Chapter 11 Unsaturated Hydrocarbons 1 11 1 Alkenes

Chapter 11 Unsaturated Hydrocarbons 1 11. 1 Alkenes and Alkynes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Saturated Hydrocarbons 2 Saturated hydrocarbons • have the maximum number of hydrogen atoms attached to each carbon atom • are alkanes and cycloalkanes with single C–C bonds CH 3–CH 2–CH 3 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Unsaturated Hydrocarbons 3 Unsaturated hydrocarbons • have fewer hydrogen atoms attached to the carbon chain than alkanes • are alkenes with double bonds • are alkynes with triple bonds Ball-and-stick models of ethene and ethyne show functional groups of double or triple bonds and bond angles. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Bond Angles in Alkenes and Alkynes 4 According to VSEPR theory: • the three groups bonded to carbon atoms in a double bond are at 120 angles • alkenes are flat because the atoms in a double bond all lie in the same plane • the two groups bonded to each carbon in a triple bond are at 180 angles Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Ethene (Ethylene) 5 Ethene, or ethylene, • is the simplest alkene, with the formula C 2 H 4 • has two carbon atoms connected by a double bond • has two H atoms bonded to each C atom • is flat, with all the C and H atoms in the same plane • is a plant hormone used to accelerate the ripening of fruits Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Fragrant Alkenes 6 The odors associated with the smell of lemons, oranges, roses, and lavender are due to volatile compounds that are synthesized by the plants. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Ethyne (Acetylene) 7 The simplest alkyne is ethyne, which has the common name of acetylene. Acetylene is a fuel used in welding. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Naming Alkenes and Alkynes 8 When the carbon chain of an alkene or alkyne has four or more C atoms, number the chain to give the lowest number to the first carbon in the double or triple bond. CH 2=CH CH 2 CH 3 1 2 3 4 1 -butene CH 3 CH=CH CH 3 1 2 3 4 2 -butene CH 3 CH 2 C=C CH 3 5 4 3 2 1 2 -pentyne Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
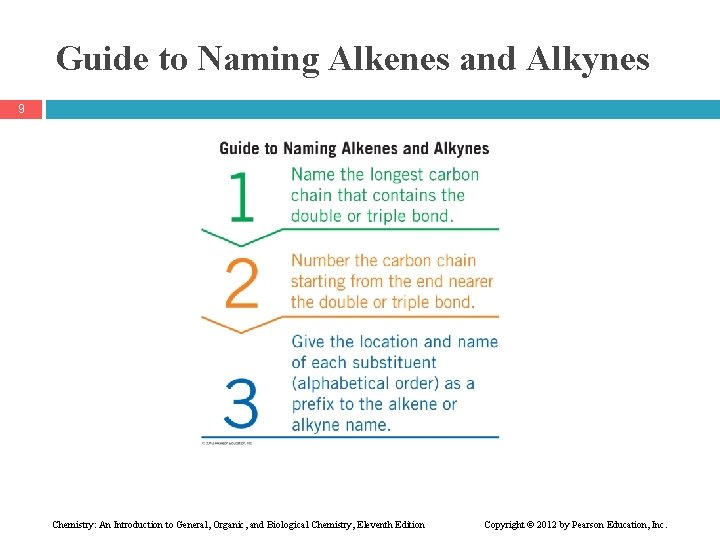
