Chapter 10 Section 1 Be able to name

Chapter 10

Section 1 • Be able to name cations and anions • Indicate the charge an ion will likely take from elements of group A • Indicate how you identify the charge of a transition metal.

Section 2 • Be able to name and write formulas for binary ionic compounds and ternary ionic compounds. • Criss-Cross Rule • Polyatomic ions are on the back of the periodic table.

Example: Write the formula for the following: 1. Magnesium ion reacting with Fluoride ion Mg. F 2 __________ 2. Aluminum ion reacting with Sulfite ion Al 2 SO 3 ___________ 3. Boron ion reacting with Sulfide ion B 2 S 3 _____________ 4. Calcium ion reacting with Bromide Ca. Br 2 _______________
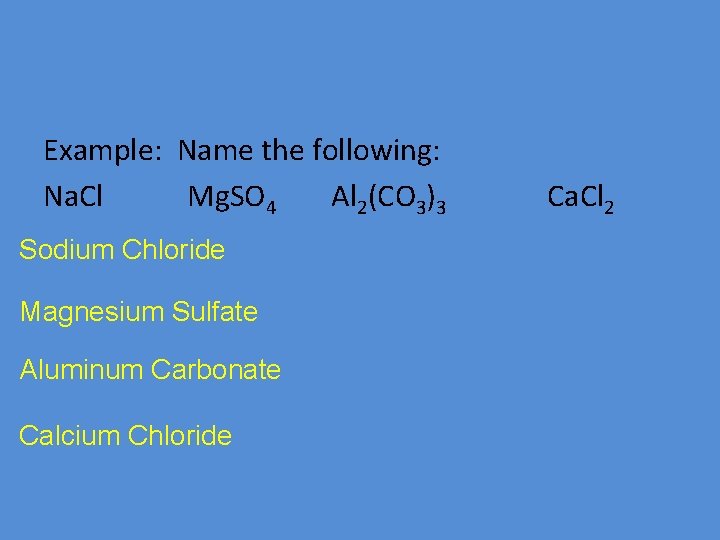
Example: Name the following: Na. Cl Mg. SO 4 Al 2(CO 3)3 Sodium Chloride Magnesium Sulfate Aluminum Carbonate Calcium Chloride Ca. Cl 2

Section 3 • Be able to name and write the formula for a binary molecular compound • 2 non-metallic elements
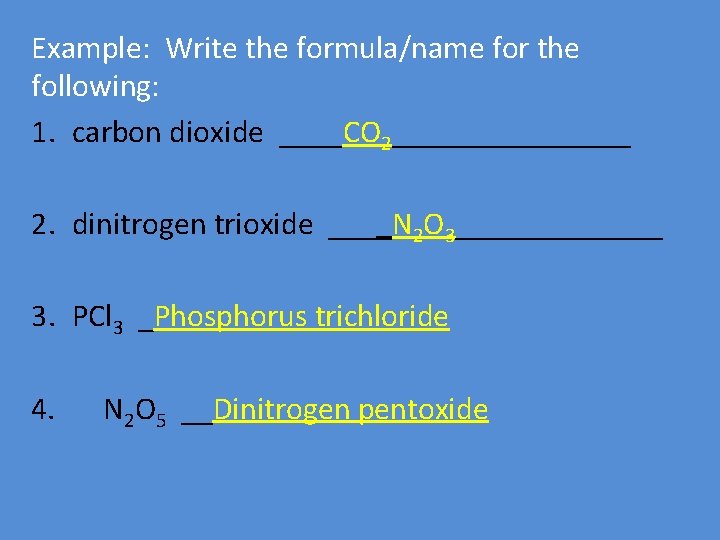
Example: Write the formula/name for the following: 1. carbon dioxide ____CO 2________ 2. dinitrogen trioxide ____N 2 O 3_______ 3. PCl 3 _Phosphorus trichloride 4. N 2 O 5 __Dinitrogen pentoxide

• Section 4 • Be able to name acids and write the formula if given the name • Example: Name/write formula for the following: – H 2 SO 3 __Sulfurous Acid – HNO 2 ___Nitrous Acid – Hydrobromic acid __HBr – Nitric Acid __HNO 3

Q 1 • Calculate the molar mass of the following compounds: a) Na. OH - 40 g/mol b) Au. Cl 3 – 303. 33 g/mol c) (NH 4)2 SO 4 – 132. 14 g/mol

