All rights reserved.
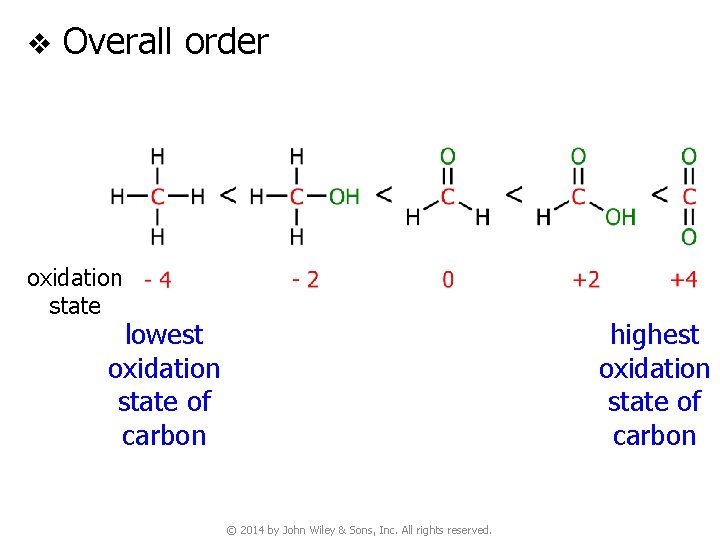
v Overall order oxidation state lowest oxidation state of carbon highest oxidation state of carbon © 2014 by John Wiley & Sons, Inc. All rights reserved.
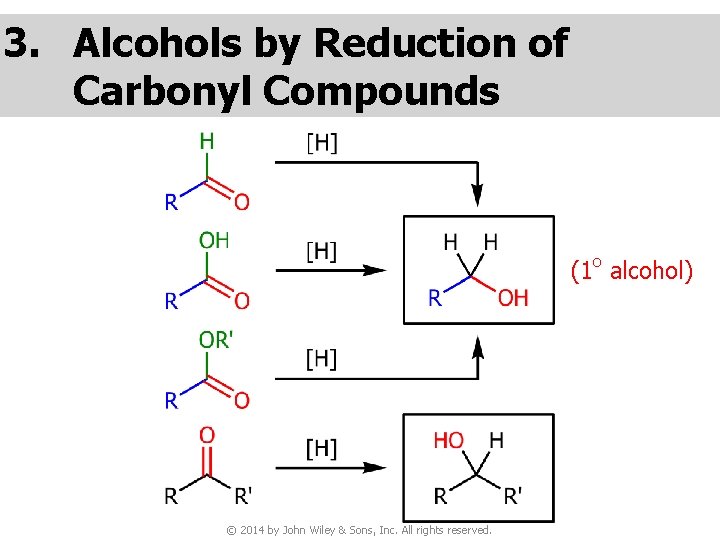
3. Alcohols by Reduction of Carbonyl Compounds (1 o alcohol) © 2014 by John Wiley & Sons, Inc. All rights reserved.

3 A. Lithium Aluminum Hydride v Li. Al. H 4 (LAH) ● Not only nucleophilic, but also very basic ● Reacts violently with H 2 O or acidic protons (e. g. ROH) ● Usually reactions run in ethereal solvents (e. g. Et 2 O, THF) ● Reduces all carbonyl groups © 2014 by John Wiley & Sons, Inc. All rights reserved.
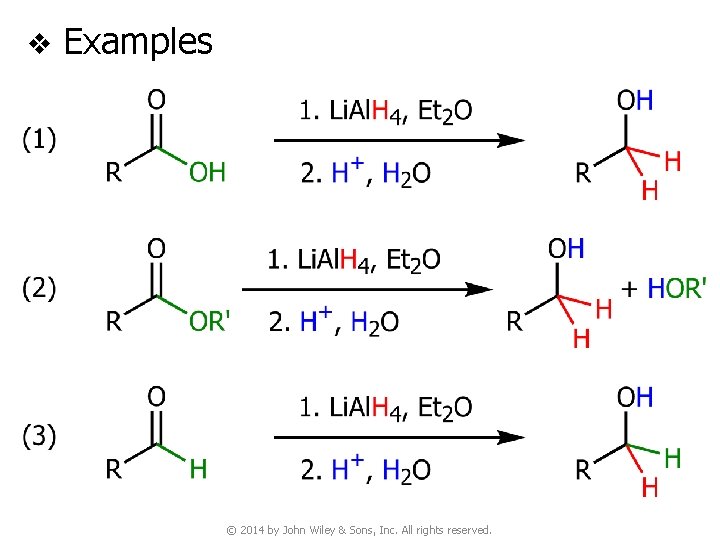
v Examples © 2014 by John Wiley & Sons, Inc. All rights reserved.
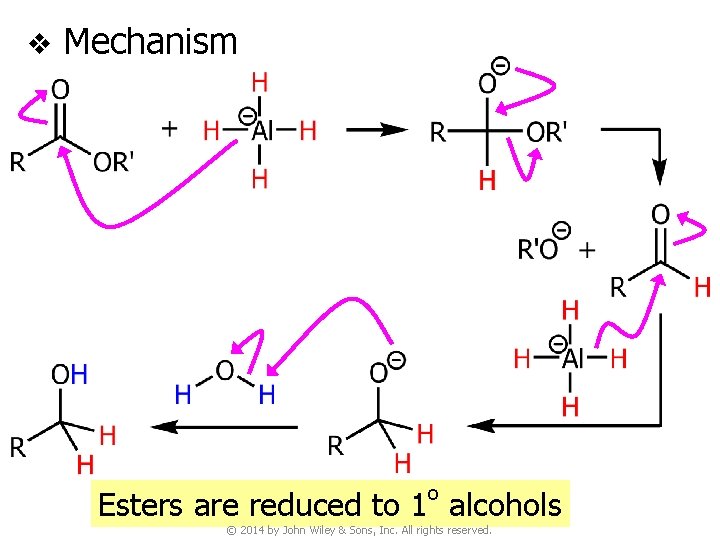
v Mechanism o Esters are reduced to 1 alcohols © 2014 by John Wiley & Sons, Inc. All rights reserved.

