CHAPTER 11 PART 1 BALANCING EQUATIONS Keefe 1415

CHAPTER 11 PART 1: BALANCING EQUATIONS Keefe 1415

WHAT ARE CHEMICAL REACTIONS? • The process by which one or more substances are rearranged to form different substances.

COMPONENTS FOR CHEMICAL EQUATIONS • Reactants Like INGREDIENTS for a reaction Products END RESULT or GOAL for a reaction Sample: 2 Mg (s) + O 2(g) 2 Mg. O(s) • Coefficients: Numerals used in an equation to indicate relative amounts of reactants and products.

WAYS TO DENOTE CONDITIONS FOR REACTIONS.

DIATOMIC MOLECULES “THE MAGNIFICENT SEVEN” • When on their own, they exist as a pair of atoms to form a molecule. • Br 2 bromine gas or liquid, I 2 iodine gas or crystal , N 2 nitrogen gas, Cl 2 chlorine gas, H 2 hydrogen gas, O 2 oxygen gas, F 2 fluorine gas “Br. INCl. HOF”

REPRESENTING CHEMICAL EQUATIONS. • Word problem: Hydrogen gas and oxygen gas react to yield water vapor Word equation: Hydrogen (g) + Oxygen(g) Water (l) • Skeleton equation: H 2(g) + O 2(g) H 2 O(l)

BALANCE THE REACTION: 2 H 2(g) + H 2 O(l) O 2(g) 2

BALANCING EQUATIONS

BALANCING EQUATIONS • Key Law: “Conservation of Matter”! • Must have the same number (#) of atoms (mass) on the reactant side as on the product side!!!

QUESTION USING THE LAW OF CONSERVATION OF MATTER. • Which windows represent (1) the reactants and (2) the products for the following reaction?

GENERAL SUMMARY STEPS TO BALANCING EQUATIONS. • Determine the correct formulas and physical states of reactants & products. • Write a skeleton equation with reactants & products. • Count atoms for each element in the reactants & products. • Balance two sides of chemical reaction with whole number coefficients. (NEVER change subscripts for ANY reason!!!) • Check each element or polyatomic ion is balanced on both sides. Make sure the coefficients are the SIMPLEST whole number

BALANCING USING A GRID Al(s) + HCl(aq) H 2(g) + Al. Cl 3(s) Draw the chart: Elements Al H Cl Reactants Products

USING A GRID WITH POLYATOMICS Pb(NO 3)4 (aq) + Cu. SO 4 Components Pb NO 31 Cu SO 42 - (aq) Reactant Pb(SO 4)2(s) +Cu(NO 3)2(aq) Product

CAN COEFFICIENTS BE REPRESENTED AS FRACTIONS? • Yes, but we use the common denominator to convert them to whole numbers. • Remember, the coefficients are ratios, just like the subscripts for each compound.
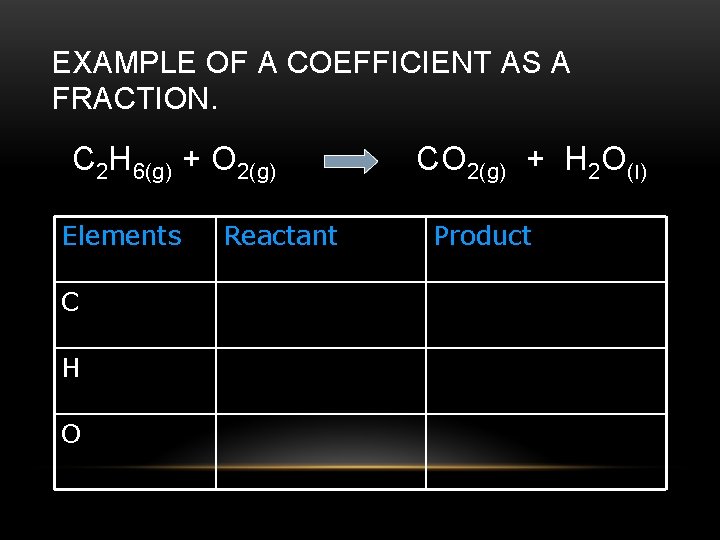
EXAMPLE OF A COEFFICIENT AS A FRACTION. C 2 H 6(g) + O 2(g) Elements C H O Reactant CO 2(g) + H 2 O(l) Product

BALANCE THE FOLLOWING REACTIONS:

BALANCE THE FOLLOWING REACTIONS:

WHAT WE MAY LOOK FOR IN A CHEMICAL REACTION!! • Temperature change (energy change) • Precipitate forming (solid from two aqueous solutions) • Formation of a gas • Smoke (for when it buuuuuurns) • Color change • Odor (not best, but possible indicator) • p. H change

TYPES OF REACTIONS 1. Synthesis reactions 2. Decomposition reactions 3. Single displacement reactions 4. Double displacement reactions 5. Combustion reactions 6. Neutralization reactions You need to be able to identify each type.