Q 2 • Convert 3. 4 moles of NH 4 into grams of NH 4. 3. 4 mole of NH 4 18. 05 g of NH 4 1 mol of NH 4 = 61. 37 g
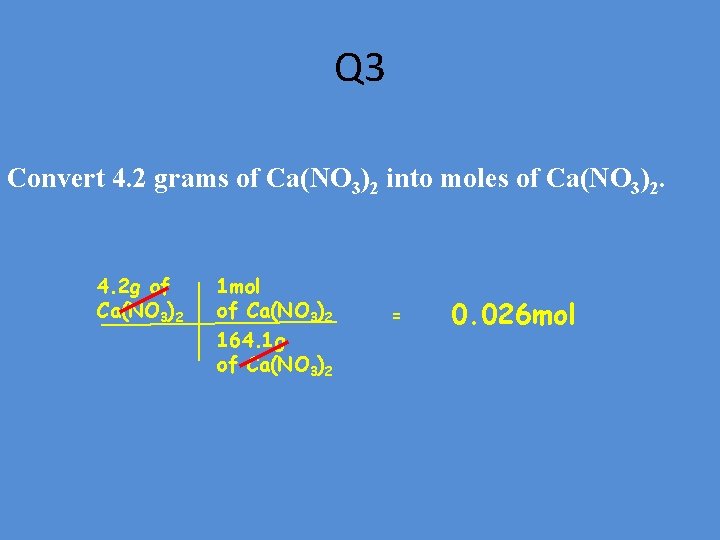
Q 3 Convert 4. 2 grams of Ca(NO 3)2 into moles of Ca(NO 3)2. 4. 2 g of Ca(NO 3)2 1 mol of Ca(NO 3)2 164. 1 g of Ca(NO 3)2 = 0. 026 mol
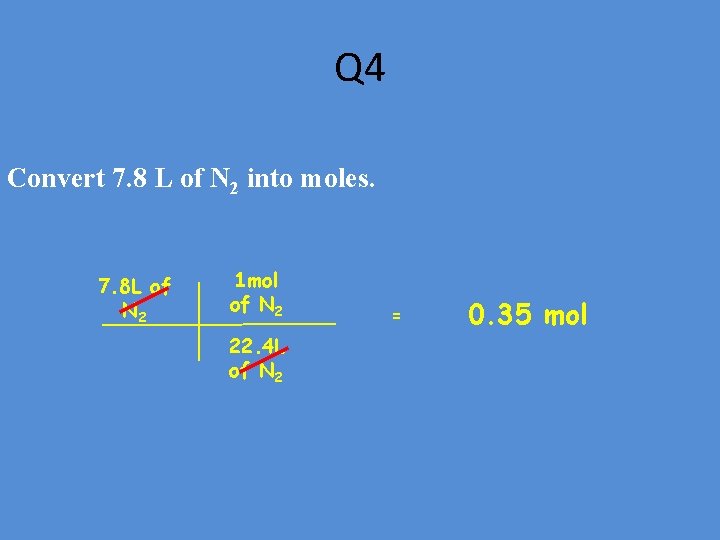
Q 4 Convert 7. 8 L of N 2 into moles. 7. 8 L of N 2 1 mol of N 2 22. 4 L of N 2 = 0. 35 mol
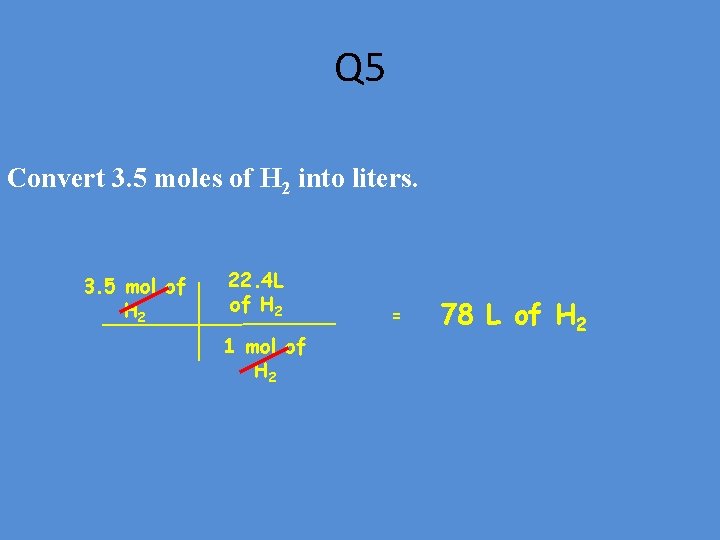
Q 5 Convert 3. 5 moles of H 2 into liters. 3. 5 mol of H 2 22. 4 L of H 2 1 mol of H 2 = 78 L of H 2
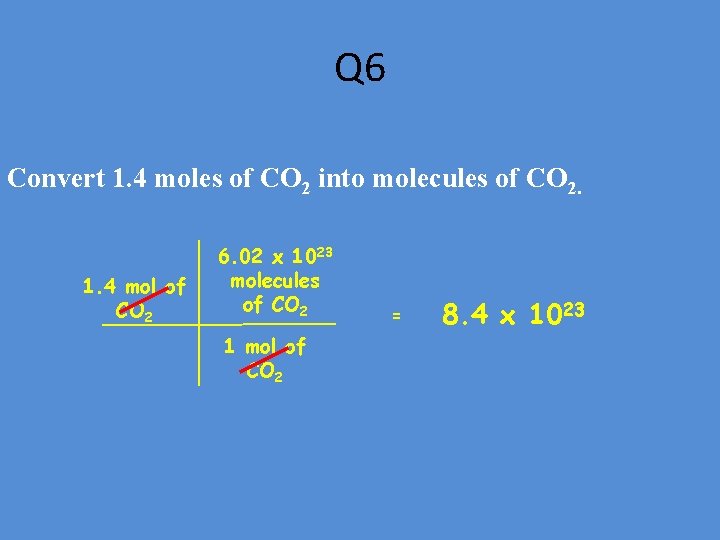
Q 6 Convert 1. 4 moles of CO 2 into molecules of CO 2. 1. 4 mol of CO 2 6. 02 x 1023 molecules of CO 2 1 mol of CO 2 = 8. 4 x 1023
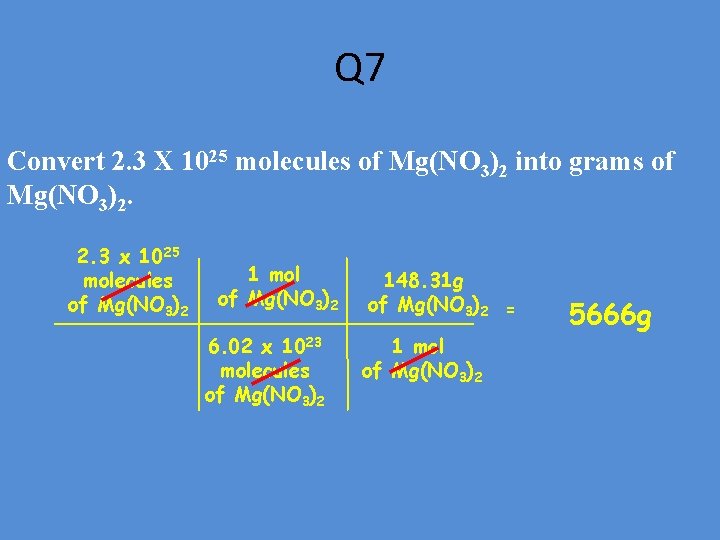
Q 7 Convert 2. 3 X 1025 molecules of Mg(NO 3)2 into grams of Mg(NO 3)2. 2. 3 x 1025 molecules of Mg(NO 3)2 1 mol of Mg(NO 3)2 6. 02 x 1023 molecules of Mg(NO 3)2 148. 31 g of Mg(NO 3)2 = 1 mol of Mg(NO 3)2 5666 g

