Chapter 11 Downstream Processing Northeast Biomanufacturing Center and

Chapter 11 -Downstream Processing Northeast Biomanufacturing Center and Collaborative

Downstream Processing Goals The overall goal of the downstream purification process is to produce a drug with required purity, efficacy, and reasonable cost per unit Starts with harvest from bioreactor and finishes with a highly purified and appropriately concentrated product ready for final formulation and packaging 3 stages – Capture or Recovery – separation of the product of interest from the cells of the bioreactor – Intermediate Purification – removal of bulk contaminants (host cell proteins, adventitious viruses, traces of DNA, endotoxin) – Polishing – elimination of trace contaminants and impurities (inactive or unwanted isoforms of the target molecule, common impurities)

Downstream Processing in Biopharmaceutical Manufacturing § Harvest by Centrifugation § Clarification by Depth Filtration § Sterile Filtration (MF) § Tangential Flow Filtration (UF/DF) § Low Pressure Liquid Column Chromatography
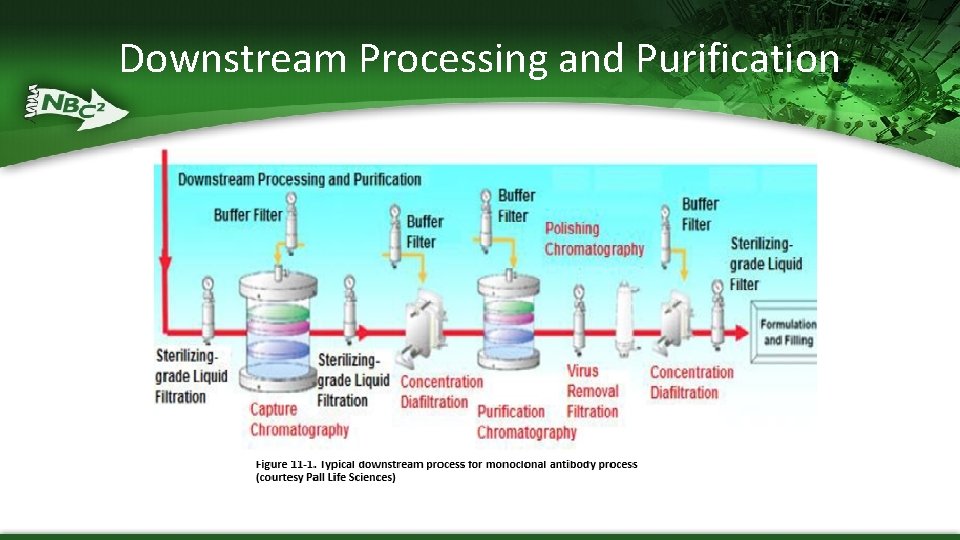
Downstream Processing and Purification

Process Filtration § Used at several stages of the downstream process § Use membrane filters to separate the protein of interest from other proteins and molecules in the sterile filtered, clarified medium. 3 General Types – Microfiltration – used at the start of the downstream processing – Ultrafiltration – used between chromatography steps to concentrate the products and change the buffer conditions (diafiltration) – Sterilizing grade direct flow filtration – eliminates microbial organisms and insoluble proteins, removes adventitious and endogenous viruses, and sterile filters the product for final formulation

Process Filtration- Flow Types Two general flow types: § Direct Flow Filtration (DFF) – process fluid crosses the membrane in a perpendicular flow direction – provides little or no prevention of particulate build up or concentration of other elements that do not fit through the pore structure – Often used for particulate removal, sterile filtration, and virus removal/clearance filtration § Tangential Flow Filtration (TFF) –flow sweeps across the active filtration surface – minimizes pore plugging and surface fouling

