Chapter 10 Lecture Basic Chemistry Fourth Edition Chapter

Chapter 10 Lecture Basic Chemistry Fourth Edition Chapter 10 Properties of Solids and Liquids 10. 2 Shapes of Molecules and Ions (VSEPR Theory) Learning Goal Predict the threedimensional structure of a molecule or a polyatomic ion and classify it as polar or nonpolar. © 2014 Pearson Education, Inc.

VSEPR In the valence shell electron repulsion theory (VSEPR), the three-dimensional shape of a molecule is determined by • drawing the electron-dot formula • counting the number of electron groups (one or more electron pairs) around the central atom • placing the electron groups as far apart as possible around the central atom © 2014 Pearson Education, Inc.

VSEPR According to VSEPR theory, the shape of a molecule is determined by the • number of electron groups surrounding the central atom • number of atoms bonded to the central atom © 2014 Pearson Education, Inc.

VSEPR—Two Electron Groups Central atoms with two electron groups (two double bonds) such as CO 2 have a • linear electron-group geometry • linear shape with a bond angle of 180° to minimize repulsion © 2014 Pearson Education, Inc.

VSEPR—Three Electron Groups Central atoms with three electron groups (two single bonds, one double bond) such as H 2 CO have a • trigonal planar electron-group geometry • trigonal planar shape with a bond angle of 120° to minimize repulsion © 2014 Pearson Education, Inc.

