Chapter 10 Molecular Geometry and Bonding Theories The


Chapter 10 Molecular Geometry and Bonding Theories
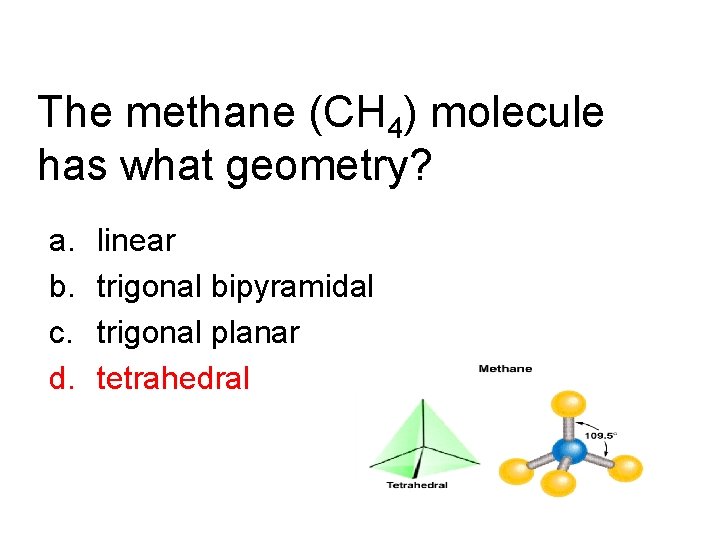
The methane (CH 4) molecule has what geometry? a. b. c. d. linear trigonal bipyramidal trigonal planar tetrahedral
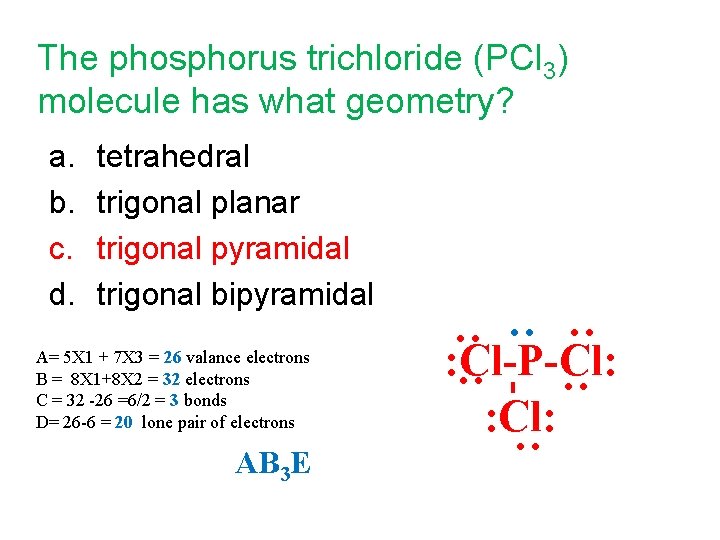
The phosphorus trichloride (PCl 3) molecule has what geometry? a. b. c. d. tetrahedral trigonal planar trigonal pyramidal trigonal bipyramidal : Cl-P-Cl: : AB 3 E : : : A= 5 X 1 + 7 X 3 = 26 valance electrons B = 8 X 1+8 X 2 = 32 electrons C = 32 -26 =6/2 = 3 bonds D= 26 -6 = 20 lone pair of electrons

