Chapter 10 Alkanes and Aromatic Hydrocarbons Cycloalkanes Aromatic

Chapter 10 Alkanes and Aromatic Hydrocarbons Cycloalkanes Aromatic Hydrocarbons Timberlake Lecture. PLUS 1999 1
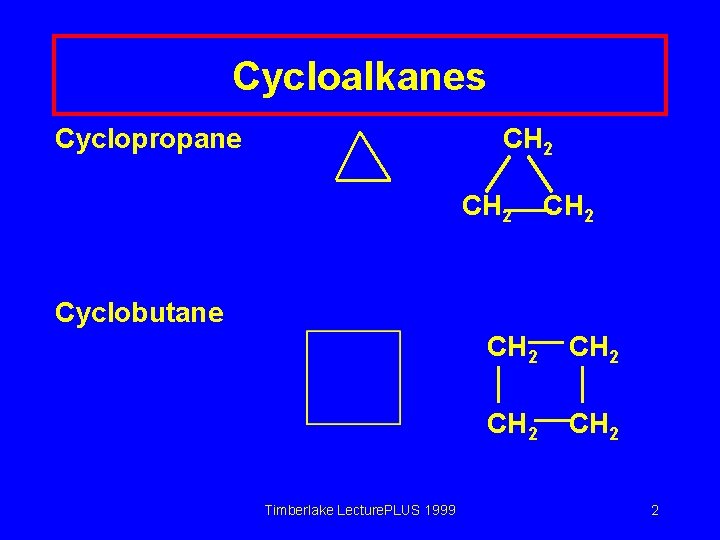
Cycloalkanes Cyclopropane CH 2 Cyclobutane Timberlake Lecture. PLUS 1999 CH 2 2
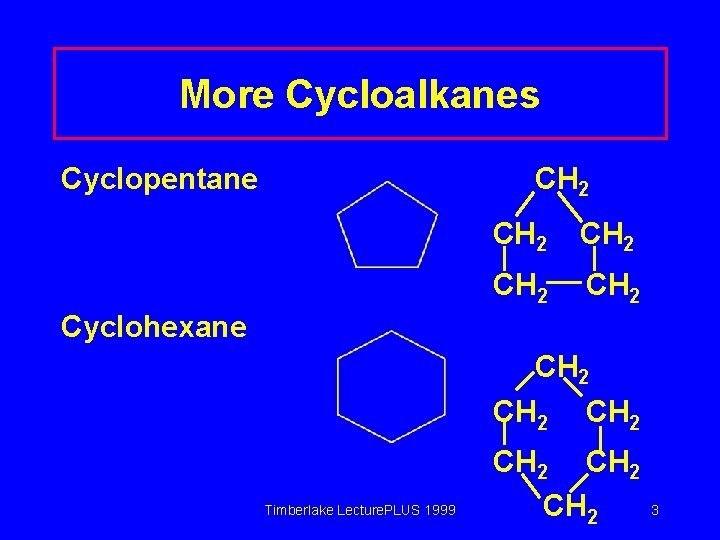
More Cycloalkanes Cyclopentane CH 2 CH 2 Cyclohexane CH 2 Timberlake Lecture. PLUS 1999 CH 2 3