VSEPR—Three Electron Groups Central atoms with three electron groups (a single bond, double bond, lone pair) such as SO 2 have a • trigonal planar electron-group geometry • bent shape with a bond angle of 120° to minimize repulsion © 2014 Pearson Education, Inc.
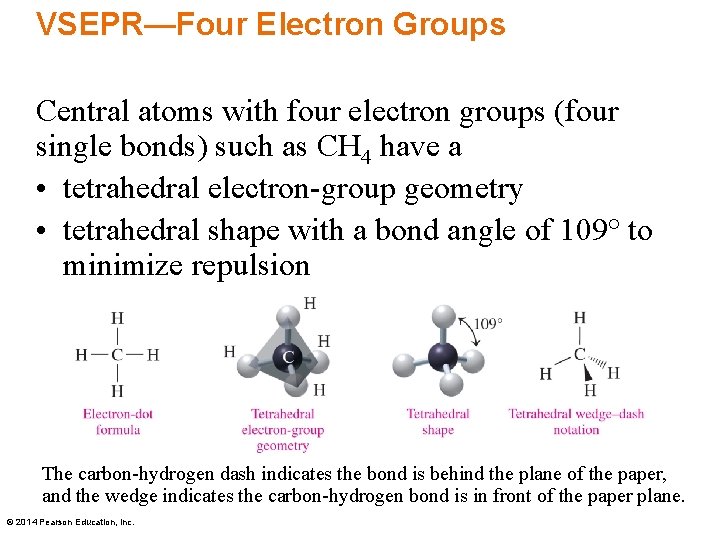
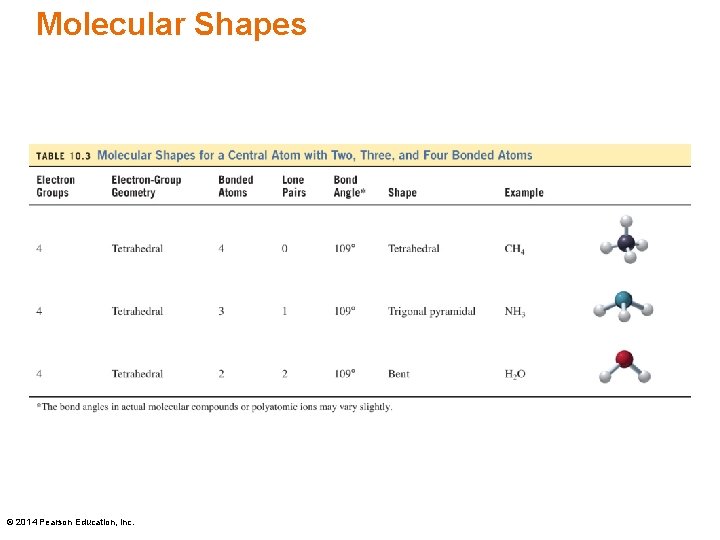
VSEPR—Four Electron Groups Central atoms with four electron groups (four single bonds) such as CH 4 have a • tetrahedral electron-group geometry • tetrahedral shape with a bond angle of 109° to minimize repulsion The carbon-hydrogen dash indicates the bond is behind the plane of the paper, and the wedge indicates the carbon-hydrogen bond is in front of the paper plane. © 2014 Pearson Education, Inc.
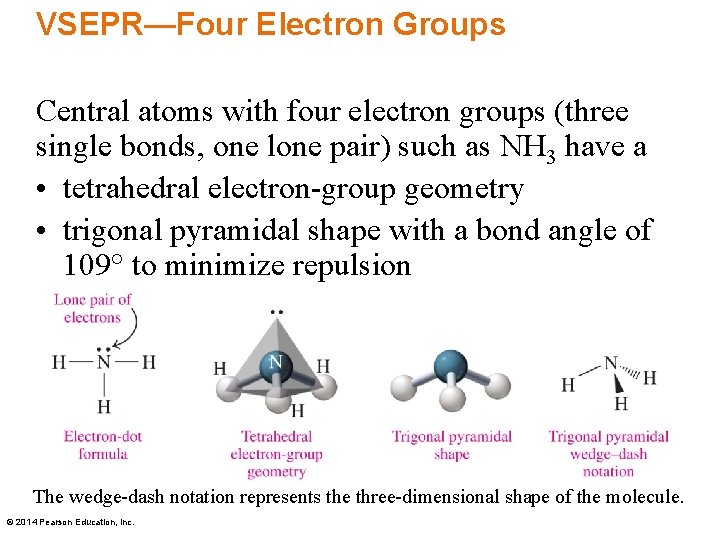
VSEPR—Four Electron Groups Central atoms with four electron groups (three single bonds, one lone pair) such as NH 3 have a • tetrahedral electron-group geometry • trigonal pyramidal shape with a bond angle of 109° to minimize repulsion The wedge-dash notation represents the three-dimensional shape of the molecule. © 2014 Pearson Education, Inc.
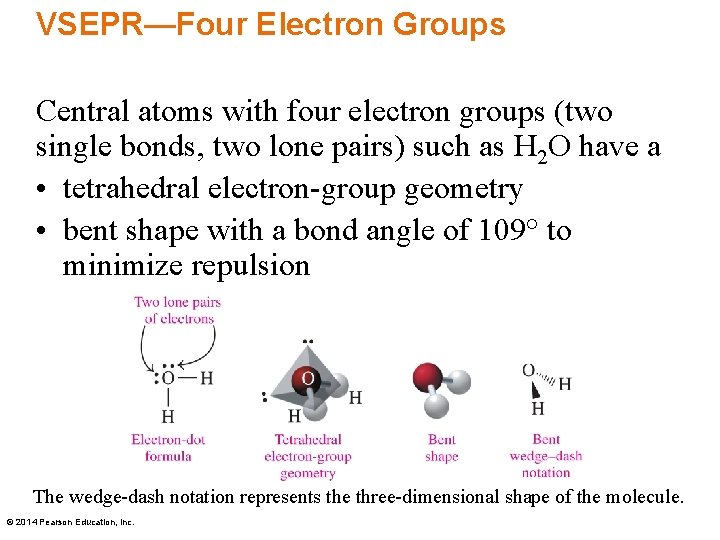
VSEPR—Four Electron Groups Central atoms with four electron groups (two single bonds, two lone pairs) such as H 2 O have a • tetrahedral electron-group geometry • bent shape with a bond angle of 109° to minimize repulsion The wedge-dash notation represents the three-dimensional shape of the molecule. © 2014 Pearson Education, Inc.
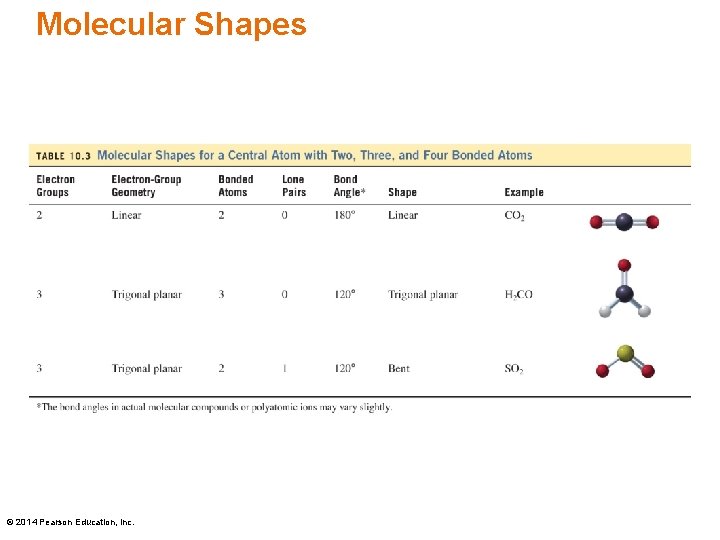
Molecular Shapes © 2014 Pearson Education, Inc.
Molecular Shapes © 2014 Pearson Education, Inc.
Guide to Predicting Molecular Shape (VSEPR Theory) © 2014 Pearson Education, Inc.

