Chapter 11 Lecture Basic Chemistry Fourth Edition Chapter

Chapter 11 Lecture Basic Chemistry Fourth Edition Chapter 11 Gases 11. 3 Pressure and Volume (Boyle’s Law) Learning Goal Use the pressure-volume relationship (Boyle’s law) to determine the final pressure or volume when the temperature and amount of gas are constant. © 2014 Pearson Education, Inc.

Boyle’s Law Boyle’s law states that • the pressure (P) of a gas is inversely related to its volume (V) when T and n are constant • if the pressure (P) increases, then the volume (V) decreases © 2014 Pearson Education, Inc. Figure 11. 4 Boyle’s law: As Volume decreases, gas molecules become more crowded, which causes the pressure to increase. Pressure and volume are inversely related.

Boyle’s Law Boyle’s law is an indirect relationship between pressure and volume. An indirect relationship between variables means as one variable increases the second decreases. P 1 V 1 = P 2 V 2 Temperature and moles are constant. © 2014 Pearson Education, Inc.

Boyle’s Law Equation The equation for Boyle’s law can be rearranged to solve for any factor. At a constant temperature and amount of gas: P 1 V 1 = P 2 V 2 Boyle’s law To obtain V 2, divide both sides by P 2. © 2014 Pearson Education, Inc.
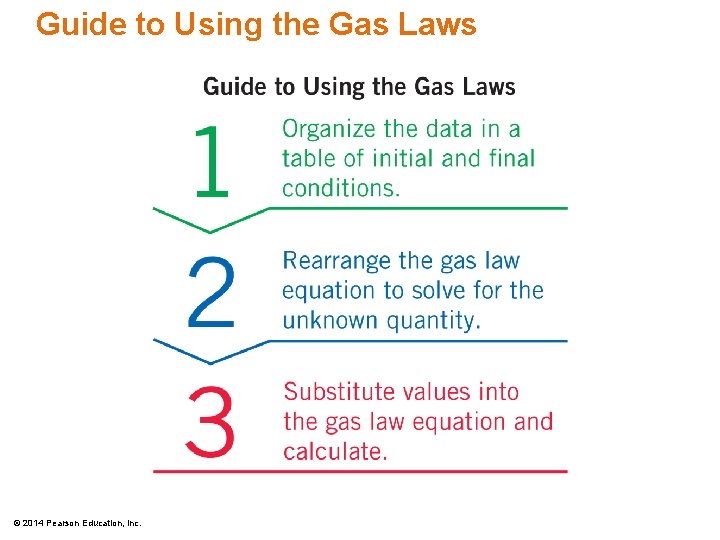
Guide to Using the Gas Laws © 2014 Pearson Education, Inc.

Learning Check Freon-12, CCl 2 F 2, is a gas used in refrigeration systems. What is the new volume (L) of an 8. 0 -L sample of Freon gas initially at 550 mm. Hg after its pressure is changed to 2200 mm. Hg at constant T? © 2014 Pearson Education, Inc.

Solution What is the new volume (L) of an 8. 0 -L sample at 550 mm. Hg after its pressure is changed to 2200 mm. Hg at constant T? Step 1 Organize the data in a table of initial and final conditions. We can predict that as P increases, V decreases. Conditions 1 P 1 = 550 mm. Hg Conditions 2 V 1 = 8. 0 L V 2 = ? © 2014 Pearson Education, Inc. P 2 = 2200 mm. Hg

Solution What is the new volume (L) of an 8. 0 -L sample at 550 mm. Hg after its pressure is changed to 2200 mm. Hg at constant T? Step 2 Rearrange the gas law equation to solve for the unknown quantity. Solve Boyle’s law for V 2. Increasing P decreases V when n and T are constant. © 2014 Pearson Education, Inc.