Naming Cycloalkanes with Side Groups Number of side groups One Two Naming Side group name goes in front of the cycloalkane name. Number the ring in the direction that gives the lowest numbers to the side groups. Timberlake Lecture. PLUS 1999 4
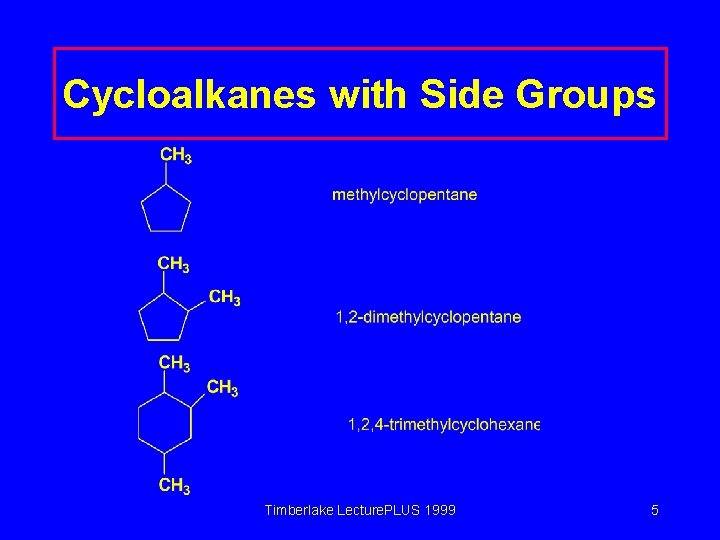
Cycloalkanes with Side Groups Timberlake Lecture. PLUS 1999 5
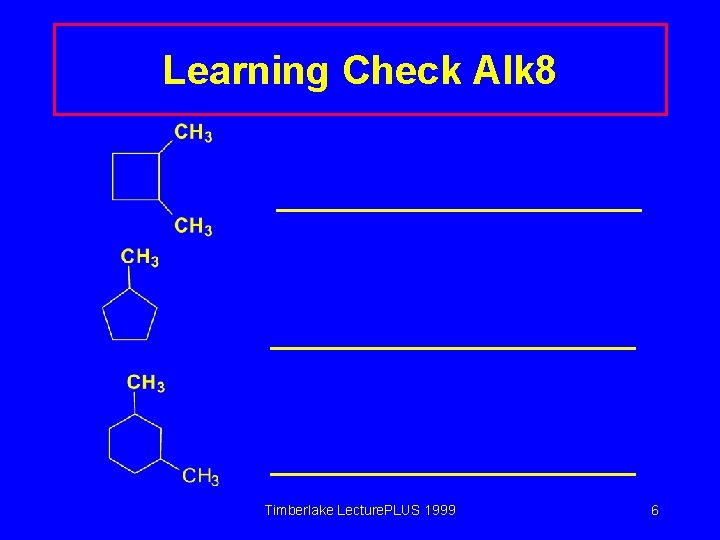
Learning Check Alk 8 Timberlake Lecture. PLUS 1999 6
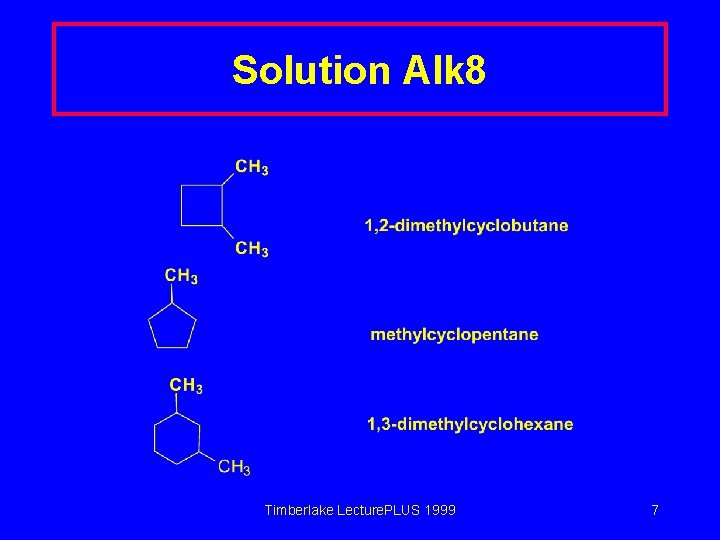
Solution Alk 8 Timberlake Lecture. PLUS 1999 7

Aromatic Compounds and Benzene Aromatic compounds contain benzene. Benzene, C 6 H 6 , is represented as a six carbon ring with 3 double bonds. Two possible can be drawn to show benzene in this form. Timberlake Lecture. PLUS 1999 8

Benzene Structure The structures for benzene can also be written as a single structure where the alternating double bonds are written as a circle within the ring. Benzene structure Timberlake Lecture. PLUS 1999 9

Aromatic Compounds in Nature and Health Many aromatic compounds are common in nature and in medicine. Timberlake Lecture. PLUS 1999 10

Naming Aromatic Compounds Aromatic compounds are named with benzene as the parent chain. One side group is named in front of the name benzene. methylbenzene (toluene) chlorobenzene Timberlake Lecture. PLUS 1999 11

Naming Aromatic Compounds When two groups are attached to benzene, the ring is numbered to give the lower numbers to the side groups. The prefixes ortho (1, 2), meta (1, 3 -) and para (1, 4 -) are also used. Timberlake Lecture. PLUS 1999 12

