Chapter 10 Chemical Quantities Yes you will need



































- Slides: 36

Chapter 10 “Chemical Quantities” Yes, you will need a calculator for this chapter! 1

Section 10. 1 Da Mole: A Measurement of Matter 2

How do we measure items? 3 § Measure mass in grams. § Measure volume in liters. § Measure amount in MOLES.

What is the mole? 4 We’re not talking about this kind of mole!

Moles (mol) § It is an amount. Defined as the number of carbon atoms in exactly 12 grams of carbon-12. 1 mole = 6. 02 x 1023 of representative particles. 6. 02 x 1023 is called: Avogadro’s number. § 1 mole = 6. 02 x 1023 = Avogadro’s number § § § 5

Similar Words for amount Dozen: § § § 6 1 dozen muffins = 12 muffins 2 dozen puppies = 24 puppies 36 m&m stuffed donuts = 3 dozen donuts Mole: § 1 mol muffins = 6. 02 x 1023 muffins § 2 mol puppies = 1. 2 x 1024 puppies § 2. 4 x 1024 m&m stuffed donuts = 4 mol donuts

What are Representative Particles? The smallest pieces of a substance: § § n 7 Changes based on what we’re looking at 1) For a molecular compound: it is the molecule. 2) For an ionic compound: it is the formula unit (ex: Na. Cl, Mg. S). 3) For an element: it is the atom. 1 mol of CO 2 molecules, 1 mol of Na. Cl formula units, and 1 mol of H atoms all equal 6. 02 x 1023 of that thing.

Measuring Moles § The atomic mass on the PT is also the molar mass (g/mol) – mass (in grams) of 1 mole of that atom § 8 = 16. 0 g/mol

Find the Molar Mass (g/mol) of the following: 9 n Nitrogen 14. 01 g/mol n Aluminum 26. 98 g/mol n Zinc 65. 39 g/mol

What about compound mass? In 1 mole of H 2 O, there are 2 moles of H atoms and 1 mole of O atoms § To find the mass of a compound: – Determine number of each element present – Multiply the number times their mass (from the periodic table) – Add them up for the total mass 10

Calculating Compound Mass Calculate the mass of magnesium carbonate, Mg. CO 3. 24. 31 g/mol + 12. 01 g/mol + 3 x (16. 00 g/mol) = 84. 32 g/mol Mg. CO 3 11

Practice Problem: n What is the mass of one mole of CH 4? 1 mole of C = 12. 01 g/mol 4 mole of H x 1. 01 g = 4. 04 g/mol 1 mole CH 4 = 12. 01 + 4. 04 = 16. 05 g/mol 12

Mole Practice 1. 2. 1 mol = particles 2. 5 mol Mg. Cl 2 = ? particles Mg. Cl 2 2. 5 mol 24 1. 5 x 10 6. 02 x 1023 particles = particles 1 mol of Mg. Cl 2 3. 7. 2 x 1024 Fe particles = ? mol Fe 7. 2 x 1024 particles 13 1 mol 6. 02 x 1023 particles = 12 mol Fe

Molar Mass Practice 4. Molar mass of Be. F 2 = Be = 9. 0 g/mol F = 19. 0 g/mol 9. 0 + 2(19. 0) = 47 g/mol 5. Molar mass of C 6 H 12 O 6 = C = 12. 0 g/mol H = 1. 0 g/mol O= 16. 0 g/mol 14 6(12. 0) + 12(1. 0) + 6(16. 0) = 180 g/mol

Section 10. 2 Mole-Mass and Mole-Volume Relationships 15

Since Molar Mass is… § Number of grams in 1 mole. § grams per mole (g/mol) § Use to make conversion factors from these. - Use molar mass to convert to grams or mols of a substance. 16

For example n How many moles is 5. 69 g of Na. OH? 5. 69 g Na. OH n 1 mol Na. OH =. 142 mol Na. OH 40. 0 g Na. OH How many grams in. 53 mol of Na. OH? . 53 mol Na. OH 17 21. 2 g 40. 0 g Na. OH = Na. OH mol Na. OH 1

Practice Problems: § How much would 2. 34 moles of carbon weigh? 28. 1 grams C § How many moles of magnesium is 24. 31 g of Mg? 1. 000 mol Mg 18

The Mole-Volume Relationship § Under different circumstances, gases can change. § Two things effect the volume of a gas: a) Temperature and b) Pressure **We need to compare all gases at the same temperature and pressure. 19

Standard Temperature and Pressure n abbreviated STP n 0ºC (273 K) and 1. 0 atm pressure n At STP 1 mole of gas occupies 22. 4 L= molar volume • 22. 4 L/mol of any gas at STP 20

Practice Problems: § What is the volume of 4. 59 mole of CO 2 gas at STP? 4. 59 mol CO 2 § 22. 4 L CO 2 1 mol CO 2 = 103 L CO 2 How many moles is 5. 67 L of O 2 at STP? 5. 67 L O 2 1 mol O 2 = 0. 253 mol O 2 22. 4 L O 2 21