The most stable geometry • The hybridization of for six electron domains the sulfur atom in a is: SF 6 molecule is: a. square planar. a. sp b. octahedral. b. sp 2 c. square pyramidal. c. sp 3 d 2 d. tetrahedral. d. sp 3 d
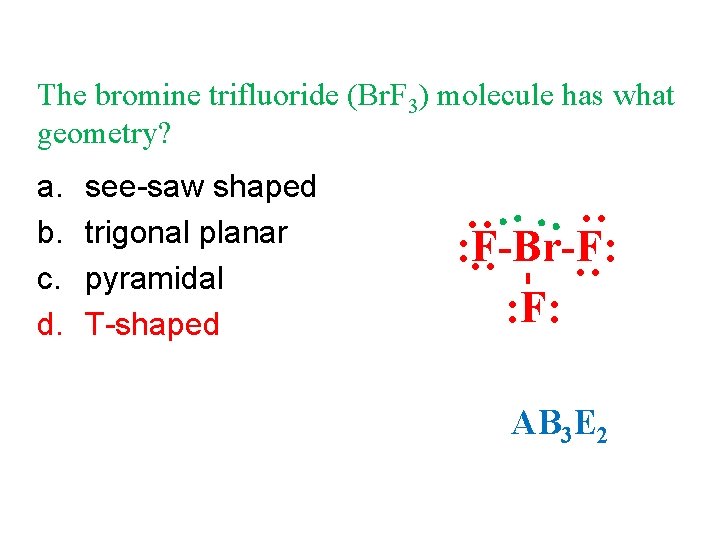
The bromine trifluoride (Br. F 3) molecule has what geometry? see-saw shaped trigonal planar pyramidal T-shaped : : : a. b. c. d. : F-Br-F: : F: AB 3 E 2

What is the molecular geometry of NO 2? 1. Linear 2. Bent 3. Trigonal planar 4. Tetrahedral 5. Pyramidal
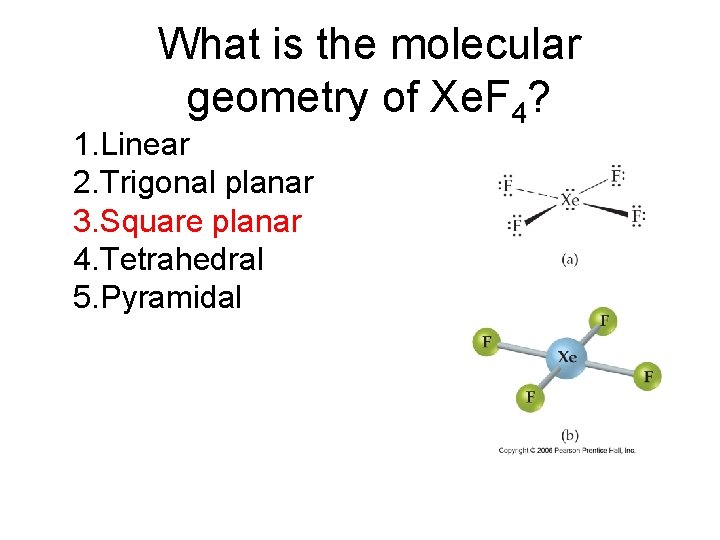
What is the molecular geometry of Xe. F 4? 1. Linear 2. Trigonal planar 3. Square planar 4. Tetrahedral 5. Pyramidal
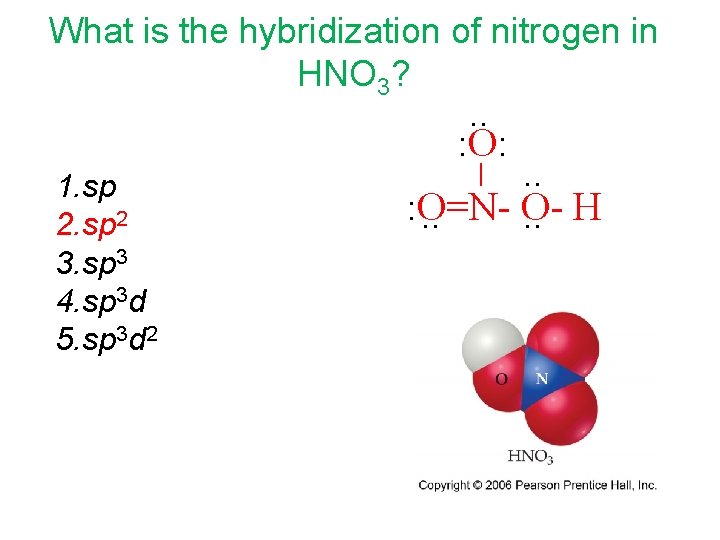
What is the hybridization of nitrogen in HNO 3? . . : O: . . : O=NO. . H _ 1. sp 2 3. sp 3 4. sp 3 d 5. sp 3 d 2
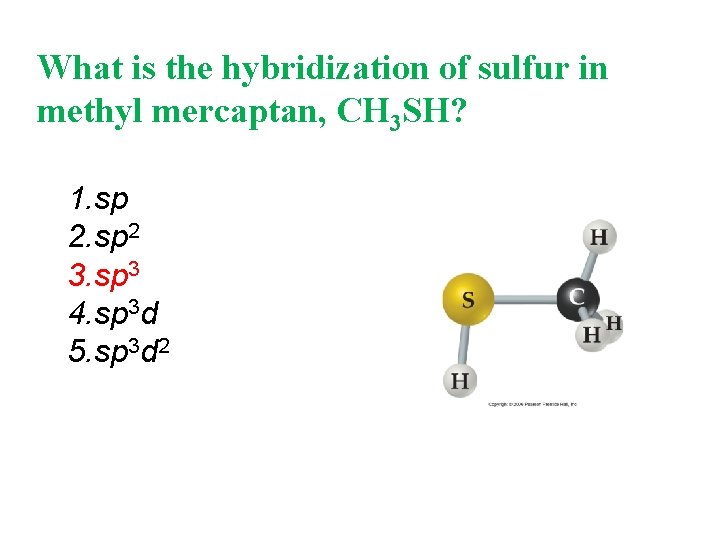
What is the hybridization of sulfur in methyl mercaptan, CH 3 SH? 1. sp 2 3. sp 3 4. sp 3 d 5. sp 3 d 2

