Chapter 11 Gases 11 1 Gases Pressure Defining















































- Slides: 59

Chapter 11 Gases 11. 1 Gases & Pressure

Defining Gas Pressure • • Pressure Increases • Number of Particles Increases • How are number of particles and pressure related? Pressure –force per unit area that particles exert on walls of their container Gas particles collide with walls = greater pressure Pressure is directly proportional to number of particles.

Temperature & Pressure • • Kelvin Temperature Increases Pressure of Gas Increases • Higher temperature results in more kinetic energy! IF the volume of container remains constant and IF the amount of gas remains constant: the pressure of a gas increases in direct proportion to the Kelvin temperature. (Kelvin Temp = Celsius Temp + 273) Volume of a gas at constant pressure is directly proportional to Kelvin temp.

Temperature Conversions • Kelvin & Celsius Tk = (Tc + 273) Tc = (Tk - 273) • Fahrenheit & Celsius Tf = (9/5 Tc) + 32 Tc = (Tf - 32) 5/9

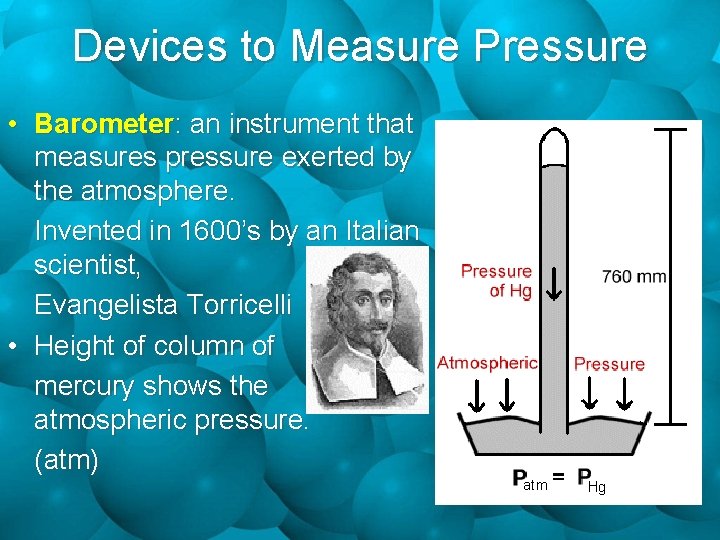
Devices to Measure Pressure • Barometer: an instrument that measures pressure exerted by the atmosphere. Invented in 1600’s by an Italian scientist, Evangelista Torricelli • Height of column of mercury shows the atmospheric pressure. (atm)

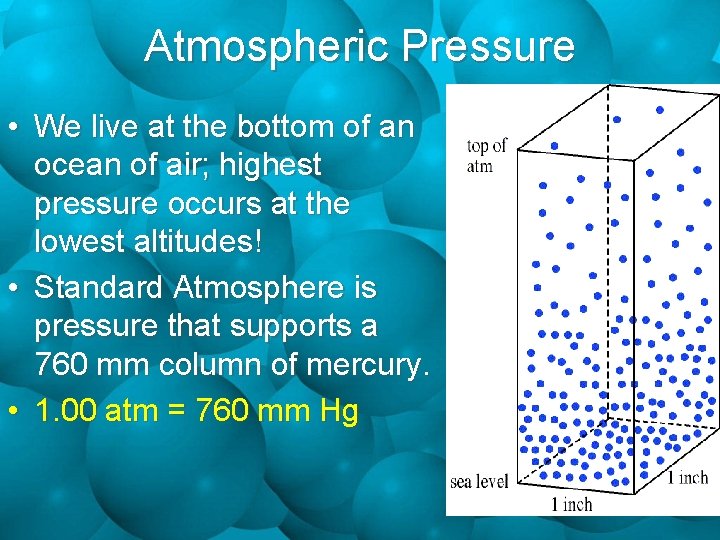
Atmospheric Pressure • We live at the bottom of an ocean of air; highest pressure occurs at the lowest altitudes! • Standard Atmosphere is pressure that supports a 760 mm column of mercury. • 1. 00 atm = 760 mm Hg

Devices to Measure Pressure • Pressure Gauge: instrument used to measure pressure inside a tire or oxygen tank. Tire Pressure Blood Pressure

Absolute Pressure • When measuring tire pressure; you measure pressure ABOVE atmosphere pressure. Recommended pressures for tires are gauge pressures. • Absolute pressure – the TOTAL pressure of all gases including the atmosphere. Q: How would you figure it for an inflated tire? A: Add barometric pressure to the gauge pressure.

