Chapter 10 Holt Chemistry SOL Mr Bollinger FDHS

Chapter 10 Holt Chemistry SOL Mr. Bollinger FDHS Student Notes

What is Energy? • Energy: the ability to do work or produce heat • There are many forms of Energy: – Potential Energy: Stored energy, often in chemical bonds – Kinetic Energy: Energy of motion, • Units of Energy: – Joules (J), and calories (cal)

Law of Conservation of Energy • In any chemical reaction, energy can be converted from one form to another, but cannot be created nor destroyed. • This Unit covers Thermodynamics: – Thermo: heat/temperature – Dynamics: properties, behavior • However, heat and temperature have very different meanings

Temperature • A measure of how hot or cold something is • Measures the average kinetic energy of all the particles in an object • Measured in degrees Celsius, Kelvin, and degrees Fahrenheit • Temperature is an Intensive property; meaning that is doesn’t matter how much of the substance there is

Heat (Q) • The energy transferred between objects because of their difference in temperature. • Energy always flows from warmer to cooler objects • Because it is a transfer of energy, we measure heat in Joules (J) or Calories (cal) • Heat is an extensive property, meaning that the measurement is dependent on the amount of the sample

Energy Units: 1 Joule (J) = 0. 2390 calories (cal) 1 calorie (cal) = 4. 184 Joules (J) 1 kilojoule (k. J) = 1000 Joules (J) 1 Calorie (Cal) = 1 kilocalorie (kcal) = 1000 calories (cal)

Practice converting Joules & Calories • When doing conversions, we use conversion factors: Use fractions! • In this case we can use a T-chart or multiplication: 207 J = ____ calories? 195, 300 calories = _____ kilo. Joules (k. J)?

Enthalpy (H) • The heat content of a system at constant pressure • This measurement includes the kinetic energy of all of its particles – Finding the exact enthalpy of something is impossible to know exactly, so instead we mostly use…. • Change in Enthalpy or ΔH • In a reaction: ΔHrxn = ΔHproducts - ΔHreactants

Heat capacity (c) • The amount of heat energy needed to increase the temperature of a substance by 10 C • Every substance can have a different heat capacity. • Heat capacity is influenced by the type of substance, and the amount of substance

Specific Heat Capacity (c) • The same thing, but for each specific material. • The higher a material’s specific heat, the more energy needed to change the temperature. • Common Specific Heats (J/g 0 C) – Water (l) = 4. 184 (or 1 cal/g 0 C) – Water (s) = 2. 03 – Water (g) = 2. 01 – Aluminum = 0. 897 – Iron = 0. 449

We can use specific heat to calculate the heat (q) Q= m c ΔT Q = heat in Joules (J) or calories (cal) m = mass of the substance (grams) c = specific heat capacity (J/g*C or cal/g*0 C) ΔT = change in temperature: Tfinal – Tinitial (0 C)

• How much heat is needed to change the temperature of 12 g of silver with a specific heat of 0. 057 cal/g C from 25 C to 83 C? • Q = m c T • m = 12 g • c = 0. 057 cal/g C • T = 58 C (no need to change to Kelvin)

• If you put 6500 J of heat into a 15 g piece of Al at 25 C , what will the final temperature be? ( c = 0. 90 J/g C ) • Q = m c T • m = 15 g • c = 0. 90 J/g C • Q = 6500 J
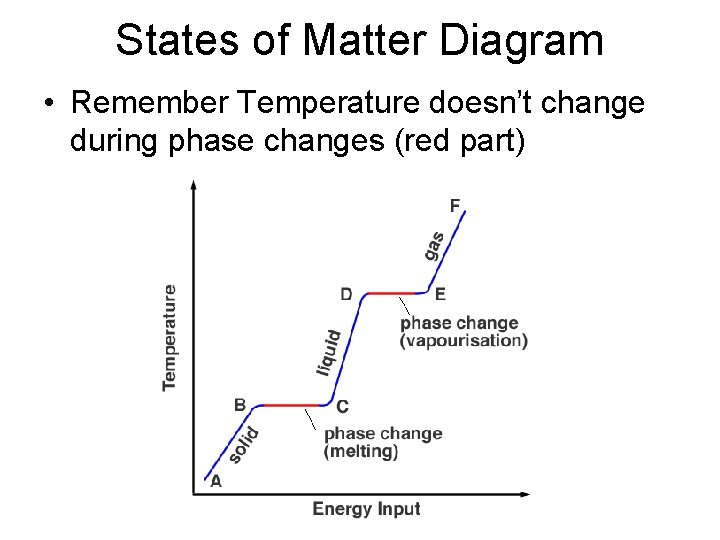
States of Matter Diagram • Remember Temperature doesn’t change during phase changes (red part)