3 B. Sodium Borohydride v Na. BH 4 ● less reactive and less basic than Li. Al. H 4 ● can use protic solvent (e. g. ROH) ● reduces only more reactive carbonyl groups (i. e. aldehydes and ketones) but not reactive towards esters or carboxylic acids © 2014 by John Wiley & Sons, Inc. All rights reserved.
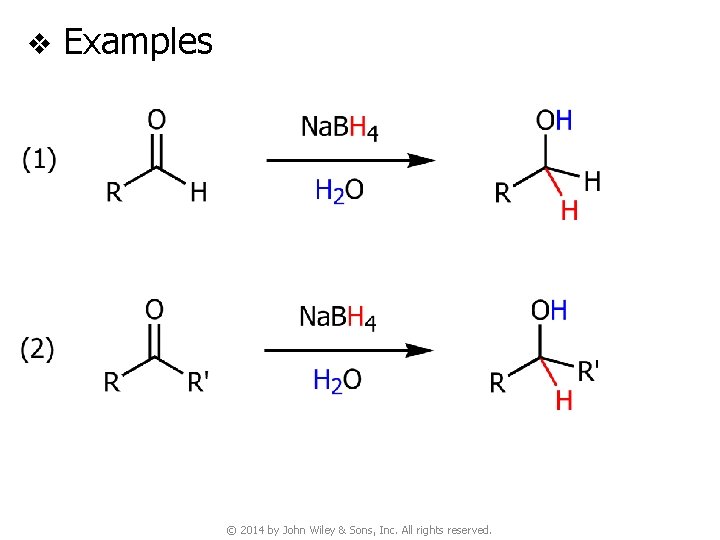
v Examples © 2014 by John Wiley & Sons, Inc. All rights reserved.
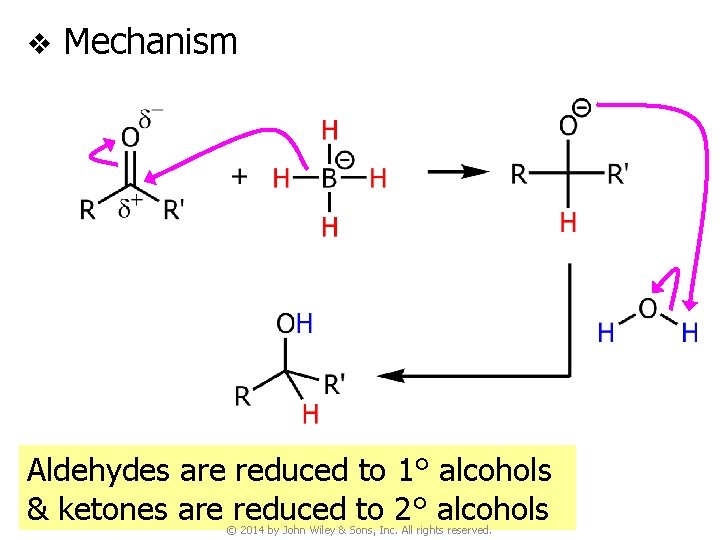
v Mechanism Aldehydes are reduced to 1° alcohols & ketones are reduced to 2° alcohols © 2014 by John Wiley & Sons, Inc. All rights reserved.
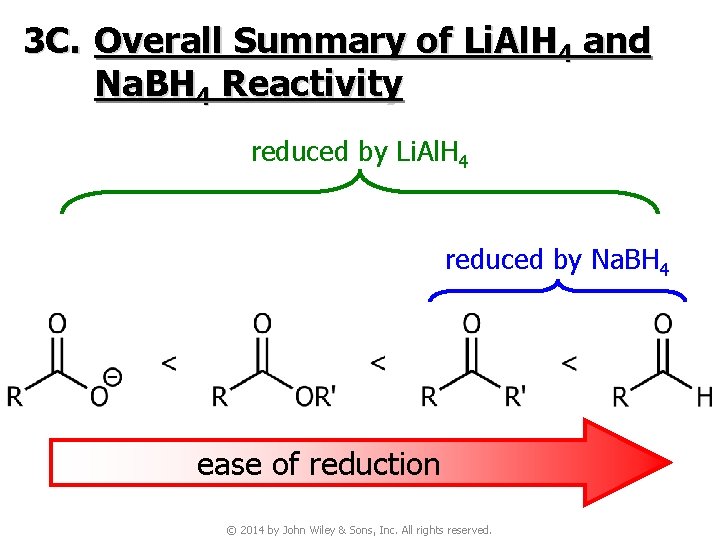
3 C. Overall Summary of Li. Al. H 4 and Na. BH 4 Reactivity reduced by Li. Al. H 4 reduced by Na. BH 4 ease of reduction © 2014 by John Wiley & Sons, Inc. All rights reserved.
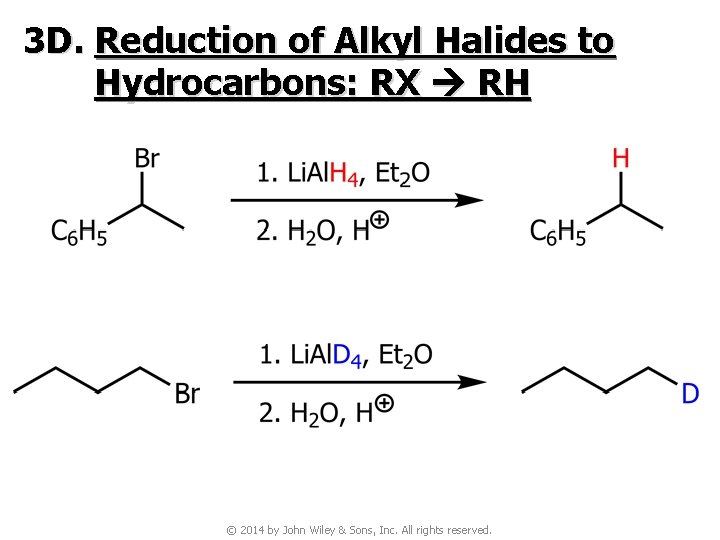
3 D. Reduction of Alkyl Halides to Hydrocarbons: RX RH © 2014 by John Wiley & Sons, Inc. All rights reserved.
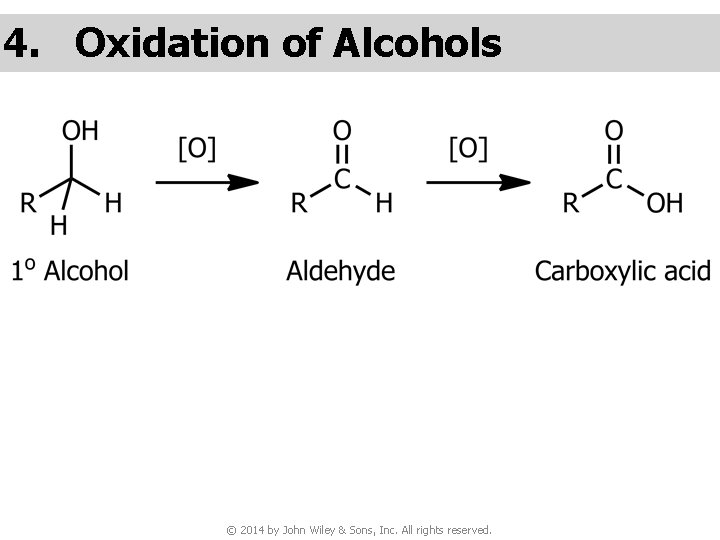
4. Oxidation of Alcohols © 2014 by John Wiley & Sons, Inc. All rights reserved.
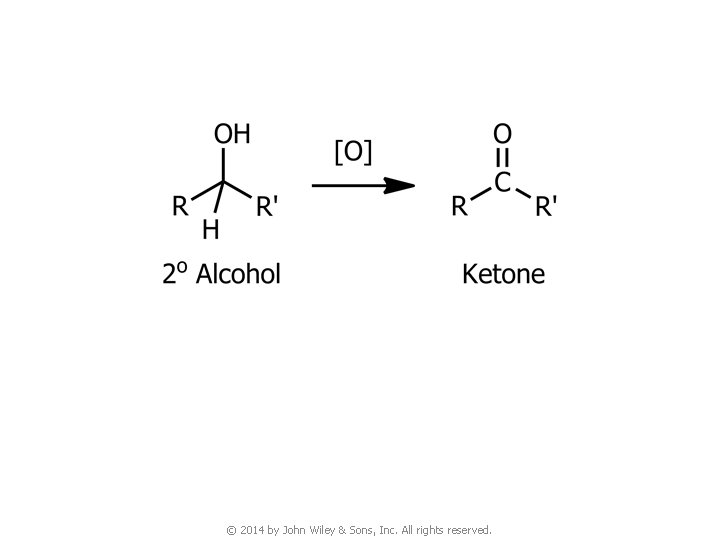
© 2014 by John Wiley & Sons, Inc. All rights reserved.
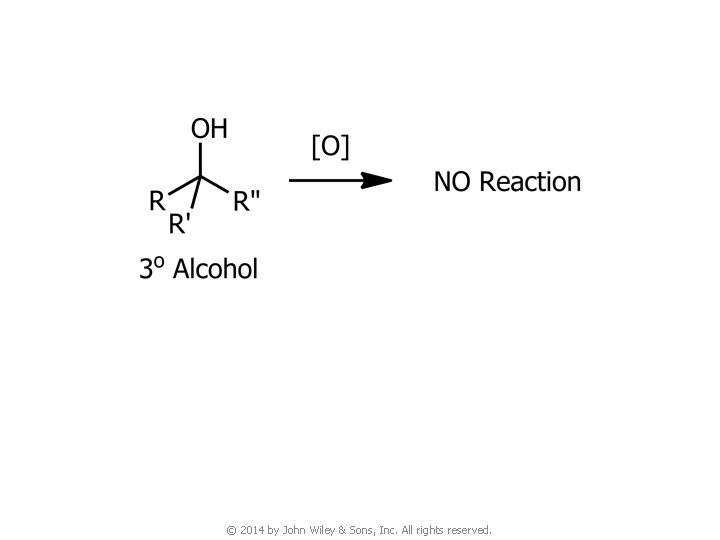
© 2014 by John Wiley & Sons, Inc. All rights reserved.