14. The Cl-C-Cl bond angle in the CCl 2 O molecule (C is the central atom) is slightly (a). greater than 109. 5° (b). less than 109. 5° (c). less than 120° (d). greater than 120° A = 4+2 X 7+6 = 24 B= 8+2 X 8+8 = 32 C=32 -24=8/2 = 4 D= 24 -8 = 16 : : : The Lewis structure of the CCl 2 O molecule shows that there is a double bond between the O and the central C atom. A double bond represents a slightly larger electron domain which disrupts the ideal 120° Cl-C-Cl bond angle, making it smaller

Chapter 14 Chemical Equilibrium

A large value of the equilibrium constant indicates that when the reaction reaches equilibrium, mostly ______ will be present. a. b. c. d. reactants products catalysts shrapnel

When equilibrium is achieved: a. b. c. d. Q>K Q<K Q=K Q 2 = K
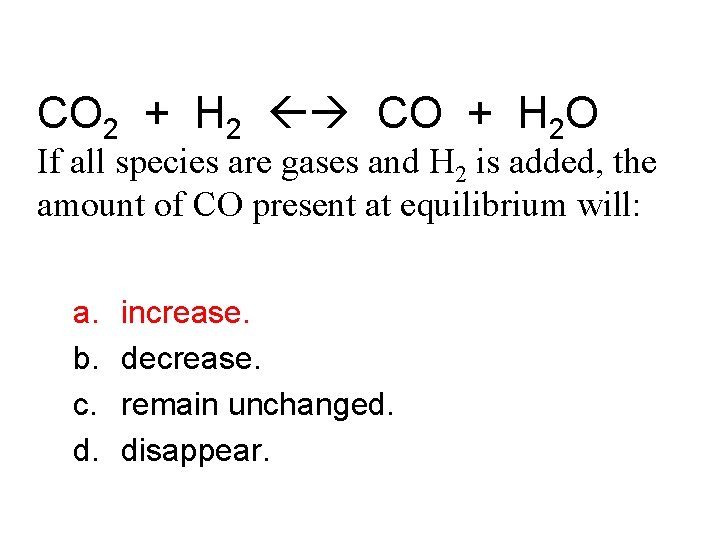
CO 2 + H 2 CO + H 2 O If all species are gases and H 2 is added, the amount of CO present at equilibrium will: a. b. c. d. increase. decrease. remain unchanged. disappear.
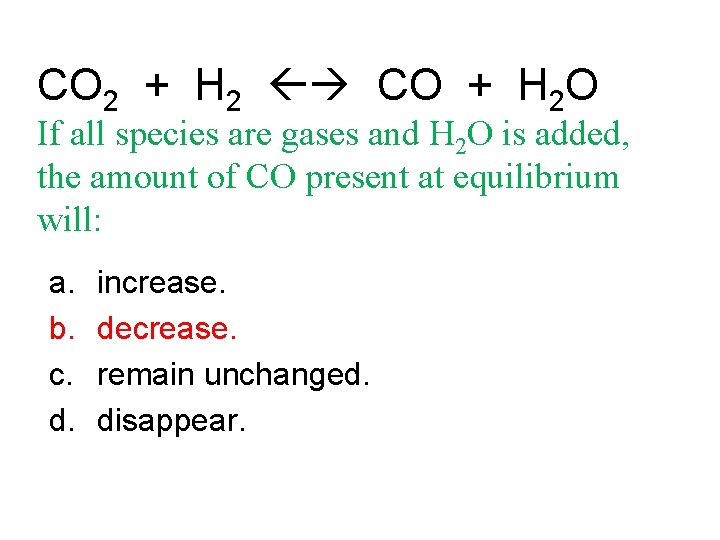
CO 2 + H 2 CO + H 2 O If all species are gases and H 2 O is added, the amount of CO present at equilibrium will: a. b. c. d. increase. decrease. remain unchanged. disappear.