• We have the formula for determing the amount of energy to change the temperature while in a phase (blue lines) Q = m c ΔT Or ΔH = m c ΔT • This applies to each section where the phase is the same
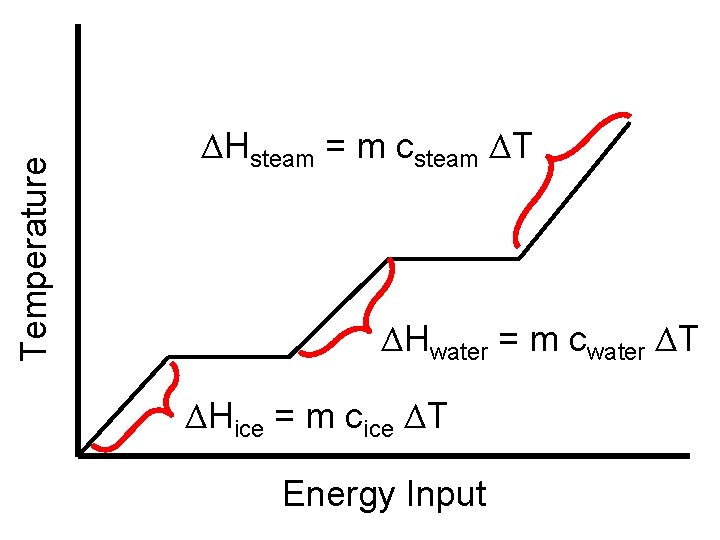
Temperature ΔHsteam = m csteam ΔT ΔHwater = m cwater ΔT ΔHice = m cice ΔT Energy Input

What about the phase changes? • Latent Heat: heat absorbed or released by a substance during a process that does not involve a change in temperature • Solid Liquid = Latent Heat of fusion (Lf) – OR ΔHfus = ΔHf = Heat of fusion • Liquid Gas = Latent Heat of vaporization (Lv) – OR ΔHvap = ΔHv = Heat of vaporization

Calculating Latent Heat ΔH = m ΔHfus AND ΔH = m ΔHvap • Just like specific heat, different materials have different ΔHfus and ΔHvap. For water: • ΔHfus = 334 J/g • ΔHvap = 2260 J/g
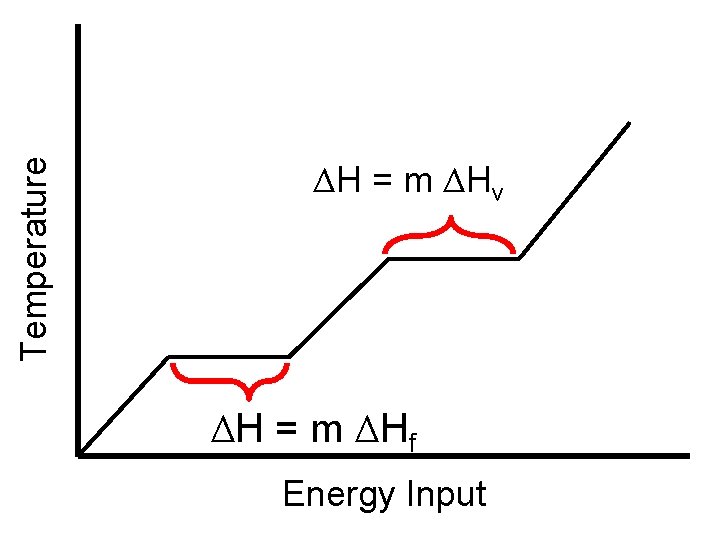
Temperature ΔH = m ΔHv ΔH = m ΔHf Energy Input