Direct Flow Filtration • • • Random Internal plugging Dramatic flux decline over time
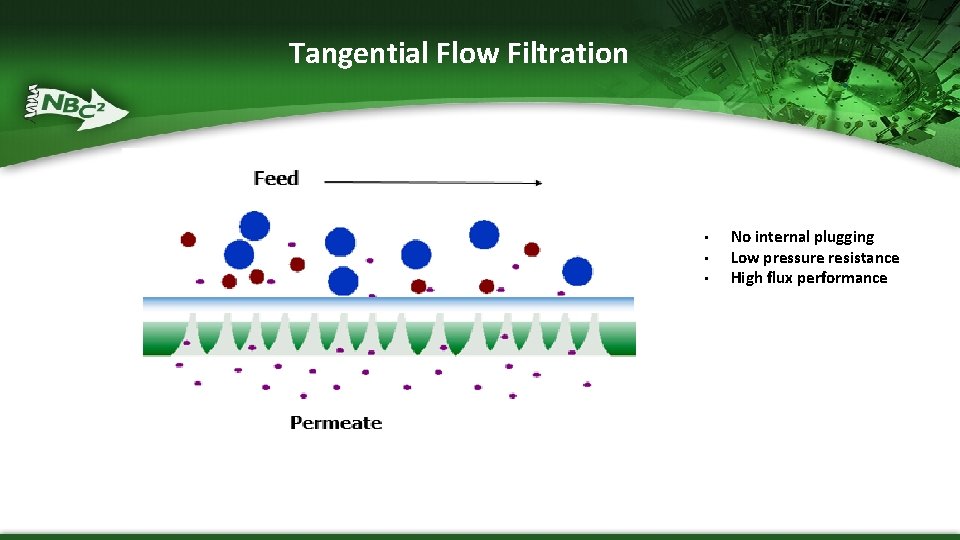
Tangential Flow Filtration • • • No internal plugging Low pressure resistance High flux performance
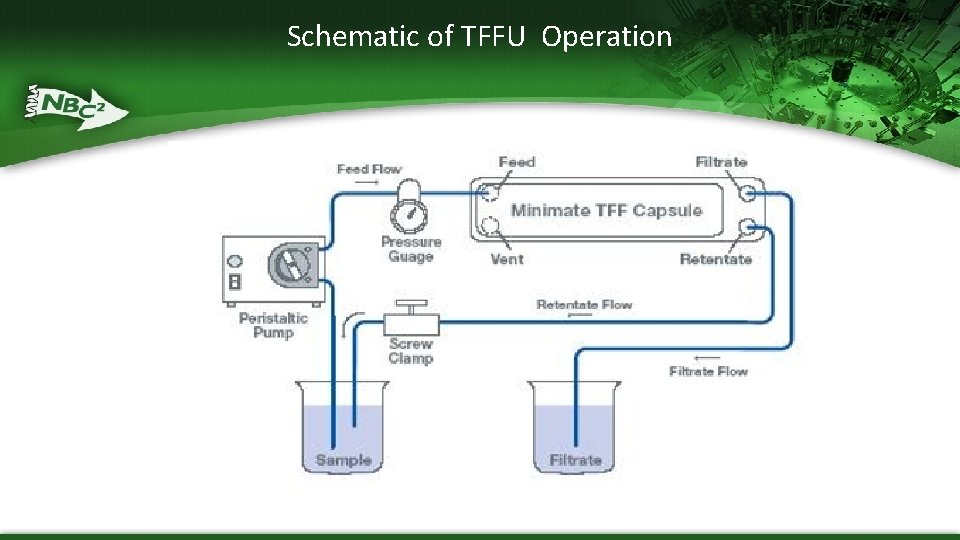
Schematic of TFFU Operation

Direct Filtration Systems

Tangential Flow Filtration Units

Downstream Processing – Liquid Chromatography – Separation techniques based on the differential interaction of molecules between a stationary and a mobile phases – In Greek "Chroma" means "color“ and "Graphein" means "to write” – Some constituents bind or interact with the stationary phase (packed resin made up with immobile matrix such as cellulose or agarose, synthetic polymers and silica) while others pass through the stationary phase with no interaction – Molecules move past stationary phase in different rates based on their difference in their relative attraction to the mobile phase – The bound or interacting components are then removed or eluted by the gradual or step-wise change of the composition of the mobile phase (liquid buffer or gas that moves pass the stationary phase).

