Chapter 10 Gases Overview u Pressure o Barometer

Chapter 10: Gases

Overview u Pressure o. Barometer & Atmospheric Pressure u u Standard Conditions Gas Laws o. Boyle’s Law o. Charles’ Law o. Avogadro’s Law o. Ideal Gas Law

u u Gas Laws under Two Conditions Gas Densities Darlton’s Law of Partial Pressure Kinetic Molecular Theory o. Molecular Effusion/Diffusion o. Graham’s Law u Deviation from Ideality

Characteristics u Solids ohave own shape and volume oparticles close together with strong interaction u Liquids ohave own volume but assume shape of container oparticles farther apart but have moderate interaction u Gases oassume shape and volume of container oparticles far apart with little/no interaction ohighly compressible

Pressure u P = F/A o. Force in Newtons o. Area in m 2 u Barometer o. P in N/m 2 = Pascal unit o 1 x 105 N/m 2 = 1 x 105 Pa or 100 k. Pa u Standard Pressure o 1 atm = 760 mm Hg = 1. 01325 x 105 Pa = 101. 325 k. Pa (or torr)

force of the atmosphere force of the column h when atmospheric force equals the force of the column the atmospheric pressure is measured as “h”

Gas Laws u Boyle’s Law o. P µ 1/V constant T, n ovolume increases as pressure decreases u Charles’ Law o. V µ T constant P, n ovolume increases as temperature increases u Avogadro’s Law o. V µ n constant P, T ovolume increases as moles of gas (n) increases

u Ideal Gas Law ocombines all gas laws PV = n. RT • R = 0. 0821 L-atm mol-K • any volumes must be in liters • any temperatures must be in kelvin • any pressures must be in atmospheres o. STP or SC -- standard temperature/pressure • P = 1 atm (same as 760 mm Hg) • T = 273 K (same as 0° C)

u Problem 10. 3: A flashbulb contains 2. 4 x 10 -4 mol of O 2 gas at 1. 9 atm and 19°C. What is the volulme? • PV = n. RT or • V = 2. 4 x 10 -4 mol V = 3. 0 x 10 -3 L V = n. RT P x 0. 0821 L-atm x mol-K 1. 9 atm or 3. 0 m. L or 292 K 3. 0 cm 3

Gas Laws Under Two Conditions u u P 1 V 1 = P 2 V 2 T 1 T 2 Problem 10. 4: Pressure in a tank is kept at 2. 20 atm. When the temp. is -15°C the volume is 28, 500 ft 3. What is the volume is the temp. is 31°C o. P 1 = P 2 = 2. 20 atm T 1 = 258 K V 1 = 28, 500 ft 3 T 2 = 304 K o V 2 = P 1 V 1 T 2 P 2 T 1 o. V 2 = 28, 500 ft 3 x 304 K = 33, 600 ft 3 258 K
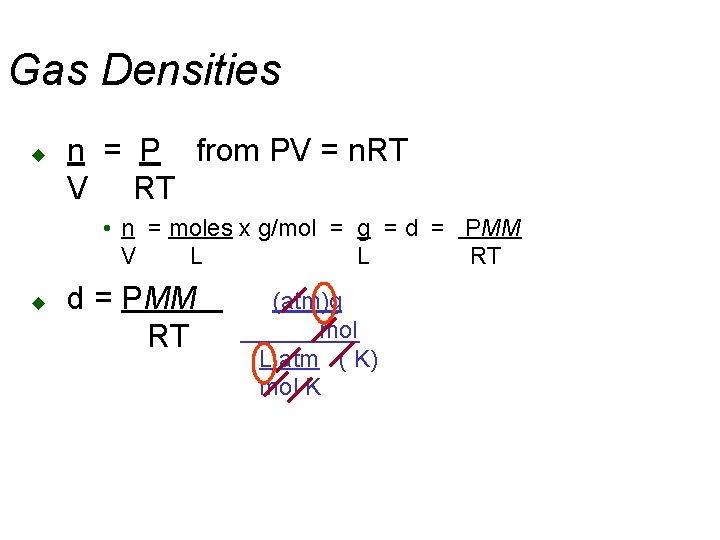
Gas Densities u n = P from PV = n. RT V RT • n = moles x g/mol = g = d = PMM V L L RT u d = PMM RT (atm)g mol L atm ( K) mol K