Pressure Units • • SI unit for measuring pressure is the pascal (pa) after the French physicist Blaise Pascal (1600 s) A kilopascal (k. Pa) is 1000 pascals and is more commonly used. Equivalent Pressures 1. 00 atm 101300 Pa 101. 3 k. Pa 760 mm Hg 760 Torr 14. 7 psi

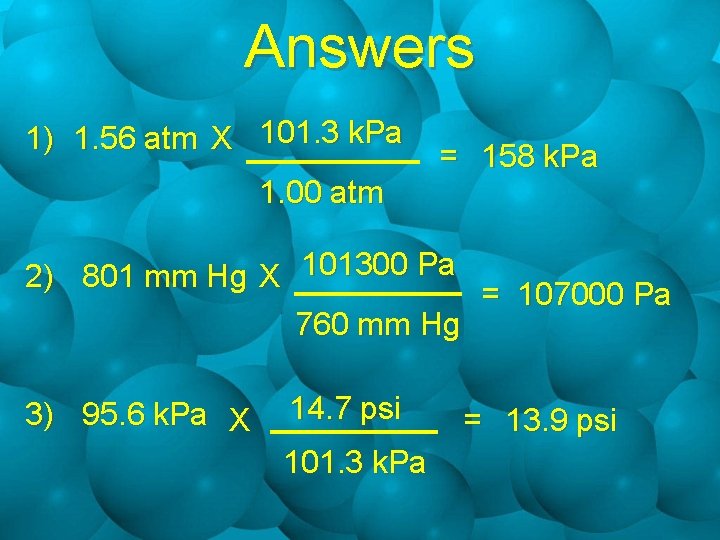
Sample Calculations 1. Express 1. 56 atm in k. Pa. 2. Convert 801 mm Hg to Pa. 3. How many psi are equivalent to 95. 6 k. Pa?

Answers 1) 1. 56 atm X 101. 3 k. Pa 1. 00 atm = 158 k. Pa 2) 801 mm Hg X 101300 Pa = 107000 Pa 760 mm Hg 3) 95. 6 k. Pa X 14. 7 psi 101. 3 k. Pa = 13. 9 psi

Dalton’s Law of Partial Pressures • Partial Pressure The pressure exerted by each gas in a mixture The t mixtu otal pres sure re of the s gases is of a um o e q press ual to f t h ures e par of the tial c gase omponen s t Dalton’s Law: PT = P 1 + P 2 + P 3 + …

Practice • Calculate the partial pressure in mm Hg exerted by the four main gases in air at 760 mm Hg: nitrogen, oxygen, argon and carbon dioxide. Their abundance by volume is 78. 08%, 20. 95%, 0. 934% and 0. 035%, respectively. N 2= 593. 4 mm Hg O 2= 159. 2 mm Hg Ar = 7. 10 mm Hg CO 2= 0. 27 mm Hg

Gases Collected by Water Displacement • Gases produced in the lab are often collected by the displacement of water in a collection bottle • Water vapor will be present in the collected gas, and it exerts a pressure • Water vapor pressure = PH 20 • Water vapor pressure increases with temperature (Appendix A, Table-8) • Pressure of the dry gas P atm = P gas + P H 20 so… P gas = P atm – P H 2 O

Practice • A student has stored 100. 0 m. L of neon gas over water on a day when the temperature if 27. 0 °C. If the barometer in the room reads 743. 3 mm Hg, what is the pressure of the neon gas in its container? P atm = P Ne + P H 2 O P Ne = P atm – P H 2 O P Ne = 743. 3 mm Hg – 26. 7 mm Hg =716. 6 mm Hg