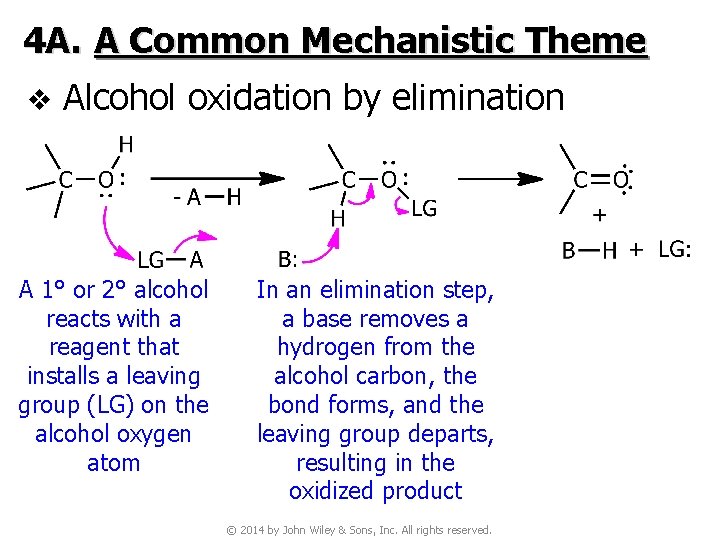
4 A. A Common Mechanistic Theme v Alcohol oxidation by elimination A 1° or 2° alcohol reacts with a reagent that installs a leaving group (LG) on the alcohol oxygen atom In an elimination step, a base removes a hydrogen from the alcohol carbon, the bond forms, and the leaving group departs, resulting in the oxidized product © 2014 by John Wiley & Sons, Inc. All rights reserved.
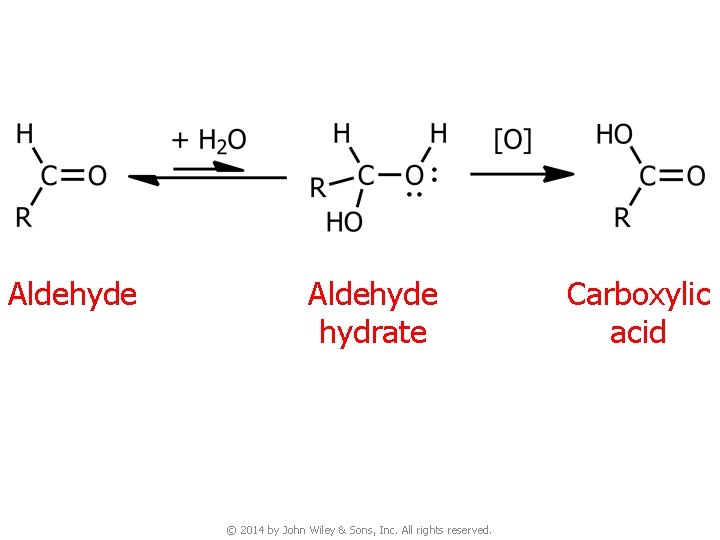
Aldehyde hydrate © 2014 by John Wiley & Sons, Inc. All rights reserved. Carboxylic acid
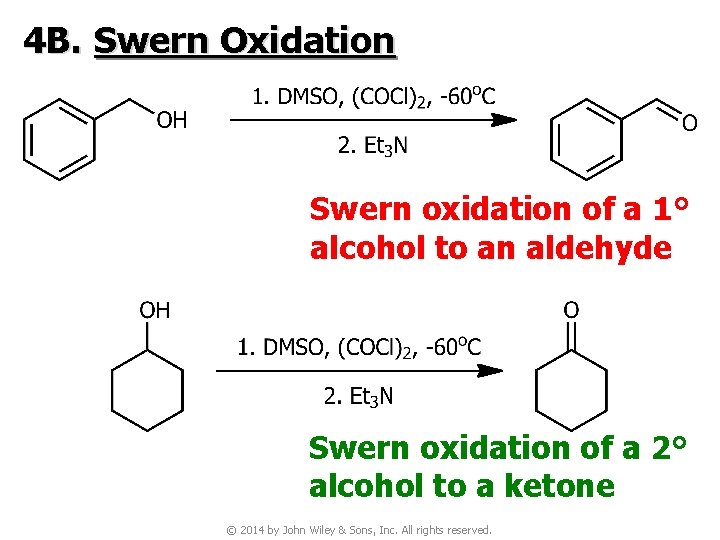
4 B. Swern Oxidation Swern oxidation of a 1° alcohol to an aldehyde Swern oxidation of a 2° alcohol to a ketone © 2014 by John Wiley & Sons, Inc. All rights reserved.
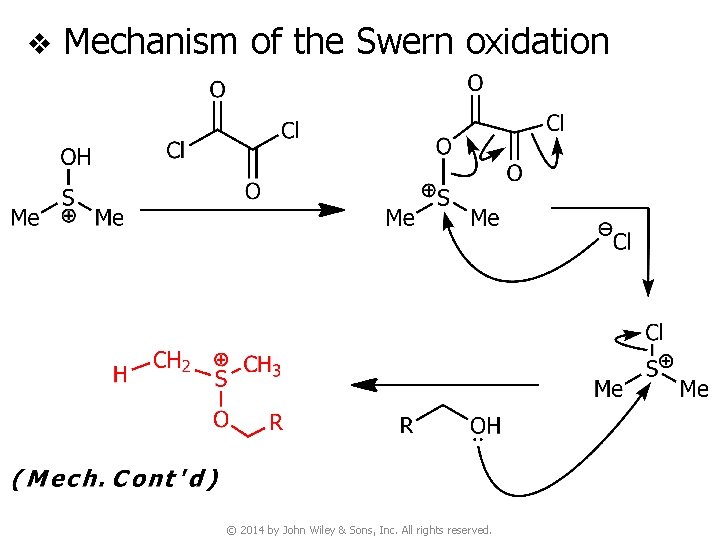
v Mechanism of the Swern oxidation © 2014 by John Wiley & Sons, Inc. All rights reserved.
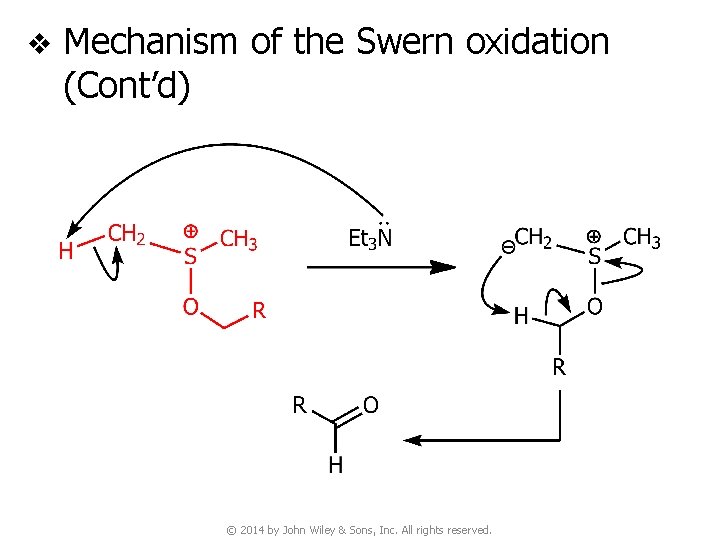
v Mechanism of the Swern oxidation (Cont’d) © 2014 by John Wiley & Sons, Inc. All rights reserved.
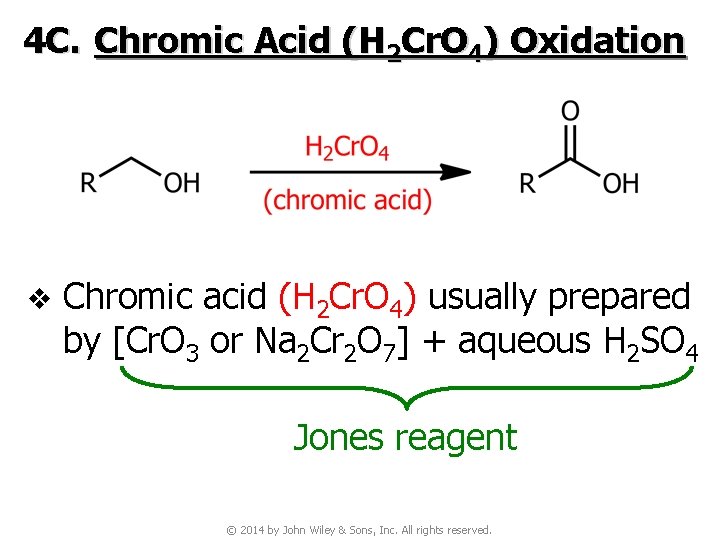
4 C. Chromic Acid (H 2 Cr. O 4) Oxidation v Chromic acid (H 2 Cr. O 4) usually prepared by [Cr. O 3 or Na 2 Cr 2 O 7] + aqueous H 2 SO 4 Jones reagent © 2014 by John Wiley & Sons, Inc. All rights reserved.

