Chapter 11 Reactions of Carboxylic Acids and Carboxylic

Chapter 11 Reactions of Carboxylic Acids and Carboxylic Acid Derivatives Paula Yurkanis Bruice University of California, Santa Barbara

Contents of Chapter 11 n n Naming Carboxylic Acids and Derivatives Physical Properties of Carbonyl Compounds Reactivity of Carboxylic Acid Derivatives Reactions of Carboxylic Acids and Derivatives Chapter 11 2

IUPAC Nomenclature of Carboxylic Acids Ø Ø Ø Select as the parent compound the longest chain of carbon atoms that contains the carboxyl group Change the suffix –e of the parent name to –oic acid Carboxyl carbon is carbon 1 Chapter 11 3
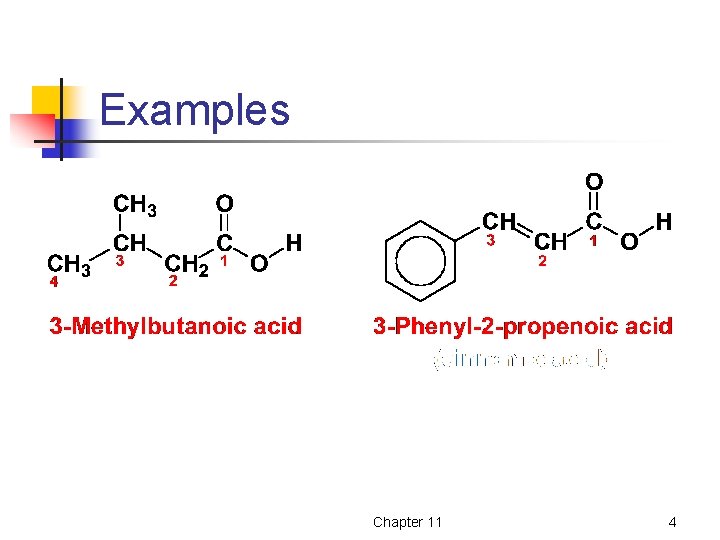
Examples Chapter 11 4