Temperature ΔHsteam = m csteam ΔT ΔH = m ΔHv ΔH = m ΔHf ΔHwater = m cwater ΔT ΔHice = m cice ΔT Energy Input
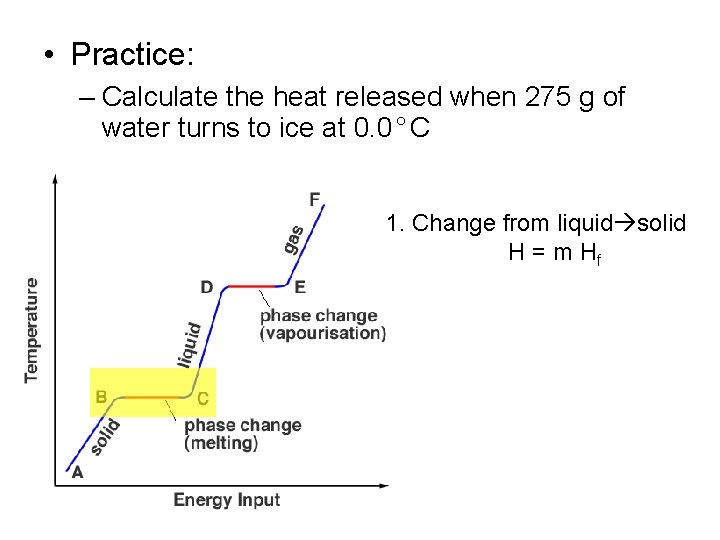
• Practice: – Calculate the heat released when 275 g of water turns to ice at 0. 0°C 1. Change from liquid solid H = m Hf

• Practice: – Calculate the amount of heat released to convert 137 g of steam at 135°C to water at 53°C. 1. Change in Temp of Steam H = m c ΔT 2. Change from gas liquid H = m Hv 3. Change in Temp of liquid H = m c ΔT

Calorimetry • The experimental measurement of an enthalpy change of a reaction • We can use calorimetry to determine ΔH, which can be used interchangeably with heat (Q) at constant pressure. • A calorimeter is the device used to measure the energy change. – It accomplishes this by measuring the temperature before and after.
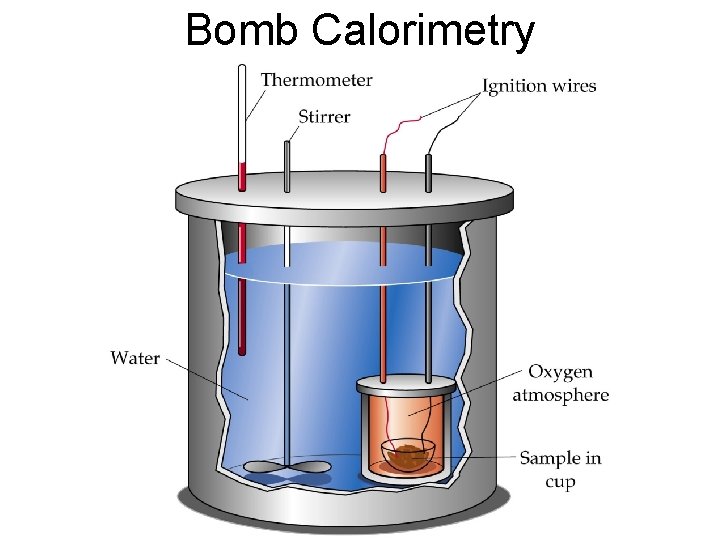
Bomb Calorimetry


• We then use the change in temperature, the mass of water, and the specific heat of water to calculate how much energy we produced. • Bomb Calorimetry is used by nutritionists to determining the caloric content of foods.

