Chapter 11 Stoichiometry The Math of Chemistry Using













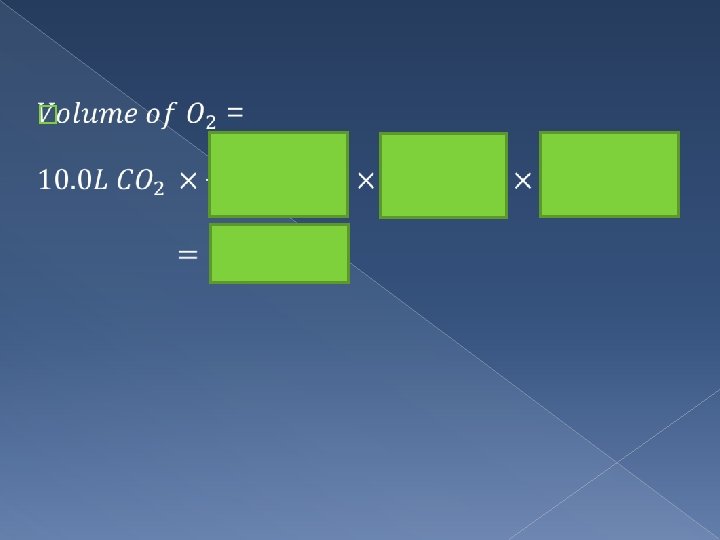










- Slides: 30

Chapter 11 - Stoichiometry The Math of Chemistry

Using Mole Ratios From a Balanced Equation 1. Recall that the coeffcients in a balanced equation can represent: 1. The number of atoms/molecules 2. The number of moles of atoms/molecules 1 mol of CH 4 molecules 2 mols of O 2 molecules 1 mol of CO 2 molecules 2 moles of H 20 molecules

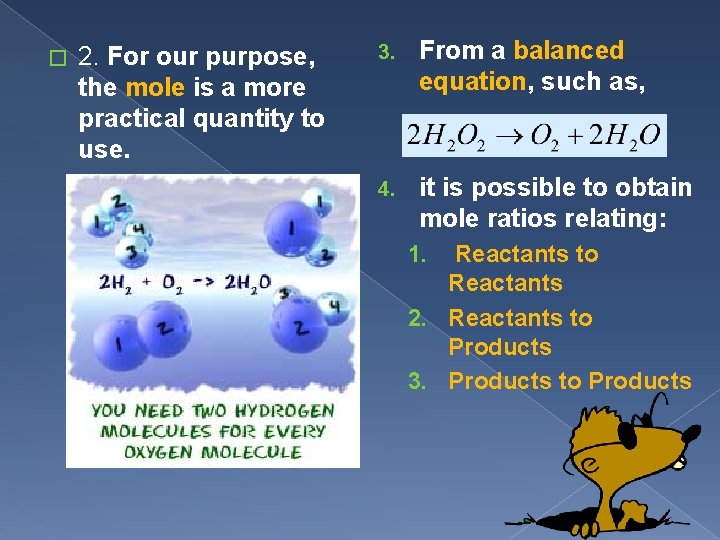
� 2. For our purpose, the mole is a more practical quantity to use. 3. From a balanced equation, such as, 4. it is possible to obtain mole ratios relating: Reactants to Reactants 2. Reactants to Products 3. Products to Products 1.

4. Using Mole Ratios in a chemical reaction problem. E. g. Hydrazine (N 2 H 4) and hydrogen peroxide (H 2 O 2) are used for rocket fuel and combine to produce N 2 and H 2 O gas plus a large amount of energy according to: If it produces a large amount of energy what type of reaction is this? ? ?

Q#1: How many moles of hydrazine are needed to produce 12 moles of water? The standard method of solving this is using unitary rates or factors: *Think this through in terms of the equation we just saw. . . Does this make sense? ? ? X 3 = 3 X 3 = 12

Now Try This!? ! 4. 76 moles of H 2 O 2 produces how many moles of water?

1. The mole ratio used must come from the balanced chemical equation. 2. In the mole ratio, the quantity that you want to find goes in the numerator (e. g. 1 mole N 2 H 4).

