Chapter 10 Phase Transformations ISSUES TO ADDRESS Transforming








- Slides: 30

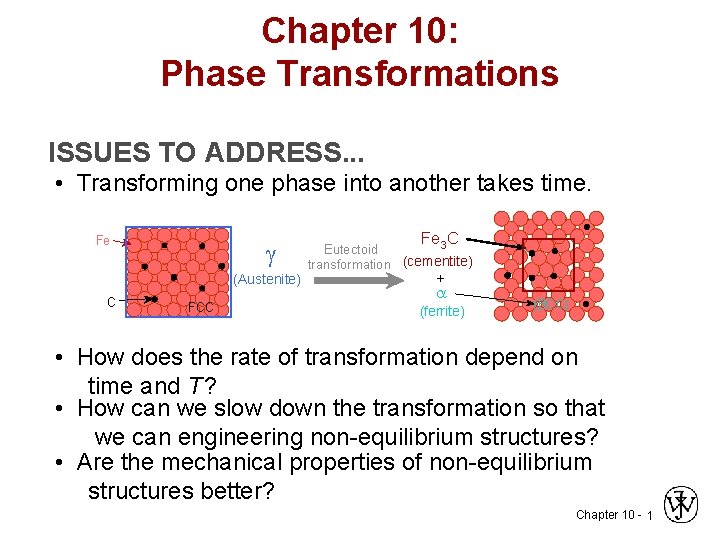
Chapter 10: Phase Transformations ISSUES TO ADDRESS. . . • Transforming one phase into another takes time. Fe (Austenite) C FCC Fe C 3 Eutectoid transformation (cementite) + (ferrite) (BCC) • How does the rate of transformation depend on time and T ? • How can we slow down the transformation so that we can engineering non-equilibrium structures? • Are the mechanical properties of non-equilibrium structures better? Chapter 10 - 1

Phase Transformations Nucleation – nuclei (seeds) act as template to grow crystals – for nucleus to form rate of addition of atoms to nucleus must be faster than rate of loss – once nucleated, grow until reach equilibrium Driving force to nucleate increases as we increase T – supercooling (eutectic, eutectoid) – superheating (peritectic) Small supercooling few nuclei - large crystals Large supercooling rapid nucleation - many nuclei, small crystals Chapter 10 - 2

Solidification: Nucleation Processes • Homogeneous nucleation – nuclei form in the bulk of liquid metal – requires supercooling (typically 80 -300°C max) • Heterogeneous nucleation – much easier since stable “nucleus” is already present • Could be wall of mold or impurities in the liquid phase – allows solidification with only 0. 1 -10ºC supercooling Chapter 10 - 3

Homogeneous Nucleation & Energy Effects Surface Free Energy- destabilizes the nuclei (it takes energy to make an interface) = surface tension GT = Total Free Energy = GS + GV Volume (Bulk) Free Energy – stabilizes the nuclei (releases energy) r* = critical nucleus: nuclei < r* shrink; nuclei>r* grow (to reduce energy) Adapted from Fig. 10. 2(b), Callister 7 e. Chapter 10 - 4

Solidification r* = critical radius = surface free energy Tm = melting temperature HS = latent heat of solidification T = Tm - T = supercooling Note: HS = strong function of T = weak function of T r* decreases as T increases For typical T r* ca. 100Å Chapter 10 - 5

Rate of Phase Transformations Kinetics - measure approach to equilibrium vs. time • Hold temperature constant & measure conversion vs. time How is conversion measured? X-ray diffraction – have to do many samples electrical conductivity – follow one sample sound waves – one sample Chapter 10 - 6

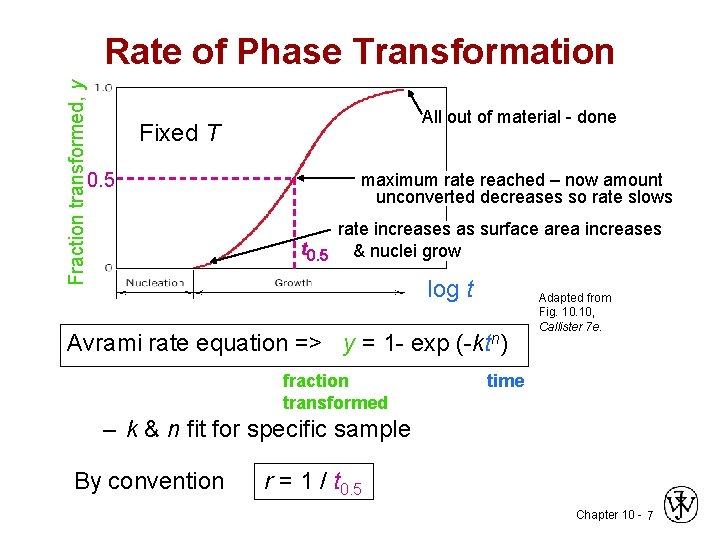
Fraction transformed, y Rate of Phase Transformation All out of material - done Fixed T 0. 5 maximum rate reached – now amount unconverted decreases so rate slows rate increases as surface area increases t 0. 5 & nuclei grow log t Avrami rate equation => y = 1 - exp (-ktn) fraction transformed Adapted from Fig. 10, Callister 7 e. time – k & n fit for specific sample By convention r = 1 / t 0. 5 Chapter 10 - 7