Guide to Naming Alkenes and Alkynes 9 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Naming Alkenes 10 Using the IUPAC system, name the following compounds: CH 3 A. CH 3 CH H 2 C=CH 2 B. H 2 C=CH CH 3 Step 1 Name the longest carbon chain that contains the double bond. Use the ending ene for alkenes. CH 3 A. CH 3 CH H 2 C=CH 2 B. H 2 C=CH CH 3 butene pentene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Naming Alkenes 11 Step 2 Number the longest carbon chain starting from the end nearer the double bond. CH 3 A. CH 3 CH H 2 C=CH 2 B. H 2 C=CH CH 3 1 -butene 1 -pentene Step 3 Give the location and name of each substituent (in alphabetical order) as a prefix to the alkene name. CH 3 A. CH 3 CH H 2 C=CH 2 3 -methyl-1 -butene B. H 2 C=CH CH 3 3, 4 -dimethyl-1 -pentene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Naming Alkynes 12 Using the IUPAC system, name the following compound: CH 3 HC C CH CH CH 3 Step 1 Name the longest carbon chain that contains the triple bond. Use the ending yne for alkynes. CH 3 HC C CH CH CH 3 pentyne Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
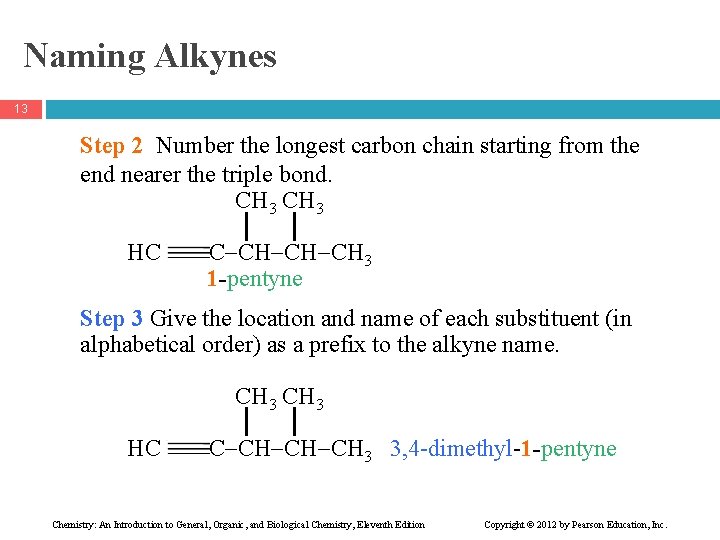
Naming Alkynes 13 Step 2 Number the longest carbon chain starting from the end nearer the triple bond. CH 3 HC C CH CH CH 3 1 -pentyne Step 3 Give the location and name of each substituent (in alphabetical order) as a prefix to the alkyne name. CH 3 HC C CH CH CH 3 3, 4 -dimethyl-1 -pentyne Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 14 Write the IUPAC name for each of the following: A. CH 2=CH CH 2 CH 3 B. CH 3 CH=CH CH 3 CH=C CH 3 D. CH 3 C C CH 3 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 15 Write the IUPAC name for each of the following: A. CH 2=CH CH 2 CH 3 1 -butene B. CH 3 CH=CH CH 3 2 -butene C. CH 3 CH=C CH 3 2 -methyl-2 -butene D. CH 3 C 2 -butyne C CH 3 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 16 Write the IUPAC name for each of the following: A. CH 3 CH 2 C C CH 3 B. CH 3 CH 2 C=CH CH 3 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 17 Write the IUPAC name for each of the following: A. CH 3 CH 2 C C CH 3 2 -hexyne CH 3 B. CH 3 CH 2 C=CH CH 3 3 -methyl-2 -pentene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Chapter 11 Unsaturated Hydrocarbons 11. 2 Cis–Trans Isomers Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
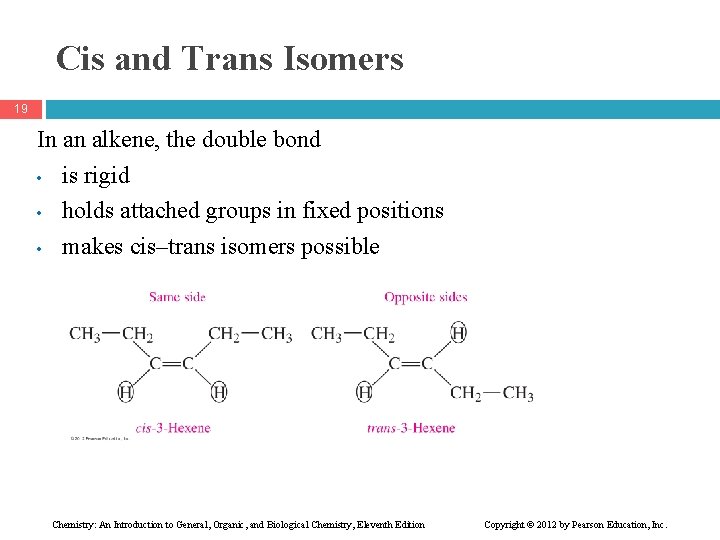
Cis and Trans Isomers 19 In an alkene, the double bond • is rigid • holds attached groups in fixed positions • makes cis–trans isomers possible Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Cis–Trans Isomers 20 In cis–trans isomers, • there is no rotation around the double bond in alkenes • groups attached to the double bond are fixed relative to each other You can make a “double bond” with your fingers with both thumbs on the same side or opposite from each other. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
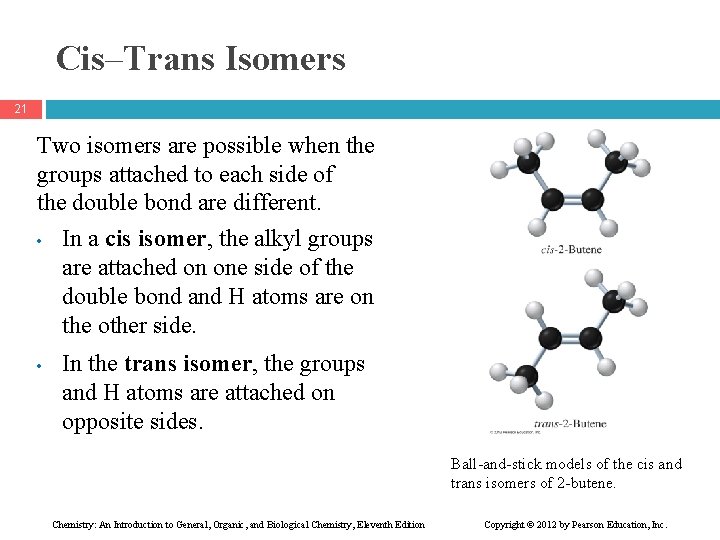