Chapter 11 Gases 11. 2 The Gas Laws

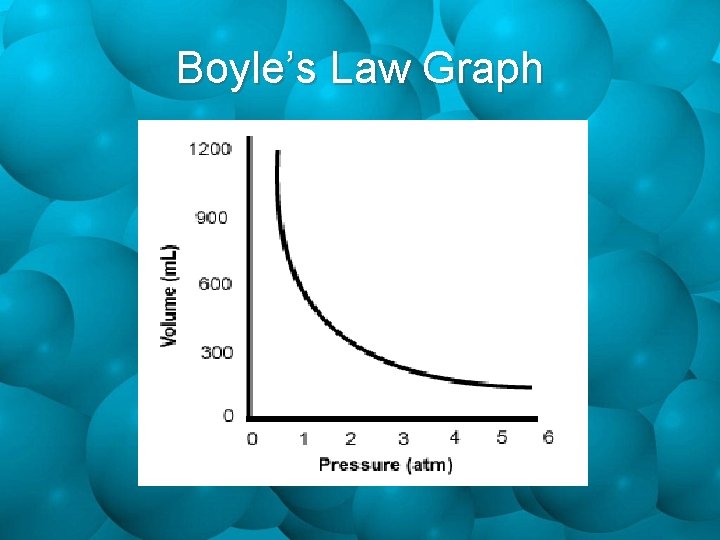
Pressure & Volume • In the 1600 s, Robert Boyle did many experiments involving gases. • He did these experiments at constant temperature. if pressure increa ses, volume dec reases if pressure decre ases, volume inc reases Pressure & Volume are Inversely Proportional!

Boyle’s Law Graph

Boyle’s Law V 1 P 1=V 2 P 2 Where: V 1 = initial volume P 1 = initial pressure V 2 = final volume P 2 = final pressure

Kinetic Explanation of Boyle’s Law • As volume is reduced, number of particles and temperature remains constant but number of collisions with the walls of the container increases. • There is a smaller area of space for the same number of particles to move around, so pressure increases.


Practice • If you have 5. 5 L of gas at a pressure of 1. 6 atm, and the pressure changes to 1. 2 atm, what is your new volume? V 1 P 1 = V 2 P 2 (5. 5 L) x (1. 6 atm) = (x L) x (1. 2 atm) x = 7. 3 L

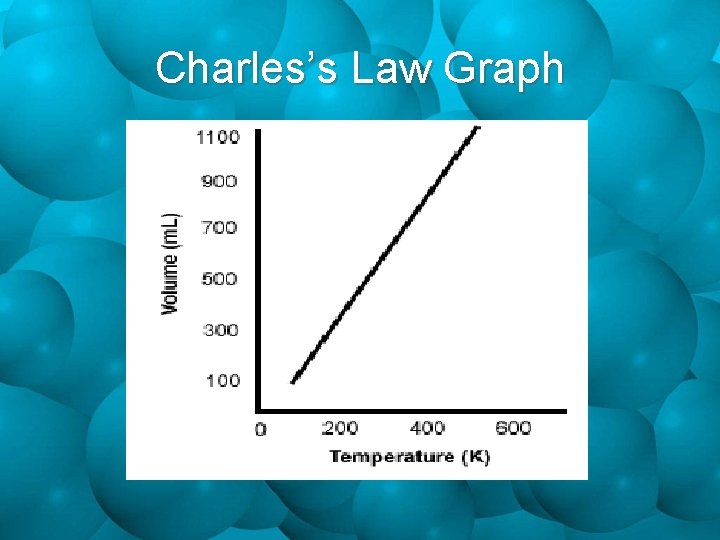
Temperature & Volume • Jacques Charles did experiments concerning gases held at constant pressure, while varying temperature. As Kelvin tem perature increases, vo lume increase s. As Kelvin tem perature decreases, vo lume decreas es. Temperature & Volume are Directly Proportional!

Charles’s Law Graph

Charles’s Law V 1 V 2 = T 1 T 2 Where: V 1 = initial volume T 1 = initial temperature V 2 = final volume T 2 = final temperature