v Jones oxidation ● Reagent: Cr. O 3 + aqueous H 2 SO 4 ● A Cr(VI) oxidant © 2014 by John Wiley & Sons, Inc. All rights reserved.
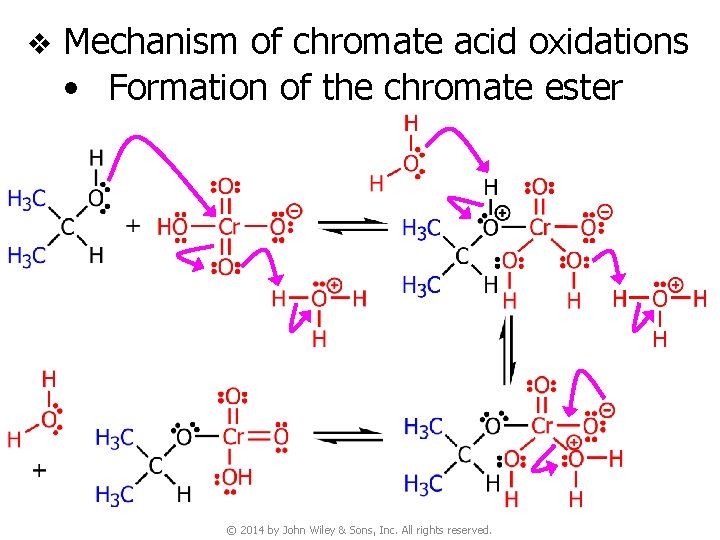
v Mechanism of chromate acid oxidations • Formation of the chromate ester © 2014 by John Wiley & Sons, Inc. All rights reserved.
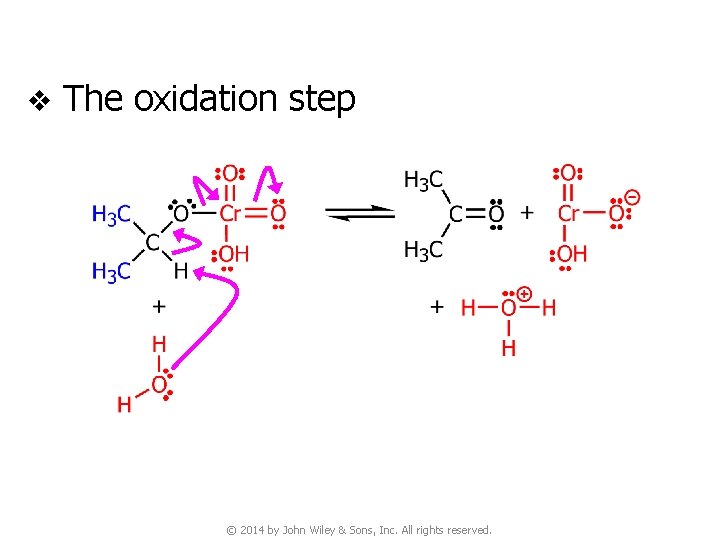
v The oxidation step © 2014 by John Wiley & Sons, Inc. All rights reserved.
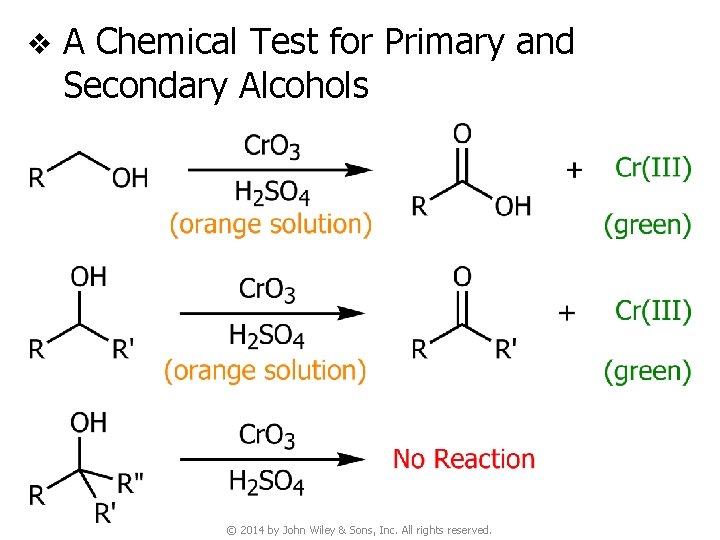
v A Chemical Test for Primary and Secondary Alcohols © 2014 by John Wiley & Sons, Inc. All rights reserved.
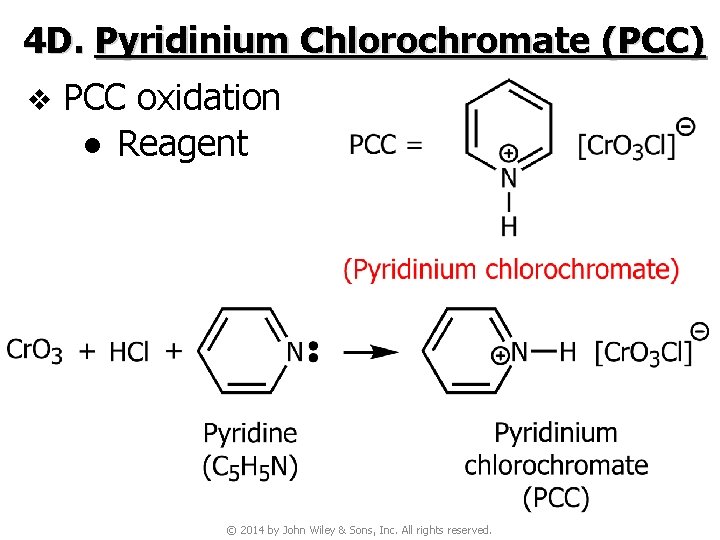
4 D. Pyridinium Chlorochromate (PCC) v PCC oxidation ● Reagent © 2014 by John Wiley & Sons, Inc. All rights reserved.
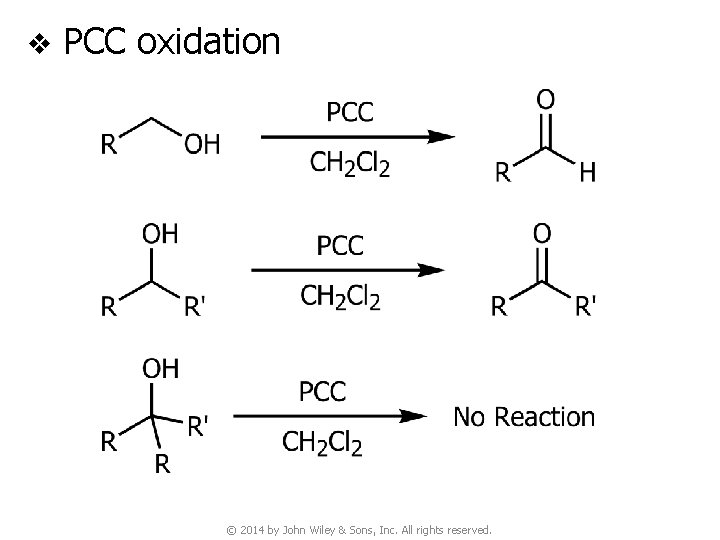
v PCC oxidation © 2014 by John Wiley & Sons, Inc. All rights reserved.
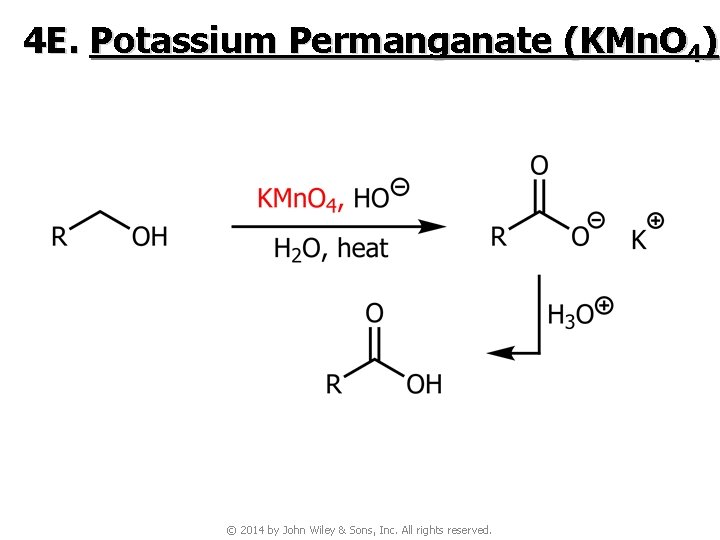
4 E. Potassium Permanganate (KMn. O 4) © 2014 by John Wiley & Sons, Inc. All rights reserved.