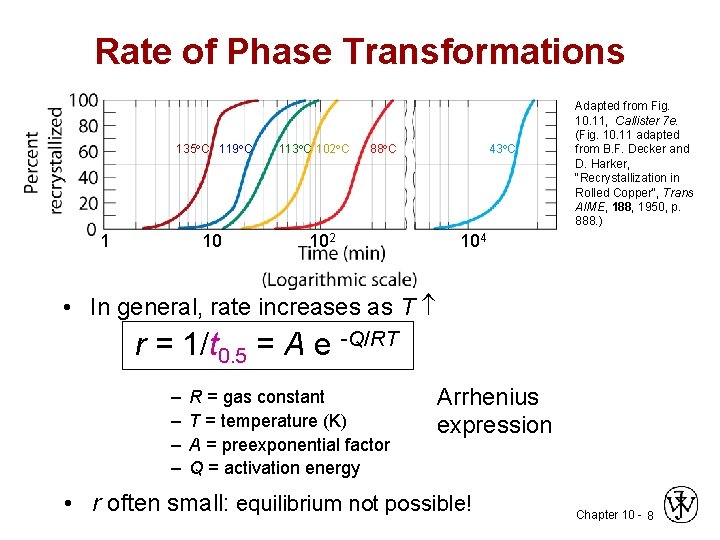
Rate of Phase Transformations 135 C 119 C 1 10 113 C 102 C 88 C 102 43 C Adapted from Fig. 10. 11, Callister 7 e. (Fig. 10. 11 adapted from B. F. Decker and D. Harker, "Recrystallization in Rolled Copper", Trans AIME, 188, 1950, p. 888. ) 104 • In general, rate increases as T r = 1/t 0. 5 = A e -Q/RT – – R = gas constant T = temperature (K) A = preexponential factor Q = activation energy Arrhenius expression • r often small: equilibrium not possible! Chapter 10 - 8

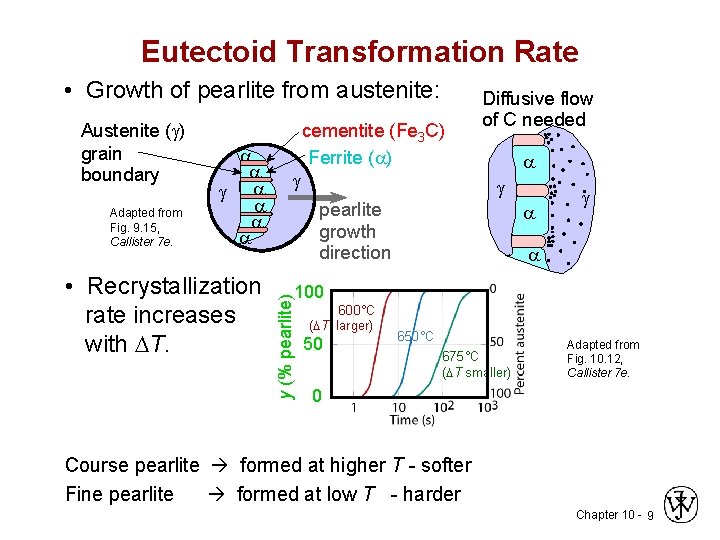
Eutectoid Transformation Rate • Growth of pearlite from austenite: Adapted from Fig. 9. 15, Callister 7 e. • Recrystallization rate increases with T. cementite (Fe 3 C) Ferrite ( ) pearlite growth direction 100 y (% pearlite) Austenite ( ) grain boundary Diffusive flow of C needed 600°C ( T larger) 50 650°C 675°C ( T smaller) Adapted from Fig. 10. 12, Callister 7 e. 0 Course pearlite formed at higher T - softer Fine pearlite formed at low T - harder Chapter 10 - 9

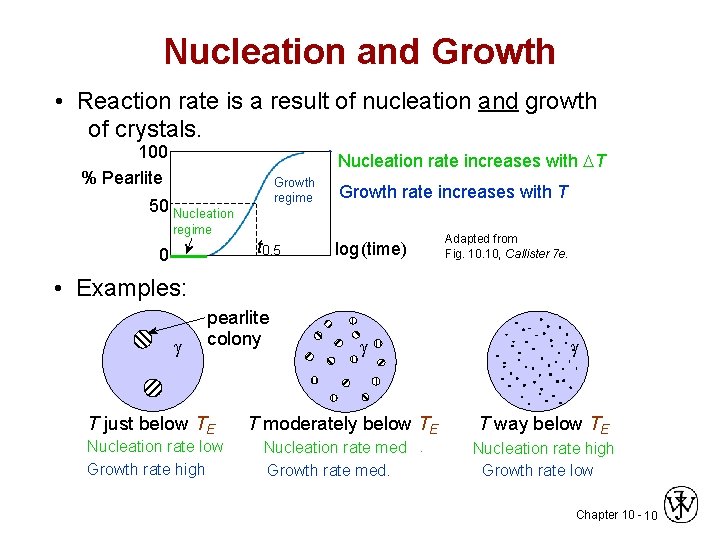
Nucleation and Growth • Reaction rate is a result of nucleation and growth of crystals. 100 % Pearlite Nucleation rate increases with T Growth regime 50 Nucleation regime 0 t 0. 5 Growth rate increases with T log (time) Adapted from Fig. 10, Callister 7 e. • Examples: pearlite colony T just below TE Nucleation rate low Growth rate high T moderately below TE Nucleation rate med. Growth rate med. T way below TE Nucleation rate high Growth rate low Chapter 10 - 10