Q 8 What is the percent composition of sulfur in Al 2(SO 4)3? Part (3 x molar mass of S) X 100% Whole (molar mass of Al 2(SO 4)3) 96. 18 g 342. 15 g X 100% = % Comp = 28. 1% S

Q 9 What is the percent composition of carbon in pentane, C 5 H 12? Part (5 x molar mass of C) Whole (molar mass of C 5 H 12) 60. 05 g 72. 15 g X 100% = % Comp = 83. 23% C

Q 10 Find the molecular formula of ethylene glycol, which is used as antifreeze. The molar mass is 62 g/mol and the empirical formula is CH 3 O. Molecular Formula = Step 1: Find the Empirical Molar Mass of CH 3 O 12. 01 + (3 * 1. 01) + 16 = 31. 01 Step 2: Molecular Molar mass divided by Empirical molar mass – 62 =2 31. 04 Step 3: Multiply the Empirical formula by the answer in 2 – 2 (CH 3 O) = C 2 H 6 O 2

Chapter 11
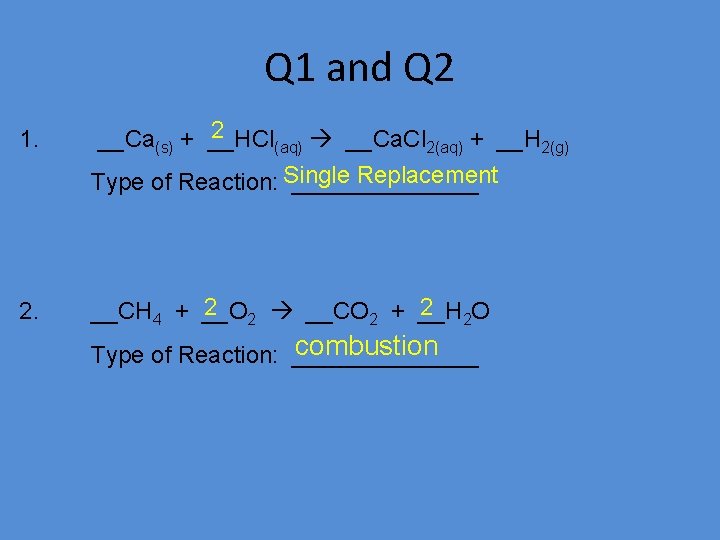
Q 1 and Q 2 1. 2 __Ca(s) + __HCl (aq) __Ca. Cl 2(aq) + __H 2(g) Replacement Type of Reaction: Single _______ 2. 2 2 __CO 2 + __H 2 2 O __CH 4 + __O combustion Type of Reaction: _______

Q 3 and Q 4 3. 2 2 __Mg + __Ag. NO __Ag + __Mg(NO 3)2 3 Replacement Type of Reaction: Single _______ 4. 2 __H 2 O 2 2 __H 2 O + __O 2 Decomposition Type of Reaction: _______