1. SYNTHESIS Example C + O 2 C + O O General: O C O A + B AB

EX. SYNTHESIS REACTION

PRACTICE • Predict the products and balance the reactions. Na(s) + Cl 2(g) Mg(s) + F 2(g) • . Al(s) + F 2(g)

MORE PRACTICE: GIVE THE FORMULAE, PREDICT PRODUCTS, AND BALANCE • Sulfur dioxide gas is bubbled through water • Solid potassium oxide is added to a container of carbon dioxide gas • A piece of sodium is added to a container of iodine vapor

2. DECOMPOSITION Example: Na. Cl Cl Na General: Cl + Na AB A + B
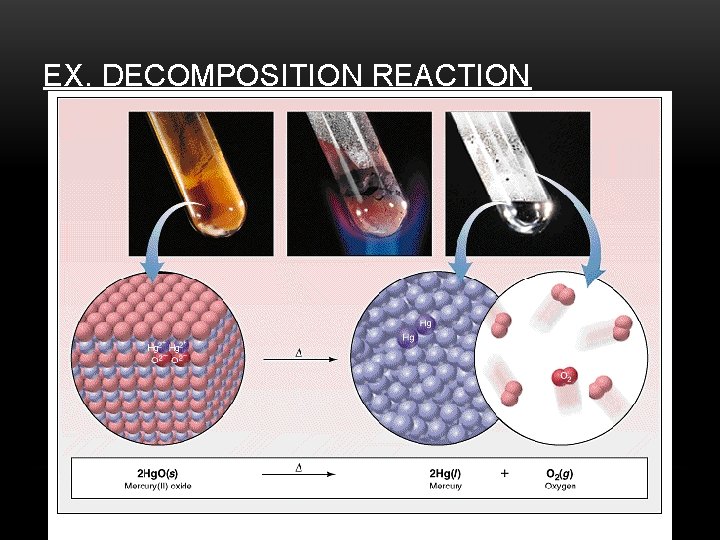
EX. DECOMPOSITION REACTION

DECOMPOSITION PRACTICE: • balance: • H 2 O ---> H 2 + O 2

GIVE THE FORMULA, PREDICT THE PRODUCTS, AND BALANCE:

3. SINGLE DISPLACEMENT (AKA SINGLE REPLACEMENT) Example: Zn + Cu. Cl 2 Zn was oxidized Went from neutral (0) to (+2) Cl Cu + Cl Zn + Cu Cl Cu was reduced Went from (+2) to Neutral (0) General: AB + C AC + B
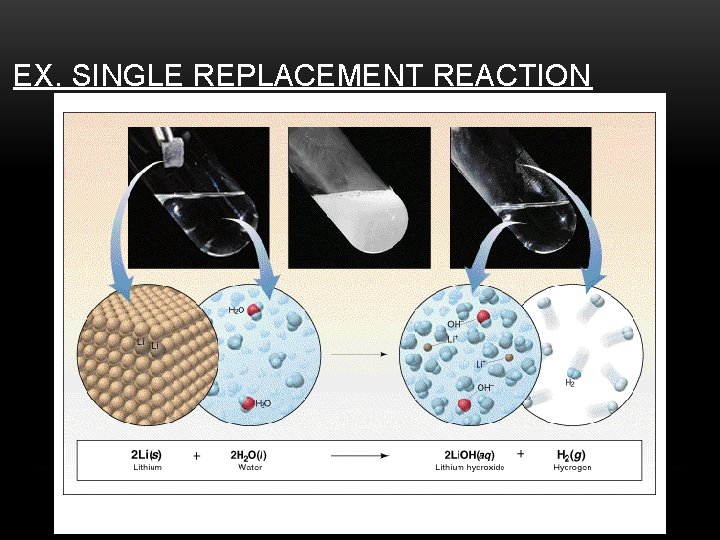
EX. SINGLE REPLACEMENT REACTION

SINGLE REPLACEMENT REACTIONS • Predict the product and balance the following single replacement reaction equations: • Zn(s) + HCl(aq) • Na. Cl(s) + F 2(g) • Al(s)+ Cu(NO 3)2(aq)