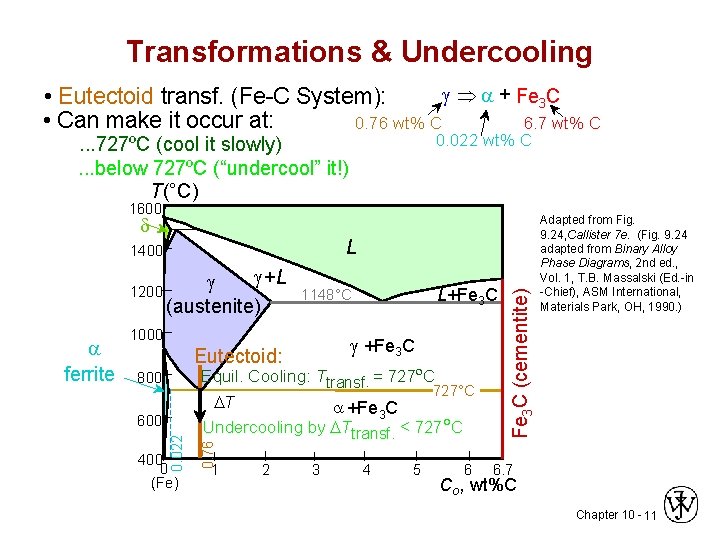
Transformations & Undercooling Þ + Fe 3 C • Eutectoid transf. (Fe-C System): • Can make it occur at: 0. 76 wt% C 6. 7 wt% C 0. 022 wt% C . . . 727ºC (cool it slowly). . . below 727ºC (“undercool” it!) T(°C) 1600 d +L 1200 (austenite) 1000 +Fe 3 C Eutectoid: Equil. Cooling: Ttransf. = 727ºC 800 T 400 0 (Fe) 727°C +Fe 3 C Undercooling by Ttransf. < 727 C 0. 76 600 0. 022 ferrite L+Fe 3 C 1148°C 1 2 3 4 5 6 Fe 3 C (cementite) L 1400 Adapted from Fig. 9. 24, Callister 7 e. (Fig. 9. 24 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in -Chief), ASM International, Materials Park, OH, 1990. ) 6. 7 Co , wt%C Chapter 10 - 11

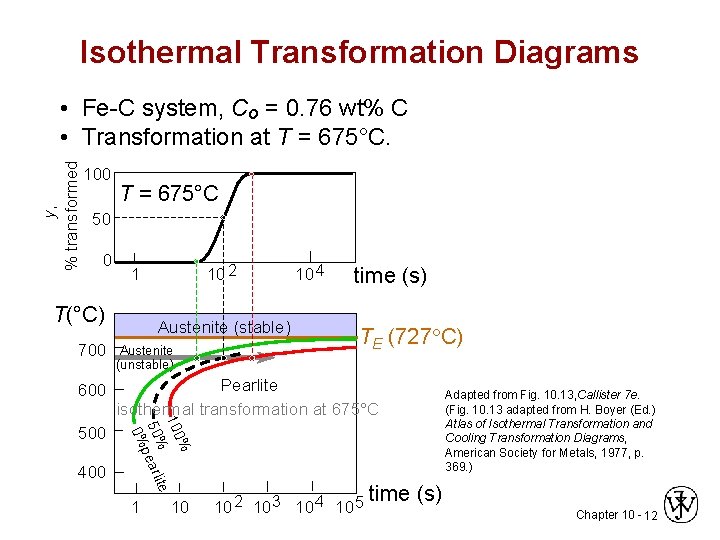
Isothermal Transformation Diagrams y, % transformed • Fe-C system, Co = 0. 76 wt% C • Transformation at T = 675°C. 100 T = 675°C 50 0 10 2 1 T(°C) Austenite (stable) 10 4 time (s) TE (727 C) 600 Pearlite isothermal transformation at 675°C 500 400 % 100 te 50%pearli 0% 700 Austenite (unstable) 1 10 10 2 10 3 10 4 10 5 Adapted from Fig. 10. 13, Callister 7 e. (Fig. 10. 13 adapted from H. Boyer (Ed. ) Atlas of Isothermal Transformation and Cooling Transformation Diagrams, American Society for Metals, 1977, p. 369. ) time (s) Chapter 10 - 12

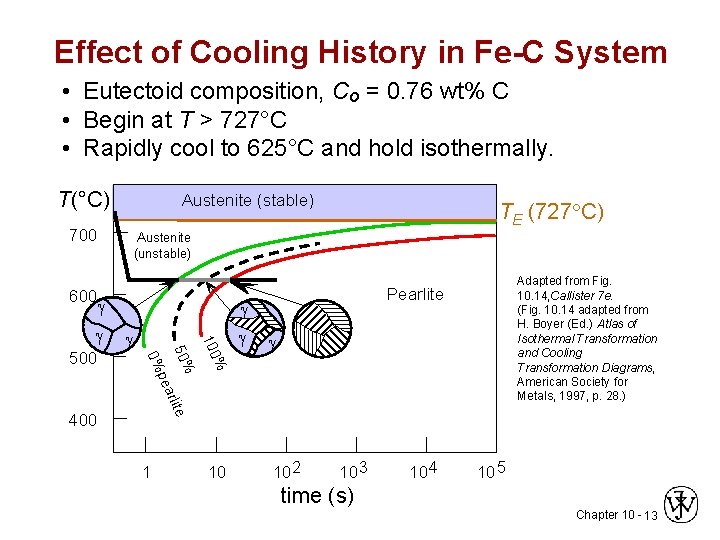
Effect of Cooling History in Fe-C System • Eutectoid composition, Co = 0. 76 wt% C • Begin at T > 727°C • Rapidly cool to 625°C and hold isothermally. T(°C) Austenite (stable) 700 600 TE (727 C) Austenite (unstable) % 100 pea 50% 500 Adapted from Fig. 10. 14, Callister 7 e. (Fig. 10. 14 adapted from H. Boyer (Ed. ) Atlas of Isothermal Transformation and Cooling Transformation Diagrams, American Society for Metals, 1997, p. 28. ) Pearlite 400 1 10 10 2 10 3 10 4 10 5 time (s) Chapter 10 - 13

