Chapter 10 Alkanes and Aromatic Hydrocarbons Branched Alkanes

Chapter 10 Alkanes and Aromatic Hydrocarbons Branched Alkanes Structural Formulas Robert J. O'Reilly 1
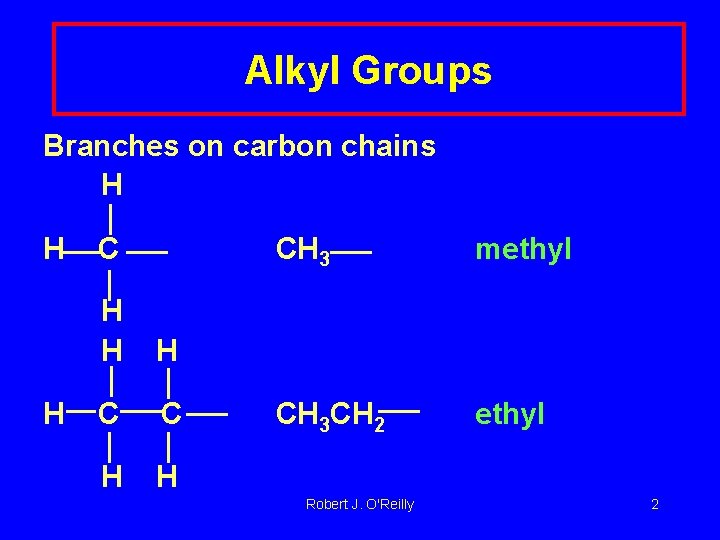
Alkyl Groups Branches on carbon chains H H H C C H H CH 3 methyl CH 3 CH 2 ethyl Robert J. O'Reilly 2
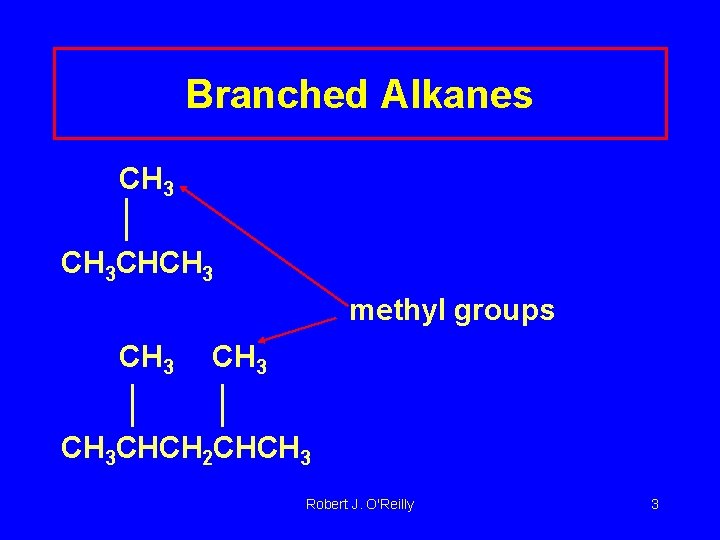
Branched Alkanes CH 3 CHCH 3 methyl groups CH 3 CHCH 2 CHCH 3 Robert J. O'Reilly 3
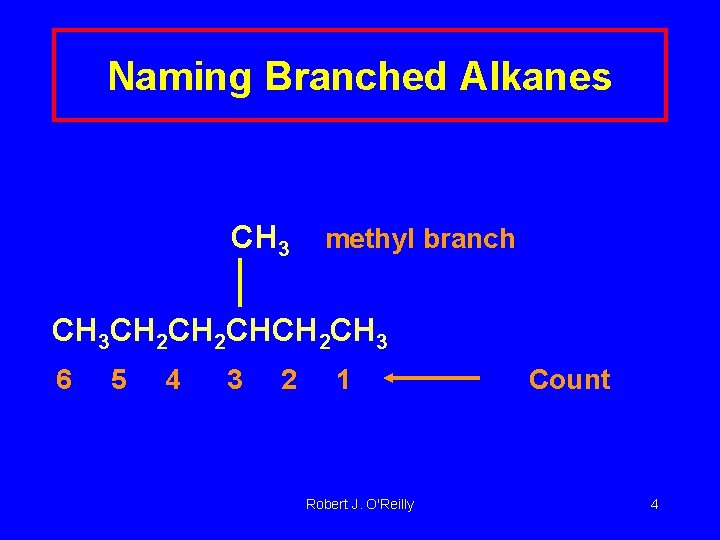
Naming Branched Alkanes CH 3 methyl branch CH 3 CH 2 CHCH 2 CH 3 6 5 4 3 2 1 Robert J. O'Reilly Count 4
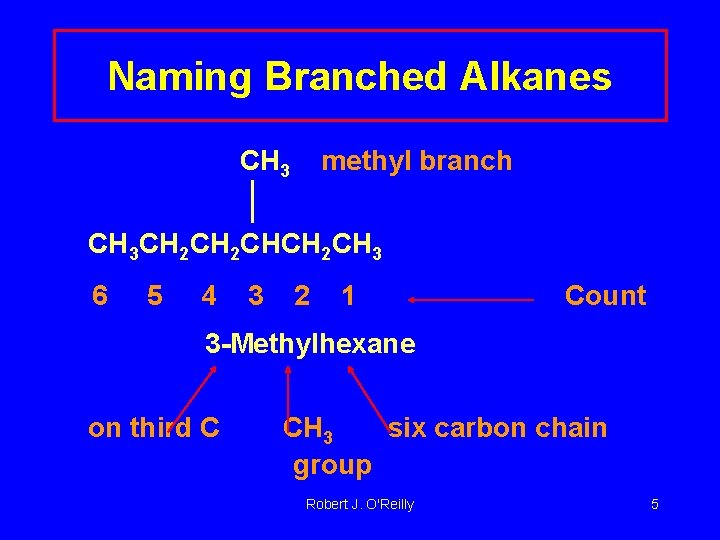