Exothermic vs. Endothermic Exothermic: • Change in matter where E is released, often as heat • Surroundings get hotter • Q or H of the system is negative Endothermic: • Change in matter where E is absorbed, often as heat • Surroundings get cooler • Q or H of the system is positive
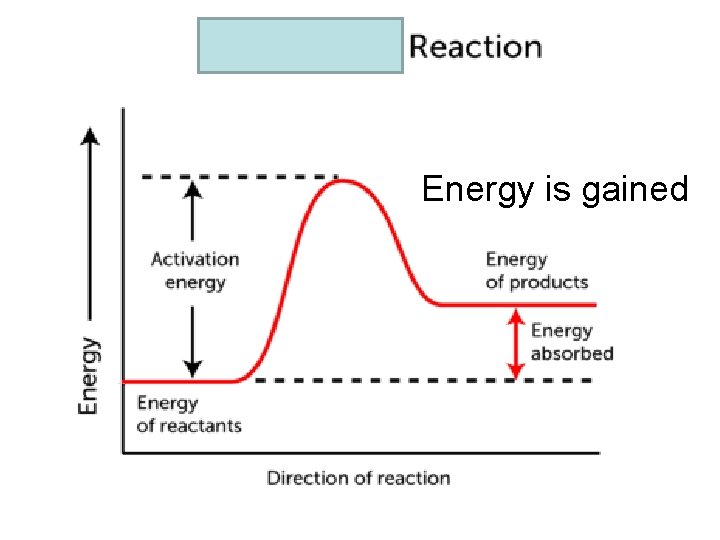
Energy is gained
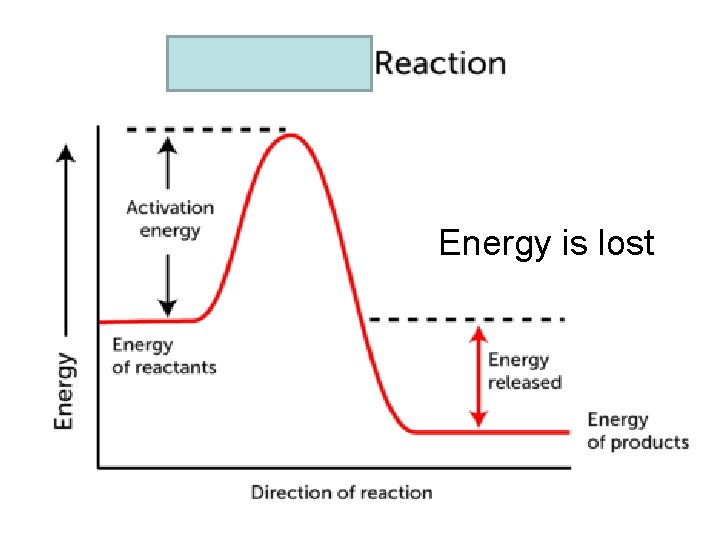
Energy is lost

Exactly how much Energy is exchanged? • One way to find out is with Calorimetry! • Another uses known ΔH value from other reactions to put together the reaction you are looking at • Hess’s Law: – The amount of heat released or absorbed (ΔH) for a reaction is the same if it takes one, or several steps – The path a reaction takes doesn’t matter, it only matters where you started, and where you end.
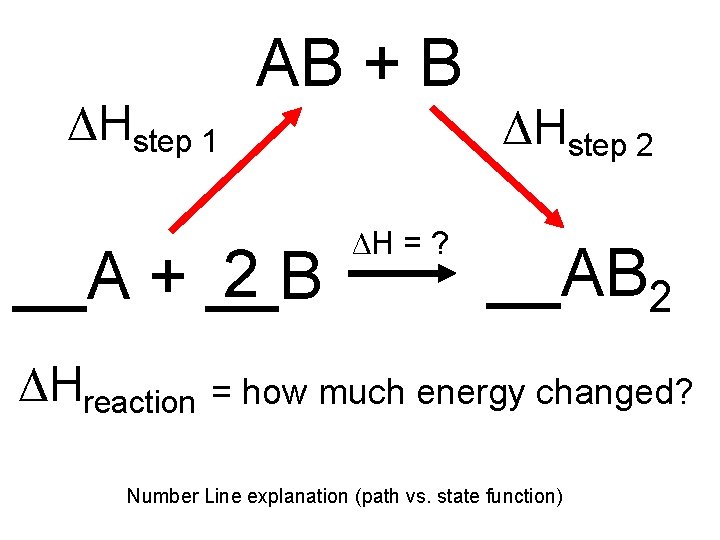
ΔHstep 1 AB + B 2 __A + __B ΔHreaction ΔHstep 2 ΔH = ? __AB 2 = how much energy changed? Number Line explanation (path vs. state function)

• We might not know the Enthalpy (ΔH) for a reaction, be we can put the pieces together using math! • Sometimes, Enthalpy is negative, so it all evens out in the end. • If I gave you: ΔHstep 1 = 5 J ΔHstep 2 = -1. 5 J • ΔHreaction = Step 1 then Step 2 = (5 Joules) + (-1. 5 Joules) = 3. 5 Joules

Enthalpies of Formation • Enthalpy of Formation: – Heat lost or gained when 1 mole of a compound is formed from its constituent elements • Each compound can be broken down into its simplest elements: H 2 O H 2 + O 2 The ΔH values are given in tables in your textbook.