Cis–Trans Isomers 21 Two isomers are possible when the groups attached to each side of the double bond are different. • In a cis isomer, the alkyl groups are attached on one side of the double bond and H atoms are on the other side. • In the trans isomer, the groups and H atoms are attached on opposite sides. Ball-and-stick models of the cis and trans isomers of 2 -butene. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
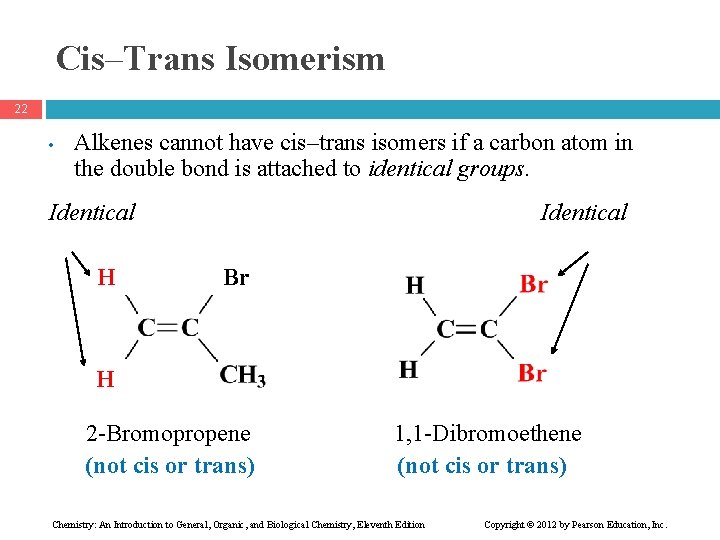
Cis–Trans Isomerism 22 • Alkenes cannot have cis–trans isomers if a carbon atom in the double bond is attached to identical groups. Identical H Identical Br H H 2 -Bromopropene (not cis or trans) 1, 1 -Dibromoethene (not cis or trans) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Cis–Trans Isomers in Nature 23 • • Insects emit tiny quantities of pheromones, which are chemicals that send messages. The silkworm moth attracts other moths by emitting bombykol, which has one cis and one trans double bond. Pheromones allow insects to attract mates from a great distance. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
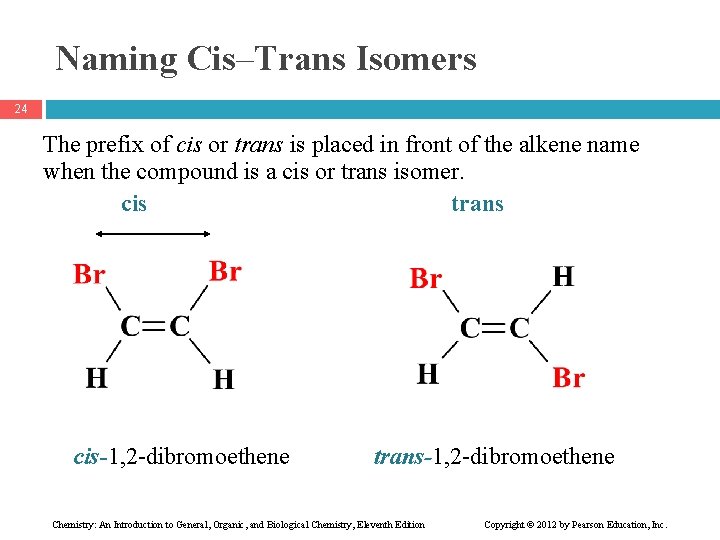
Naming Cis–Trans Isomers 24 The prefix of cis or trans is placed in front of the alkene name when the compound is a cis or trans isomer. cis trans cis-1, 2 -dibromoethene trans-1, 2 -dibromoethene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
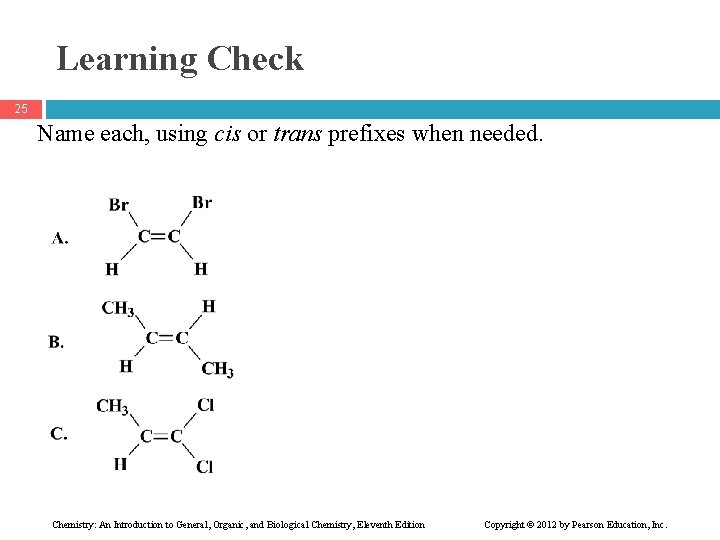
Learning Check 25 Name each, using cis or trans prefixes when needed. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
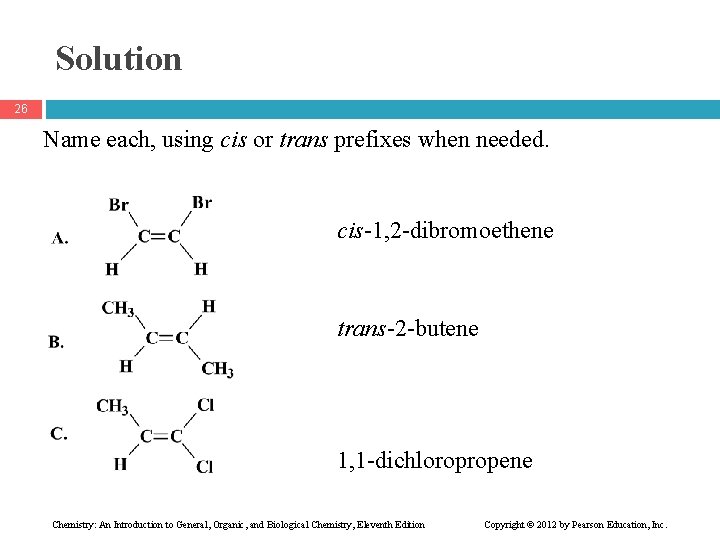
Solution 26 Name each, using cis or trans prefixes when needed. cis-1, 2 -dibromoethene trans-2 -butene 1, 1 -dichloropropene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Chapter 11 Unsaturated Hydrocarbons 27 11. 3 Addition Reactions Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Addition Reactions 28 In alkene and alkynes, • the double or triple bond is easily broken, providing electrons to form new bonds • double and triple bonds are very reactive • in addition reactions, reactants are added to the carbon atoms in the double or triple bond Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Hydrogenation 29 In hydrogenation, • hydrogen atoms add to the carbon atoms of a double bond or triple bond • a catalyst such as Pt or Ni is used to speed up the reaction Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Hydrogenation of Oils 30 Adding H 2 to double bonds in vegetable oils produces • compounds with higher melting points • solids at room temperature, such as margarine, soft margarine, and shortening Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 31 Write an equation for the hydrogenation of 1 -butene using a platinum catalyst. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 32 Write an equation for the hydrogenation of 1 -butene using a platinum catalyst. CH 2=CH CH 2 CH 3 + H 2 Pt CH 3 CH 2 CH 3 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Trans Fats 33 The process of hydrogenation is used commercially to convert the double bonds in the unsaturated fats in vegetable oils to saturated fats such as margarine, which are more solid. Adjusting the amount of added hydrogen produces partially hydrogenated fats such as soft margarine, solid margarine in sticks, and shortenings, which are used in cooking. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
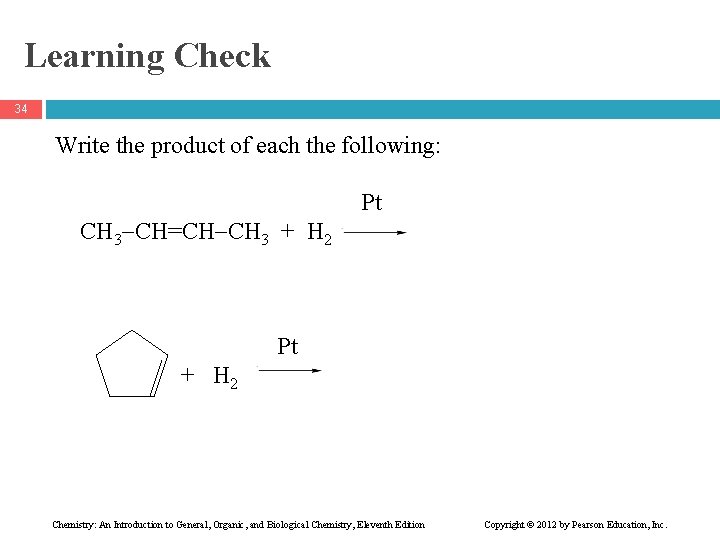
Learning Check 34 Write the product of each the following: CH 3 CH=CH CH 3 + H 2 Pt Pt + H 2 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
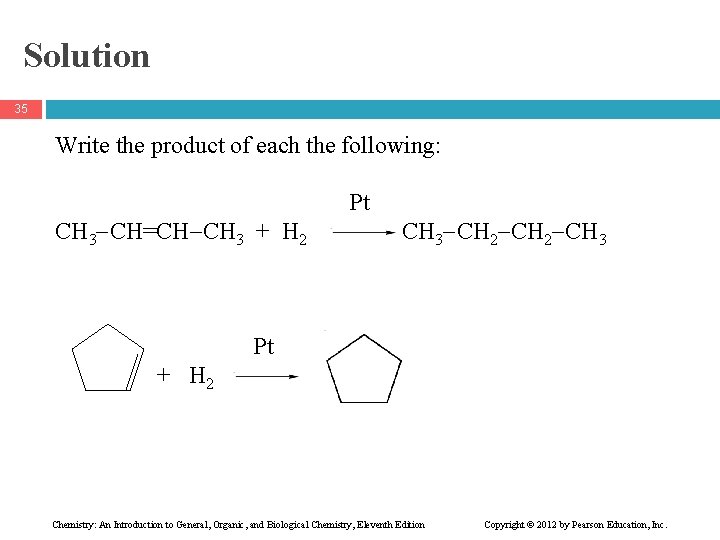
Solution 35 Write the product of each the following: CH 3 CH=CH CH 3 + H 2 Pt CH 3 CH 2 CH 3 Pt + H 2 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Halogenation Reactions 36 In halogenation reactions, • halogen atoms such as chlorine or bromine add across a double bond • the reaction occurs without a catalyst • a dihaloalkane product results In the general equation for halogenation, X 2 is used for either Cl 2 or Br 2. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Halogenation Reactions 37 The addition reaction of Br 2 across a double bond is used to test for the presence of double bonds. A positive result turns a clear solution red. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Hydration 38 In the addition reaction called hydration, • an acid H+ catalyst is required • water (H−OH) adds to a double bond • an H atom bonds to one C in the double bond • an OH bonds to the other C • an alkene is converted to an alcohol H OH H+ CH 3 CH=CH CH 3 + H OH CH 3 CH CH CH 3 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Hydration 39 When hydration occurs with a double bond that has an unequal number of H atoms, • the H atom bonds to the C in the double bond with the more H • the OH bonds to the C in the double bond with the fewer H atoms Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
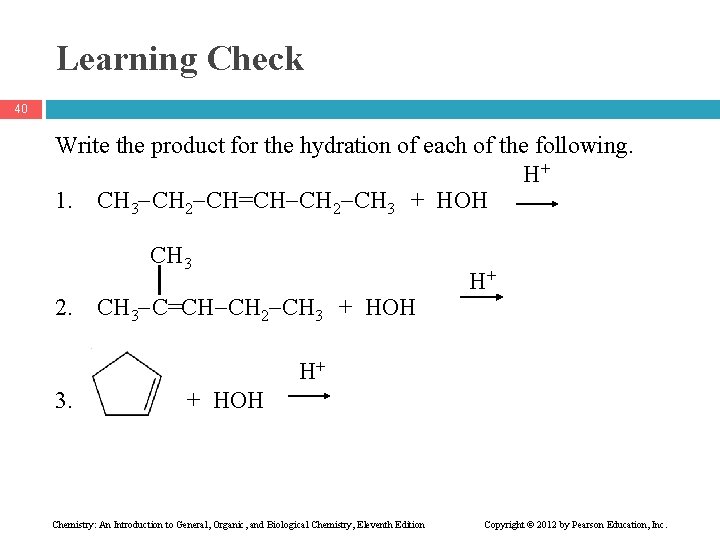
Learning Check 40 Write the product for the hydration of each of the following. H+ 1. CH 3 CH 2 CH=CH CH 2 CH 3 + HOH CH 3 2. CH 3 C=CH CH 2 CH 3 + HOH H+ H+ 3. + HOH Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 41 Write the product for the hydration of each of the following. 1. CH 3 CH 2 CH=CH CH 2 CH 3 + HOH H+ H OH CH 3 CH 2 CH CH CH 2 CH 3 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