Q 5 and Q 6 5. nitrogen trifluoride nitrogen + fluorine 2 3 F ___ NF 3 ___ N 2 + ___ 2 Decomposition Type of Reaction: _______ 6. sulfur trioxide sulfur and oxygen 2 SO ___ 2 S + ___ 3 O ___ 3 2 Decomposition Type of Reaction: _______
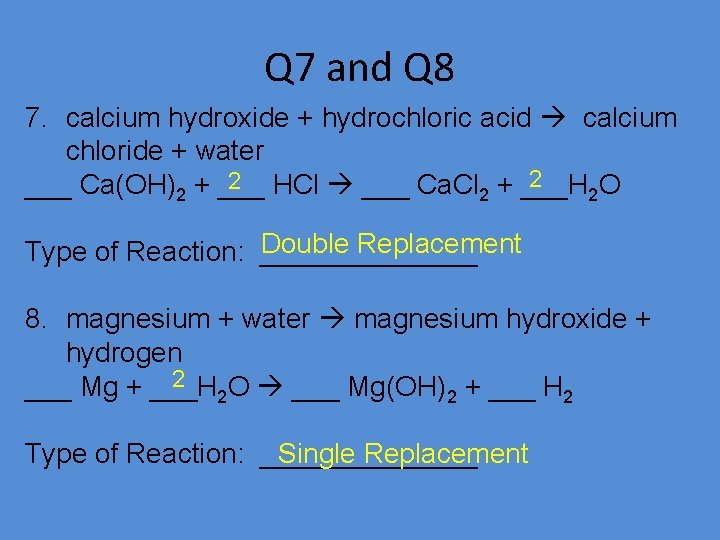
Q 7 and Q 8 7. calcium hydroxide + hydrochloric acid calcium chloride + water 2 2 HCl ___ Ca. Cl + ___H ___ Ca(OH)2 + ___ 2 2 O Double Replacement Type of Reaction: _______ 8. magnesium + water magnesium hydroxide + hydrogen 2 ___ Mg + ___H 2 O ___ Mg(OH)2 + ___ H 2 Type of Reaction: _______ Single Replacement
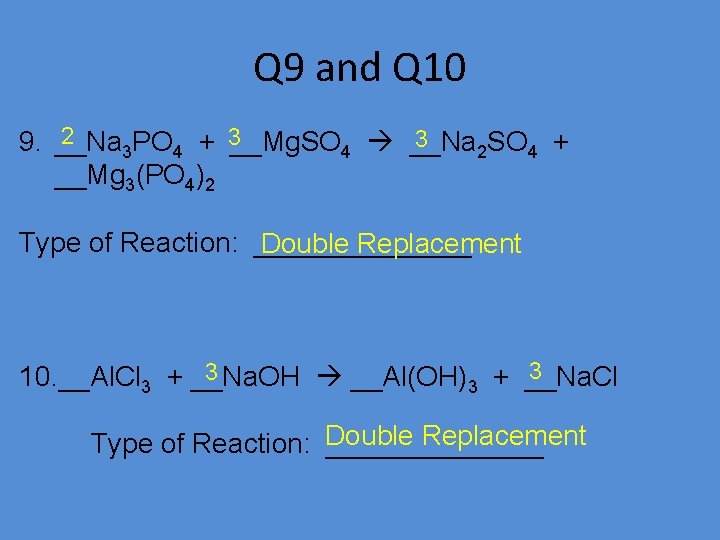
Q 9 and Q 10 2 3 3 9. __Na 3 PO 4 + __Mg. SO 4 __Na 2 SO 4 + __Mg 3(PO 4)2 Type of Reaction: _______ Double Replacement 3 3 10. __Al. Cl 3 + __Na. OH __Al(OH)3 + __Na. Cl Replacement Type of Reaction: Double _______

Q 11 and Q 12 11. _2_Al + _3_Cl 2 2 Al. Cl 3 combination reaction 12. _2_Hg. O 2 Hg + O 2 decomposition reaction

13. _2_C 4 H 10 + _13_O 2 8 CO 2 + 10 H 2 O complete combustion reaction 14. _3_Ca. SO 4 + _2_Al(NO 3 )3 3 Ca(NO 3)2 + Al 2(SO 4)3 double replacement

15. silver nitrate + sodium chloride double replacement

16. What is the activity series of metals? According to the activity series of metals found on page 333, table 11. 2, 17. which metal is the most reactive metal listed? least reactive metal?

Q 18 and Q 19 18. What metal would be the best material for use in jewelry? 19. Based on the activity series of metals, which metal would you expect to find in its elemental form?

Chapter 12

(Ch 12) Q 1 Mole-Mole Problem: According to the following equation, how many moles of Al react with 5. 2 moles of H 2 SO 4? 2 Al + 3 H 2 SO 4 Al 2(SO 4)3 + 3 H 2 5. 2 moles of H 2 SO 4 2 mol of Al 3 moles of H 2 SO 4 = 3. 47 mole

(Ch 12) Q 2 Mole-Mole Problem: According to the following equation, how many moles of oxygen are needed to form 3. 7 mol Al 2 O 3? 4 Al + 3 O 2 2 Al 2 O 3 3. 7 moles of Al 2 O 3 3 mol of O 2 2 moles of Al 2 O 3 = 5. 55 mole

(Ch 12) Q 3 Mass-Mole Problem: According to the equation in problem #2, how many moles of Al 2 O 3 are formed when 7. 8 grams of aluminum completely reacts with oxygen? 4 Al + 3 O 2 2 Al 2 O 3 7. 8 g of Al 1 mol of Al 2 O 3 26. 7 g of Al 4 mol of Al = 0. 145 mole

(Ch 12) Q 4 Mass-Mole-Mass Problem: According to the following equation, how many grams of CO 2 would be produced if 45 g of C 2 H 2 completely reacted with oxygen? 2 C 2 H 2 + 5 O 2 4 CO 2 + 2 H 2 O 45 g of C 2 H 2 1 mol C 2 H 2 26. 0 g of C 2 H 2 4 mol CO 2 44. 0 g of CO 2 2 mol C 2 H 2 1 mol CO 2 = 152. 1 g CO 2