Chromatography System Components – Columns containing stationary phase – Mobile phase pumps • Enable a wide selection of process fluids and/or proportional binary mixing to adjust conditions of conductivity and p. H – Series of pre- and post-column sensors • UV-Detects proteins coming out of the column by measuring absorbance at 280 nm • conductivity measures the amount of salt in the buffers coming out of the columns – high salt or low salt are often used to elute the protein of interest from the chromatography beads • p. H- measures the p. H during binding and elution • flow rate – monitors rate of flow of mobile phase pumps
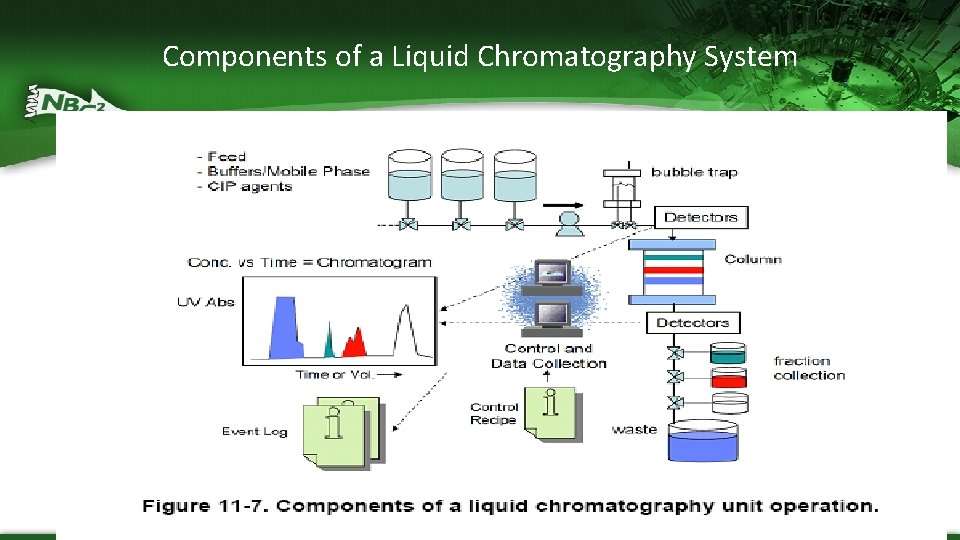
Components of a Liquid Chromatography System

