Chapter 12 5 Aromatic Hydrocarbons 1 Aromatic Compounds

Chapter 12. 5 Aromatic Hydrocarbons 1

Aromatic Compounds and Benzene • Aromatic compounds contain benzene. • Benzene, C 6 H 6 , is represented as a six carbon ring with 3 double bonds. • Two possible isomers can be drawn to show benzene in this form. 2

Aromatic Compounds and Benzene • The name aromatic came from many of the first examples discovered were pleasant smelling • Many aromatics that have recently been discovered are not pleasant smelling, though • The most common aromatic ring is benzene which includes 6 Carbons and 6 Hydrogens--it is highly unsaturated 3

Aromatic Compounds and Benzene • Strangely, aromatics do not undergo the same kinds of reactions as alkenes and alkynes • The benzene ring is planar and hexagonal • The three double bonds in benzene are shared equally between the six carbons. We say that the double bonds are delocalized • Benzene is unusually stable and resists most reactants 4

Benzene Structure The structures for benzene can also be written as a single structure where the alternating double bonds are written as a circle within the ring. Benzene structure 5
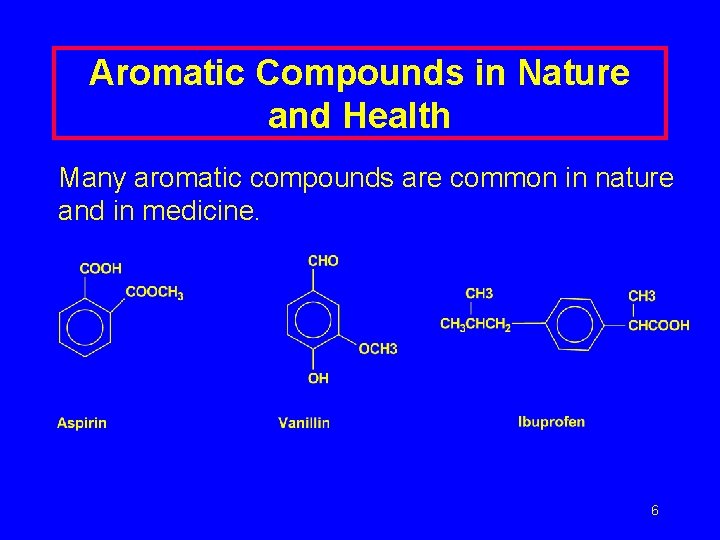
Aromatic Compounds in Nature and Health Many aromatic compounds are common in nature and in medicine. 6