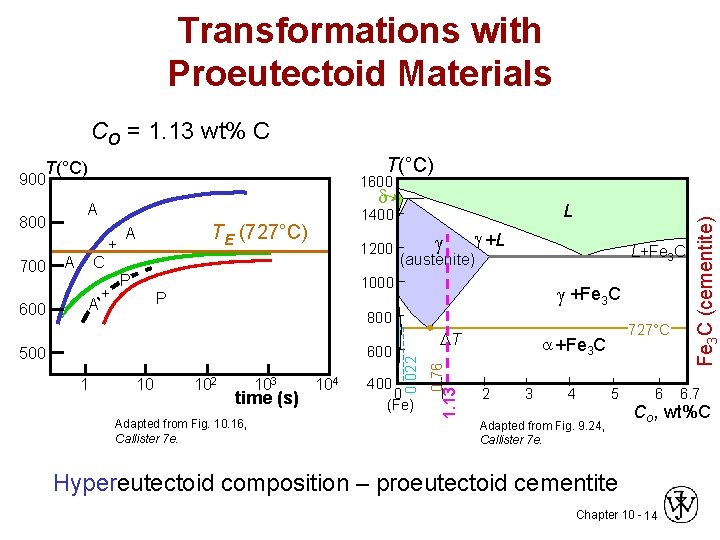
Transformations with Proeutectoid Materials CO = 1. 13 wt% C T(°C) 900 d A + C A + P P +L L+Fe 3 C (austenite) 1000 +Fe 3 C 800 600 500 1 10 102 103 time (s) Adapted from Fig. 10. 16, Callister 7 e. 104 T 400 0 (Fe) 0. 76 600 A 1200 1 1. 13 700 TE (727°C) A L 1400 0. 022 800 727°C +Fe 3 C 2 3 4 5 Adapted from Fig. 9. 24, Callister 7 e. 6 Fe 3 C (cementite) 1600 6. 7 Co , wt%C Hypereutectoid composition – proeutectoid cementite Chapter 10 - 14

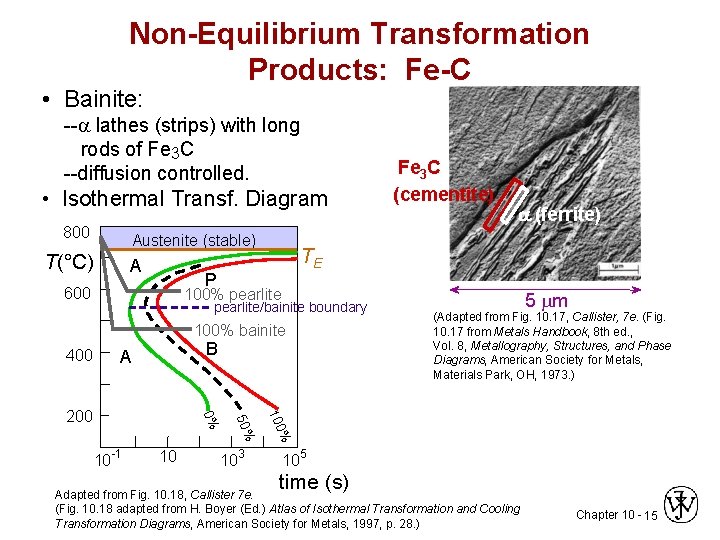
Non-Equilibrium Transformation Products: Fe-C • Bainite: -- lathes (strips) with long rods of Fe 3 C --diffusion controlled. • Isothermal Transf. Diagram 800 Austenite (stable) T(°C) A 100% pearlite/bainite boundary 100% bainite 400 a (ferrite) TE P 600 Fe 3 C (cementite) B A 10 103 (Adapted from Fig. 10. 17, Callister, 7 e. (Fig. 10. 17 from Metals Handbook, 8 th ed. , Vol. 8, Metallography, Structures, and Phase Diagrams, American Society for Metals, Materials Park, OH, 1973. ) % 100 10 -1 50% 0% 200 5 m 105 time (s) Adapted from Fig. 10. 18, Callister 7 e. (Fig. 10. 18 adapted from H. Boyer (Ed. ) Atlas of Isothermal Transformation and Cooling Transformation Diagrams, American Society for Metals, 1997, p. 28. ) Chapter 10 - 15

Spheroidite: Fe-C System • Spheroidite: (ferrite) -- grains with spherical Fe 3 C --diffusion dependent. --heat bainite or pearlite for long times Fe 3 C --reduces interfacial area (driving force) (cementite) 60 m (Adapted from Fig. 10. 19, Callister, 7 e. (Fig. 10. 19 copyright United States Steel Corporation, 1971. ) Chapter 10 - 16