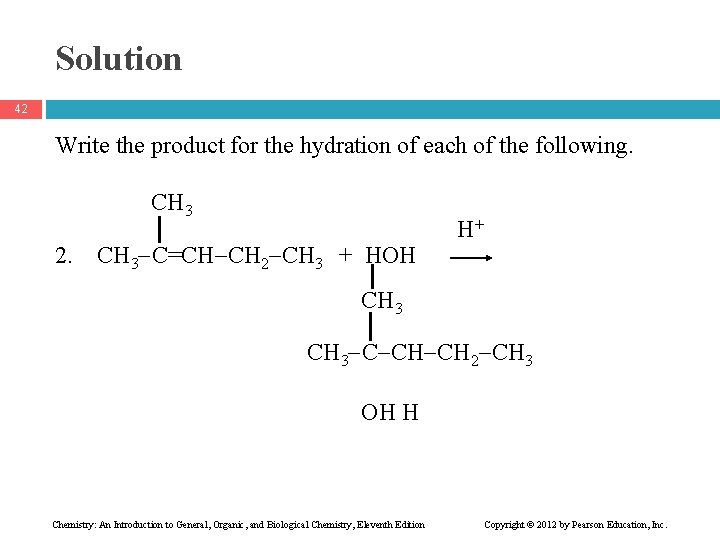
Solution 42 Write the product for the hydration of each of the following. CH 3 2. CH 3 C=CH CH 2 CH 3 + HOH H+ CH 3 C CH CH 2 CH 3 OH H Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
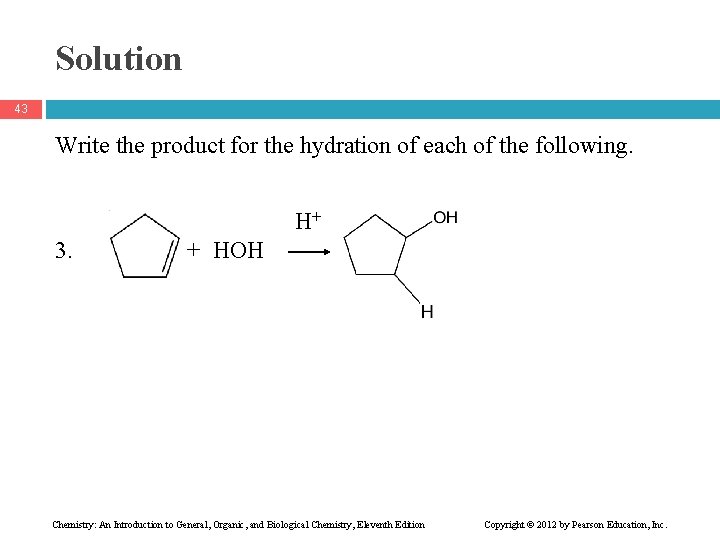
Solution 43 Write the product for the hydration of each of the following. H+ 3. + HOH Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Chapter 11 Unsaturated Hydrocarbons 44 11. 4 Polymers of Alkenes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Polymers 45 Polymers are • • • large, long-chain molecules found in nature, including cellulose in plants, starches in food, proteins, and DNA in the body also synthetic, such as polyethylene and polystyrene, Teflon, and nylon composed of small repeating units called monomers made from reaction of small alkenes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
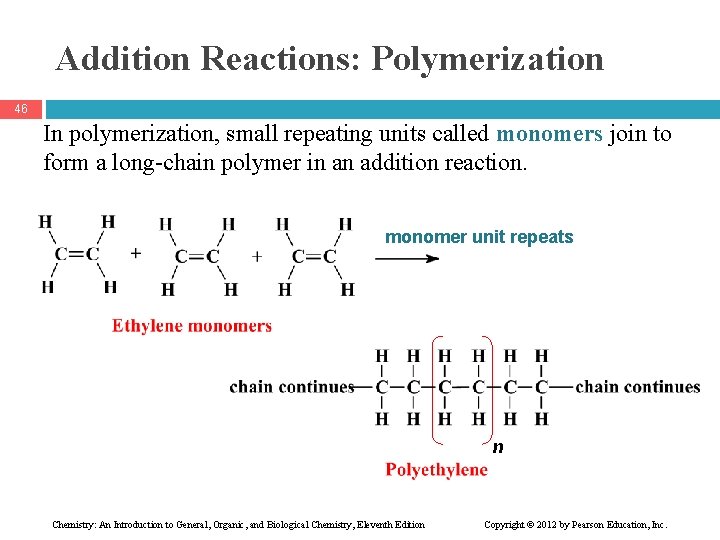
Addition Reactions: Polymerization 46 In polymerization, small repeating units called monomers join to form a long-chain polymer in an addition reaction. monomer unit repeats n Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Common Synthetic Polymers 47 Synthetic polymers provide a wide variety of items that we use every day. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Common Synthetic Polymers: Polyethylene 48 Polyethylene is made using an addition reaction with the monomer unit ethylene. Common uses for polyethylene include plastic bottles, film, and insulation materials. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Common Synthetic Polymers: Polytetrafluoroethylene 49 Polytetrafluoroethylene, primarily used in nonstick coatings, is made using an addition reaction with the monomer unit of tetrafluoroethene polytetrafluoroethylene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Common Synthetic Polymers: Polypropylene 50 Polypropylene is used for ski and hiking clothing, carpets, and artificial joints. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Common Synthetic Polymers: Polystyrene 51 Polystyrene is used for plastic coffee cups, cartons, and insulation. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Common Synthetic Polymers: Polyvinylchloride 52 Polyvinylchloride, also known as PVC, is used for plastic pipes and tubing, garden hoses, and garbage bags. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Common Synthetic Polymers: Polydichloroethylene 53 Polydichloroethylene is used in plastic film and wraps. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 54 Name the starting monomer and give its structure for each of the following polymers: A. polyethylene B. polyvinylchloride C. polystyrene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 55 Name the starting monomer and give its structure for each of the following polymers: A. polyethylene ethene B. polyvinylchloride chloroethene C. polystyrene phenylethene, styrene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 56 Name the monomer used to make polypropylene, and write a portion of a polypropylene polymer using three monomers. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 57 Name the monomer used to make polypropylene, and write a portion of a polypropylene polymer using three monomers. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Recycling Plastics 58 Recycling is simplified by using codes on plastic items. 1 2 3 4 5 6 7 PETE Polyethylene terephtalate HDPE High-density polyethylene PV Polyvinyl chloride LDPE Low-density polyethylene PP Polypropylene PS Polystyrene OTHER Other plastic: fiberglass, polycarbonate, etc. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 59 What types of plastic are indicated by the following codes? A. 3 PV B. 5 PP C. 6 PS Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 60 What types of plastic are indicated by the following codes? A. B. C. 3 PV 5 PP 6 PS polyvinylchloride polypropylene polystyrene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Chapter 11 Unsaturated Hydrocarbons 61 11. 5 Aromatic Compounds Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Aromatic Compounds 62 Benzene is • an aromatic compound • a ring of six C atoms and six H atoms • a flat ring structure drawn with three double bonds • represented by two structures because the electrons are shared among all the C atoms Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
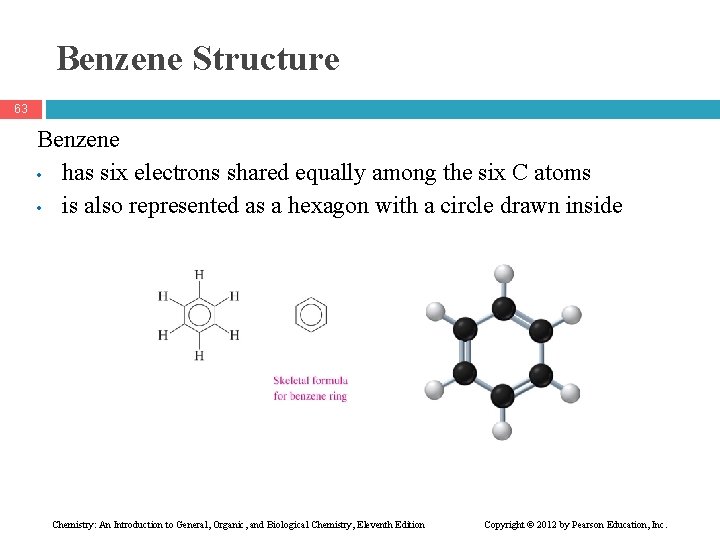
Benzene Structure 63 Benzene • has six electrons shared equally among the six C atoms • is also represented as a hexagon with a circle drawn inside Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
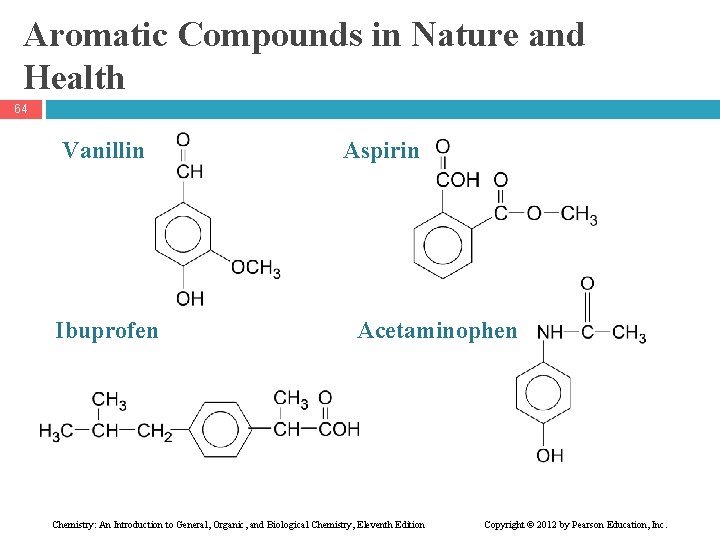
Aromatic Compounds in Nature and Health 64 Vanillin Ibuprofen Aspirin Acetaminophen Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
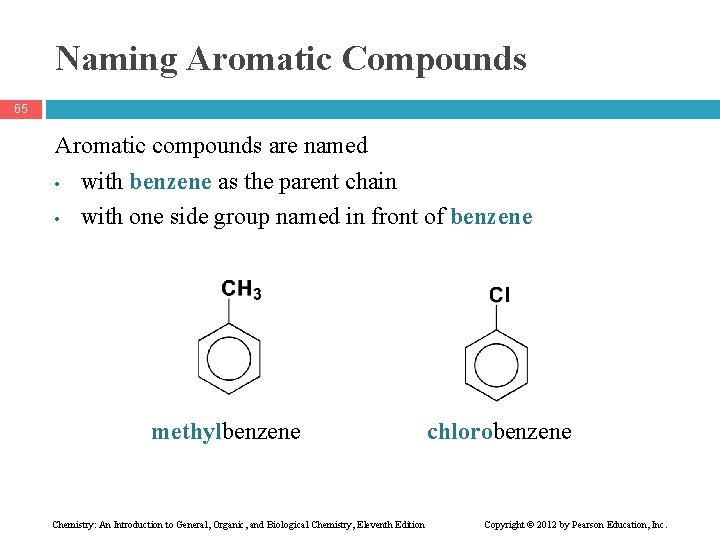
Naming Aromatic Compounds 65 Aromatic compounds are named • with benzene as the parent chain • with one side group named in front of benzene methylbenzene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition chlorobenzene Copyright © 2012 by Pearson Education, Inc.