Types of Chromatography – Bind-elute methods – product binds to resin and bound components removed (eluted) by the gradual or step-wise change of the composition of the mobile phase (buffers) – Flow-Through methods – product passes through while unwanted elements are bound to resin
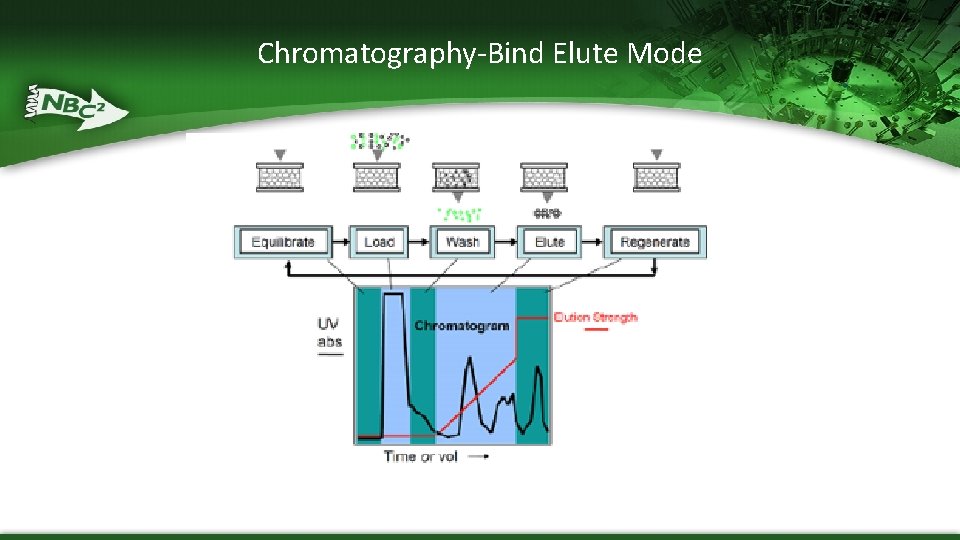
Chromatography-Bind Elute Mode
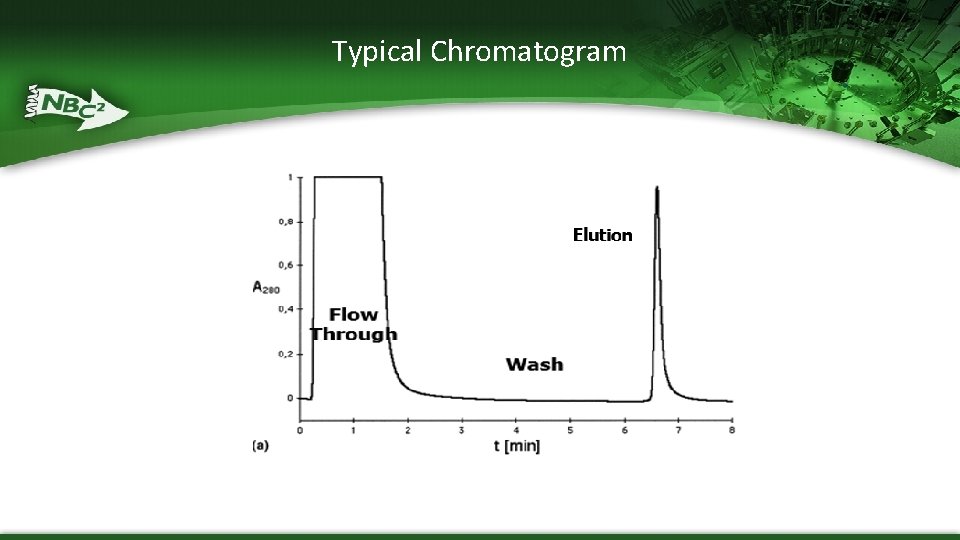
Typical Chromatogram

Liquid Column Chromatography Process Ø PURGE air from Column use Equilibration Buffer Ø PACK column with Beads (e. g. ion exchange, HIC, affinity or gel filtration beads/media Ø EQUILIBRATE column with Equilibration Buffer Ø LOAD column with Protein of Interest in Equilibration Buffer Ø WASH column with Equilibration Buffer Ø ELUTE protein of Interest with Elution Buffer of High or Low Salt or p. H Ø REGENERATE column or Clean and Store (Na. OH)
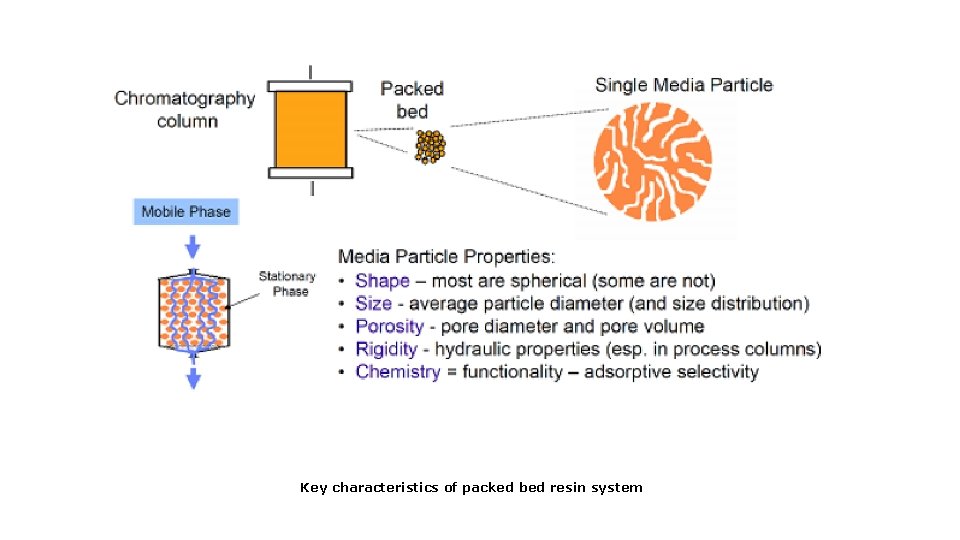
Key characteristics of packed bed resin system