Hebden p. 124 #1 -4 Then, do the Review and Practise sheet # 1 -6

. . . GETTING DOWN TO BUSINESS. . . Building on the mole ratio we looked at. . .

STOICHIOMETRY Stoichiometry involves measuring or calculating the amounts of reactants and/or products in a chemical reaction from a balanced equation 2. Usually the amounts are in units of moles or grams (if measuring for an experiment). 1.

The steps involved in solving a stoichiometric question are ones that you are familiar with: Balancing the equation 2. Mass to Mole Conversions 3. Mole to Mass Conversions 4. Mole Ratios from a balanced equation 1.

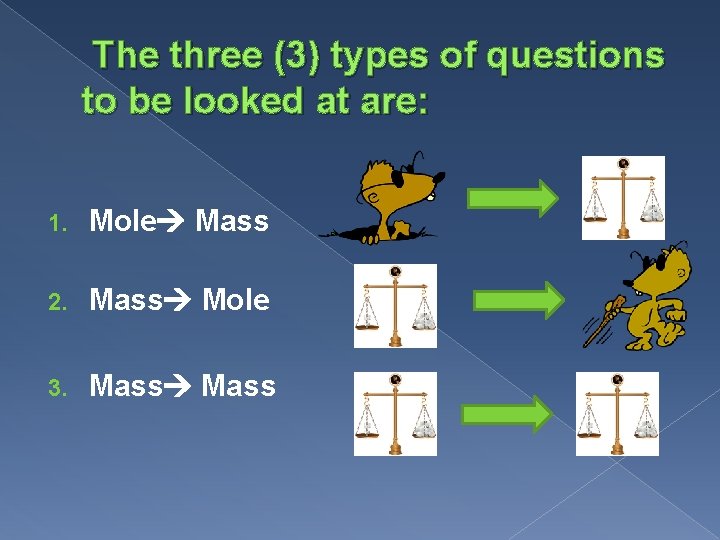
The three (3) types of questions to be looked at are: 1. Mole Mass 2. Mass Mole 3. Mass

1. Moles Mass E. g. How many grams of water will be produced when 5. 00 moles of hydrogen react with sufficient oxygen? � Note: If the other reactants are not mentioned then it’s assumed that there is enough for the reaction to go to completion.

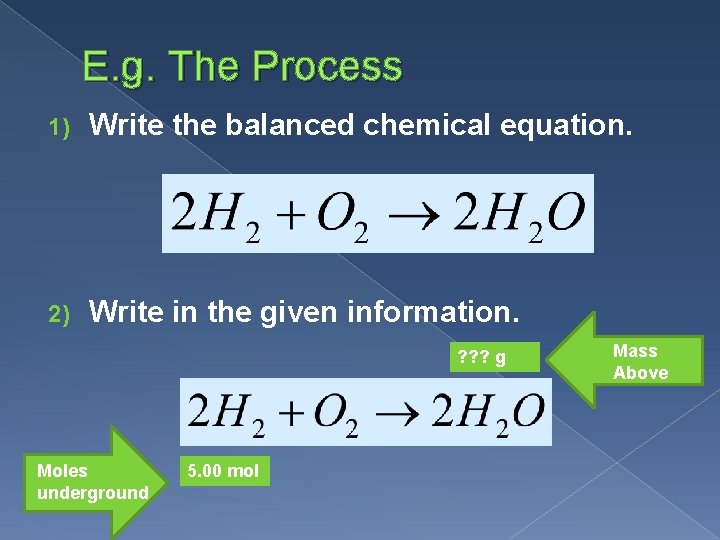
E. g. The Process 1) Write the balanced chemical equation. 2) Write in the given information. ? ? ? g Moles underground 5. 00 mol Mass Above

E. g. The Process (cont. ) 3) Set up a flow diagram for the steps needed. ? ? ? g 5. 00 mol 4) ? ? ? mol Perform the appropriate operations. Note: Each arrow is a calculation--- doing unit analysis the unit you are going “from” goes on the bottom, and the unit you are going “to” goes on the top!