Solution What is the new volume (L) of an 8. 0 -L sample at 550 mm. Hg after its pressure is changed to 2200 mm. Hg at constant T? Step 3 Substitute values into the gas law equation and calculate. The pressure ratio decreases the volume. © 2014 Pearson Education, Inc.
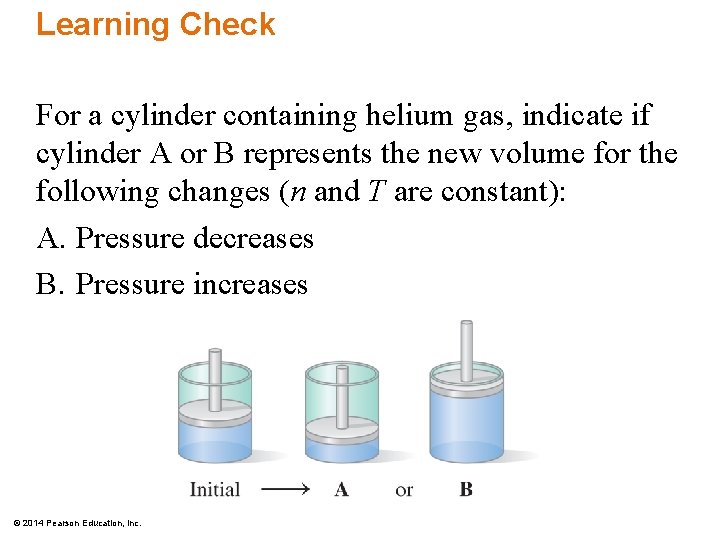
Learning Check For a cylinder containing helium gas, indicate if cylinder A or B represents the new volume for the following changes (n and T are constant): A. Pressure decreases B. Pressure increases © 2014 Pearson Education, Inc.

Solution For a cylinder containing helium gas, indicate if cylinder A or B represents the new volume for the following changes (n and T are constant): A. Pressure decreases Cylinder B B. Pressure increases Cylinder A © 2014 Pearson Education, Inc.

Learning Check A balloon of helium gas has a volume of 6. 4 L at a pressure of 0. 70 atm. At 1. 40 atm (T and n constant), is the new volume represented by A, B, or C? © 2014 Pearson Education, Inc.

Solution A balloon of helium gas has a volume of 6. 4 L at a pressure of 0. 70 atm. At 1. 40 atm (T and n constant), is the new volume represented by A, B, or C? At a higher pressure (T and n constant), the new volume is represented by the smaller balloon A. © 2014 Pearson Education, Inc.
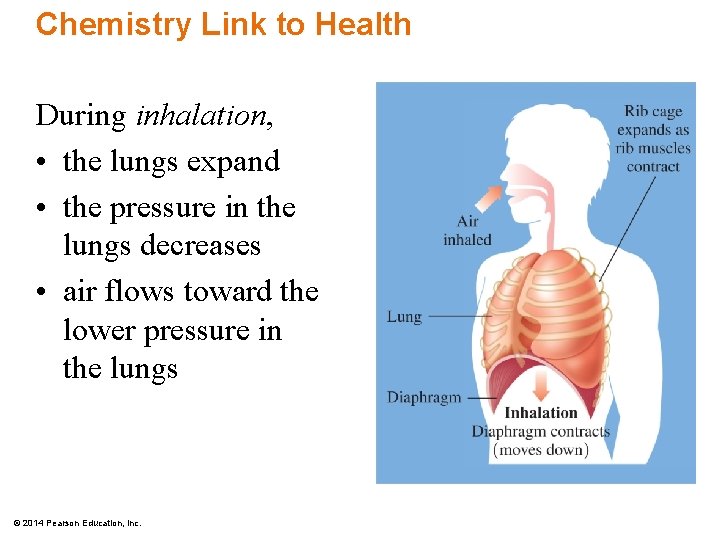
Chemistry Link to Health During inhalation, • the lungs expand • the pressure in the lungs decreases • air flows toward the lower pressure in the lungs © 2014 Pearson Education, Inc.
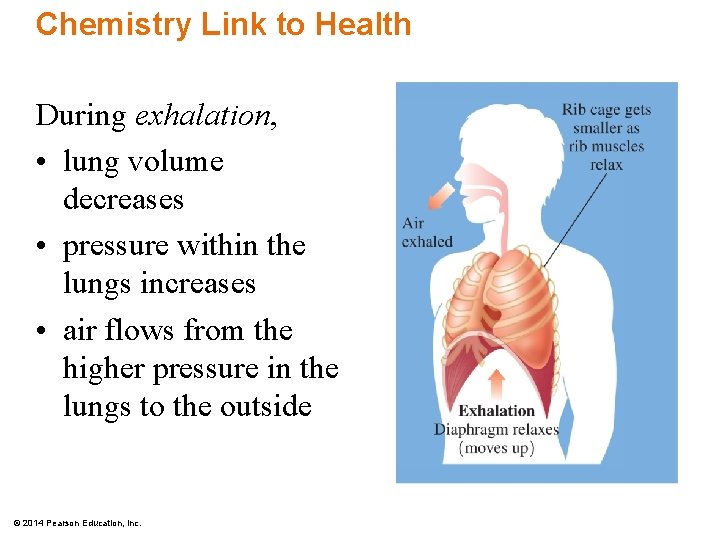
Chemistry Link to Health During exhalation, • lung volume decreases • pressure within the lungs increases • air flows from the higher pressure in the lungs to the outside © 2014 Pearson Education, Inc.
- Slides: 15