Dicarboxylic Acids Dicarboxylic acids are named by adding the suffix –dioic acid to the name of the parent compound that contains both carboxyl groups Chapter 11 5
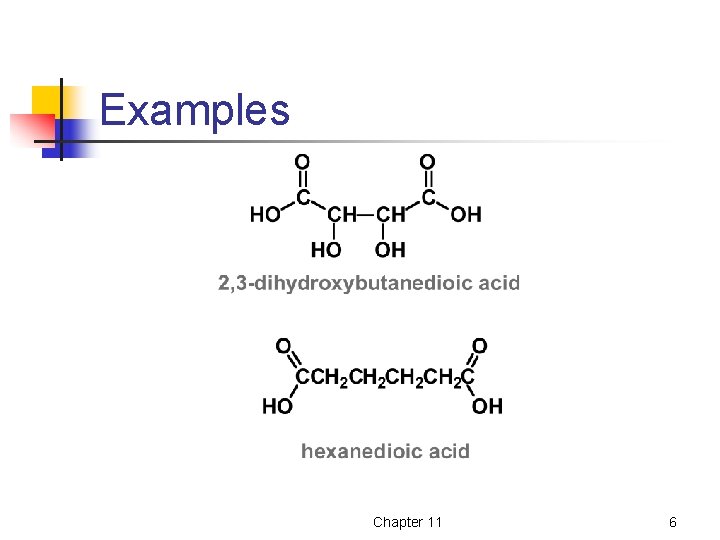
Examples Chapter 11 6
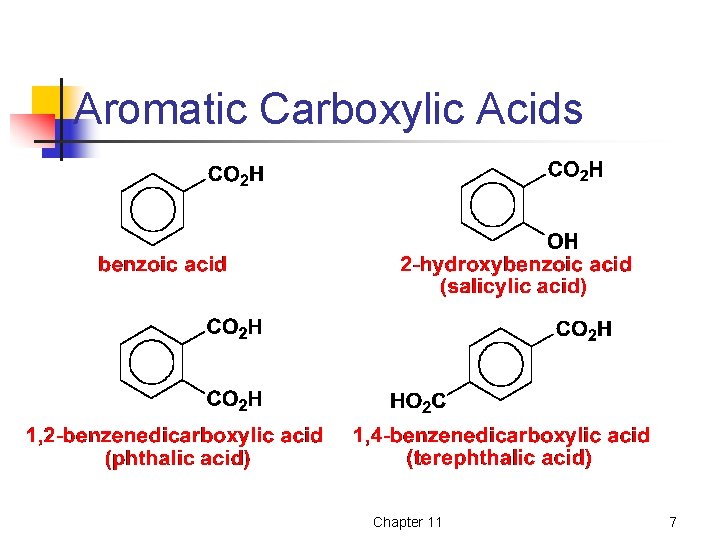
Aromatic Carboxylic Acids Chapter 11 7
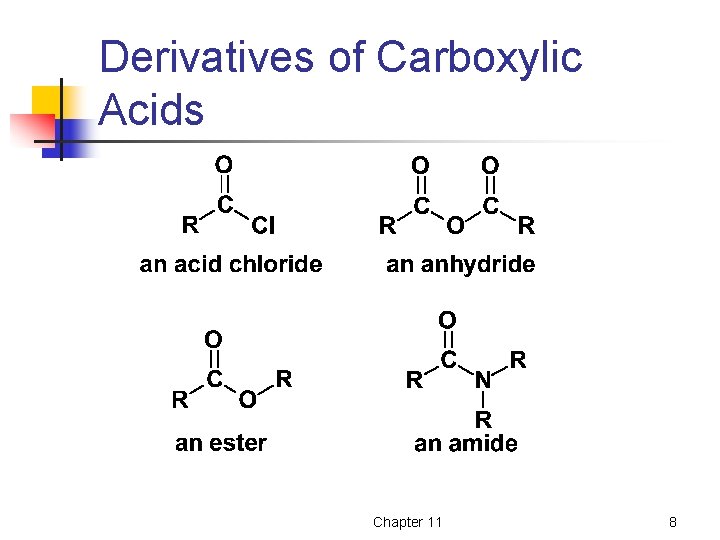
Derivatives of Carboxylic Acids Chapter 11 8
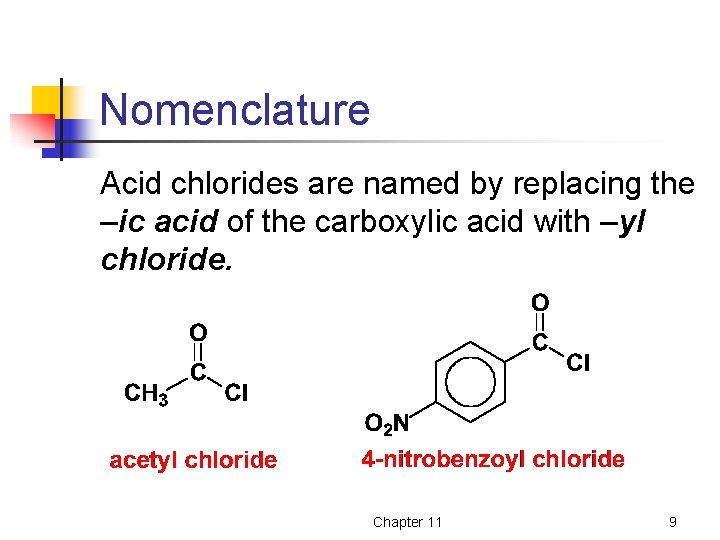
Nomenclature Acid chlorides are named by replacing the –ic acid of the carboxylic acid with –yl chloride. Chapter 11 9
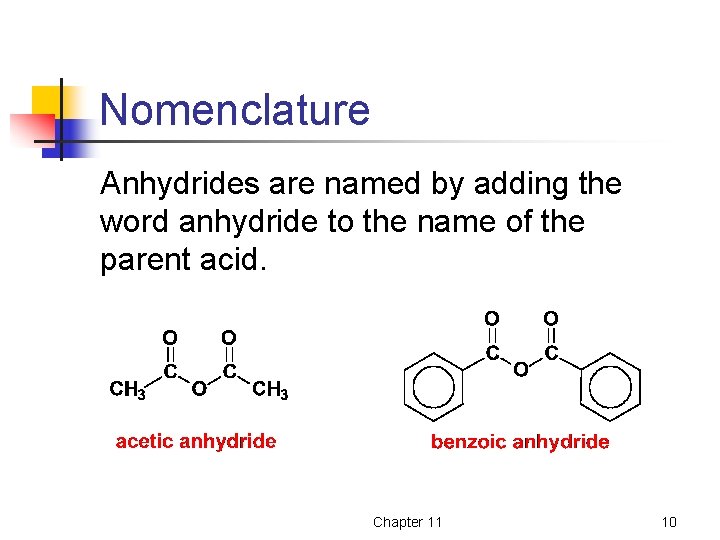
Nomenclature Anhydrides are named by adding the word anhydride to the name of the parent acid. Chapter 11 10
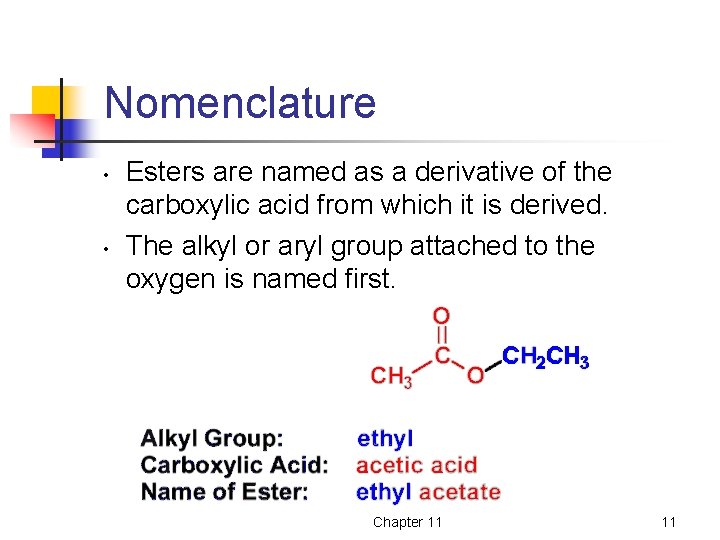
Nomenclature • • Esters are named as a derivative of the carboxylic acid from which it is derived. The alkyl or aryl group attached to the oxygen is named first. Chapter 11 11
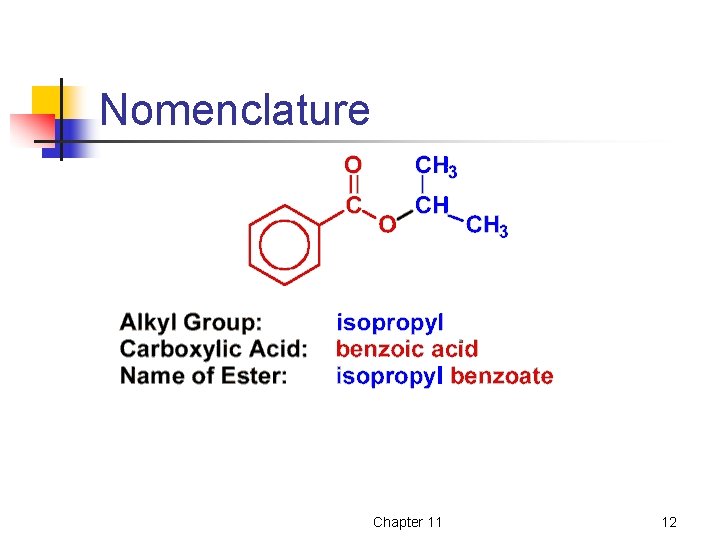
Nomenclature Chapter 11 12