Naming Branched Alkanes CH 3 methyl branch CH 3 CH 2 CHCH 2 CH 3 6 5 4 3 2 1 Count 3 -Methylhexane on third C CH 3 six carbon chain group Robert J. O'Reilly 5

Naming Summary 1. Count the C’s in the longest chain 2. Name each attached group 3 Count the longest carbon chain to give the first attached group the smallest number 4. Name and locate each group Robert J. O'Reilly 6
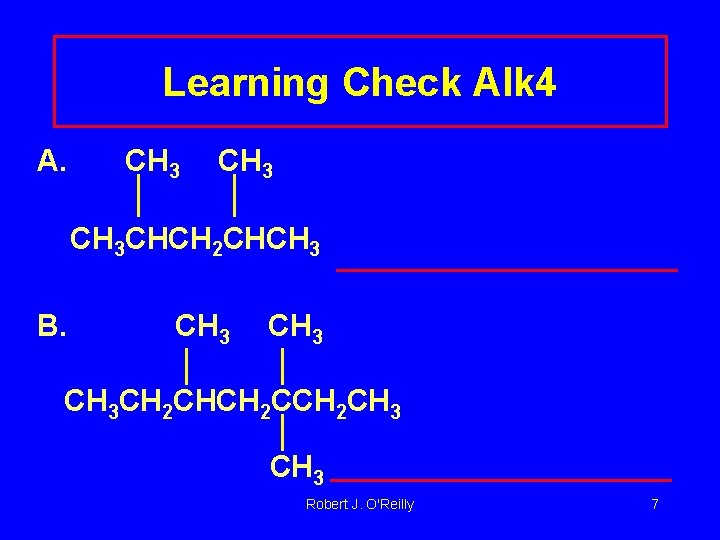
Learning Check Alk 4 A. CH 3 CHCH 2 CHCH 3 B. CH 3 CH 2 CHCH 2 CH 3 Robert J. O'Reilly 7
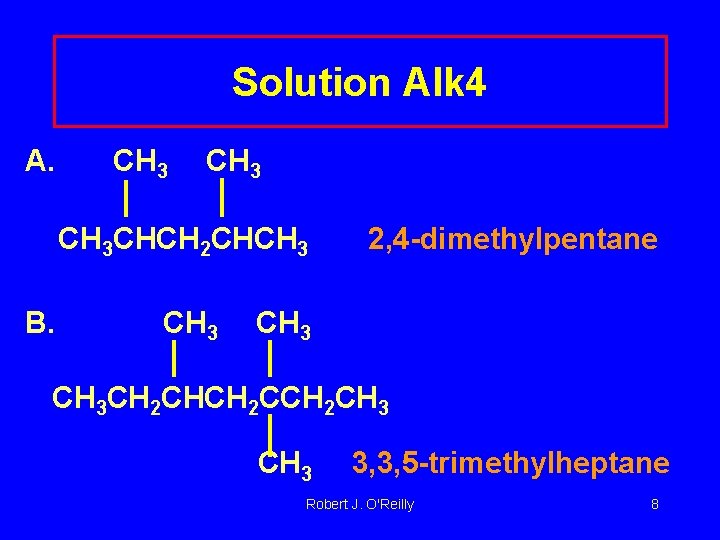
Solution Alk 4 A. CH 3 CHCH 2 CHCH 3 B. CH 3 2, 4 -dimethylpentane CH 3 CH 2 CHCH 2 CH 3 3, 3, 5 -trimethylheptane Robert J. O'Reilly 8