CO 2 + H 2 CO + H 2 O If the reaction is endothermic and the temperature is raised, the amount of CO present will: a. b. c. d. increase. decrease. remain unchanged. disappear.

CO 2 + H 2 CO + H 2 O If all species are gases and the container is compressed, the amount of CO present will: a. b. c. d. increase. decrease. remain unchanged. disappear.

CO 2 + H 2 CO + H 2 O If a catalyst is added to the equilibrium, the amount of CO present will: a. b. c. d. increase. decrease. remain unchanged. disappear.

What is Kp in terms of Kc for the following reaction ? 2 NO(g) + • • • O 2(g) Kp = Kc. RT Kp = Kc/RT Kp = Kc. R/T Kp = K c Kp = Kc/(RT) 2 2 NO 2(g) Kp = Kc (RT)∆n ∆n = 2 -3 =-1 Kp = Kc /(RT)
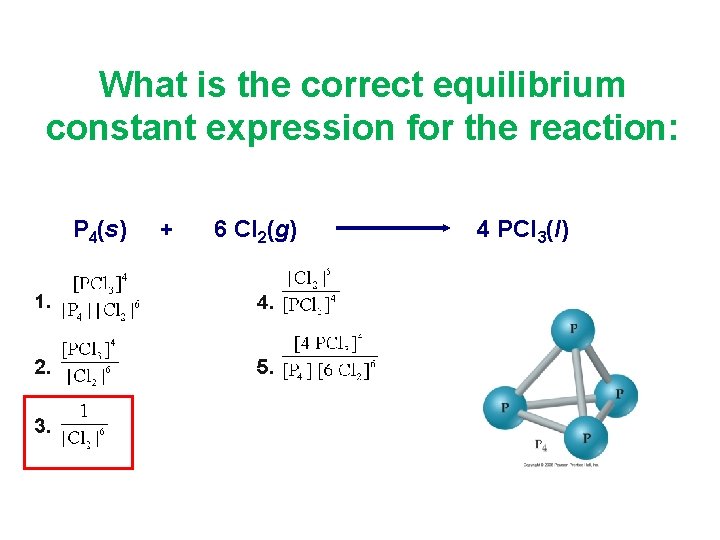
What is the correct equilibrium constant expression for the reaction: P 4(s) + 6 Cl 2(g) 4 PCl 3(l)
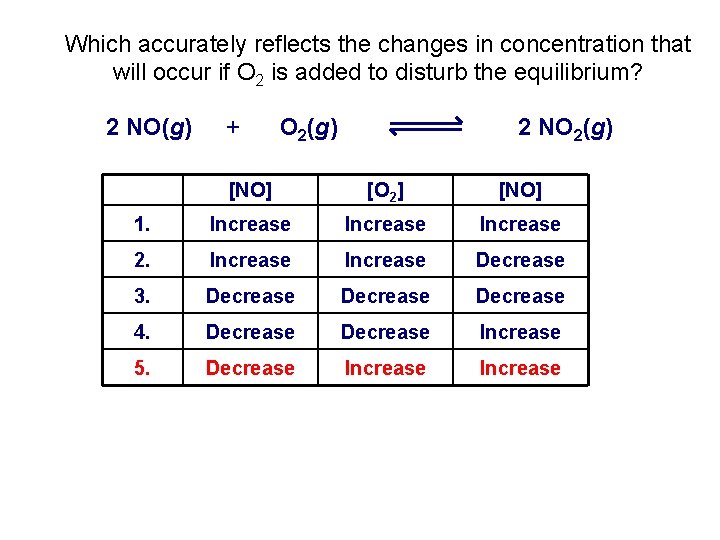
Which accurately reflects the changes in concentration that will occur if O 2 is added to disturb the equilibrium? 2 NO(g) + O 2(g) 2 NO 2(g) [NO] [O 2] [NO] 1. Increase 2. Increase Decrease 3. Decrease 4. Decrease Increase 5. Decrease Increase