5. Organometallic Compounds v Compounds that contain carbon-metal bonds are called organometallic compounds © 2014 by John Wiley & Sons, Inc. All rights reserved.

6. Preparation of Organolithium & Organomagnesium Compounds 6 A. Organolithium Compounds v Preparation of organolithium compounds v Order of reactivity of RX ● RI > RBr > RCl © 2014 by John Wiley & Sons, Inc. All rights reserved.
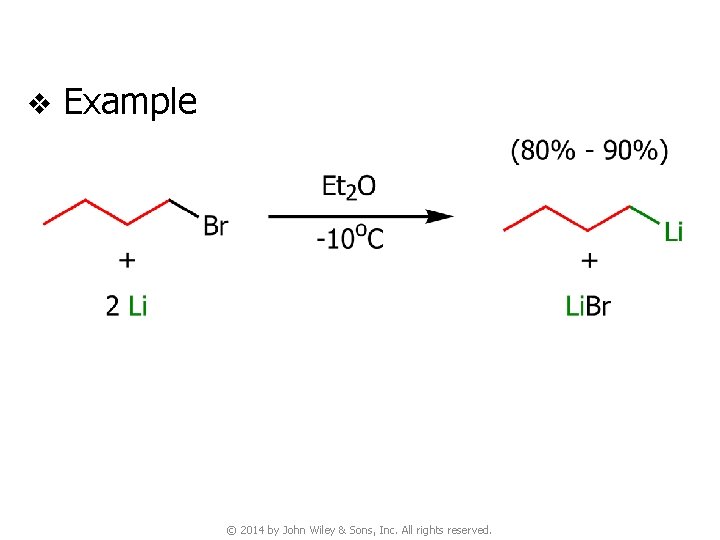
v Example © 2014 by John Wiley & Sons, Inc. All rights reserved.

6 B. Grignard Reagents v Preparation of organomagnesium compounds (Grignard reagents) v Order of reactivity of RX ● RI > RBr > RCl © 2014 by John Wiley & Sons, Inc. All rights reserved.
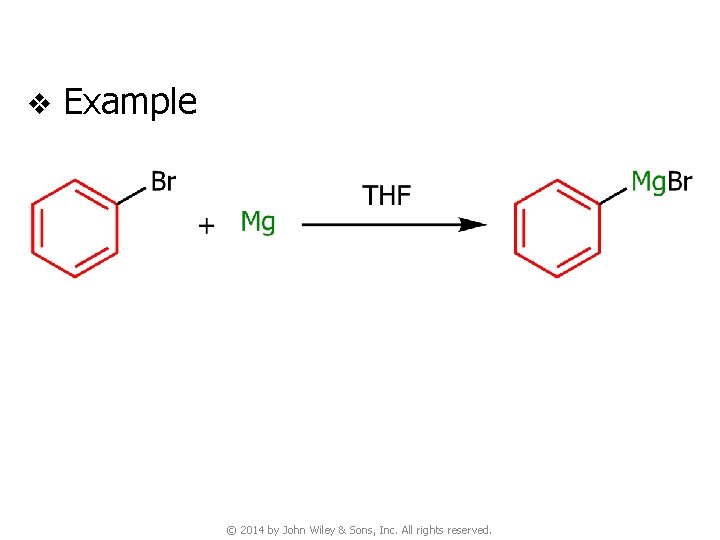
v Example © 2014 by John Wiley & Sons, Inc. All rights reserved.

7. Reactions of Organolithium and Organomagnesium Compounds 7 A. Reactions with Compounds Containing Acidic Hydrogen Atoms © 2014 by John Wiley & Sons, Inc. All rights reserved.

v Grignard reagents and organolithium compounds are very strong bases © 2014 by John Wiley & Sons, Inc. All rights reserved.
v Examples ● As base © 2014 by John Wiley & Sons, Inc. All rights reserved.
v Examples ● As base A good method for the preparation of alkynylmagnesium halides © 2014 by John Wiley & Sons, Inc. All rights reserved.