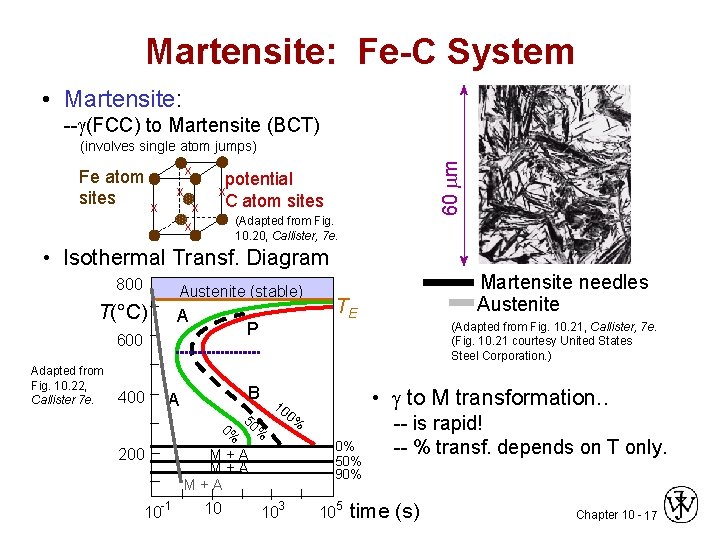
Martensite: Fe-C System • Martensite: -- (FCC) to Martensite (BCT) Fe atom sites x x x 60 m (involves single atom jumps) potential C atom sites x x x (Adapted from Fig. 10. 20, Callister, 7 e. • Isothermal Transf. Diagram 800 Austenite (stable) T(°C) A P 600 Adapted from Fig. 10. 22, Callister 7 e. 400 A 200 10 -1 B 5 0% 0% 10 10 TE 0% 50% 90% 103 (Adapted from Fig. 10. 21, Callister, 7 e. (Fig. 10. 21 courtesy United States Steel Corporation. ) • to M transformation. . 0% M+A M+A Martensite needles Austenite 105 -- is rapid! -- % transf. depends on T only. time (s) Chapter 10 - 17

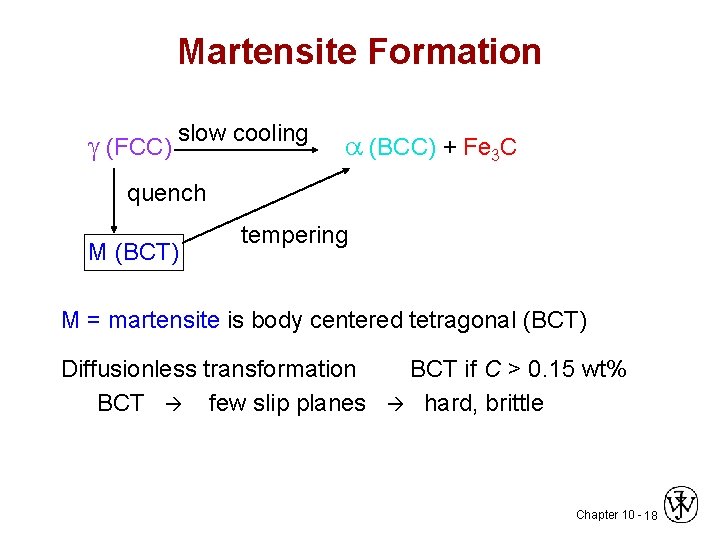
Martensite Formation (FCC) slow cooling (BCC) + Fe 3 C quench M (BCT) tempering M = martensite is body centered tetragonal (BCT) Diffusionless transformation BCT few slip planes BCT if C > 0. 15 wt% hard, brittle Chapter 10 - 18

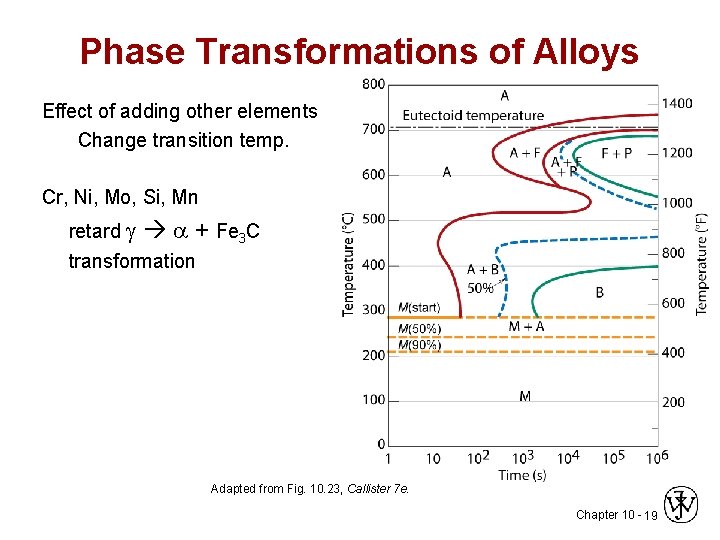
Phase Transformations of Alloys Effect of adding other elements Change transition temp. Cr, Ni, Mo, Si, Mn retard + Fe 3 C transformation Adapted from Fig. 10. 23, Callister 7 e. Chapter 10 - 19