Which of the following will result in an equilibrium shift to the right? PCl 3(g) + Cl 2(g) • • • PCl 5(g) DH° = – 87. 9 k. J/mol Increase temperature/increase volume Increase temperature/decrease volume Decrease temperature/increase volume Decrease temperature/decrease volume None of the above PCl 5

• Consider the reversible reaction at equilibrium at 392 o. C: 2 A(g) + B(g) ⇄ C(g) • The partial pressures are found to be: A: 6. 70 atm, B: 10. 1 atm, C: 3. 60 atm. Evaluate Kp for this reaction. • (a) 7. 94 x 10 -3 • • (b) 0. 146 (c) 0. 0532 (d) 54. 5 (e) 121

The equation relating Kp and Kc is Kp will be equal to Kc if A) Kp = Kc (RT)∆n A) ∆ n = 1 B) Kp = Kc RT∆n B) ∆ n = 0 C) Kc = Kp RT ∆ n C) RT = 0 D) Kc = Kp (RT) ∆n D) ∆ n = -1 Dr. Liala AL-Harbi

Chapter 15 Acid-Base Equilibria

What is the p. H of 0. 0200 M aqueous solution of HBr ? a. 1. 00 b. 1. 70 c. 2. 30 d. 12. 30 • The p. OH of a solution of Na. OH is 11. 30. What is the [H+] for this solution? • (a) 2. 0 x 10 -3 • (b) 2. 5 x 10 -3 • (c) 5. 0 x 10 -12 • (d) 4. 0 x 10 -12 • (e) 6. 2 x 10 -8

What is the p. H of a 0. 0400 M aqueous solution of KOH ? a. b. c. d. 12. 60 10. 30 4. 00 1. 40 • What is the approximate p. H of a solution labeled 6 x 10 -5 M HBr? • • • (a) 4. 2 (b) 4. 5 (c) 5. 8 (d) 9. 8 (e) 8. 2 Dr. Liala AL-Harbi

If the p. H = 2 for an HNO 3 solution, what is the concentration of HNO 3? • • • 0. 10 0. 20 0. 010 0. 020 0. 0010 • A solution in which [H+] = 10 -8 M has a p. H of ___ and is ___. • (a) 8, acidic • (b) 6, basic • (c) -6, basic • (d) -8, neutral • (e) 8, basic Dr. Liala AL-Harbi

• Which of the following solutions has the lowest p. H at 25 o. C? (No calculations required. ) • (a) 0. 2 M Na. OH • (b) 0. 2 M NH 3 • (c) 0. 2 M HCl • (d) pure water • What is the p. H of a 0. 010 M HF solution? • <1 • 2 • >2 • 7 Dr. Liala AL-Harbi