One more conversion to remember!! � 1 mole of any gas at at STP (Standard Temperature and Pressure)… has a volume of 22. 4 L. Eg. What is the volume occupied by 0. 350 mol of SO 2 at STP? #L = 0. 350 mol x 22. 4 L 1 mol = 7. 84 L

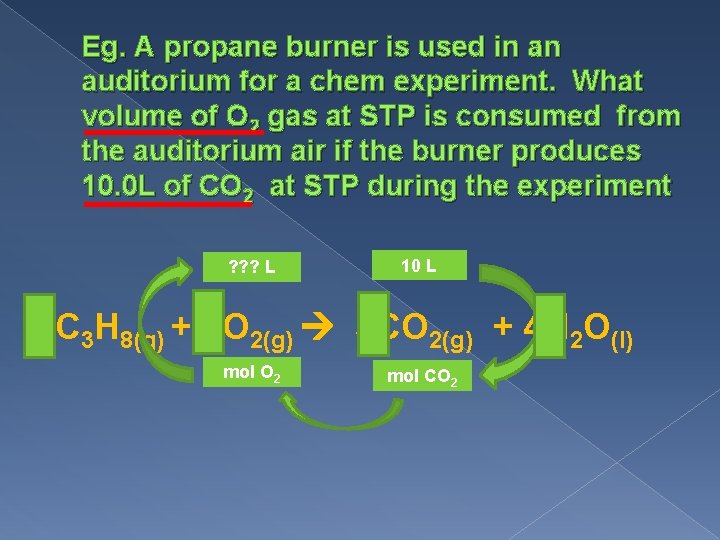
Eg. A propane burner is used in an auditorium for a chem experiment. What volume of O 2 gas at STP is consumed from the auditorium air if the burner produces 10. 0 L of CO 2 at STP during the experiment ? ? ? L 10 L C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) mol O 2 mol CO 2
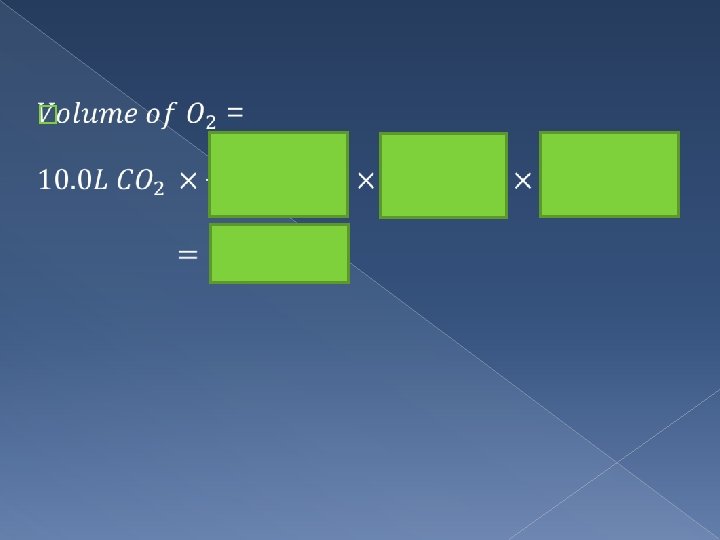

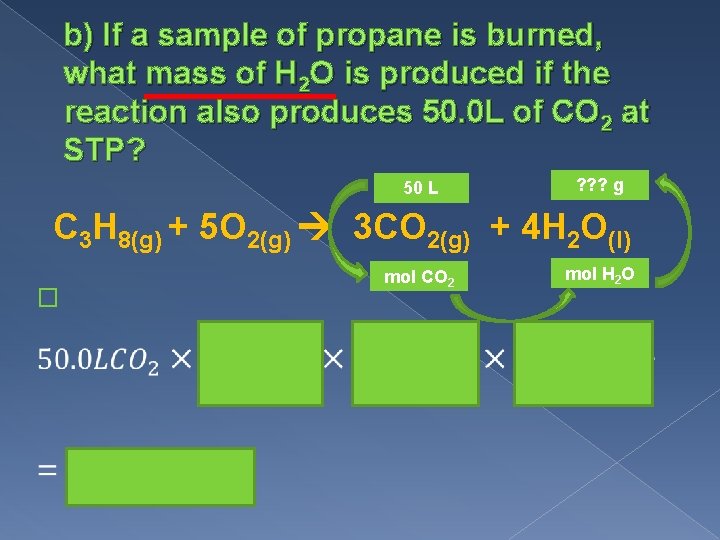
b) If a sample of propane is burned, what mass of H 2 O is produced if the reaction also produces 50. 0 L of CO 2 at STP? 50 L ? ? ? g C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) � mol CO 2 mol H 2 O