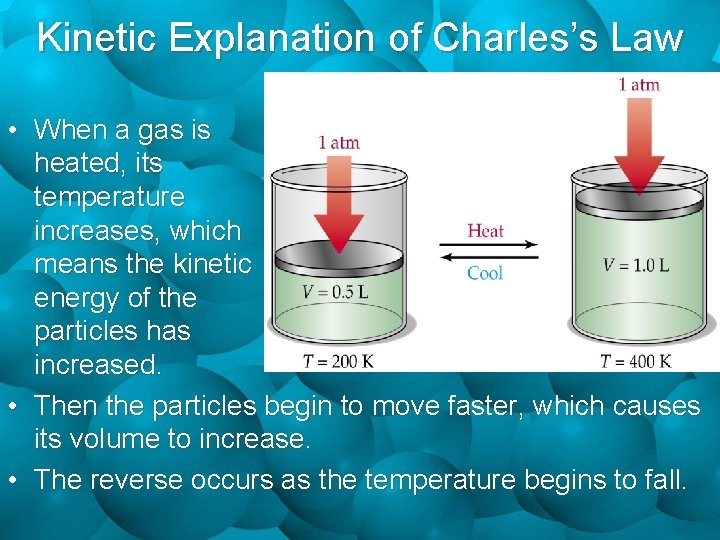
Kinetic Explanation of Charles’s Law • When a gas is heated, its temperature increases, which means the kinetic energy of the particles has increased. • Then the particles begin to move faster, which causes its volume to increase. • The reverse occurs as the temperature begins to fall.

Practice • 3. 0 L of Helium gas is in a balloon at 22 C and a pressure of 760 mm Hg. If the temperature rises to 31 C and the pressure remains constant, what will the new volume be? (remember to convert any temperatures to KELVIN!!!) V 1 V 2 = T 1 T 2 3. 0 L V 2 = (273 + 22 C) (273 + 31 C ) V 2 = (3. 0 L x 304 K) / 295 K V 2 = 3. 1 L

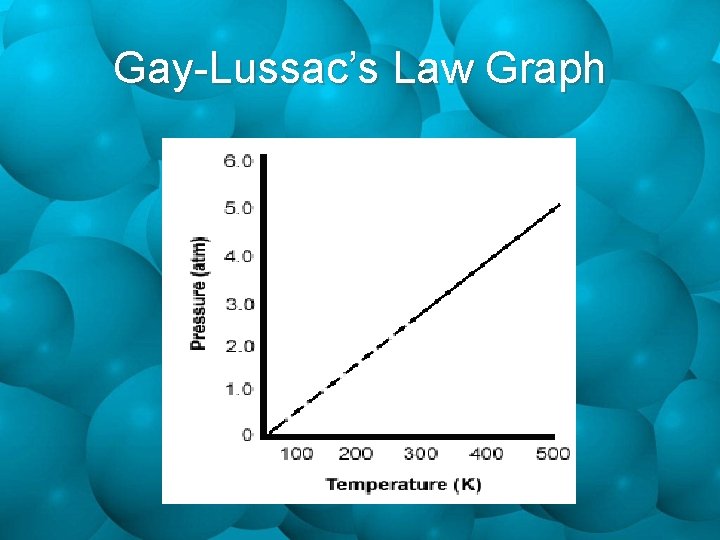
Pressure & Temperature • From the prior relationships of volume & pressure, and temperature & volume, it could be concluded that a relationship exists between pressure & temperature. For a give n mass of a dry gas, if the volume is constant, the press ure is directly pr oportiona l to the Kelvin tem perature Pressure & Temperature are Directly Proportional!

Gay-Lussac’s Law Graph

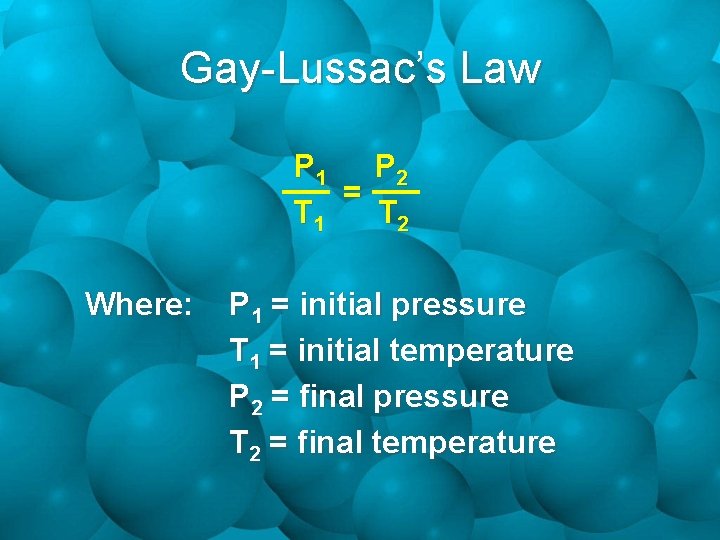
Gay-Lussac’s Law P 1 P 2 = T 1 T 2 Where: P 1 = initial pressure T 1 = initial temperature P 2 = final pressure T 2 = final temperature