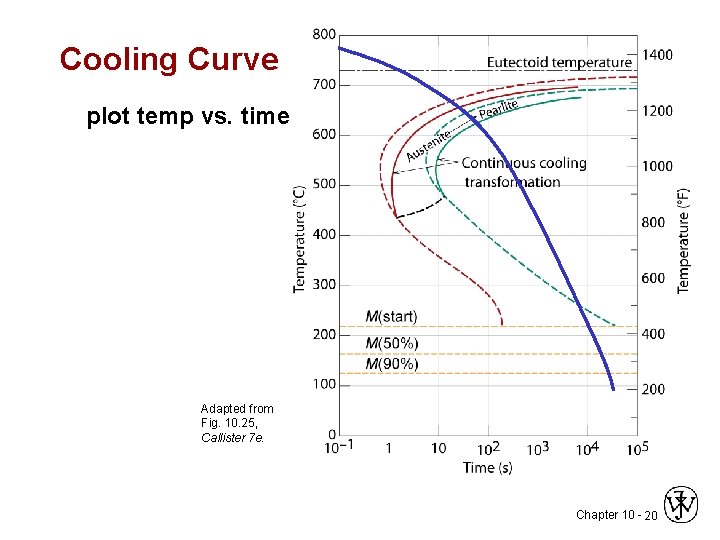
Cooling Curve plot temp vs. time Adapted from Fig. 10. 25, Callister 7 e. Chapter 10 - 20

Dynamic Phase Transformations On the isothermal transformation diagram for 0. 45 wt% C Fe-C alloy, sketch and label the time-temperature paths to produce the following microstructures: a) 42% proeutectoid ferrite and 58% coarse pearlite b) 50% fine pearlite and 50% bainite c) 100% martensite d) 50% martensite and 50% austenite Chapter 10 - 21

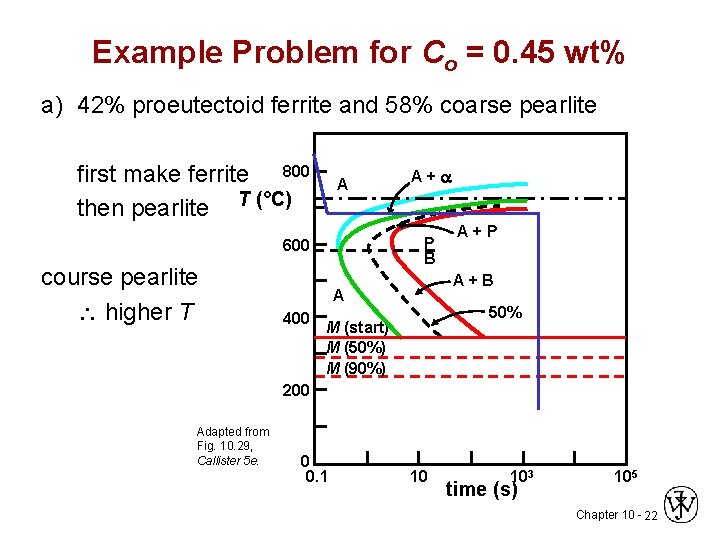
Example Problem for Co = 0. 45 wt% a) 42% proeutectoid ferrite and 58% coarse pearlite first make ferrite 800 then pearlite T (°C) A P B 600 course pearlite higher T A+a A+B A 400 A+P 50% M (start) M (50%) M (90%) 200 Adapted from Fig. 10. 29, Callister 5 e. 0 0. 1 10 103 time (s) 105 Chapter 10 - 22

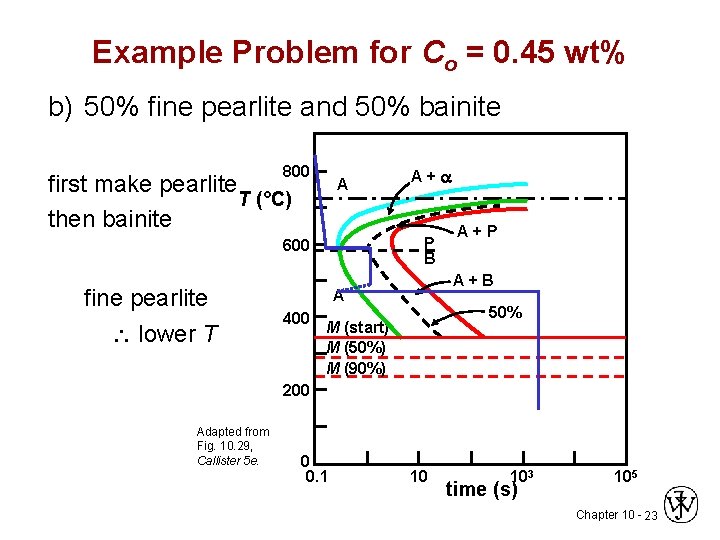
Example Problem for Co = 0. 45 wt% b) 50% fine pearlite and 50% bainite 800 first make pearlite T (°C) then bainite A P B 600 fine pearlite lower T A+a A+B A 400 A+P 50% M (start) M (50%) M (90%) 200 Adapted from Fig. 10. 29, Callister 5 e. 0 0. 1 10 103 time (s) 105 Chapter 10 - 23

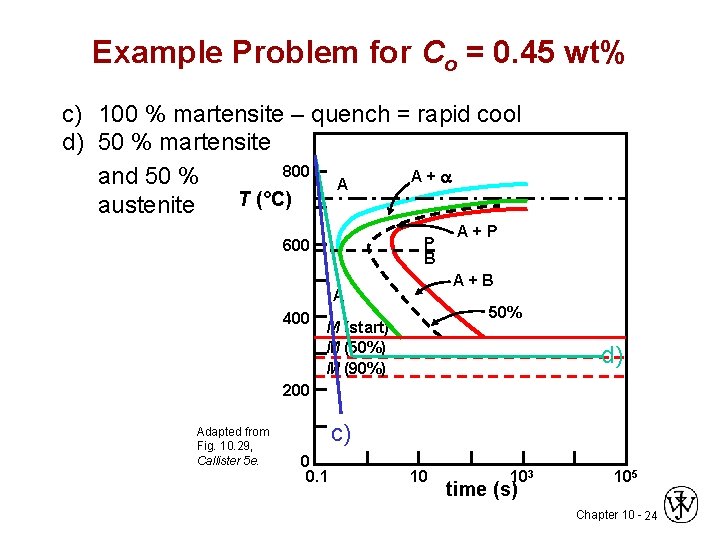
Example Problem for Co = 0. 45 wt% c) 100 % martensite – quench = rapid cool d) 50 % martensite 800 A+a and 50 % A T (°C) austenite P B 600 A+B A 400 A+P 50% M (start) M (50%) M (90%) d) 200 Adapted from Fig. 10. 29, Callister 5 e. c) 0 0. 1 10 103 time (s) 105 Chapter 10 - 24