(Ch 12) Q 5 a/b/c In the equation below 3. 4 g of Al reacts with 4. 2 g of O 2 to form aluminum oxide. a. ) What is the limiting reactant? Limiting 4 Al + 3 O 2 2 Al 2 O 3 Reagent 3. 4 g of Al 1 mol Al 27. 0 g of Al 4. 2 g of O 2 Excess Reagent 1 mol O 2 32. 0 g of O 2 2 mol Al 2 O 3 4 mol Al 2 O 3 3 mol O 2 101. 9 g Al 2 O 3 1 mol Al 2 O 3 = 6. 4 g Al 2 O 3 Theoretical Yield = 8. 9 g Al 2 O 3

(Ch 12) Q 5 d d. ) How much of the excess reactant is left over? 4 Al + 3 O 2 2 Al 2 O 3 3. 4 g of Al 1 mol Al 27. 0 g of Al 6. 4 g of Al 2 O 3 1 mol Al 2 O 3 101. 9 g of Al 2 O 3 3 mol O 2 4 mol Al 3 mol O 2 2 mol Al 2 O 3 32. 0 g O 2 1 mol O 2 32. 9 g O 2 1 mol O 2 = 3. 02 g O 2 Excess Reactant (O 2) = 4. 2 -3. 02 g = 1. 18 g

(Ch 12) Q 5 e e) If the actual yield of aluminum oxide is 1. 24 g Al 2 O 3 what is the percent yield? 4 Al + 3 O 2 2 Al 2 O 3 Actual yield (grams) Theoretical yield (grams) 1. 24 g of Al 2 O 3 6. 4 g of Al 2 O 3 X 100% = % Yield = 19. 3%
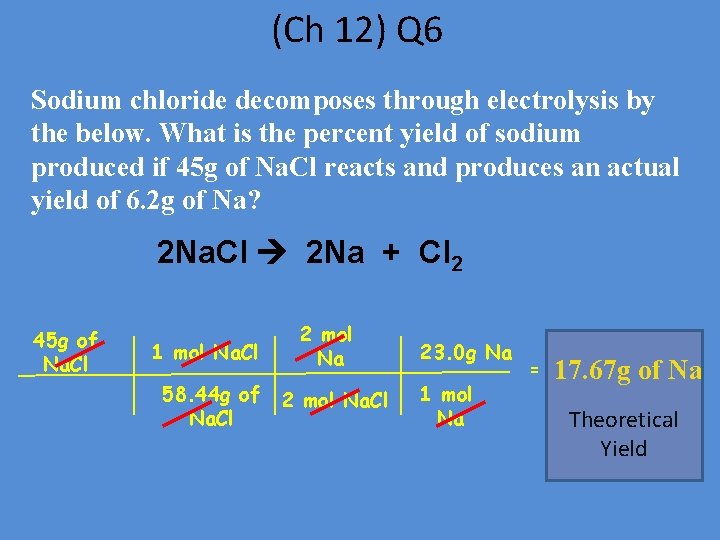
(Ch 12) Q 6 Sodium chloride decomposes through electrolysis by the below. What is the percent yield of sodium produced if 45 g of Na. Cl reacts and produces an actual yield of 6. 2 g of Na? 2 Na. Cl 2 Na + Cl 2 45 g of Na. Cl 1 mol Na. Cl 58. 44 g of Na. Cl 2 mol Na. Cl 23. 0 g Na 1 mol Na = 17. 67 g of Na Theoretical Yield
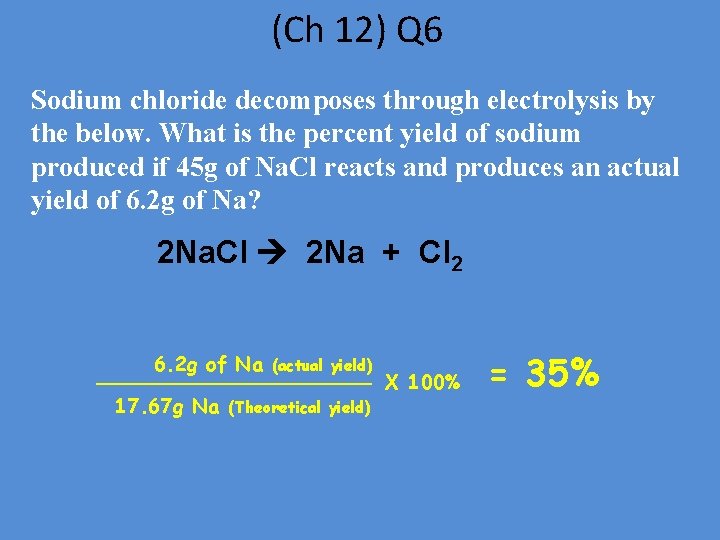
(Ch 12) Q 6 Sodium chloride decomposes through electrolysis by the below. What is the percent yield of sodium produced if 45 g of Na. Cl reacts and produces an actual yield of 6. 2 g of Na? 2 Na. Cl 2 Na + Cl 2 6. 2 g of Na 17. 67 g Na (actual yield) (Theoretical yield) X 100% = 35%
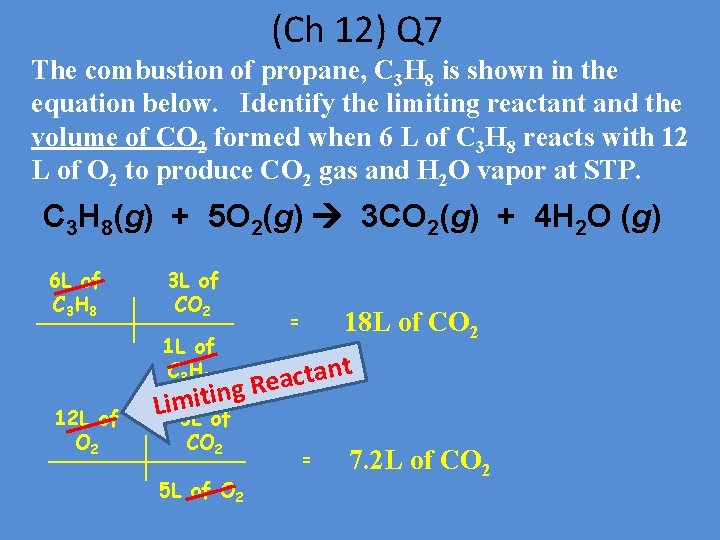
(Ch 12) Q 7 The combustion of propane, C 3 H 8 is shown in the equation below. Identify the limiting reactant and the volume of CO 2 formed when 6 L of C 3 H 8 reacts with 12 L of O 2 to produce CO 2 gas and H 2 O vapor at STP. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) 6 L of C 3 H 8 3 L of CO 2 1 L of C 3 H 8 12 L of O 2 18 L of CO 2 = t n a t c g Rea itin Lim 3 L of CO 2 5 L of O 2 = 7. 2 L of CO 2