• Calculate the p. H of a • 3. 5 × 10 -3 M HNO 3 solution. • • A. -2. 46 • • B. 0. 54 • • C. 2. 46 • • D. 3. 00 • • E. 3. 46 Calculate the p. H of 2. 6 × 10 -2 M KOH. A. 12. 41 B. 15. 59 C. 2. 06 D. 7. 00 E. 1. 59 Dr. Liala AL-Harbi

• What is the in a 4. 8 × 10 -2 M KOH solution? • What is the [OH-] ion in a 5. 2 × 10 -4 M HNO 3 solution? a) b) c) d) e) [H+]ion 10 -2 4. 8 × M 1. 0 × 10 -7 M 4. 8 × 10 -11 M 4. 8 × 10 -12 M 2. 1 × 10 -13 M 1. 9 × 10 -11 M 1. 0 × 10 -7 M 5. 2 × 10 -4 M zero 1. 0 × 10 -4 M Dr. Liala AL-Harbi

Chapter 25 The Chemistry of Life: Organic and Biological Chemistry

C 10 H 22 is the formula of an: a. b. c. d. alkane. alkene. alkyne. aromatic hydrocarbon. • A molecule with the formula C 3 H 8 is a(n): • (a) hexane • (b) propane • (c) decane • (d) butane • (e) ethane

Which compound below does not havegeometric isomers (cis-trans isomers)? a. 1 -butene b. 2 -butene c. 2 -pentene d. 3 -hexene • The hybridization of carbon atoms in alkanes is • (a) sp • (b) sp 2 • (c) sp 3 • (d) sp 3 d • (e) sp 3 d 2
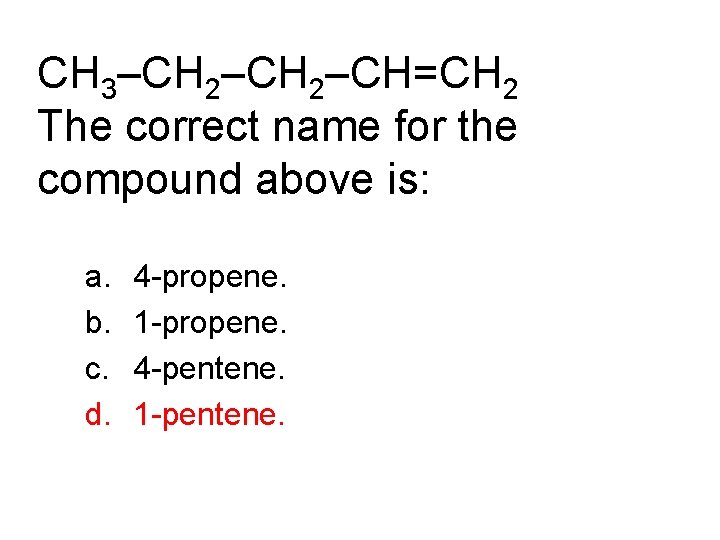
CH 3–CH 2–CH=CH 2 The correct name for the compound above is: a. b. c. d. 4 -propene. 1 -propene. 4 -pentene. 1 -pentene.