Try One on your own: �

2. Mass Moles This procedure is exactly the same once you have a flowchart you just follow the arrows! ü

E. g. How many moles of water will be produced if 100. 0 grams of oxygen reacts with sufficient hydrogen? 1) Write the balanced chemical equation. 2) Write in the given information. Masses Here 100. 0 g Moles Here ? ? ? mol

E. g. The Process (cont. ) 3) Set up a flow diagram for the steps needed. 100. 0 g ? ? ? mol 4) ? ? ? mol Perform the appropriate operations. Note: Each arrow is a calculation---

3. Mass Although this is technically the most difficult type of question, once again the procedure is exactly the same if you can set-up a flowchart you just follow the arrows!

E. g. What mass of hydrogen is needed to produce 400. 0 g of water? 1) Write the balanced chemical equation. 2) Write in the given information. ? ? ? g Moles Here 400. 0 g Masses Here

E. g. The Process (cont. ) 3) Set up a flow diagram for the steps needed. ? ? ? g ? ? ? mol 4) 400. 0 g ? ? ? mol Perform the appropriate operations.

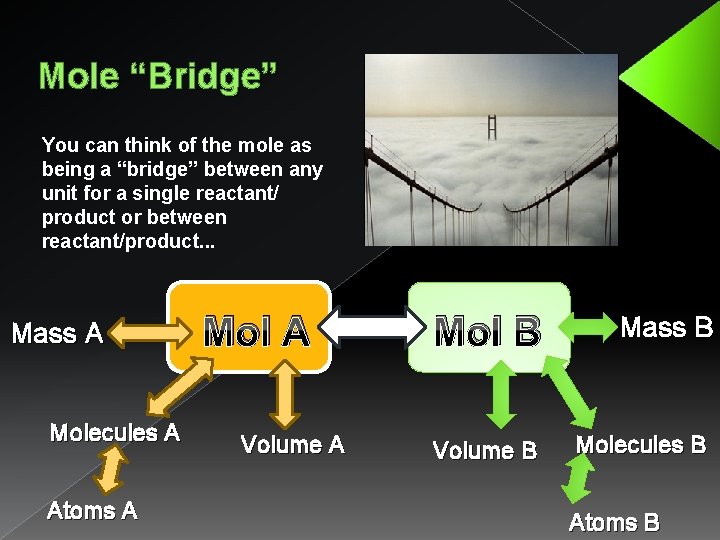
Mole “Bridge” You can think of the mole as being a “bridge” between any unit for a single reactant/ product or between reactant/product. . . Mass A Molecules A Atoms A Mol A Volume A Mol B Mass B Volume B Molecules B Atoms B

What are the connections between units and the mole? � Moles and mass › Molecular/ Molar Mass �From the periodic table �Mass of ONE MOLE!!! � Moles and Molecules › Avagadro’s number �Items in ONE MOLE!!! � Moles and Volume (more on these soon) › Concentration of solutions › Molar volume of gases �The moles in ONE LITER!!! � Moles and Moles › Mole Ratio �From the BALANCED CHEMICAL EQUATION!!!

Try One More: A student wants to put 50. 0 L of Hydrogen gas at STP into a plastic bag by reacting excess aluminum metal with 3. 00 Molar (mol/L) Sodium Hydroxide… 2 Al(s) + 2 Na. OH(aq) + 2 H 2 O(l) 2 Na. Al. O 2 + 3 H 2(g) What volume of Na. OH solution is required? Answer: 0. 496 L Na. OH

Work for Today: � Do Review and Practise Questions Sheet (the Real stuff!) � For Extra Practice at home… � Hebden Pg 127 # 7 -12 � Hebden Pg 131 # 18 -24