Chromatography- Stationary Phases § Stationary phase (resin) functional groups – Charged – Specific biochemical make-up – Hydrophobic – Combination of above (mixed mode)

Types of Column Media/ Resins • Hydrophobic Interaction (HIC) • Ion Exchange (anion- AEX and cation exchange -CEX) • Affinity • Gel Filtration (Size Exclusion)

Column Qualification § When packing columns, flow of the resin and pressure should be controlled and monitored § After column is packed, integrity and efficiency of column bed should be determined § Efficiency calculated from number of theoretical plates (N) and height equivalent of theoretical plates (HETP) – HETP normalizes the column of the column to length of the column
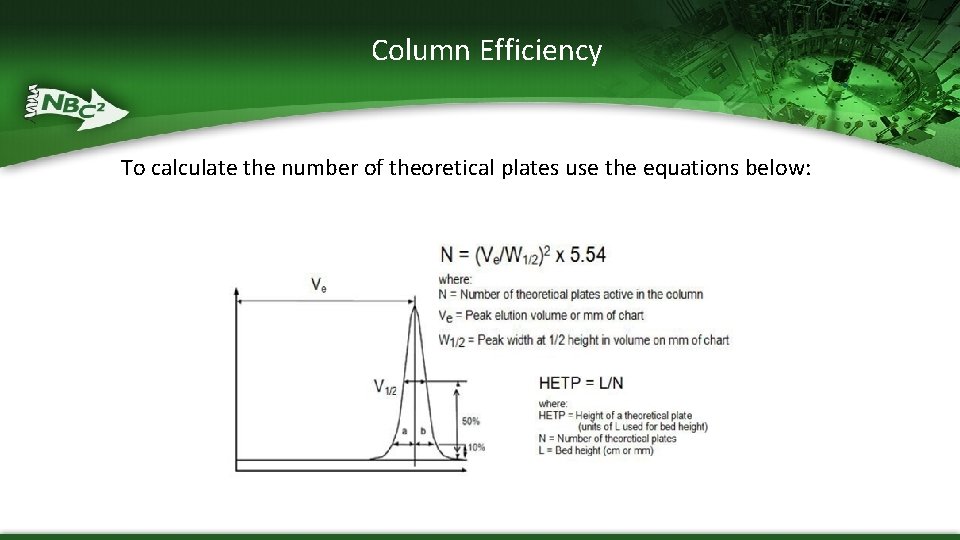
Column Efficiency To calculate the number of theoretical plates use the equations below:

Validation /Qualification of Solvents and Media Sterilizing grade filter compatibility studies on filtration devices • Spike a sample of the drug product with bacteria or bacterial spores at various stages in the process to demonstrate microbial clearance as well as the maintenance of the drugs’ chemical stability and biological activity Detection and quantification of leaching ligand during affinity chromatography to prove the drug substance is free of leaching ligand such as protein A Validation of contaminant removal • spike and track a known and excessive quantity of the contaminant through the downstream process

Viral Clearance and Inactivation – According to FDA, viral clearance on the final product will not only be from direct testing for their presence, but also from demonstration that the purification regimen is capable of removing and inactivating the viruses – For each downstream processing step, the effectiveness of viral removal or inactivation must be validated using virus challenge or spiking studies where model viruses are added at known high titers than tracked through each step of the process typically via infectivity assay – For m. Ab purification processes, a low p. H exposure at p. H 2. 5 - 4. 0 such as the elution buffer of the protein A column combined with a viral filtration is accepted by regulatory agencies as a virus clearance method.
- Slides: 25