7 B. Reactions of Grignard Reagents with Epoxides (Oxiranes) v Grignard reagents react as nucleophiles with epoxides (oxiranes), providing convenient synthesis of alcohols © 2014 by John Wiley & Sons, Inc. All rights reserved.
v Via SN 2 reaction © 2014 by John Wiley & Sons, Inc. All rights reserved.
v Also works for substituted epoxides © 2014 by John Wiley & Sons, Inc. All rights reserved.
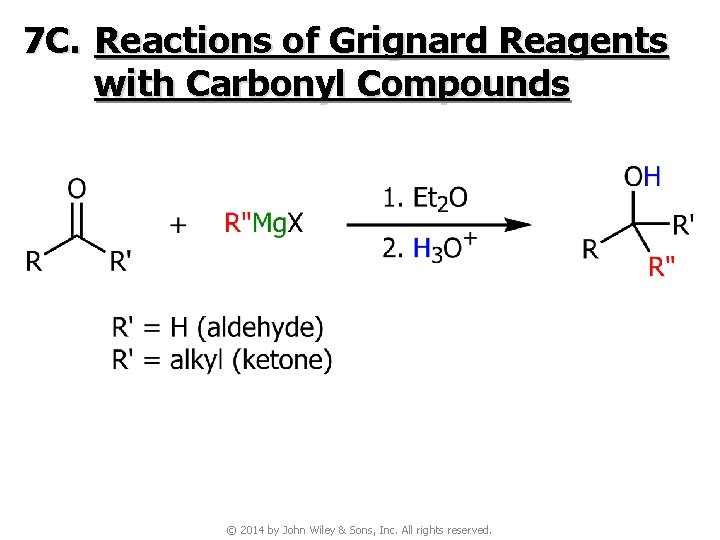
7 C. Reactions of Grignard Reagents with Carbonyl Compounds © 2014 by John Wiley & Sons, Inc. All rights reserved.
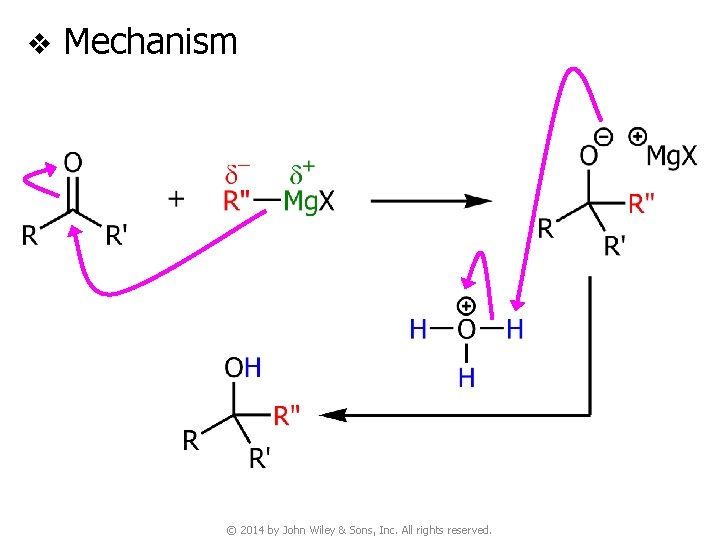
v Mechanism © 2014 by John Wiley & Sons, Inc. All rights reserved.
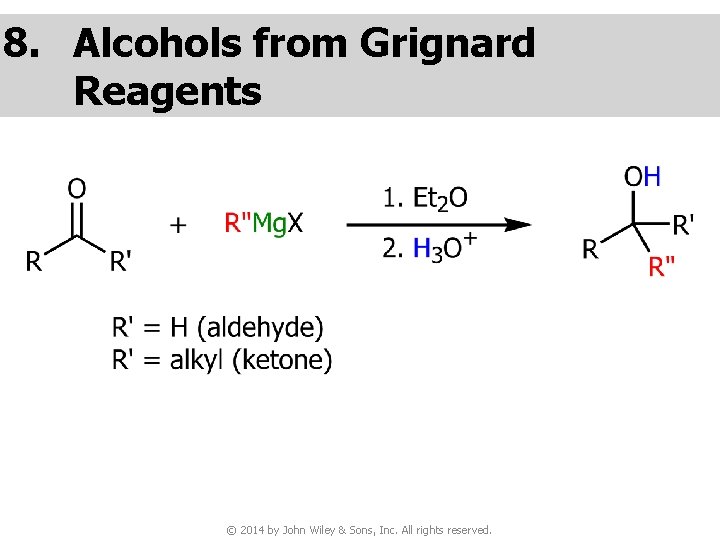
8. Alcohols from Grignard Reagents © 2014 by John Wiley & Sons, Inc. All rights reserved.
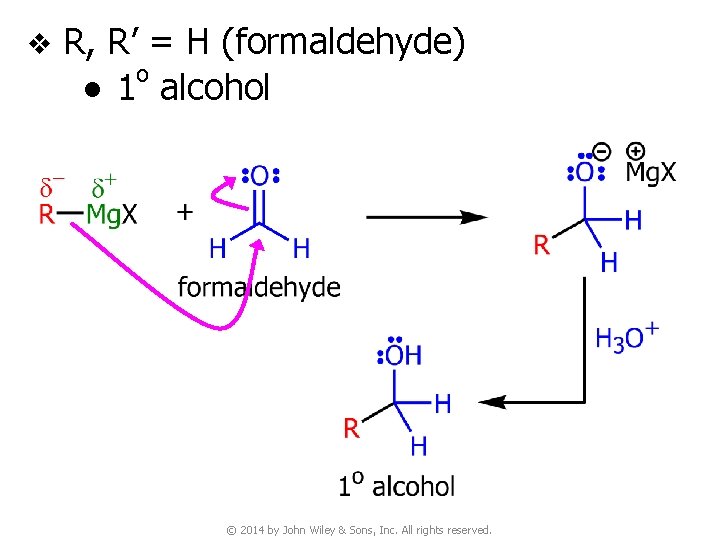
v R, R’ = H (formaldehyde) o ● 1 alcohol © 2014 by John Wiley & Sons, Inc. All rights reserved.
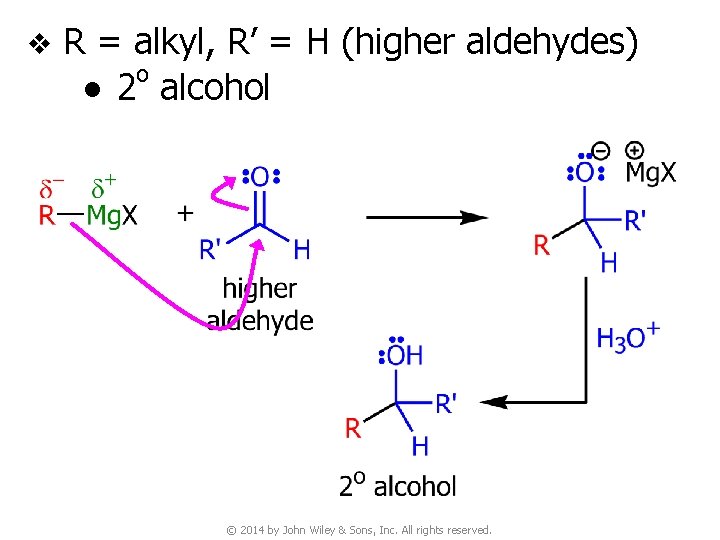
v R = alkyl, R’ = H (higher aldehydes) o ● 2 alcohol © 2014 by John Wiley & Sons, Inc. All rights reserved.
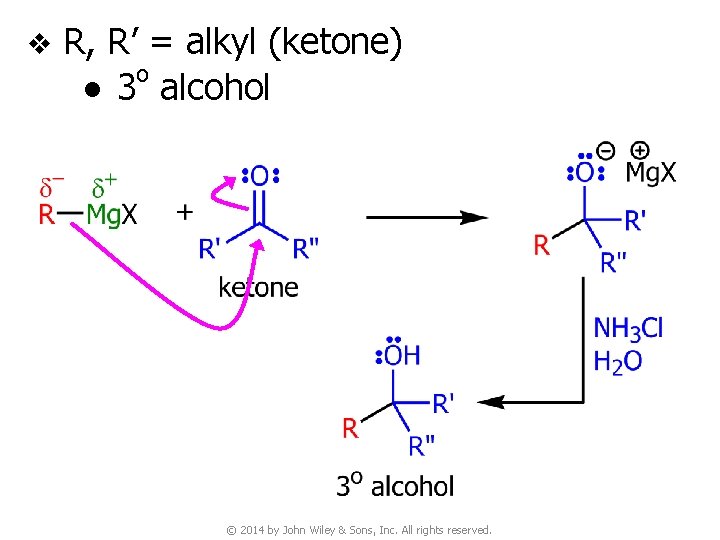
v R, R’ = alkyl (ketone) o ● 3 alcohol © 2014 by John Wiley & Sons, Inc. All rights reserved.
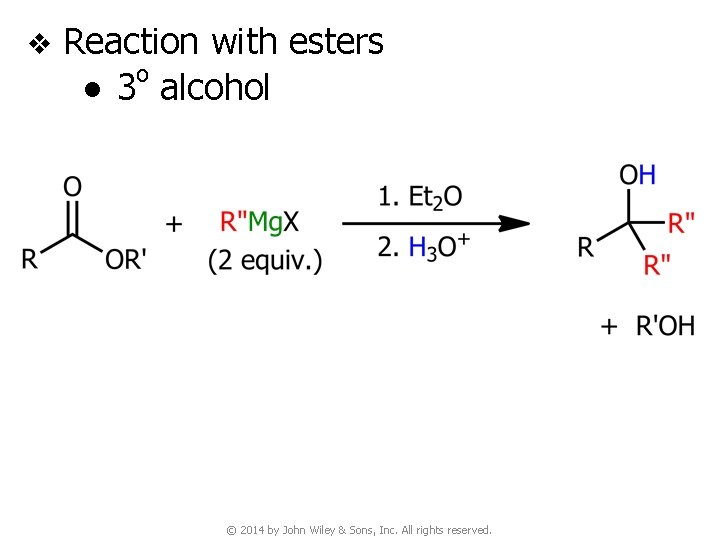
v Reaction with esters o ● 3 alcohol © 2014 by John Wiley & Sons, Inc. All rights reserved.
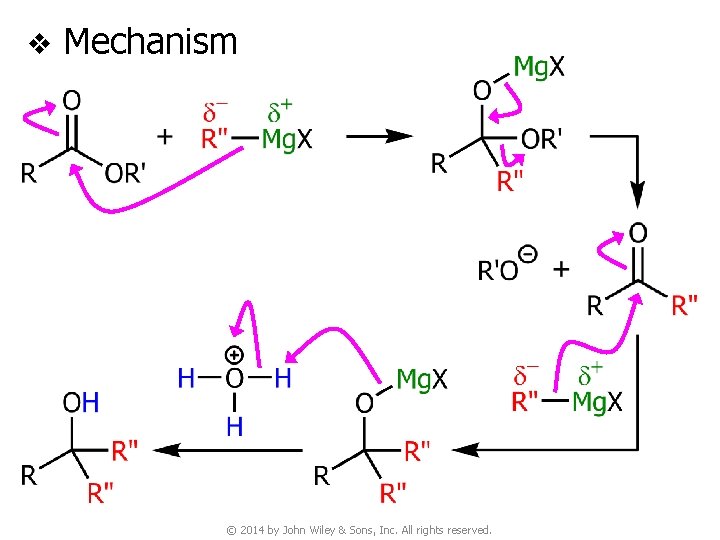
v Mechanism © 2014 by John Wiley & Sons, Inc. All rights reserved.
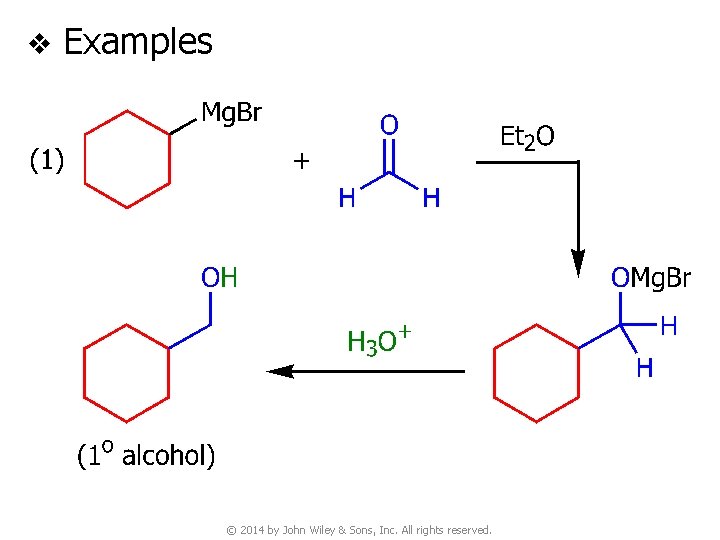
v Examples © 2014 by John Wiley & Sons, Inc. All rights reserved.