• Calculate the Enthalpy of reaction using: ΔHreaction = Σ ΔHproducts - Σ ΔHreactants Find the ΔH for the reaction below given the Standard Enthalpies of Formation: C(s) + CO 2(g) 2 CO(g) ΔH = ? Enthalpies of Formation: C(s) = 0 k. J/mol CO 2(g) = -393. 5 k. J/mol CO(g) = -110. 5 k. J/mol Multipl y by ho w many m oles there a re!

Steps for Hess’s Law • Write the equations needed to make up the target equation when added up • When a substance is on the left and right side (even from different equations) they cancel out • You can multiply and divide the whole equations • You can flip the entire equation • Write what you did next to the equation • What you do to the equation, you do to the ΔH value

Example: • From the known enthalpy changes: Cu + Cl 2 Cu. Cl 2 ΔH = -206 k. J 2 Cu + Cl 2 2 Cu. Cl ΔH = -136 k. J Find the enthalpy change of: Cu. Cl 2 + Cu 2 Cu. Cl ΔH = ? Cu. Cl 2 Cu + Cl 2 Flipped 2 Cu + Cl 2 2 Cu. Cl Same Cu. Cl 2 + Cu 2 Cu. Cl

Cu. Cl 2 Cu + Cl 2 Flipped ΔH = (-206 k. J) x -1 2 Cu + Cl 2 2 Cu. Cl Same Cu. Cl 2 + Cu 2 Cu. Cl ΔH = -136 k. J ΔHreaction = (206 k. J) + (-136 k. J) = 70 k. J

Example: • I will give you the equations and enthalpies: – Rearrange the equations so that the sum is equal to following: C + H 2 O CO + H 2 C + O 2 CO 2 ΔH = -395. 3 k. J 2 CO + O 2 2 CO 2 ΔH = -566. 0 k. J 2 H 2 + O 2 2 H 2 O ΔH = -483. 6 k. J
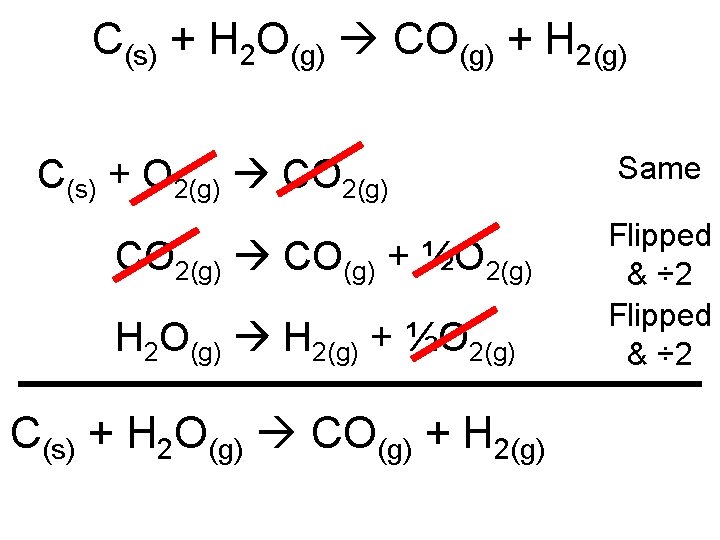
C(s) + H 2 O(g) CO(g) + H 2(g) C(s) + O 2(g) CO(g) + ½O 2(g) H 2 O(g) H 2(g) + ½O 2(g) C(s) + H 2 O(g) CO(g) + H 2(g) Same Flipped & ÷ 2

ΔH = -395. 3 k. J ΔH = -566. 0 k. J ΔH = -483. 6 k. J Same Flipped & ÷ 2 = -395. 3 k. J = +283. 0 k. J = +241. 8 k. J ΔHreaction = 129. 5 k. J

• Hess’s Law video: – https: //www. brightstorm. com/science/chemistr y/thermochemistry/hesss-law/

Entropy (S) • Entropy is the measure of disorder in a system. It is a measure of randomness – It is not a form of energy, but has the unit Joules/Kelvin (J/K) • A process (or reaction) is more likely to occur if entropy increases (meaning randomness increases) – (This is considered the 2 nd Law of Thermodynamics: entropy of spontaneous processes always increase)

Disorder & Randomness • What we call “order” is just the specific circumstance we choose. • Think about cleaning your room. – When your room is “ordered” or clean, that is just because your definition of clean is for clothes to be put away, sheets to be covering the bed, etc. – That is just one possible configuration of all the stuff in your room. – We call each configuration a “microstate”

How many possible “microstates” are there? • When I have 1 magnet, and 3 circles, how many different microstates are there? • When I have 2 magnets and 3 circles, how many different microstates are there? • When I have 3 magnets and 3 circles, how many different microstates are there?