(Ch 12) Q 8 From problem #7, how much of excess reactant is left unreacted? C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) 12 L of O 2 1 L of C 3 H 8 5 L of O 2 = 2. 4 L of C 3 H 8 needed Excess Reactant (C 3 H 8) = 6 -2. 4 = 3. 6 L

(Ch 12) Q 9 From problem #7, if the actual yield was 6. 9 L of CO 2, what is the percent yield? C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) 6. 9 L of CO 2 7. 2 L CO 2 (actual yield) (Theoretical yield) X 100% = 95. 8%

(Ch 12) Q 10 a In the reaction below, 5. 50 g of Mg reacts with 3. 25 g of O 2 to produce Mg. O. a) Balance this equation: 2 Mg + 1 O 2 2 Mg. O

(Ch 12) Q 10 b/c/d In the reaction below, 5. 50 g of Mg reacts with 3. 25 g of O 2 to produce Mg. O. b) Which is theoretical yield of Mg. O? 2 Mg + 1 O 2 2 Mg. O 5. 5 g of Mg Excess Reactant 3. 25 g of O 2 Limiting Reactant 1 mol Mg 24. 3 g of Mg 1 mol O 2 32. 0 g of O 2 2 mol Mg. O 1 mol O 2 40. 3 g Mg. O 1 mol Mg. O = 9. 12 g = Mg. O 8. 19 g Mg. O

(Ch 12) Q 10 e In the reaction below, 5. 50 g of Mg reacts with 3. 25 g of O 2 to produce Mg. O. e) How much of the excess reactant is left unreacted? 2 Mg + 1 O 2 2 Mg. O 3. 25 g of O 2 1 mol O 2 32. 0 g of O 2 2 mol Mg 1 mol O 2 24. 3 g Mg 1 mol Mg = 4. 94 g Mg Excess Reactant (Mg) = 5. 50 -4. 94= 0. 56 g

(Ch 12) Q 10 f In the reaction below, 5. 50 g of Mg reacts with 3. 25 g of O 2 to produce Mg. O. f) Calculate the percent yield of Mg. O if the actual yield was 6. 80 g Mg. O. 2 Mg + 1 O 2 2 Mg. O 6. 8 g of Mg. O 8. 19 g Mg. O (actual yield) (Theoretical yield) X 100% = 83. 0%

Chapter 14

V 1 Ch 14 Q 5 P 1 V 1 = P 2 V 2 P 1 A gas has a volume of 50 cm 3 at a pressure of 200 mm Hg. If the pressure is changed to 190 mm Hg, what is the new volume of that sample of gas? P 2 P 1 V 1 = P 2 V 2 P 1 V 1 = V 2= P 2 V 2 = 52. 6 cm 3 200 x 50 190