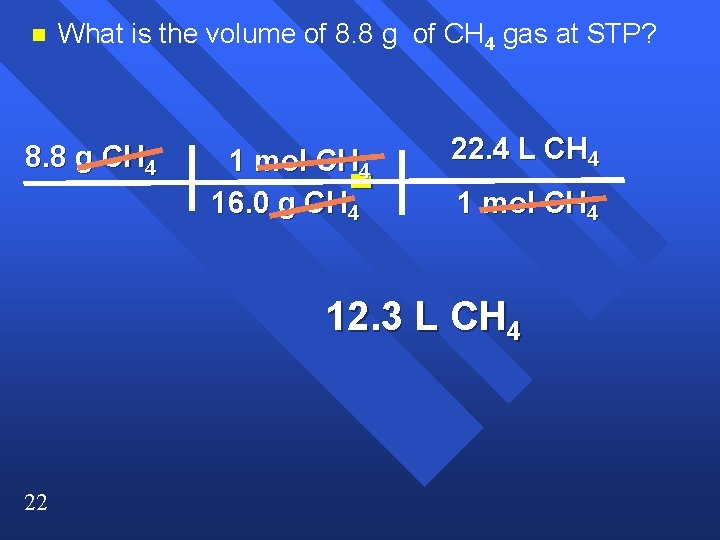
n What is the volume of 8. 8 g of CH 4 gas at STP? 8. 8 g CH 4 1 mol CH 4 = 16. 0 g CH 4 22. 4 L CH 4 1 mol CH 4 12. 3 L CH 4 22

Summary: n These four items are all equal: a) 1 mole b) molar mass (in grams/mol) c) 6. 02 x 1023 particles (atoms, molecules, or formula units) d) 22. 4 L of a gas at STP **Thus, we can make conversion factors from them. 23

Avog. # Practice problems: § How many molecules of CO 2 are in 4. 6 moles of CO 2? 2. 8 x 1024 molecules of CO 2 § How many moles of water is in 5. 87 x 1022 molecules? 0. 0975 mol H 2 O (or 9. 75 x 10 -2) § How many atoms of carbon are in 1. 230 moles of Carbon? 7. 405 x 1023 atoms C § How many moles is 7. 78 x 1024 formula units of Mg. Cl 2? 12. 9 moles Mg. Cl 2 24

Mixed Practice Problems: § How many atoms of lithium is 1. 0 g of Li? 8. 7 x 1022 atoms Li § How much would 3. 45 x 1022 atoms of U weigh? 13. 6 g U § 25 What is the volume of 10. 0 g of CH 4 gas at STP? 14. 0 L CH 4

Section 10. 3 Percent Composition and Chemical Formulas 26

Percentage Composition n Percentage by mass of each element in a compound 27

Calculating Percent Composition of a Compound § Like all percent problems: part x 100 % = percent whole 1) Find mass of each element (from the periodic table) 2) Next, divide by total mass of compound; then x 100 28

Percentage Composition n Find the % composition of Cu 2 S. %Cu = %S = 29 127. 10 g Cu 159. 17 g Cu 2 S 32. 07 g S 159. 17 g Cu 2 S 100 = 79. 85% Cu 100 = 20. 15% S

Percentage Composition n Find the mass percentage of water in calcium chloride dihydrate, Ca. Cl 2 • 2 H 2 O? %H 2 O = 36. 04 g 100 =24. 51% H 2 O 147. 02 g 30

Empirical Formula (EF) n Lowest whole number ratio of atoms in a compound C 2 H 6 simplify subscripts 31 CH 3

Empirical Formula (EF) § Just find lowest whole number ratio C 6 H 12 O 6 = CH 2 O CH 4 N = this is already the lowest ratio. § Formula is not just ratio of atoms, it is also ratio of moles. § In 1 mole of CO 2 there is 1 mole of carbon and 2 moles of oxygen. § In one molecule of CO 2 there is 1 atom 32 of C and 2 atoms of O.

Formulas (continued) Formulas for ionic compounds are ALWAYS empirical (the lowest whole number ratio = cannot be reduced). Examples: Na. Cl Mg. Cl 2 Al 2(SO 4)3 *Remember, we simplify beforehand 33 K 2 CO 3

Formulas (continued) Formulas for molecular compounds MIGHT be empirical (lowest whole number ratio). Molecular: (Correct formula) Empirical: (Lowest whole number ratio) 34 H 2 O C 6 H 12 O 6 C 12 H 22 O 11 H 2 O CH 2 O C 12 H 22 O 11

Formulas n Empirical Formula (EF) = lowest whole number ratio of elements in a compound. n Molecular Formula (MF) = the actual ratio of elements in a compound. n The two can be the same. n CH 2 = EF C 2 H 4 = MF n C 3 H 6 = MF H 2 O = EF 35

Last extra practice slide n How many O atoms are in 5 mol of Al 2 O 3? 5 mol Al 2 O 3 3 mol O 1 mol Al 2 O 3 6. 02 e 23 atoms O 1 mol O 9. 03 e 24 atoms O n How many Al atoms are in 5 mol of Al 2 O 3? 5 mol Al 2 O 3 2 mol Al 1 mol Al 2 O 3 36 6. 02 e 23 atoms Al 1 mol Al 6. 02 e 24 atoms Al