• For the example with 3 magnets: How many microstates have: 1 microstate – 3 magnets in circle 1? 6 microstates – 2 magnets in circle 1? – 1 magnet in circle 1? 12 microstates – 0 magnets in circle 1? 8 microstates • It was therefore a higher probability for the magnets to spread out to each circle! • The universe follows probabilities; entropy increases (without adding energy/work)

Factors that affect Entropy (S): • Diffusion: dispersing increases entropy • Dilution: more space = increase entropy • Increased Temperature: move faster, increased entropy • Phase change: Gas is more disordered than liquids. Liquids are usually more disordered than solids • In reactions: Increase in number of moles of gas means increased Entropy 2 Na(s) + Cl 2(g) 2 Na. Cl(s) ∆S = -181 J/K

• The tendency for a reaction to occur depends on both ∆H and ∆S. • A reaction is more likely to occur if – ∆H is negative (the system loses energy) – ∆S is positive (the system becomes more random) • What happens if we have to deal with ∆H and ∆S at the same time? – What if ∆H is negative, but ∆S is also negative?

Gibbs Energy (G) • Gibbs energy = the free energy: energy available in a system to do work. • It can help us determine if a process is spontaneous or not. • When the change in Gibbs energy (ΔG) is: – Negative: the reaction is spontaneous – Positive: the reaction is NOT spontaneous – Zero: the reaction is at equilibrium

Spontaneous Reactions: • Does NOT mean random • In chemistry, spontaneous reactions are ones that are able, or do occur without assistance – No catalyst, or additional energy added. • An avalanche is a spontaneous occurance, because it may or may not occur, but it always can occur.

Back to Gibbs: • Gibbs Energy, and the change in Gibbs (ΔG) energy can be found by using Enthalpy (ΔH) and Entropy (ΔS) as well as Temperature (T) ΔG = ΔH – T ΔS • Given the values for change in enthalpy, temperature, and change in entropy, we can determine the free energy (ΔG) • Since Entropy (S) is in J/K, temperature must also be in Kelvin

Practice: • Find the free energy for the reaction below at 75°C, and determine if it is spontaneous ΔH = -392 k. J ΔS = 0. 047 k. J/K 2 K(s) + 2 H 2 O(l) 2 K+(aq) + H 2(g) Remember: ΔG = ΔH – T ΔS ΔG = (-392 k. J) – (75°C) (0. 047 k. J/K) ΔG = (-392 k. J) – (348 K) (0. 047 k. J/K) = = - 408. 4 k. J Negative = spontaneous

Think-Pair-Share • Try the next problem on your own for 2 min. • Then work with a neighbor for 1 minute • Then be prepared to share your answer with the class Determine Gibbs free energy of the reaction at 25 degrees Celsius: CH 3 OH(l) + 3/2 O 2(g) CO 2(g) + 2 H 2 O(g) ΔH = -638. 4 k. J ΔS = 156. 9 J/K

What do you need to know? • Energy Conversions (Joule calorie, etc. ) • Temperature vs. Heat • Calculating heat (from phase diagram) – During a phase change (ΔH=mΔHfus or ΔH=mΔHvap) – While in a phase (solid/liquid/gas ΔH = m c ΔT) • Be able to rearrange equations to solve for mass(m), specific heat(c), or temp change (ΔT). • Label a phase change diagram • Identify if a reaction is Exo or Endothermic

Continued…. • Know how a calorimeter works • Use Hess’s law to rearrange and find ΔHreaction • Find ΔHreaction with = ΔHproducts – ΔHreactants • Know what Entropy is (S) • Be able to explain factors influencing Entropy • Know what Gibbs energy is (G) • Calculate Gibbs energy with H and S – Don’t forget temperature must be in Kelvin – Make sure energy units are same (both J or k. J)

I will provide: • All equations, and some we haven’t used • Specific heat values (c) for water and others • Heat of fusion & vaporization #s (ΔHf & ΔHv) • Any Enthalpy (ΔH) Entropy (ΔS), and Gibbs energy (ΔG) that are needed.
- Slides: 55