Practice • At 27 C, Helium gas is in a balloon at pressure of 760 mm Hg. If the temperature rises to 31 C, what will the new pressure be? (remember to convert any temperatures to KELVIN!!!) P 1 P 2 T 1 = T 2 760 mm Hg = P 2 (273 + 27 C) (273 + 31 C ) P 2 = (760 mm Hg x 304 K) / 300 K P 2 = 770 mm Hg

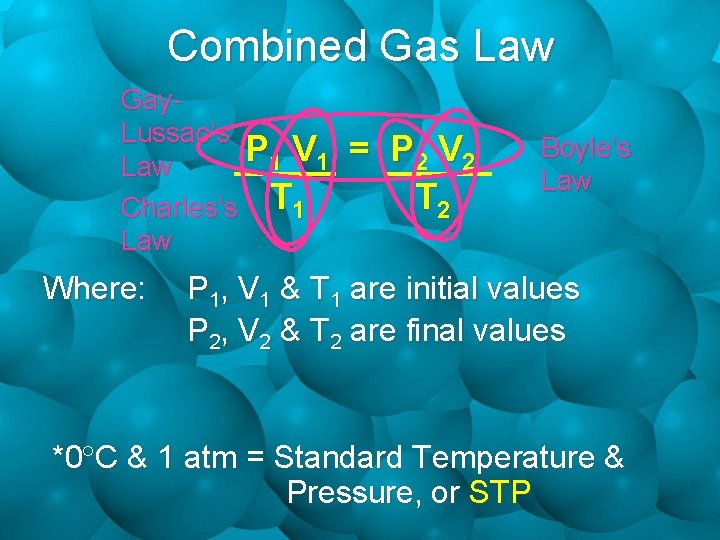
Combined Gas Law • All 3 Gas Laws require one variable to be held constant. • How can we solve a problem when all 3 variables; volume, pressure & temperature change? • Since 2 out of the 3 laws always have a variable in common, there should be a way to relate these laws into one formula. • This new formula is called the Combined Gas Law.

Combined Gas Law Gay. Lussac’s P V 1 1 Law Charles’s T 1 Law Where: = P 2 V 2 T 2 Boyle’s Law P 1, V 1 & T 1 are initial values P 2, V 2 & T 2 are final values *0 C & 1 atm = Standard Temperature & Pressure, or STP

Practice • 154 m. L of Carbon Dioxide gas is at a pressure of 121 k. Pa and a temperature of 117 C. What volume would this gas occupy at STP? (Remember to convert your temps to Kelvin!!!) 1 atm = 101. 3 k. Pa P 1 V 1/ T 1 = P 2 V 2/ T 2 (154 m. L)(121 k. Pa) = (101. 3 k. Pa)(V 2) (117 C + 273) (0 C + 273) V 2 = (154 m. L)(121 k. Pa)(273 K) (390 K)(101. 3 k. Pa) V 2 = 129 m. L

Chapter 11 Gases 11. 3 Gas Volumes & the Ideal Gas Law

The Law of Combining Gas Volumes • If one volume of water, H 2 O, is decomposed, one volume of oxygen will be formed and 2 volumes of hydrogen will be formed. • How can 3 volumes be formed from only 1 initial volume? 1 L H 2 + 1 L H 2 O 1 L H 2 + 1 LO 2

The Law of Combining Gas Volumes The law of combining volumes states that in chemical reactions involving gases, the ratio of the gas volumes is a small whole number. All of the gases are at the same temperature & pressure, each of the identical flasks contains the same number of molecules. Notice how the combining ratio: 2 volumes H 2 : 1 volume O 2 : 2 volumes H 2 O leads to a result in which all the atoms present initially are accounted for in the product.

The Law of Combining Gas Volumes • Avogadro was the first to study this and concluded a water molecule is composed of particles. • We now know that a water molecule is composed of 2 hydrogen atoms & 1 oxygen atom. When a molecule of water breaks down, it breaks down according to the ratio of particles that compose it; 2 volumes of H 2 & 1 volume of O 2 from 1 volume of H 2 O.