Naming Aromatic Compounds Aromatic compounds are named with benzene as the parent chain. One side group is named in front of the name benzene. methylbenzene (toluene) chlorobenzene 7
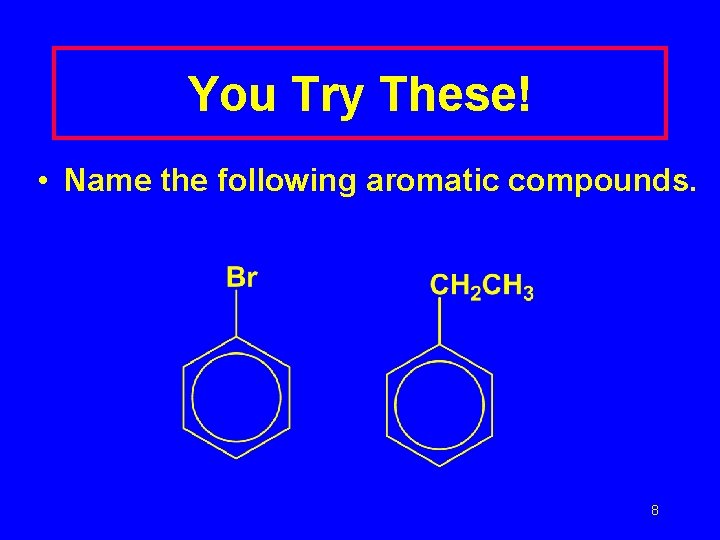
You Try These! • Name the following aromatic compounds. 8
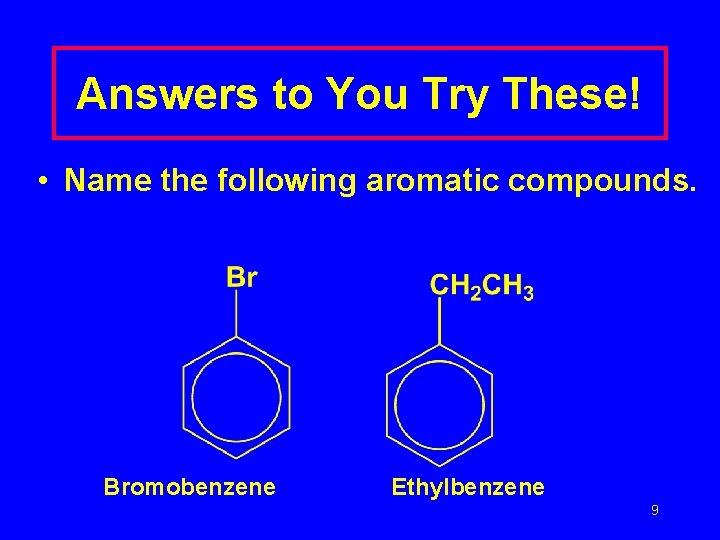
Answers to You Try These! • Name the following aromatic compounds. Bromobenzene Ethylbenzene 9
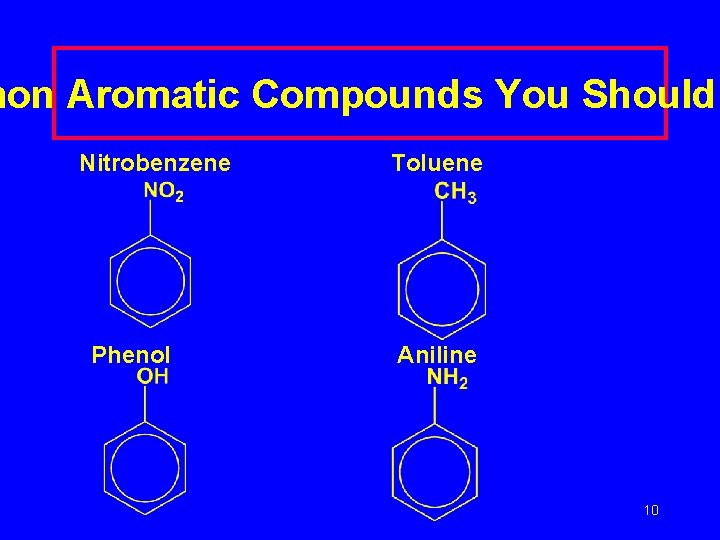
mon Aromatic Compounds You Should Nitrobenzene Phenol Toluene Aniline 10