V 1 Ch 14 WP Q 6 V 1/T 1 = V 2/T 2 T 1 If 400 m. L of nitrogen is collected at 30 o. C, what will the new volume be if the temperature is increased to 50 o. C? T 2 V 1 V 2 = T 1 T 2 400 x 323 V 1 T 2 = V 2 = T 1 303 V 1 = 426 m. L
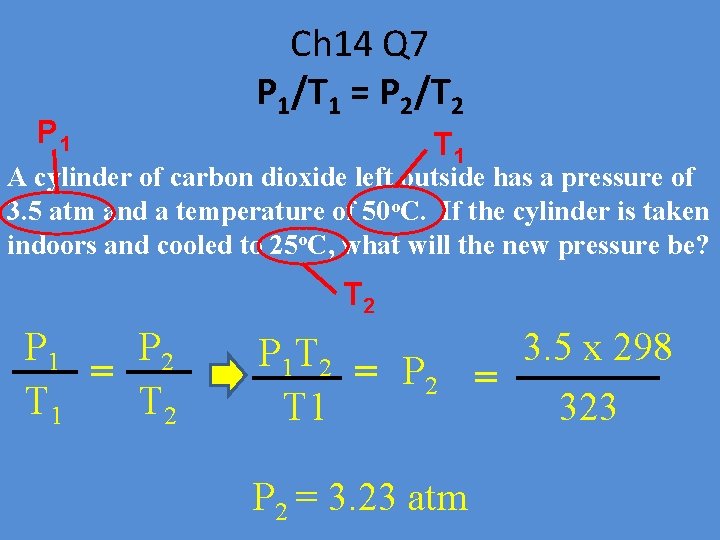
P 1 Ch 14 Q 7 P 1/T 1 = P 2/T 2 T 1 A cylinder of carbon dioxide left outside has a pressure of 3. 5 atm and a temperature of 50 o. C. If the cylinder is taken indoors and cooled to 25 o. C, what will the new pressure be? T 2 P 1 P 2 = T 1 T 2 3. 5 x 298 P 1 T 2 = P 2 = T 1 323 P 2 = 3. 23 atm

V 1 Ch 14 Q 8 P 1 V 1 = P 2 V 2 P 1 A gas has a volume of 125 m. L at a pressure of 2 atm. If the pressure is changed to 7. 2 atm, what is the new volume of that sample of gas? P 2 P 1 V 1 = P 2 V 2 P 1 V 1 = V 2= P 2 V 2 = 34. 7 m. L 2 x 125 7. 2
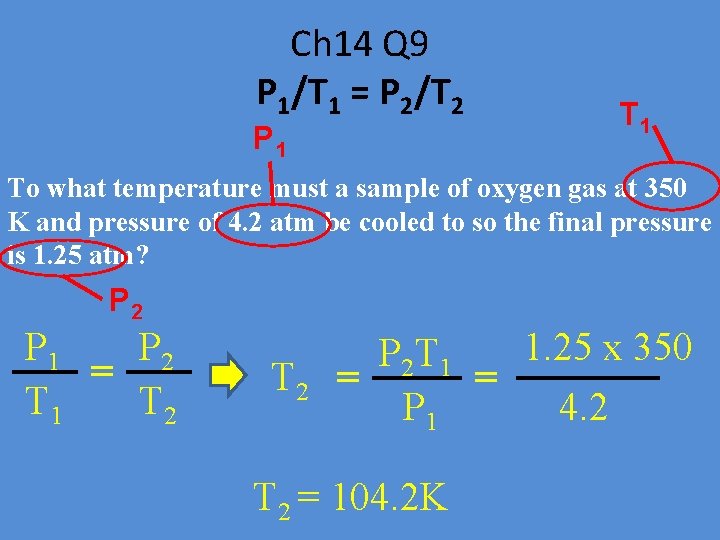
Ch 14 Q 9 P 1/T 1 = P 2/T 2 P 1 To what temperature must a sample of oxygen gas at 350 K and pressure of 4. 2 atm be cooled to so the final pressure is 1. 25 atm? P 2 P 1 P 2 = T 1 T 2 1. 25 x 350 P 2 T 1 = = P 1 4. 2 T 2 = 104. 2 K

V 1 Ch 14 Q 10 V 1/T 1 = V 2/T 2 T 1 A gas has a volume of 20 L at a temperature of 350 K. What will the volume of the gas occupy if the Kelvin temperature is increased to 423 K? T 2 V 1 V 2 = T 1 T 2 20 x 423 V 1 T 2 = V 2 = T 1 350 V 2 = 24. 2 L

Chapter 16

Ch 16 Q 1 M = mol/L Calculate the molarity of a 3. 4 mol of Na 2 CO 3 dissolved in a 100 m. L solution? 3. 4 mole 0. 100 L = 34 M
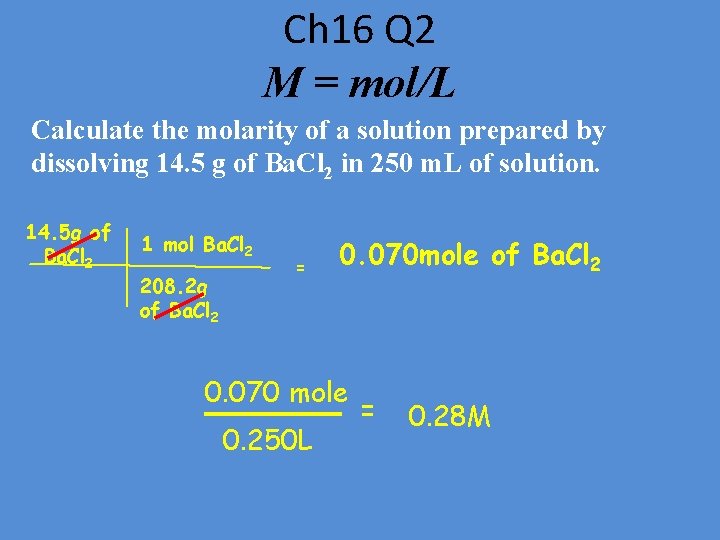
Ch 16 Q 2 M = mol/L Calculate the molarity of a solution prepared by dissolving 14. 5 g of Ba. Cl 2 in 250 m. L of solution. 14. 5 g of Ba. Cl 2 1 mol Ba. Cl 2 208. 2 g of Ba. Cl 2 = 0. 070 mole of Ba. Cl 2 0. 070 mole 0. 250 L = 0. 28 M