Learning Check Alk 5 Write a condensed structure for A. 3, 4 -dimethylheptane B. 2, 2 -dimethyloctane Robert J. O'Reilly 9
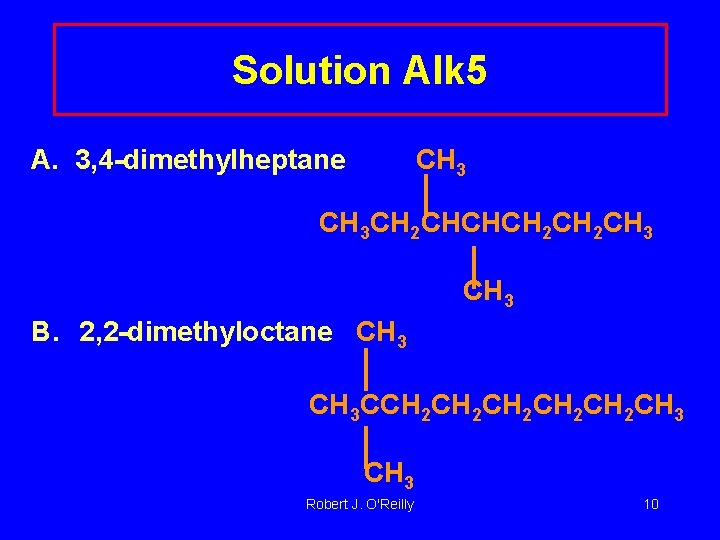
Solution Alk 5 A. 3, 4 -dimethylheptane CH 3 CH 2 CHCHCH 2 CH 3 B. 2, 2 -dimethyloctane CH 3 CCH 2 CH 2 CH 2 CH 3 Robert J. O'Reilly 10

Learning Check Alk 6 Write 3 isomers of C 5 H 12 and name each. Robert J. O'Reilly 11
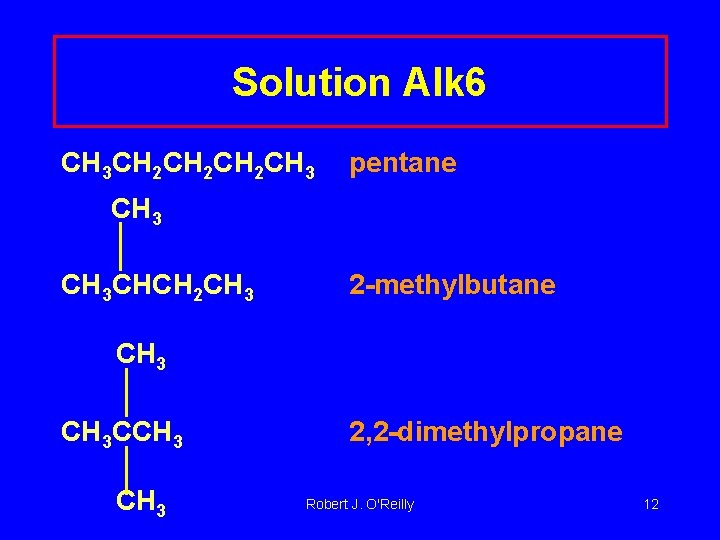
Solution Alk 6 CH 3 CH 2 CH 2 CH 3 pentane CH 3 CHCH 2 CH 3 2 -methylbutane CH 3 CCH 3 2, 2 -dimethylpropane Robert J. O'Reilly 12

Isomers =Same molecular formula =Same number and types of atoms =Different arrangement of atoms Robert J. O'Reilly 13