Nomenclature Amides are named as derivatives of their carboxylic acid by dropping the suffix of the acid and adding –amide. Note: the above amides are all primary (1 o) amides. Chapter 11 13
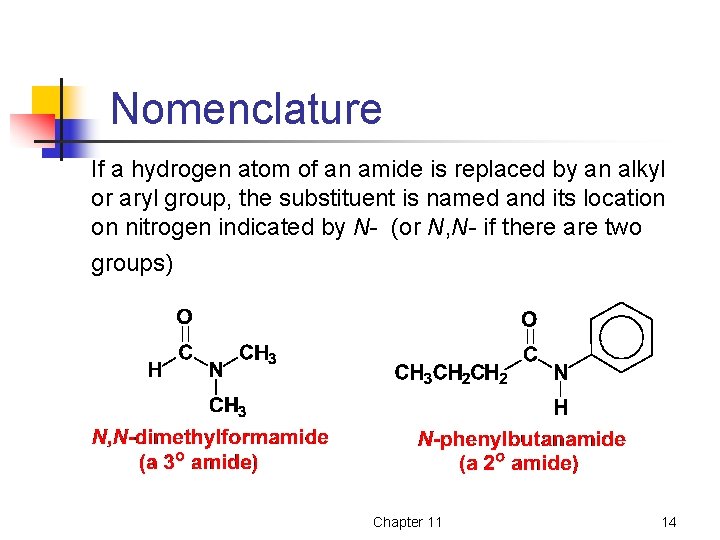
Nomenclature If a hydrogen atom of an amide is replaced by an alkyl or aryl group, the substituent is named and its location on nitrogen indicated by N- (or N, N- if there are two groups) Chapter 11 14
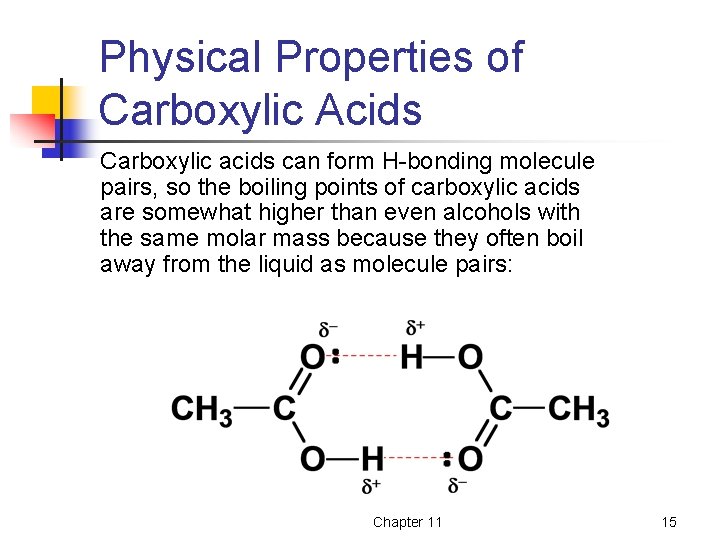
Physical Properties of Carboxylic Acids Carboxylic acids can form H-bonding molecule pairs, so the boiling points of carboxylic acids are somewhat higher than even alcohols with the same molar mass because they often boil away from the liquid as molecule pairs: Chapter 11 15