Some Common Names Some substituted benzene rings also use a common name. Then naming with additional more side groups uses the ortho-, meta-, para- system. Timberlake Lecture. PLUS 1999 13
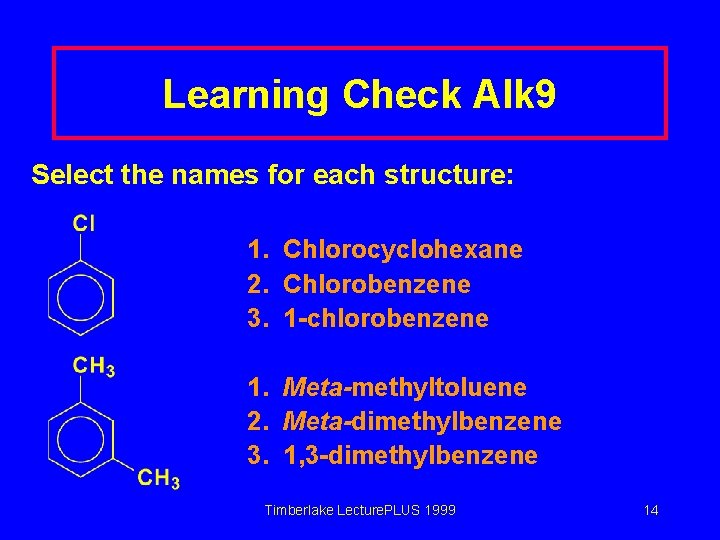
Learning Check Alk 9 Select the names for each structure: 1. Chlorocyclohexane 2. Chlorobenzene 3. 1 -chlorobenzene 1. Meta-methyltoluene 2. Meta-dimethylbenzene 3. 1, 3 -dimethylbenzene Timberlake Lecture. PLUS 1999 14
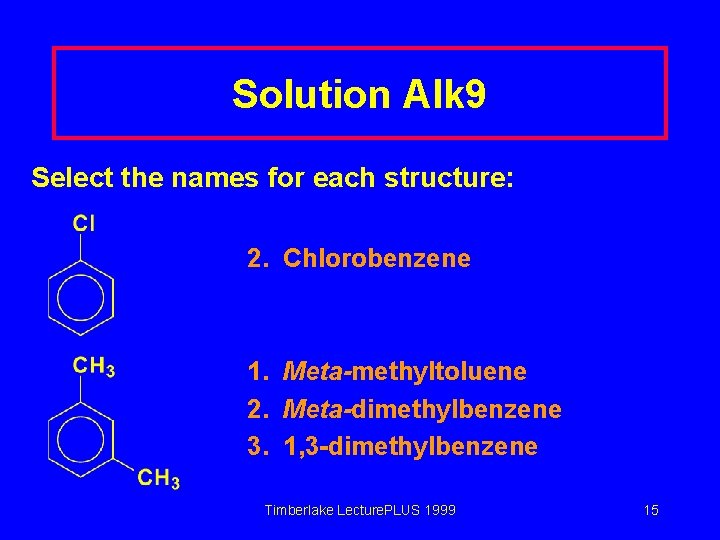
Solution Alk 9 Select the names for each structure: 2. Chlorobenzene 1. Meta-methyltoluene 2. Meta-dimethylbenzene 3. 1, 3 -dimethylbenzene Timberlake Lecture. PLUS 1999 15

Learning Check Alk 10 Write the structural formulas for each of the following: A. 1, 3 -dichlorobenzene B. Ortho-chlorotoluene Timberlake Lecture. PLUS 1999 16
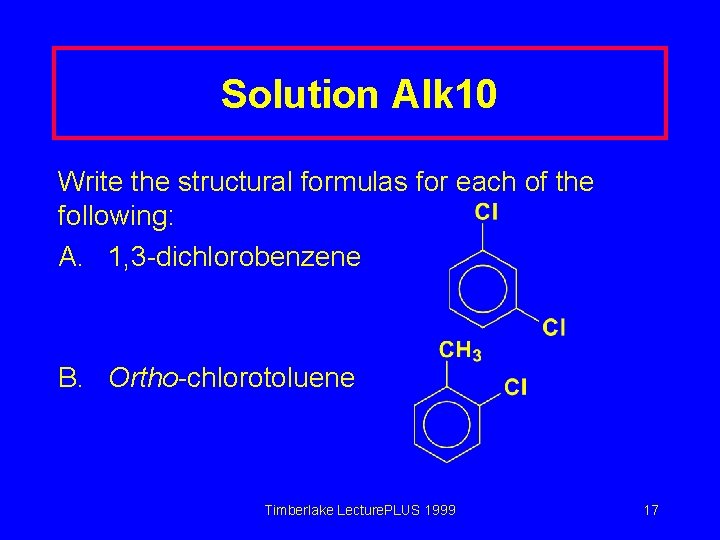
Solution Alk 10 Write the structural formulas for each of the following: A. 1, 3 -dichlorobenzene B. Ortho-chlorotoluene Timberlake Lecture. PLUS 1999 17
- Slides: 17