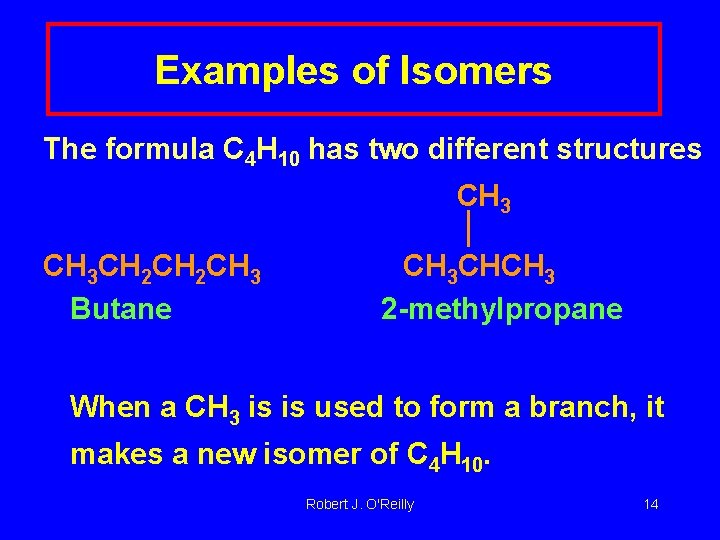
Examples of Isomers The formula C 4 H 10 has two different structures CH 3 CH 2 CH 3 Butane CH 3 CHCH 3 2 -methylpropane When a CH 3 is is used to form a branch, it makes a new isomer of C 4 H 10. Robert J. O'Reilly 14

Learning Check Alk 7 Write the structural formulas of 3 isomers that have the formula C 5 H 12. Name each. Robert J. O'Reilly 15
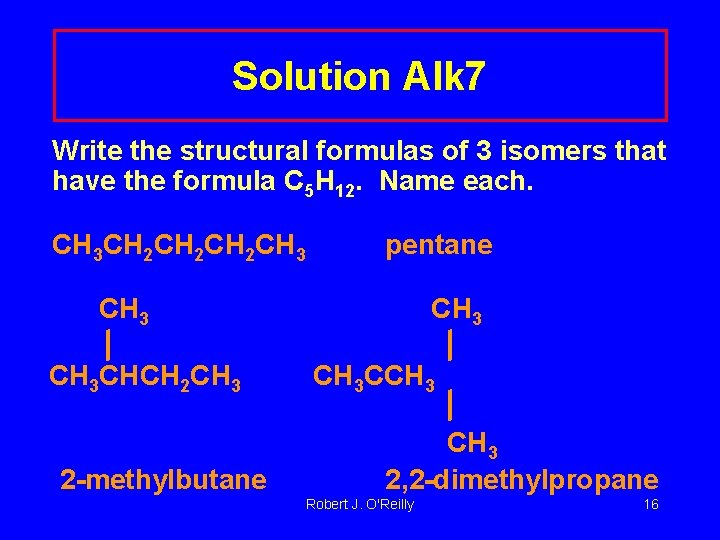
Solution Alk 7 Write the structural formulas of 3 isomers that have the formula C 5 H 12. Name each. CH 3 CH 2 CH 2 CH 3 pentane CH 3 CHCH 2 CH 3 2 -methylbutane CH 3 CCH 3 2, 2 -dimethylpropane Robert J. O'Reilly 16
- Slides: 16