Learning Check Use VSEPR theory to predict the shape of N 2 O. © 2014 Pearson Education, Inc.

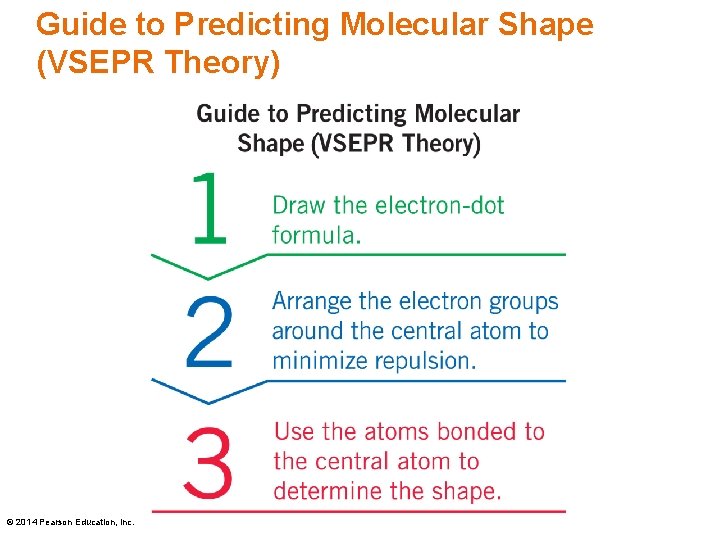
Solution Use VSEPR theory to predict the shape of N 2 O. Step 1 Draw the electron-dot formula. In the electron-dot structure with 16 e−, octets are acquired using two double bonds in the central N atom. © 2014 Pearson Education, Inc.

Solution Use VSEPR theory to predict the shape of N 2 O. Step 2 Arrange the electron groups around the central atom to minimize repulsion. Repulsion is minimized with two electron groups at 180°, a linear arrangement. © 2014 Pearson Education, Inc.

Solution Use VSEPR theory to predict the shape of N 2 O. Step 3 Use the atoms bonded to the central atom to determine the molecular shape. The shape of a N 2 O molecule with two bonded atoms and no lone pairs on the central N is linear. © 2014 Pearson Education, Inc.

Learning Check State the number of electron groups, lone pairs, and bonded atoms, and use VSEPR theory to determine the shape of the following molecules: (1) tetrahedral (2) trigonal pyramidal (3) bent A. PF 3 B. H 2 S C. CCl 4 D. PO 43– © 2014 Pearson Education, Inc.

Solution State the number of electron groups, lone pairs, and bonded atoms, and use VSEPR theory to determine the shape of the following molecules: (1) tetrahedral (2) trigonal pyramidal (3) bent A. PF 3 4 electron groups, 3 bonded atoms, 1 lone pair, trigonal pyramidal (2) B. H 2 S 4 electron groups, 2 bonded atoms, 2 lone pairs, bent (3) © 2014 Pearson Education, Inc.

Solution State the number of electron groups, lone pairs, and bonded atoms, and use VSEPR theory to determine the shape of the following molecules: (1) tetrahedral (2) trigonal pyramidal (3) bent C. CCl 4 4 electron groups, 4 bonded atoms, 0 lone pairs, tetrahedral (1) D. PO 43– 4 electron groups, 4 bonded atoms, 0 lone pairs, tetrahedral (1) © 2014 Pearson Education, Inc.
- Slides: 19