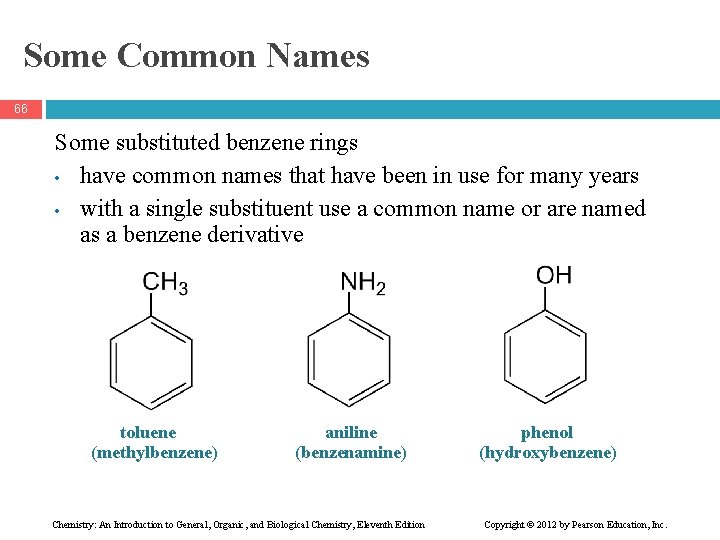
Some Common Names 66 Some substituted benzene rings • have common names that have been in use for many years • with a single substituent use a common name or are named as a benzene derivative toluene (methylbenzene) aniline (benzenamine) Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition phenol (hydroxybenzene) Copyright © 2012 by Pearson Education, Inc.
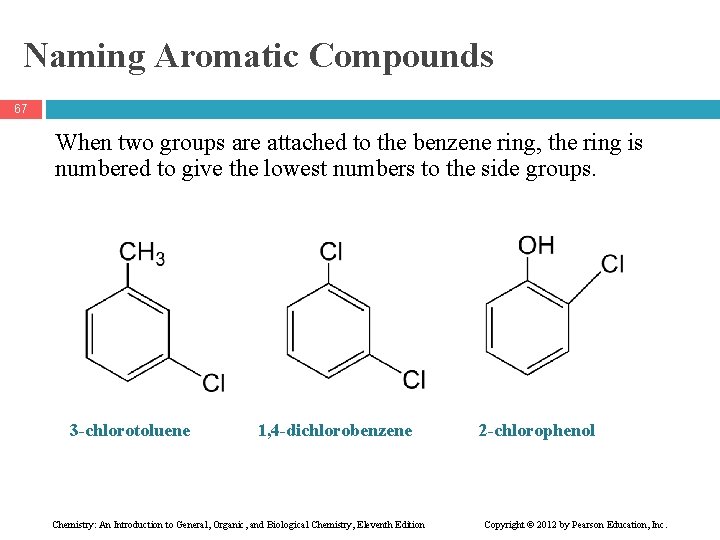
Naming Aromatic Compounds 67 When two groups are attached to the benzene ring, the ring is numbered to give the lowest numbers to the side groups. 3 -chlorotoluene 1, 4 -dichlorobenzene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition 2 -chlorophenol Copyright © 2012 by Pearson Education, Inc.
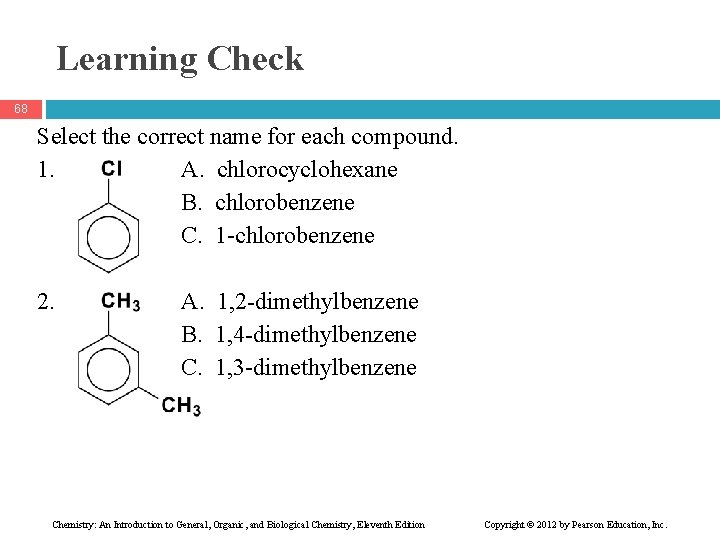
Learning Check 68 Select the correct name for each compound. 1. A. chlorocyclohexane B. chlorobenzene C. 1 -chlorobenzene 2. A. 1, 2 -dimethylbenzene B. 1, 4 -dimethylbenzene C. 1, 3 -dimethylbenzene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
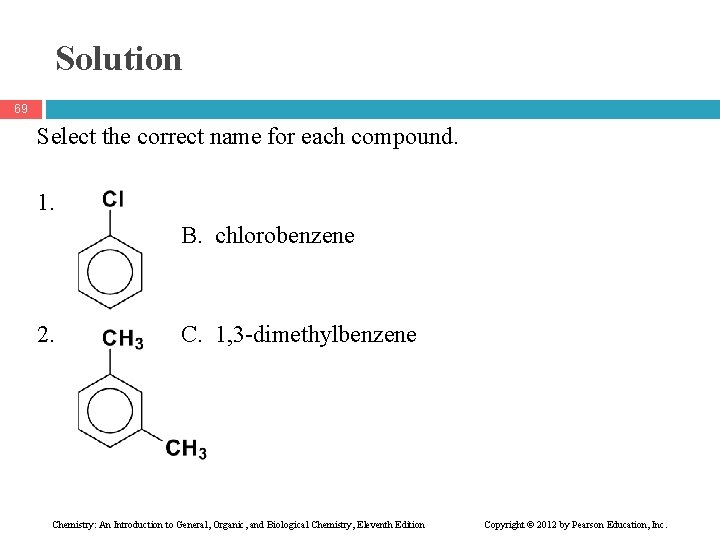
