Chapter 10 Thermal Physics Temperature Thermodynamics branch of

Chapter 10 Thermal Physics

Temperature • Thermodynamics – branch of physics studying thermal energy of systems • Temperature (T), a scalar – measure of thermal (internal) energy of a system • SI unit: K (Kelvin) • Kelvin scale has a lower limit (absolute zero) and has no upper limit William Thomson (Lord Kelvin) (1824 - 1907)

Kelvin scale • Kelvin scale is defined by the temperature of the triple point of pure water • Triple point – set of pressure and temperature values at which solid, liquid, and gas phases can coexist • International convention: T of the triple point of water is

The zeroth law of thermodynamics • If two (or more) bodies in contact don’t change their internal energy with time, they are in thermal equilibrium • 0 th law of thermodynamics: if bodies are in thermal equilibrium, their temperatures are equal

Measuring temperature • Temperature measurement principle: if bodies A and B are each in thermal equilibrium with a third body C, then A and B are in thermal equilibrium with each other (and their temperatures are equal) • The standard temperature for the Kelvin scale is measured by the constant-volume gas thermometer
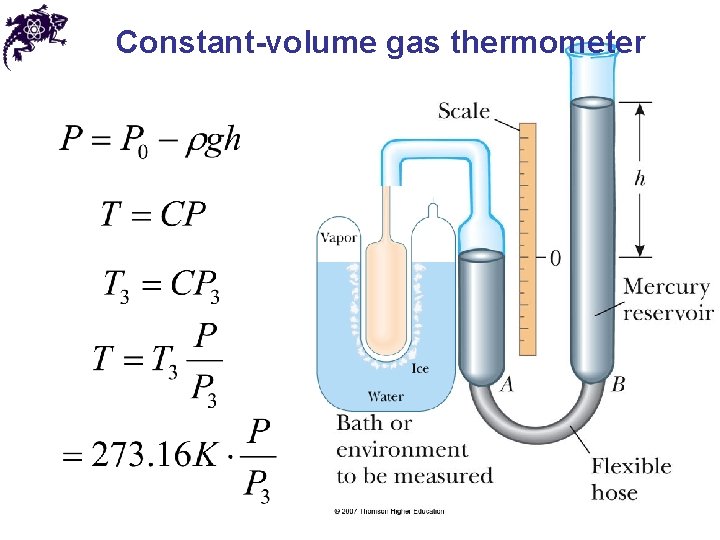
Constant-volume gas thermometer

Celsius and Fahrenheit scales • Celsius scale: • Fahrenheit scale: Anders Cornelius Celsius (1701 - 1744) Gabriel Daniel Fahrenheit (1686 - 1736)

Chapter 10 Problem 3 Convert the following temperatures to their values on the Fahrenheit and Kelvin scales: (a) the boiling point of liquid hydrogen, – 252. 87°C; (b) the temperature of a room at 20°C.

Thermal expansion • Thermal expansion: increase in size with an increase of a temperature • Linear expansion: • Volume expansion:
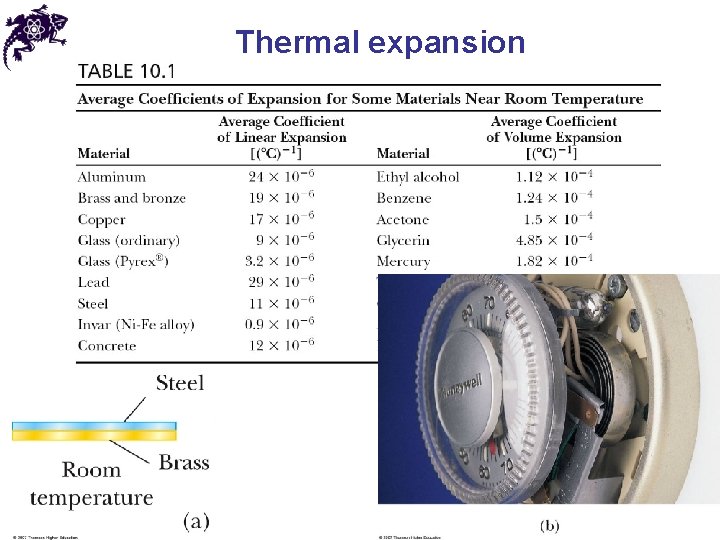
Thermal expansion

Chapter 10 Problem 14 A cube of solid aluminum has a volume of 1. 00 m 3 at 20°C. What temperature change is required to produce a 100 -cm 3 increase in the volume of the cube?