The product of the reaction of HBr with 2 -butene is named: a. b. c. d. 1 -bromobutane. 2 -bromobutane. 1, 2 -dibromobutane. 2, 3 -dibromobutane.
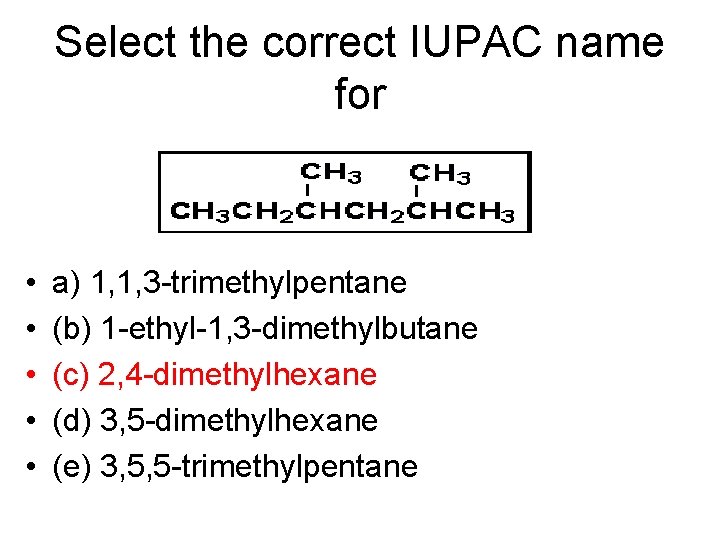
Select the correct IUPAC name for • • • a) 1, 1, 3 -trimethylpentane (b) 1 -ethyl-1, 3 -dimethylbutane (c) 2, 4 -dimethylhexane (d) 3, 5 -dimethylhexane (e) 3, 5, 5 -trimethylpentane
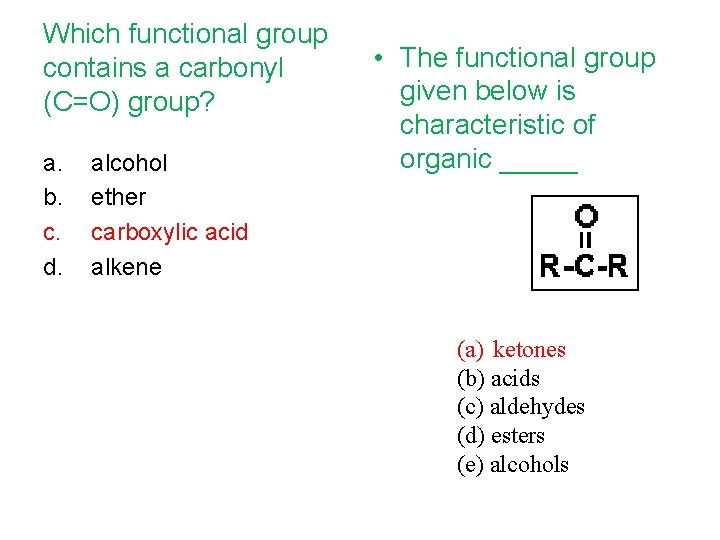
Which functional group contains a carbonyl (C=O) group? a. b. c. d. alcohol ether carboxylic acid alkene • The functional group given below is characteristic of organic _____ (a) ketones (b) acids (c) aldehydes (d) esters (e) alcohols

How many structural isomers does pentane (C 5 H 12) have? 1. 2. 3. 4. 5. 1 2 3 4 5 • Which of the following hydrocarbons does not have isomers? • • • (a) C 7 H 16 (b) C 6 H 14 (c) C 5 H 10 (d) C 4 H 8 (e) C 3 H 8
Which of the following does NOT exhibit geometric isomerism? (Hint: draw them!) • • • (a) 4 -octene (b) 2 -pentene (c) 3 -hexene (d) 2 -hexene (e) 1 -hexene • Which of the following will undergo an addition reaction with chlorine? (a) CH 3 CH 2 CH 3 (b) CH 3 CH 2 CH=CHCH 3 (c) C 6 H 6 (d) CH 3 CH 2 COOH (e) CH 3 CH 2 OH

For which of the compounds below are cistrans isomers possible? CH 3 CH=CH 2 CH 3 CH=CHCH 2 CH 3 CH=CHCH 3 (1) (2) (3) a) only 2 (b) both 1 and 2 (c) both 2 and 3 (d) all three (e) only 3