MORE PRACTIC E:
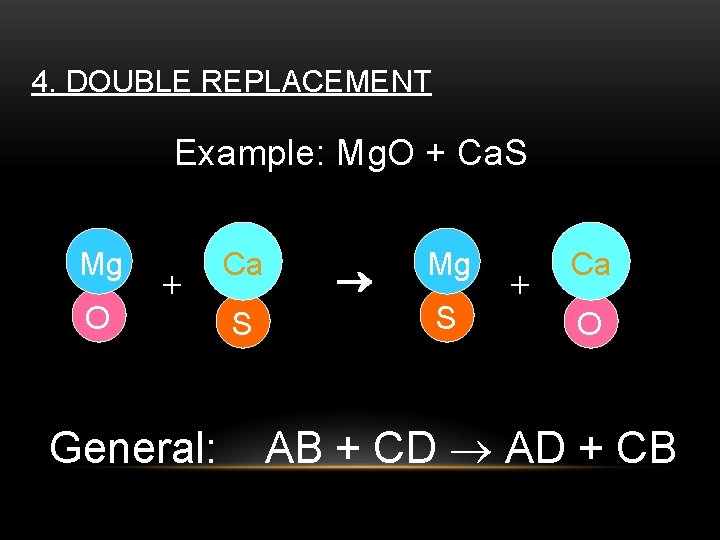
4. DOUBLE REPLACEMENT Example: Mg. O + Ca. S Mg O + General: Ca S Mg S + Ca O AB + CD AD + CB

DOUBLE REPLACEMENT REACTIONS • Think about it like “foil”ing in algebra, first and last ions go together + inside ions go together • Example: Ag. NO 3(aq) + Na. Cl(s) Ag. Cl(s) + Na. NO 3(aq) • Another example: K 2 SO 4(aq) + Ba(NO 3)2(aq) KNO 3(aq) + Ba. SO 4(s)

PRACTICE • Predict the products. Balance as necessary. 1. HCl(aq) + Ag. NO 3(aq) 2. Ca. Cl 2(aq) + Na 3 PO 4(aq) 3. Pb(NO 3)2(aq) + Ba. Cl 2(aq) 4. Fe. Cl 3(aq) + Na. OH(aq) 5. KOH(aq) + Cu. SO 4(aq)

COMBUSTION REACTIONS • In general: Cx. Hy + O 2 CO 2 + H 2 O • Products are ALWAYS carbon dioxide and water.

COMBUSTION • Example • C 5 H 12 + O 2 CO 2 + H 2 O • Write the products and balance the following combustion reaction: • C 10 H 22 + O 2

MORE PRACTICE:

NEUTRALIZATION • Acid + base a salt + water • ***acid formulae always start with H • *** bases always end in hydroxide and START with alkali or alkaline earth metals • Example: HBr + Na. OH ---> Na. Br + H 2 O
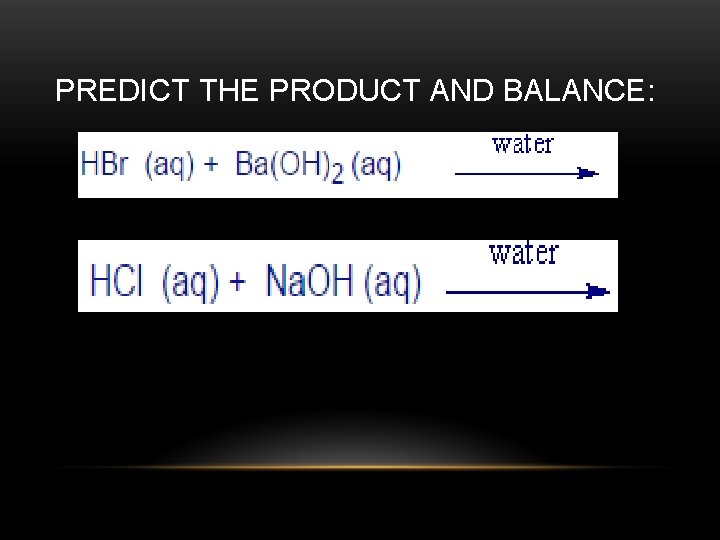
PREDICT THE PRODUCT AND BALANCE:

MIXED PRACTICE • State the type, predict the products, & balance. 1. Ba. Cl 2 + H 2 SO 4 2. C 6 H 12 + O 2 3. Zn + Cu. SO 4 4. Cs + Br 2 5. Fe. CO 3 6. NH 4 OH + HBr
- Slides: 40