Dalton’s Law of Partial Pressures u total pressure of a mixture = sum of each partial pressure o. P T = P 1 + P 2 + P 3. . u each partial pressure = the pressure each gas would have if it were alone o. P 1 = n 1 RT V 1 P 2 = n 2 RT V 2 P 3 = n 3 RT V 3 o. PT = n 1 RT + n 2 RT + n 3 RT = (n 1 + n 2 + n 3) RT V 1 V 2 V 3 V volumes are the same

o. P 1 = n 1 therefore PT n. T P 1 = n 1 P T n 1 = X 1 mole fraction n. T P 1 = X 1 P T

Kinetic Molecular Theory u u u Gases consist of particles in constant, random motion Volume of gas particles is negligible Attractive and repulsive forces are negligible Average kinetic energy is proportional to temperature Collisions are elastic

u molecular speed ou = root mean square speed or speed of molecule with average kinetic energy o • R is the gas constant (8. 314 J/mol-K), T is temp. in K & MM is molar mass o What is the rms speed of an He atom at 25°C? • u = (3 x 8. 314 kg-m 2/s 2 -mol-K x 298 K)1/2 ( 4. 00 x 10 -3 kg/mol ) • u= 1. 36 x 103 m/s
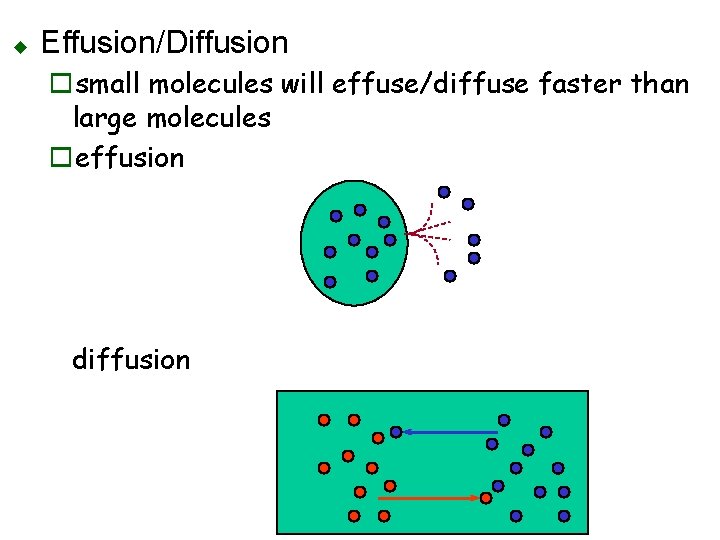
u Effusion/Diffusion osmall molecules will effuse/diffuse faster than large molecules oeffusion diffusion

u Graham’s Law where r is rate of speed & MM is the molar mass o o. Problem 10. 14: Calculate the ratio of the effusion rates of N 2 and O 2. r. N 2 = 1. 07 r. O 2

Deviation from Ideality u u Occurs at very high pressure or very low temperature Correction due to volume oideal law assumes molecules have no volume • for molecules which are far apart, this is a good assumption omust correct for the volume of the molecules themselves
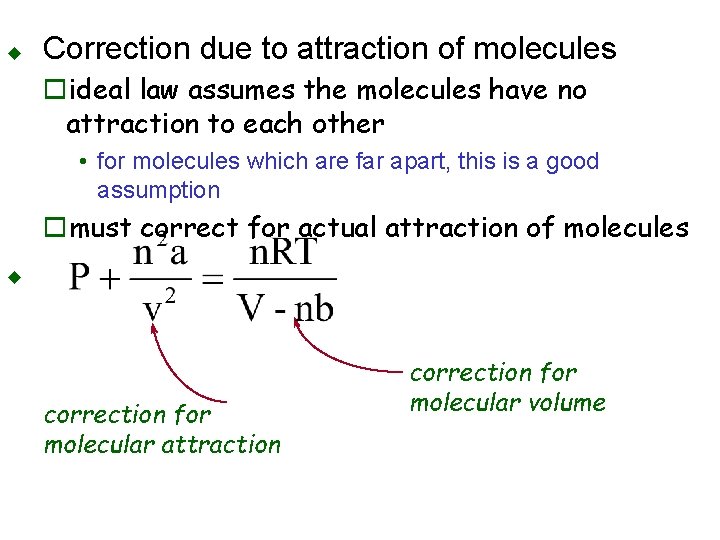
u Correction due to attraction of molecules oideal law assumes the molecules have no attraction to each other • for molecules which are far apart, this is a good assumption omust correct for actual attraction of molecules u correction for molecular attraction correction for molecular volume
- Slides: 19