Select the correct IUPAC name for a) 1, 1, 3 -trimethylpentane (b) 1 -ethyl-1, 3 -dimethylbutane (c) 2, 4 -dimethylhexane (d) 3, 5 -dimethylhexane (e) 3, 5, 5 -trimethylpentane Dr. Liala AL-Harbi
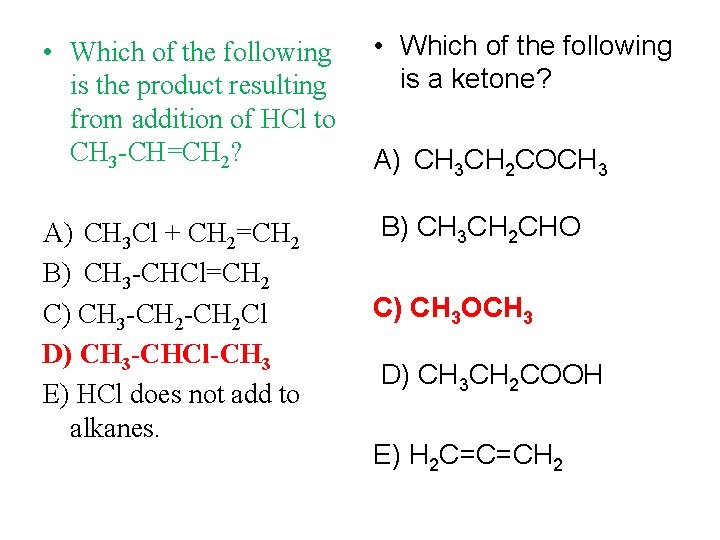
• Which of the following is the product resulting from addition of HCl to CH 3 -CH=CH 2? • Which of the following is a ketone? A) CH 3 Cl + CH 2=CH 2 B) CH 3 -CHCl=CH 2 C) CH 3 -CH 2 Cl D) CH 3 -CHCl-CH 3 E) HCl does not add to alkanes. B) CH 3 CH 2 CHO A) CH 3 CH 2 COCH 3 C) CH 3 OCH 3 D) CH 3 CH 2 COOH E) H 2 C=C=CH 2

• Which of the following is a CH 3 CH 2 CH 2 CH 2 OH ketone? represents: The formula A) an alcohol B) an alkene C) an alkyne D) an unsaturated hydrocarbon Dr. Liala AL-Harbi A) CH 3 CH 2 COCH 3 B) CH 3 CH 2 CHO C) CH 3 OCH 3 D) CH 3 CH 2 COOH E) H 2 C=C=CH 2
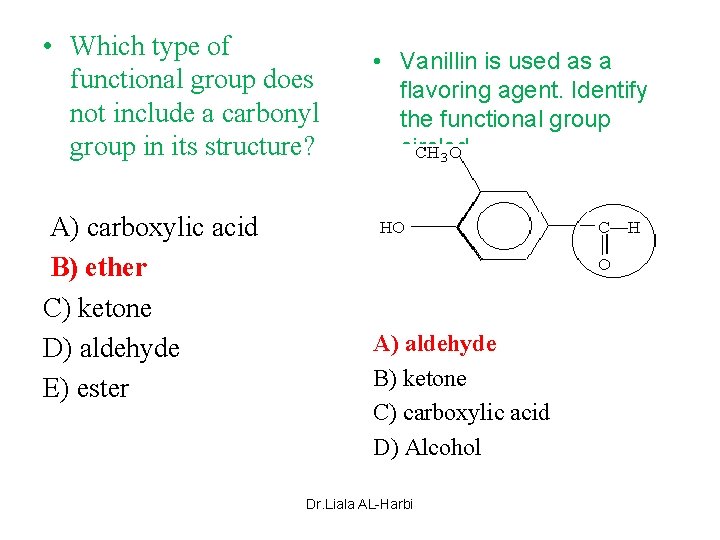
• Which type of functional group does not include a carbonyl group in its structure? A) carboxylic acid B) ether C) ketone D) aldehyde E) ester • Vanillin is used as a flavoring agent. Identify the functional group circled. A) aldehyde B) ketone C) carboxylic acid D) Alcohol Dr. Liala AL-Harbi

• An alkane with seven carbon atoms in a linear configuration is called a • • • A) hexene B) heptene C) hepylane D) heptane E) hexane A protein is: A) a polymer of amino acids B) a fatty acid ester of glycerol C) a polysaccharide D) an addition polymer E) a polymer of fatty acids
- Slides: 47