Naming Aromatic Compounds When two groups are attached to benzene, the ring is numbered to give the lower numbers to the side groups. The prefixes ortho (1, 2), meta (1, 3) and para (1, 4) are also used. ortho-dimethylbenzene meta-dichlorobenzene para-chloromethylbenzene 11
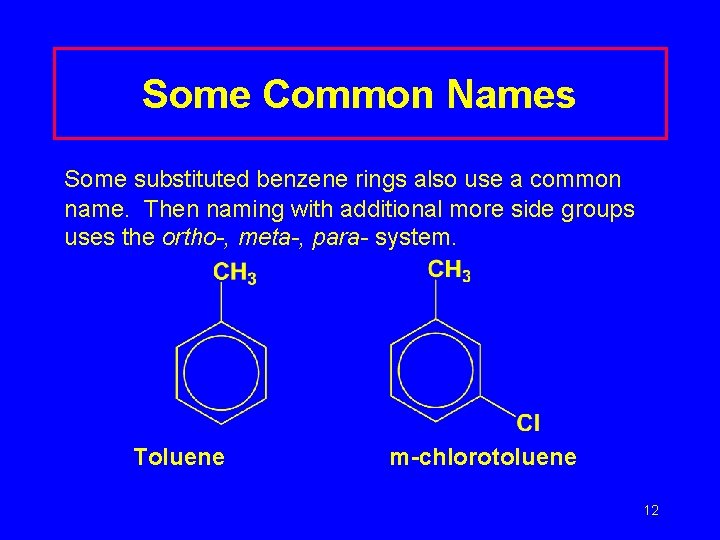
Some Common Names Some substituted benzene rings also use a common name. Then naming with additional more side groups uses the ortho-, meta-, para- system. Toluene m-chlorotoluene 12
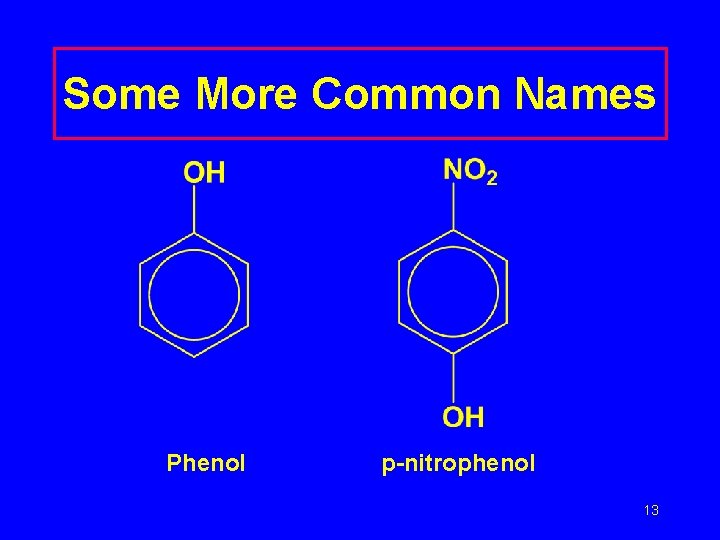
Some More Common Names Phenol p-nitrophenol 13

Other Common Aromatic Compounds You Should Know CH 3 Xylene CH 3 OH Cresol CH 3 14

You Try These! Select the names for each structure: 1. Chlorocyclohexane 2. Chlorobenzene 3. 1 -chlorobenzene 1. Meta-methyltoluene 2. Meta-dimethylbenzene 3. 1, 3 -dimethylbenzene 15

Answers to You Try These! Select the names for each structure: 2. Chlorobenzene 1. Meta-methyltoluene 2. Meta-dimethylbenzene 3. 1, 3 -dimethylbenzene 16

You Try these! Write the structural formulas for each of the following: A. 1 -bromo-3 -methylbenzene B. Ortho-chlorotoluene 17
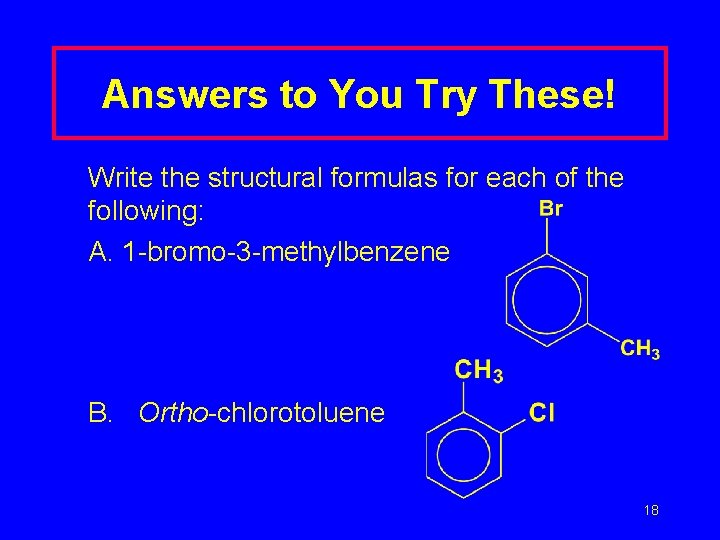
Answers to You Try These! Write the structural formulas for each of the following: A. 1 -bromo-3 -methylbenzene B. Ortho-chlorotoluene 18

Homework Assignment Pg. 345 -346 12. 11 – 12. 12 Due Tomorrow! 19
- Slides: 19