The Law of Combining Gas Volumes My princ that equ iple states al volum es of gases at temp & pthe same contain reessure qual numbers of particl es. • He reasoned that the volume of a gas depends on the number of gas particles, provided the temperature & pressure are constant.

The Law of Combining Gas Volumes • Under the same conditions of temperature and pressure, the volumes of reacting gases and their gaseous products are expressed in ratios of small whole numbers 2 L H 2 + 1 L O 2 → 2 L H 2 O (g) 2 volumes H 2 + 1 volume O 2 → 2 volumes H 2 O (g) 1 volume H 2 + 1 volume Cl 2 → 2 volumes HCl 1 volume HCl + 1 volume NH 3 → NH 4 Cl (s)

Avogadro’s Law For a gas at constant temperature and pressure, the volume is directly proportional to the number of moles of gas (at low pressures). V = an a = proportionality constant V = volume of the gas n = number of moles of gas

Standard Molar Volume Equal volumes of all gases at the same temperature and pressure contain the same number of molecules. - Amedeo Avogadro

Standard Molar Volume

Practice • You are planning an experiment that requires 0. 0580 mol of nitrogen monoxide gas at STP. What volume would you need? 0. 0580 mol x 22. 4 L = 1. 30 L 1 mol

Gas Stoichiometry Volume-Volume Calculations • Assume: All products and reactants are at the same temp and pressure • Unless otherwise stated, assume STP • Solve by normal stoichiometric processes • Volume ratios are the same as mole ratios

Volume-Mass and Mass-Volume Calculations • Order of Calculations • You are given a gas volume and asked to find a mass: gas volume A →moles B → mass B • You are given a mass and asked to find a gas volume: mass A → moles A →moles B →gas volume B

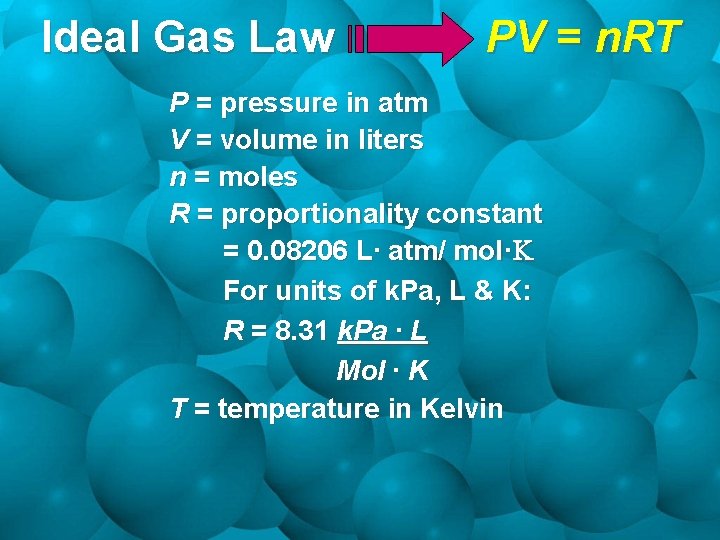
Ideal Gas Law PV = n. RT P = pressure in atm V = volume in liters n = moles R = proportionality constant = 0. 08206 L∙ atm/ mol·K For units of k. Pa, L & K: R = 8. 31 k. Pa ∙ L Mol ∙ K T = temperature in Kelvin

Calculate the Value of R • Use all standard values! P = 1 atm V = 22. 4 L n = 1 mole T = 273 K • Try substituting different standard pressures to obtain different values of R

Practice • A 2. 07 L cylinder contains 2. 88 mol of helium gas at 22. 0 °C. What is the pressure in atmospheres of the gas in the cylinder? PV = n. RT P = n. RT V = 2. 88 mol x 0. 0821 (atm∙L/mol∙K) x 295 K 2. 07 L = 33. 7 atm

