Chapter 10 11 Molecular Bonds Band Structure Semiconductors

Chapter 10 & 11 Molecular Bonds & Band Structure Semiconductors Superconductivity Lasers Harris, “Modern Physics” Eisberg & Resnick, “Quantum Physics of Atoms, Molecules, Solids, Nuclei, and Particles”

Outline • 10. 1 Molecular Bonding (~2 atoms together) – pages 334 -342 • 11. 1 Band Theory (~1023 atoms together) – pages 387 -392 • 11. 2 Semiconductor Theory – mainly pages 395 -398 • 10. 5 Superconductivity – pages 362 -381 • 10. 2 Stimulated Emission & Lasers – mainly pages 342 -347

MOLECULES (~2 atoms together) Ionic & Covalent Bonds Molecular Excitations Rotation, Vibration, Electric

Ionic Bonds ENERGY BALANCE= Ionization + Electron + Affinity Attraction of + Pauli Repulsion of Cores Electrons RNave, GSU at http: //hyperphysics. phy-astr. gsu. edu/hbase/chemical/bond. html#c 4

Ionic Bond Energy Balance Ioniz Electron Affinity Coul Attraction Pauli Repulsion Energy Balance Na. Cl 5. 14 -3. 62 -6. 10 0. 31 -4. 27 Na. F 5. 14 -3. 41 -7. 46 0. 35 -5. 34 KCl 4. 34 -3. 62 -5. 39 0. 19 -4. 49 HH 13. 6 -0. 76
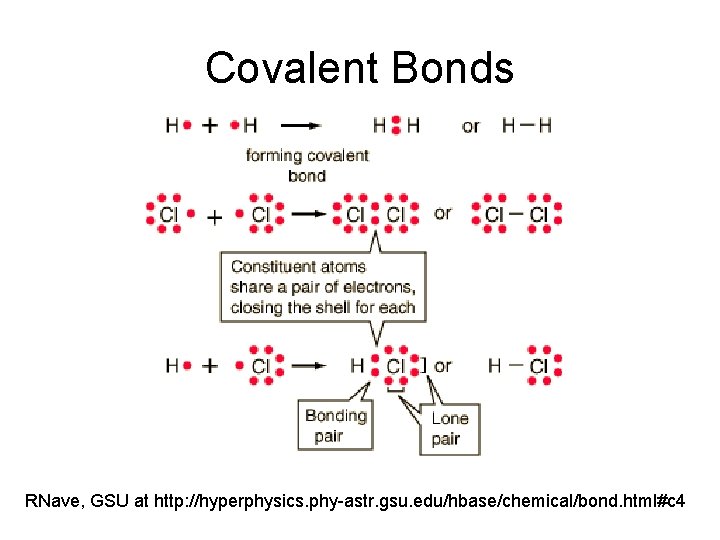
Covalent Bonds RNave, GSU at http: //hyperphysics. phy-astr. gsu. edu/hbase/chemical/bond. html#c 4
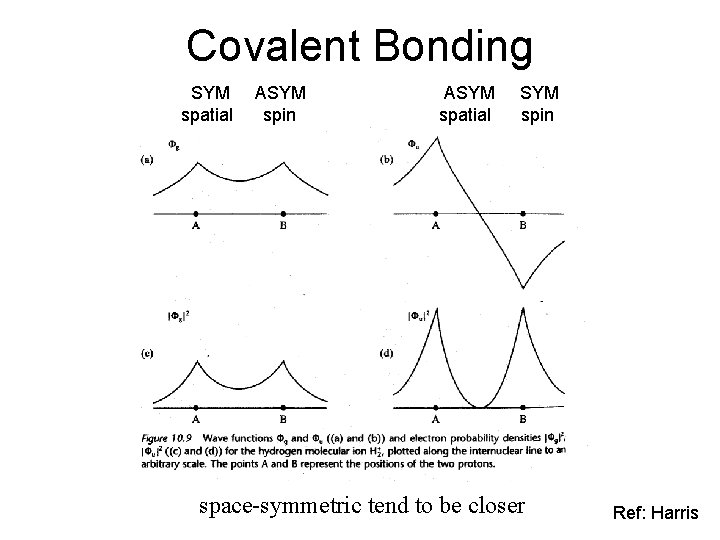
Covalent Bonding SYM spatial ASYM spin ASYM spatial SYM spin space-symmetric tend to be closer Ref: Harris

Ionic vs Covalent Bond Properties • Ionic Characteristics – Crystalline solids • Covalent Characteristics – High melting & boiling point – Gases, liquids, noncrystalline solids – Low melting & boiling point – Conduct electricity when melted – Poor conductors in all phases – Many soluble in water, but not in non-polar liquids – Many soluble in non-polar liquids but not water