Ch 16 Q 3 ML = mol Calculate the number of grams of solute needed to make a 150 m. L solution of 0. 40 M H 2 SO 4. 0. 4 M x 0. 150 L = 0. 06 mole of H 2 SO 4 0. 06 mole x 89. 08 g/mole = 5. 88 g of H 2 SO 4
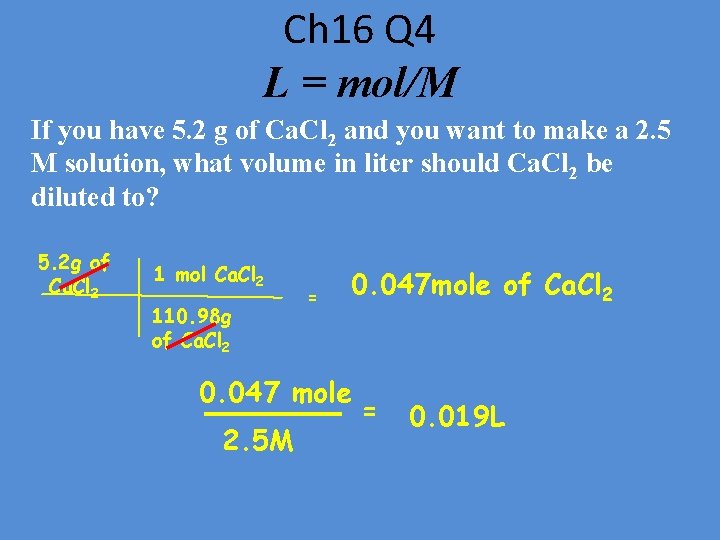
Ch 16 Q 4 L = mol/M If you have 5. 2 g of Ca. Cl 2 and you want to make a 2. 5 M solution, what volume in liter should Ca. Cl 2 be diluted to? 5. 2 g of Ca. Cl 2 1 mol Ca. Cl 2 110. 98 g of Ca. Cl 2 = 0. 047 mole of Ca. Cl 2 0. 047 mole 2. 5 M = 0. 019 L

M 2 Ch 16 Q 5 M 1 V 1 = M 2 V 2 You want to make a solution with a final concentration of 2. 25 M HCl from a stock solution of 6. 0 M HCl. How many milliliters of the stock solution are required to make a solution with a final volume of 50 m. L? V 2 M 1 V 1 = M 2 V 2 M 1 = V 1 = 18. 75 m. L 2. 25 x 50 6. 0

M 1 Ch 16 Q 8 M 1 V 1 = M 2 V 2 How many m. L of a 15. 8 M HNO 3 stock solution are required to make a 3. 0 M HNO 3 solution with a final volume of 250 m. L? M 2 V 2 M 1 V 1 = M 2 V 2 M 1 V 1 = 47. 5 m. L = 3. 0 x 250 15. 8

Ch 19

1. Review naming rules for acids and bases as describe in chapter 9. Normal ending Acid name is… ____-ide hydro-___-ic acid ____-ate _____-ic acid ____-ite _____-ous acid

What is an acid? Produces H+ ions in solution What are some properties of an acid? Tastes sour, reacts with base, p. H < 7 Give an example of an acid. HNO 3 H 2 SO 4 HCL

What is a base? Produces OH ions in solution What are some properties of a base? Bitter taste, slippery feel, p. H of > 7 Give an example of a base. Na. OH, Ca(OH)2
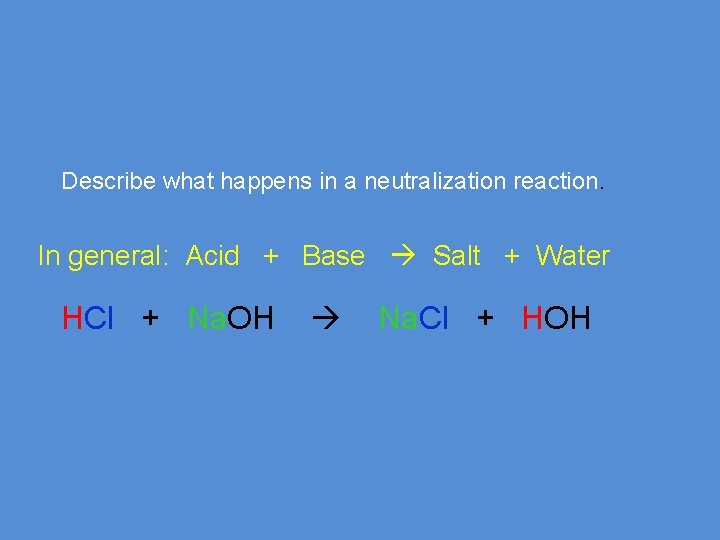
Describe what happens in a neutralization reaction. In general: Acid + Base Salt + Water HCl + Na. OH Na. Cl + HOH
- Slides: 65