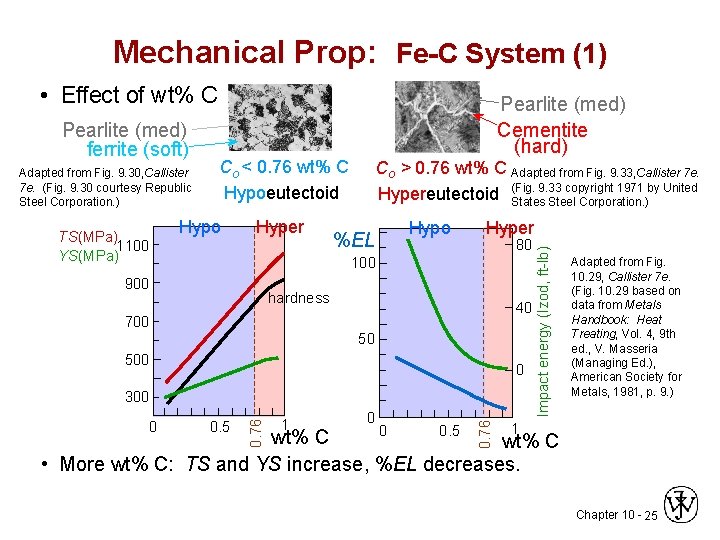
Mechanical Prop: Fe-C System (1) • Effect of wt% C Adapted from Fig. 9. 30, Callister 7 e. (Fig. 9. 30 courtesy Republic Steel Corporation. ) TS(MPa) 1100 YS(MPa) Co < 0. 76 wt% C Hypoeutectoid Hypo Hyper Co > 0. 76 wt% C Adapted from Fig. 9. 33, Callister 7 e. 9. 33 copyright 1971 by United Hypereutectoid (Fig. States Steel Corporation. ) %EL Hypo Hyper 80 100 900 hardness 40 700 50 500 0 0. 5 1 0 Adapted from Fig. 10. 29, Callister 7 e. (Fig. 10. 29 based on data from Metals Handbook: Heat Treating, Vol. 4, 9 th ed. , V. Masseria (Managing Ed. ), American Society for Metals, 1981, p. 9. ) 0. 76 0 0. 76 300 Impact energy (Izod, ft-lb) Pearlite (med) ferrite (soft) Pearlite (med) Cementite (hard) 1 0. 5 0 wt% C • More wt% C: TS and YS increase, %EL decreases. Chapter 10 - 25

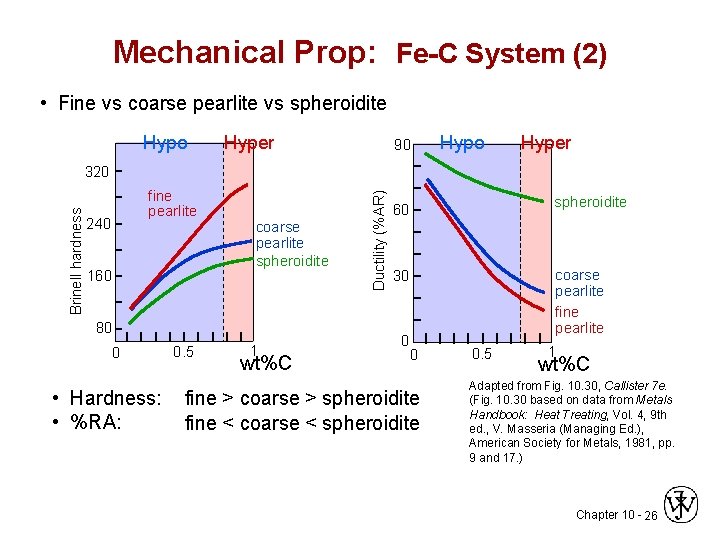
Mechanical Prop: Fe-C System (2) • Fine vs coarse pearlite vs spheroidite Hypo Hyper 90 Hypo Hyper fine pearlite 240 160 coarse pearlite spheroidite 80 0 • Hardness: • %RA: 0. 5 1 wt%C Ductility (%AR) Brinell hardness 320 spheroidite 60 coarse pearlite fine pearlite 30 0 0 fine > coarse > spheroidite fine < coarse < spheroidite 0. 5 1 wt%C Adapted from Fig. 10. 30, Callister 7 e. (Fig. 10. 30 based on data from Metals Handbook: Heat Treating, Vol. 4, 9 th ed. , V. Masseria (Managing Ed. ), American Society for Metals, 1981, pp. 9 and 17. ) Chapter 10 - 26