Temperature and heat • Heat (Q): energy transferred between a system and its environment because of a temperature difference that exists between them • SI Unit: Joule • Alternative unit: calorie (cal):

Avogadro’s number • Mole – amount of substance containing a number of atoms (molecules) equal to the number of atoms in a 12 g sample of 12 C • This number is known as Avogadro’s number (NA): NA = 6. 02 x 1023 mol -1 • The number of moles in a sample Amedeo Avogadro (1776 -1856) N – total number of atoms (molecules) m – total mass of a sample, m 0 – mass of a single atom (molecule); M – molar mass
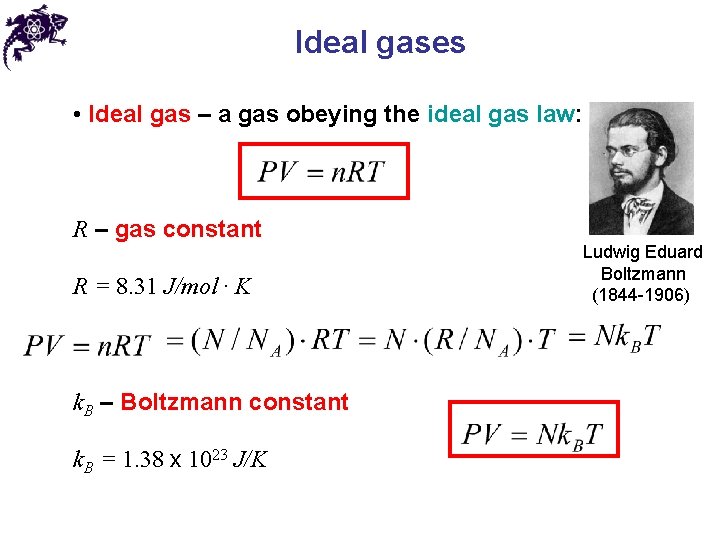
Ideal gases • Ideal gas – a gas obeying the ideal gas law: R – gas constant R = 8. 31 J/mol ∙ K k. B – Boltzmann constant k. B = 1. 38 x 1023 J/K Ludwig Eduard Boltzmann (1844 -1906)

Ideal gases • The gas under consideration is a pure substance • All molecules are identical • Macroscopic properties of a gas: P, V, T • The number of molecules in the gas is large, and the average separation between the molecules is large compared with their dimensions – the molecules occupy a negligible volume within the container • The molecules obey Newton’s laws of motion, but as a whole they move randomly (any molecule can move in any direction with any speed)

Ideal gases • The molecules interact only by short-range forces during elastic collisions • The molecules make elastic collisions with the walls and these collisions lead to the macroscopic pressure on the walls of the container • At low pressures the behavior of molecular gases approximate that of ideal gases quite well
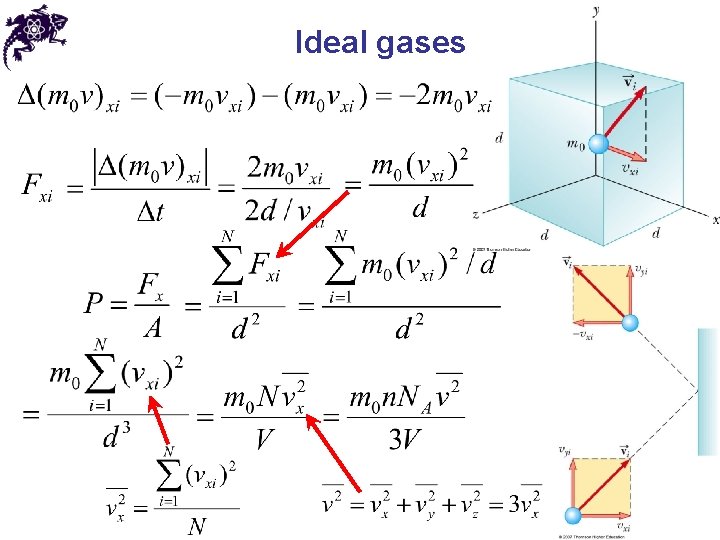
Ideal gases
Ideal gases • Root-mean-square (RMS) speed:
Translational kinetic energy • Average translational kinetic energy: • At a given temperature, ideal gas molecules have the same average translational kinetic energy • Temperature is proportional to the average translational kinetic energy of a gas

Internal energy • For the sample of n moles, the internal energy: • Internal energy of an ideal gas is a function of gas temperature only

Chapter 10 Problem 30 A tank having a volume of 0. 100 m 3 contains helium gas at 150 atm. How many balloons can the tank blow up if each filled balloon is a sphere 0. 300 m in diameter at an absolute pressure of 1. 20 atm?
Distribution of molecular speeds • Not all the molecules have the same speed • Maxwell’s speed distribution law: James Clerk Maxwell (1831 -1879) NvΔv – fraction of molecules with speeds in the range from v to v + Δv
Distribution of molecular speeds • Average speed: • RMS speed: • Most probable speed:

Questions?

Answers to the even-numbered problems Chapter 10 Problem 28 (a) 3. 0 mol (b) 1. 80 × 1024 molecules

Answers to the even-numbered problems Chapter 10 Problem 42 3. 34 × 105 Pa
- Slides: 26