Gas Density so at STP…

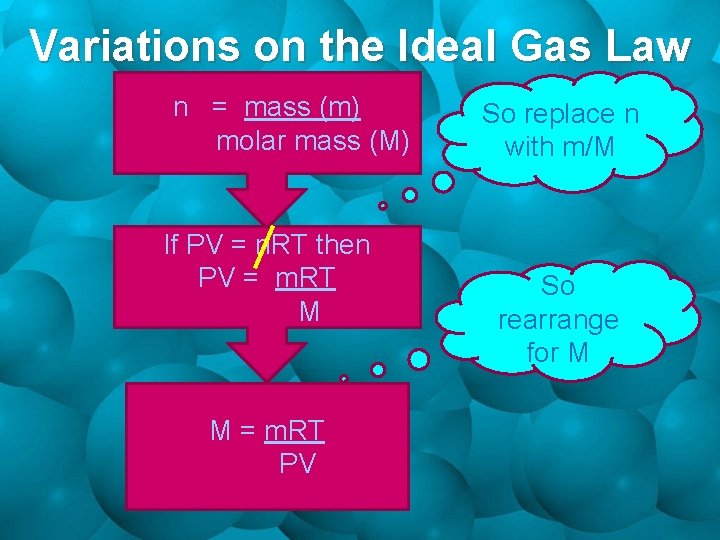
Variations on the Ideal Gas Law n = mass (m) molar mass (M) If PV = n. RT then PV = m. RT M M = m. RT PV So replace n with m/M So rearrange for M

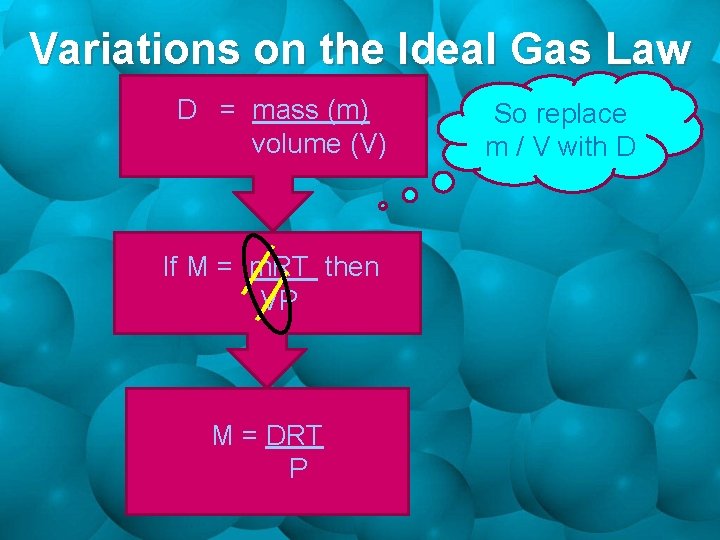
Variations on the Ideal Gas Law D = mass (m) volume (V) If M = m. RT then VP M = DRT P So replace m / V with D

Density and the Ideal Gas Law Combining the formula for density with the Ideal Gas law, substituting and rearranging algebraically: M = Molar Mass P = Pressure R = Gas Constant T = Temperature in Kelvin

Practice 1) At 28°C and 0. 974 atm, 1. 00 L of a gas has a mass of 131 g/mol 5. 16 g. What is the molar mass of this gas? 2) What is the molar mass of a gas if 0. 427 g of the gas 83. 8 g/mol occupies a volume of 125 m. L at 20. 0°C and 0. 980 atm? 3) What is the density of a sample of ammonia gas if the 0. 572 g/L pressure is 0. 928 atm and the temp is 63. 0°C? 4) The density of a gas was found to be 2. 0 g/L at 1. 50 atm 33 g/mol and 27°C. What is the molar mass of the gas? 5) What is the density of argon gas at a pressure of 551 torr 1. 18 g/L and a temp of 25°C?

Chapter 11 Gases 11. 4 Diffusion & Effusion

Effusion: describes the passage of gas into an evacuated chamber.

Graham’s Law Rates of Effusion and Diffusion Effusion: Diffusion:

Graham’s Law • Density can replace molar mass in Graham’s formula, since density is directly proportional to molar mass. • Isotopes of elements can be separated by vaporizing the element, and allowing it to effuse. • The heavier isotope effuses more slowly than the lighter isotope

Practice • At 25 °C, the average velocity of oxygen molecules is 420 m/s. What is the average velocity of helium atoms at the same temperature? Rate of O 2 is 420 m/s = √MHe Rate of He √MO 2 420 m/s = √ 4 g/mol x √ 32 g/mol 420 m/s = 0. 3536 x 1 x = 1188 m/s ≈ 1200 m/s