Physical Properties of Carboxylic Acids Chapter 11 16
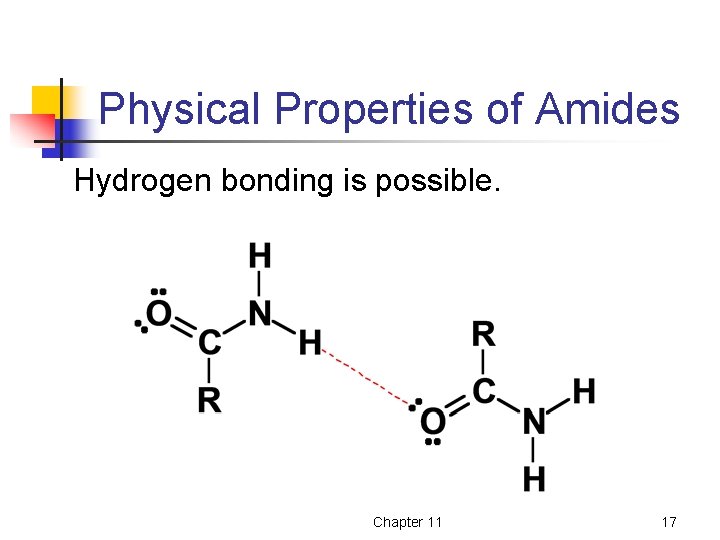
Physical Properties of Amides Hydrogen bonding is possible. Chapter 11 17
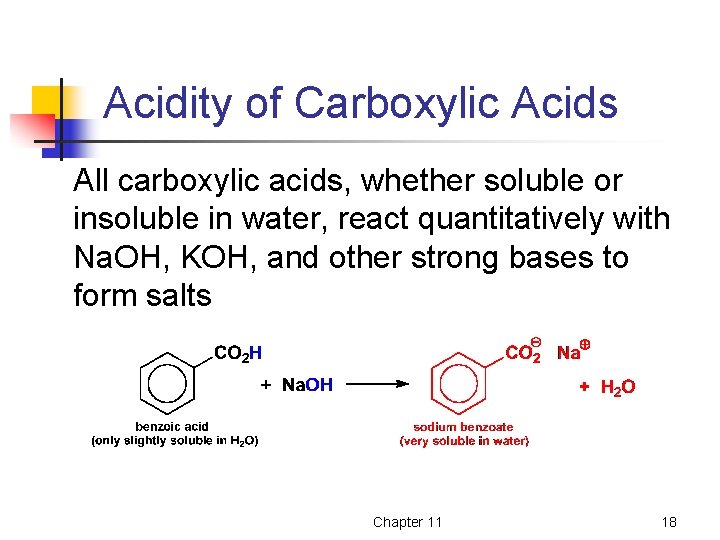
Acidity of Carboxylic Acids All carboxylic acids, whether soluble or insoluble in water, react quantitatively with Na. OH, KOH, and other strong bases to form salts Chapter 11 18
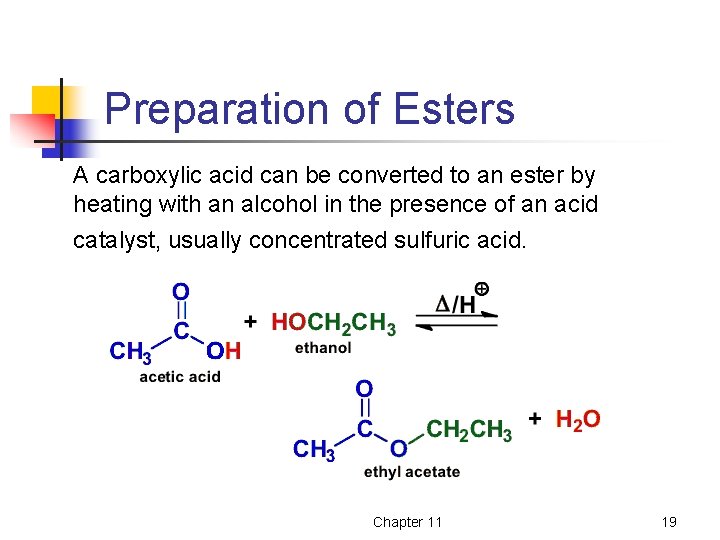
Preparation of Esters A carboxylic acid can be converted to an ester by heating with an alcohol in the presence of an acid catalyst, usually concentrated sulfuric acid. Chapter 11 19