Solution 69 Select the correct name for each compound. 1. B. chlorobenzene 2. C. 1, 3 -dimethylbenzene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 70 Write the structural formulas for each of the following: 1. 1, 3 -dichlorobenzene 2. 2 -chlorotoluene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
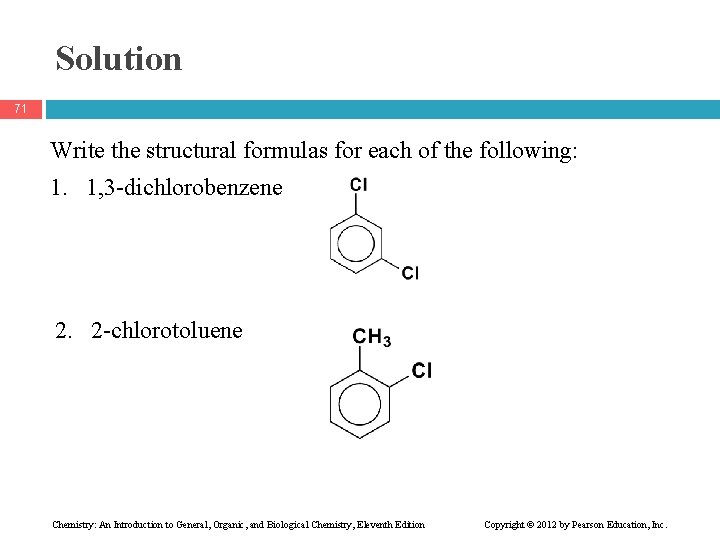
Solution 71 Write the structural formulas for each of the following: 1. 1, 3 -dichlorobenzene 2. 2 -chlorotoluene Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
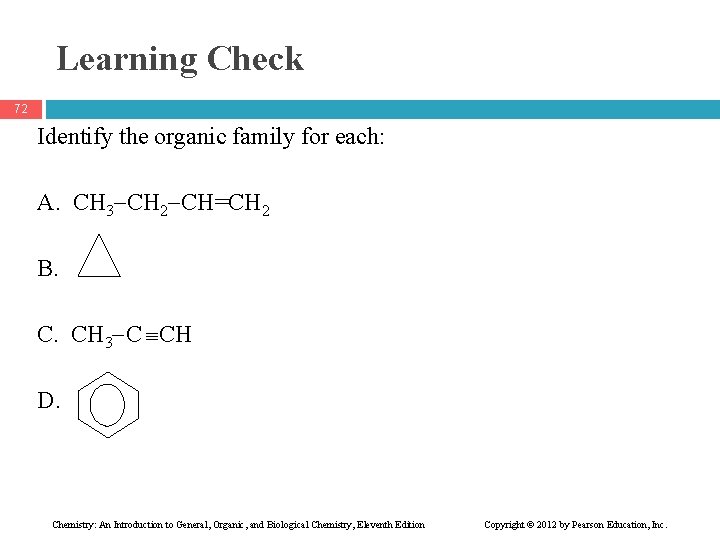
Learning Check 72 Identify the organic family for each: A. CH 3 CH 2 CH=CH 2 B. C. CH 3 C CH D. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
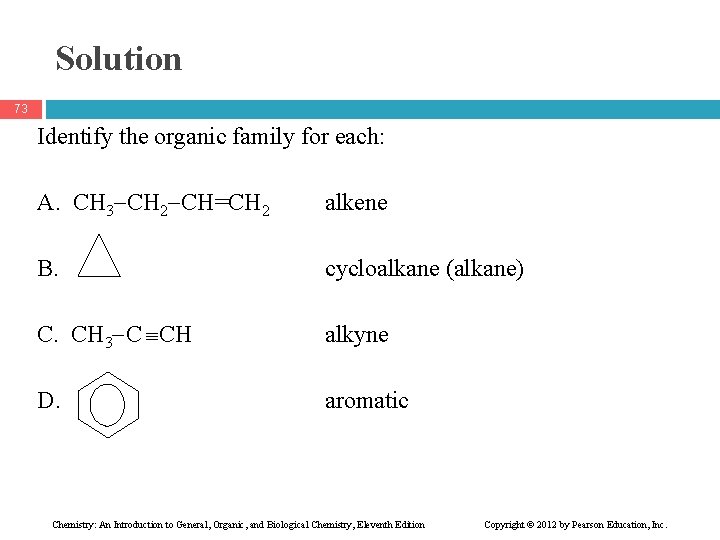
Solution 73 Identify the organic family for each: A. CH 3 CH 2 CH=CH 2 alkene B. cycloalkane (alkane) C. CH 3 C CH alkyne D. aromatic Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
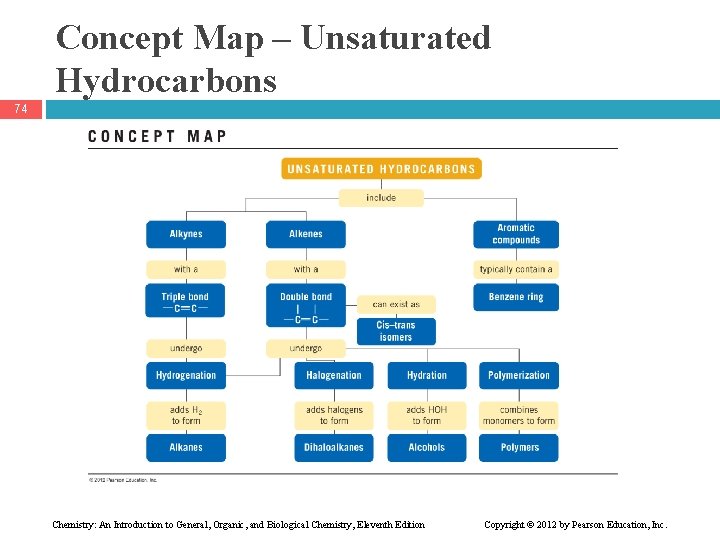
Concept Map – Unsaturated Hydrocarbons 74 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.
- Slides: 74