v Examples © 2014 by John Wiley & Sons, Inc. All rights reserved.
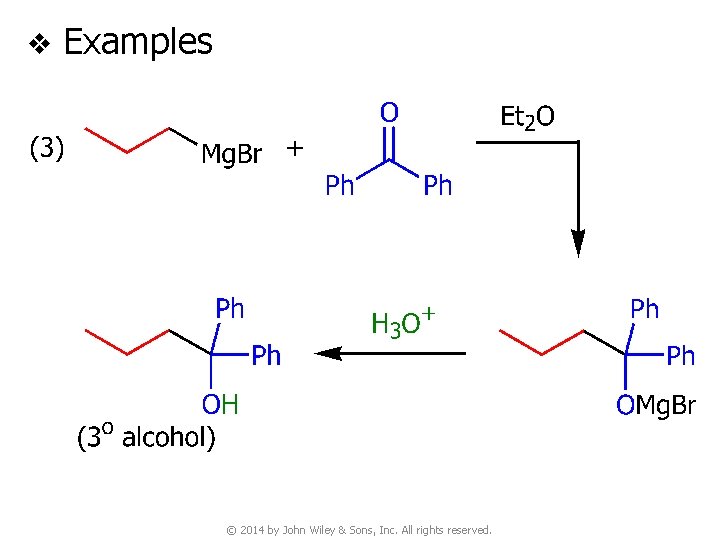
v Examples © 2014 by John Wiley & Sons, Inc. All rights reserved.
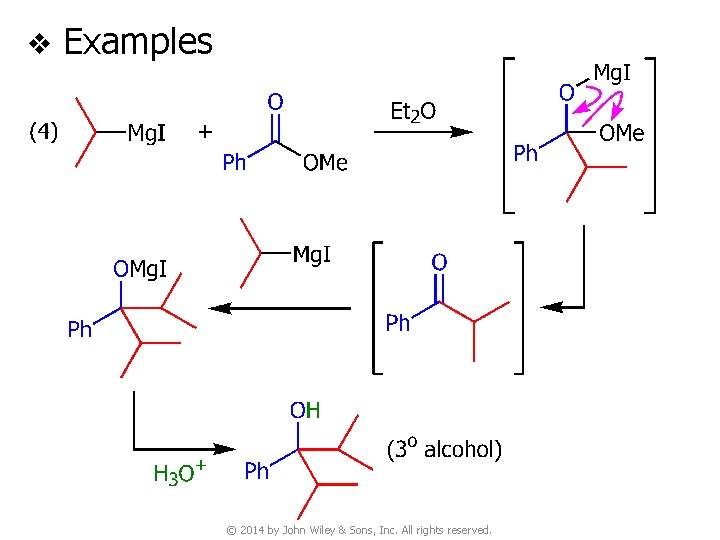
v Examples © 2014 by John Wiley & Sons, Inc. All rights reserved.
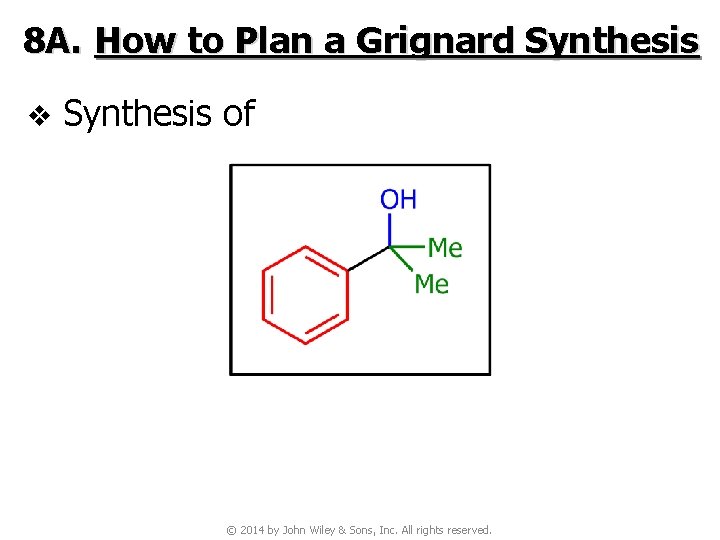
8 A. How to Plan a Grignard Synthesis v Synthesis of © 2014 by John Wiley & Sons, Inc. All rights reserved.

v Method 1 ● Retrosynthetic analysis ● Synthesis © 2014 by John Wiley & Sons, Inc. All rights reserved.
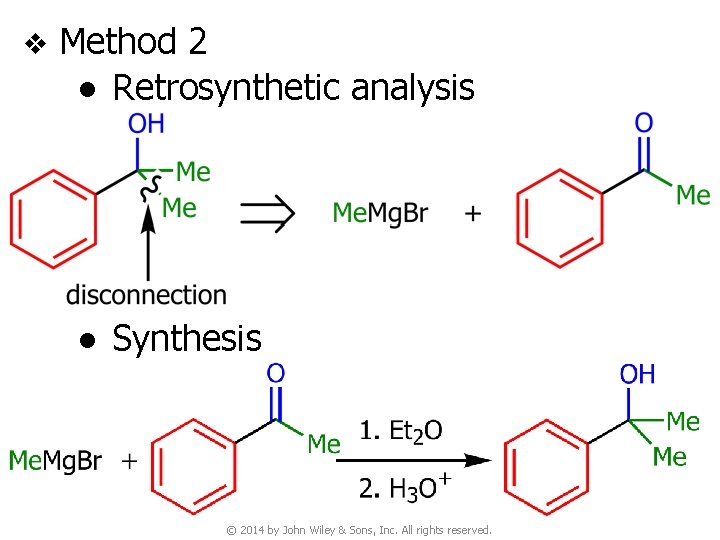
v Method 2 ● Retrosynthetic analysis ● Synthesis © 2014 by John Wiley & Sons, Inc. All rights reserved.

v Method 3 ● Retrosynthetic analysis ● Synthesis © 2014 by John Wiley & Sons, Inc. All rights reserved.