Stabilities of Carboxylic Acid Derivatives • In principle carboxylic acid derivatives should be able to be interconverted into one another using a scheme like the one shown below • In the scheme below LG represents the original group, like OR, attached to the starting compound (OR would make this an ester) • Nu (nucleophile) forces LG (leaving group) to leave and replaces it, making a new compound • In this scheme if Nu is amine and LG is alkoxy (OR) then an ester is converted into an amide Chapter 11 20

Stabilities of Carboxylic Acid Derivatives • Scheme on previous slide will only work if Nu is less stable than LG, so reaction goes downhill • Relative energy of Nu or LG given by p. Ka of conjugate acid • In table below the derivatives at the top are less stable than those beneath • In neutral or basic media you can convert a derivative higher up into one lower down, but not vice-versa Chapter 11 21

Stabilities of Carboxylic Acid Derivatives • In acidic media interconversion of carboxylic acid derivatives works by a different mechanism • In this mechanism LG can be protonated before it leaves • In this case the leaving group is no longer negatively charged when it leaves; therefore it leaves more easily (more stable) • In acidic media you can interconvert any of the carboxylic acid derivatives into one another Chapter 11 22

Reactions Carboxylic Acid Derivatives • To work out the product of a carboxylic acid derivative reaction first make sure the reaction will work • If the reaction is done in acid or goes from a less stable derivative to a more stable derivative polarize the most polar bonds in the reactants and swap partners • Always polarize the carbonyl carbon positive and the leaving group negative in these reactions Chapter 11 23
- Slides: 23