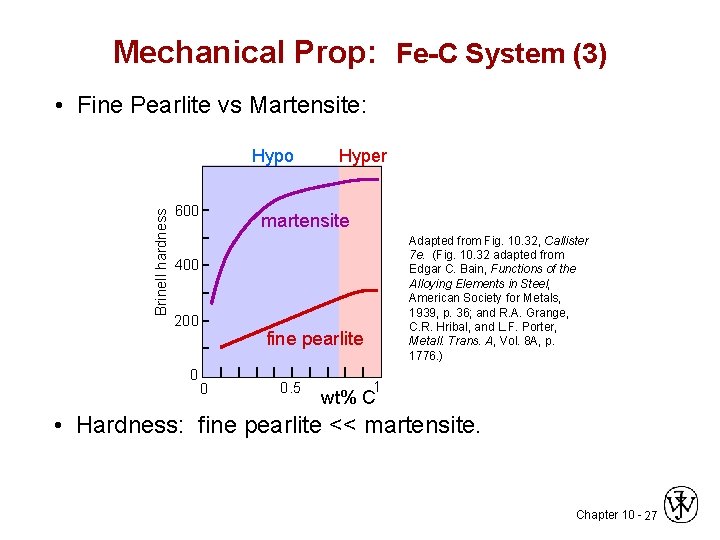
Mechanical Prop: Fe-C System (3) • Fine Pearlite vs Martensite: Brinell hardness Hypo 600 Hyper martensite Adapted from Fig. 10. 32, Callister 7 e. (Fig. 10. 32 adapted from Edgar C. Bain, Functions of the Alloying Elements in Steel, American Society for Metals, 1939, p. 36; and R. A. Grange, C. R. Hribal, and L. F. Porter, Metall. Trans. A, Vol. 8 A, p. 1776. ) 400 200 0 fine pearlite 0 0. 5 1 wt% C • Hardness: fine pearlite << martensite. Chapter 10 - 27

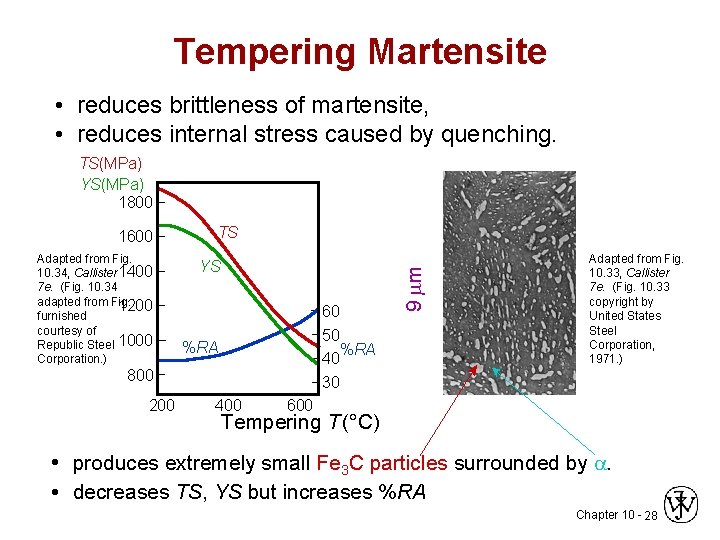
Tempering Martensite • reduces brittleness of martensite, • reduces internal stress caused by quenching. TS(MPa) YS(MPa) 1800 Adapted from Fig. 10. 34, Callister 1400 7 e. (Fig. 10. 34 adapted from Fig. 1200 furnished courtesy of Republic Steel 1000 Corporation. ) TS YS 60 50 %RA 40 30 %RA 800 200 400 9 m 1600 Adapted from Fig. 10. 33, Callister 7 e. (Fig. 10. 33 copyright by United States Steel Corporation, 1971. ) 600 Tempering T (°C) • produces extremely small Fe 3 C particles surrounded by . • decreases TS, YS but increases %RA Chapter 10 - 28

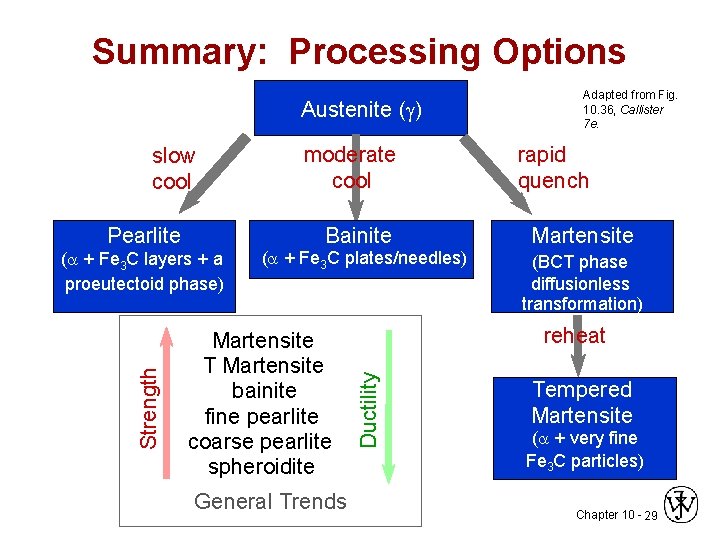
Summary: Processing Options Austenite ( ) slow cool moderate cool Adapted from Fig. 10. 36, Callister 7 e. rapid quench Bainite Martensite ( + Fe 3 C layers + a proeutectoid phase) ( + Fe 3 C plates/needles) (BCT phase diffusionless transformation) Martensite T Martensite bainite fine pearlite coarse pearlite spheroidite General Trends reheat Ductility Strength Pearlite Tempered Martensite ( + very fine Fe 3 C particles) Chapter 10 - 29

ANNOUNCEMENTS Reading: Core Problems: Self-help Problems: Chapter 10 - 30