8 B. Restrictions on the Use of Grignard Reagents Grignard reagents are useful nucleophiles but they are also very strong bases v It is not possible to prepare a Grignard reagent from a compound that contains any hydrogen more acidic than the hydrogen atoms of an alkane or alkene v © 2014 by John Wiley & Sons, Inc. All rights reserved.

v A Grignard reagent cannot be prepared from a compound containing an –OH group, an –NH– group, an –SH group, a –CO 2 H group, or an –SO 3 H group v Since Grignard reagents are powerful nucleophiles, we cannot prepare a Grignard reagent from any organic halide that contains a carbonyl, epoxy, nitro, or cyano (–CN) group © 2014 by John Wiley & Sons, Inc. All rights reserved.
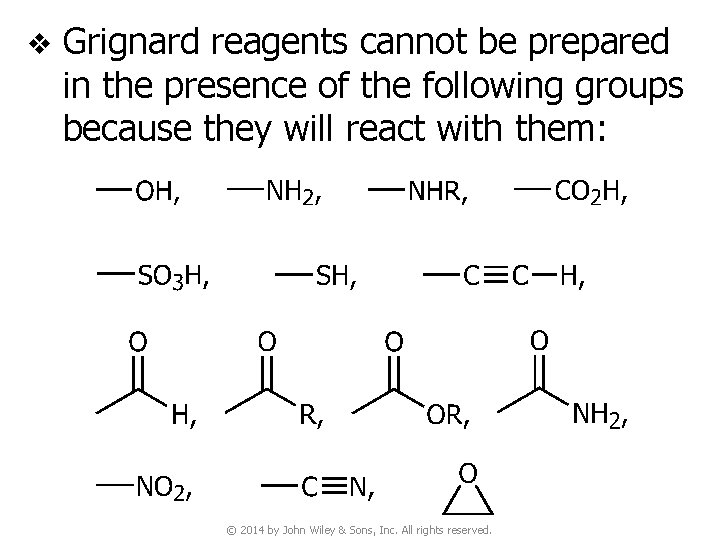
v Grignard reagents cannot be prepared in the presence of the following groups because they will react with them: © 2014 by John Wiley & Sons, Inc. All rights reserved.
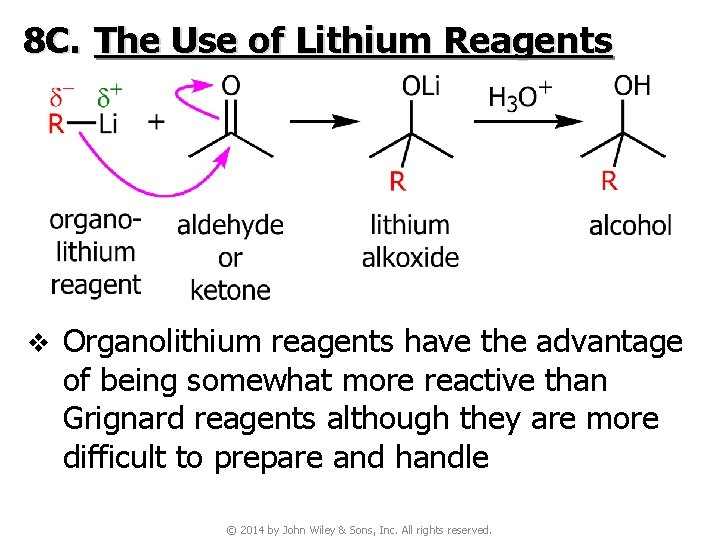
8 C. The Use of Lithium Reagents v Organolithium reagents have the advantage of being somewhat more reactive than Grignard reagents although they are more difficult to prepare and handle © 2014 by John Wiley & Sons, Inc. All rights reserved.
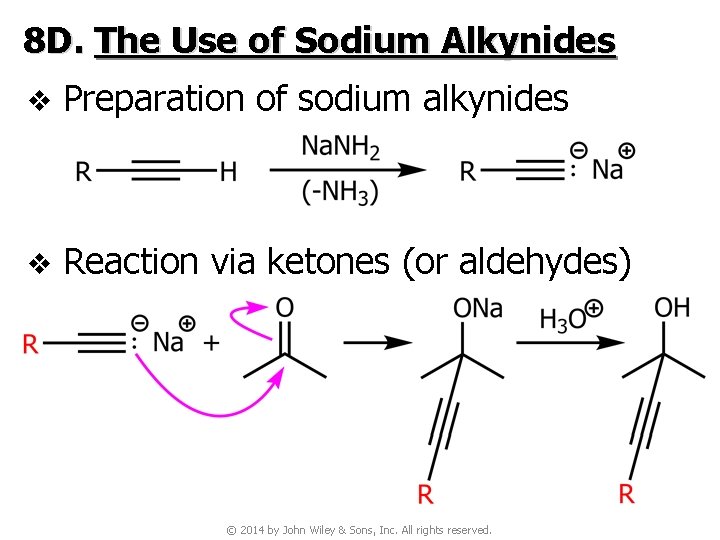
8 D. The Use of Sodium Alkynides v Preparation of sodium alkynides v Reaction via ketones (or aldehydes) © 2014 by John Wiley & Sons, Inc. All rights reserved.

9. Protecting Groups © 2014 by John Wiley & Sons, Inc. All rights reserved.
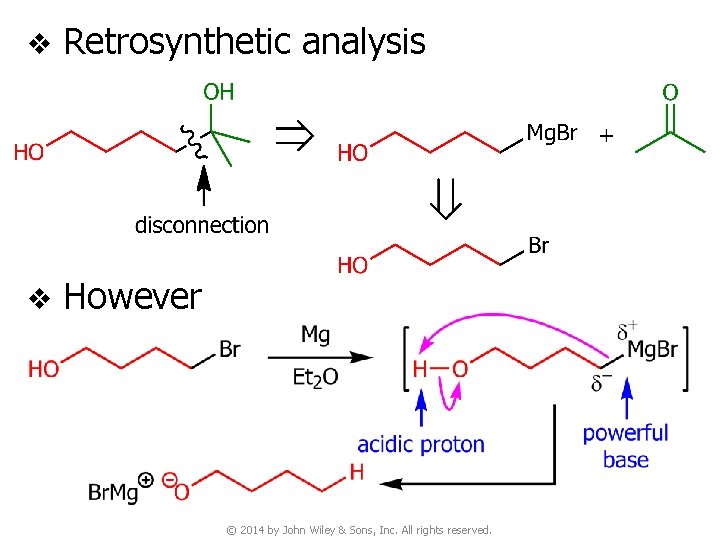
v Retrosynthetic analysis v However © 2014 by John Wiley & Sons, Inc. All rights reserved.
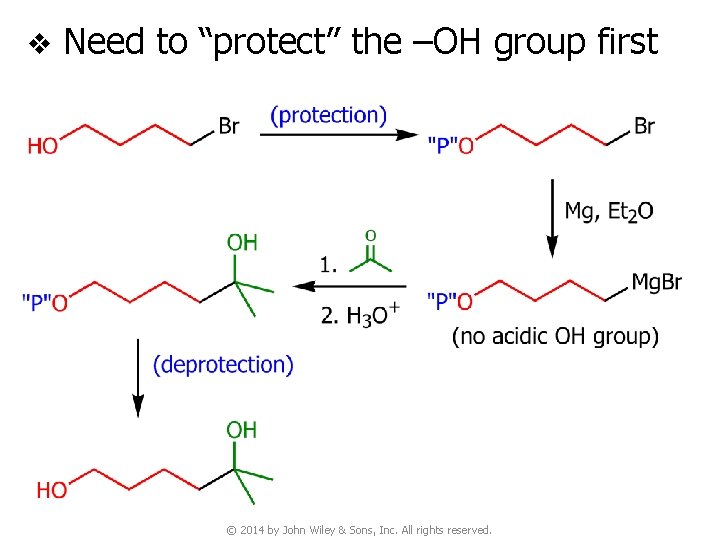
v Need to “protect” the –OH group first © 2014 by John Wiley & Sons, Inc. All rights reserved.
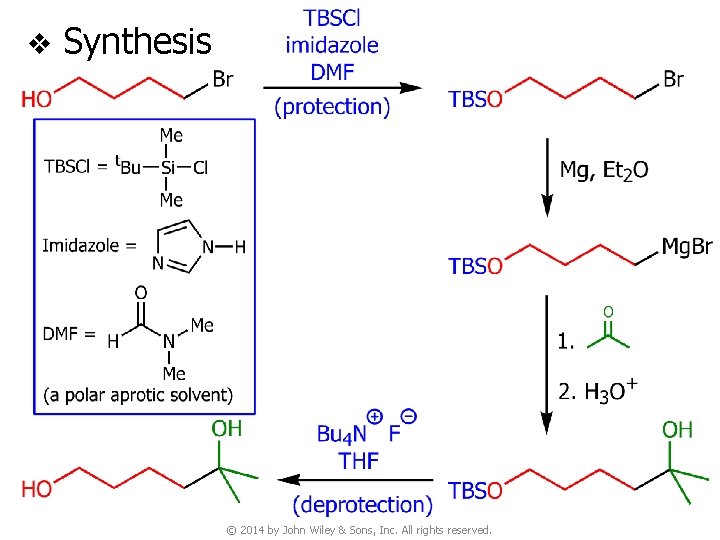
v Synthesis © 2014 by John Wiley & Sons, Inc. All rights reserved.
- Slides: 78