Molecular Excitations Rotational Spectra w moment of inertia rotational A. M.
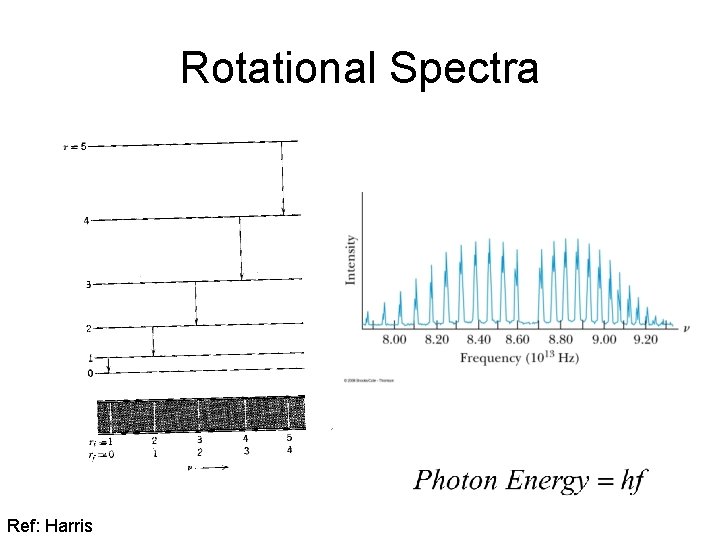
Rotational Spectra Ref: Harris

Molecular Excitations Vibration Molecule “Spring Const” ( N/m ) HF 970 HCl 480 HBr 410 Hi 320 CO 1860 NO 1530

Vibration (in an Electronic state)

Ocean Optics: Nitrogen N 2 ~ 0. 3 e. V ~ 0. 4 e. V http: //hyperphysics. phy-astr. gsu. edu/hbase/quantum/atspect. html
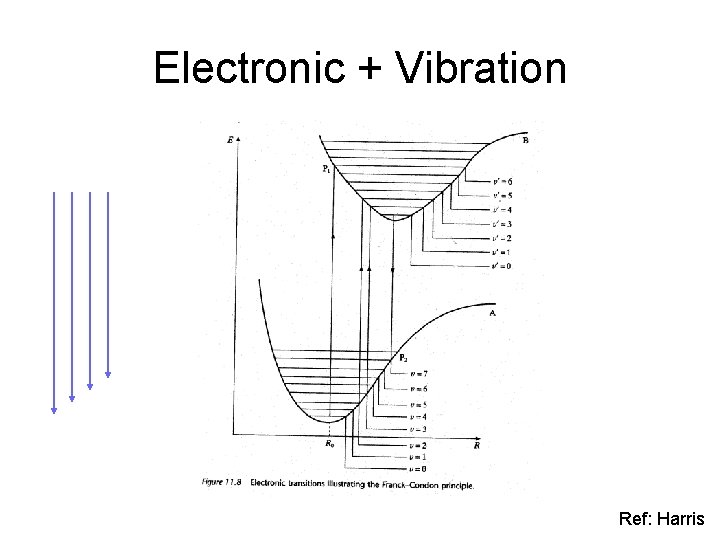
Electronic + Vibration Ref: Harris

Electronic + Vibration + Rotation 2. 550 e. V 2. 656 e. V electronic excitation gap vibrational excitation gaps Ref: Eisberg&Resnick

Electronic + Vibration + Rotation Vibrational Well 2. 656 e. V Vibrational Well depth ~ 0. 063 e. V electronic excitation gap vibrational excitation gaps Ref: Eisberg&Resnick

Electronic, Vibration, Rotation Electronic ~ optical & UV ~ 1 – 3 e. V Vibration ~ IR ~ 10 ths of e. V Rotation ~ microwave ~ 1000 ths of e. V Harris 9. 24

Some Molecular Constants Molecule Equilibrium Distance Ro (Å) Dissociation NRG Do (e. V) Vibrational freq v a (cm-1) Moment of Inertia Bb (cm-1) H 2+ 1. 06 2. 65 2297 29. 8 H 2 0. 742 4. 48 4395 60. 8 O 2 1. 21 5. 08 1580 1. 45 N 2 1. 09 9. 75 2360 2. 01 CO 1. 13 9. 60 2170 1. 93 NO 1. 15 5. 3 1904 1. 70 HCl 1. 28 4. 43 2990 10. 6 Na. Cl 2. 36 4. 22 365 0. 190 Notes: a) vibrational frequency in table is given as f / c b) moment of inertia in table is given as hbar 2/(2 I) / hc

SOLIDS x (~10 atoms together)

Isolated Atoms
Diatomic Molecule

Four Closely Spaced Atoms conduction band valence band

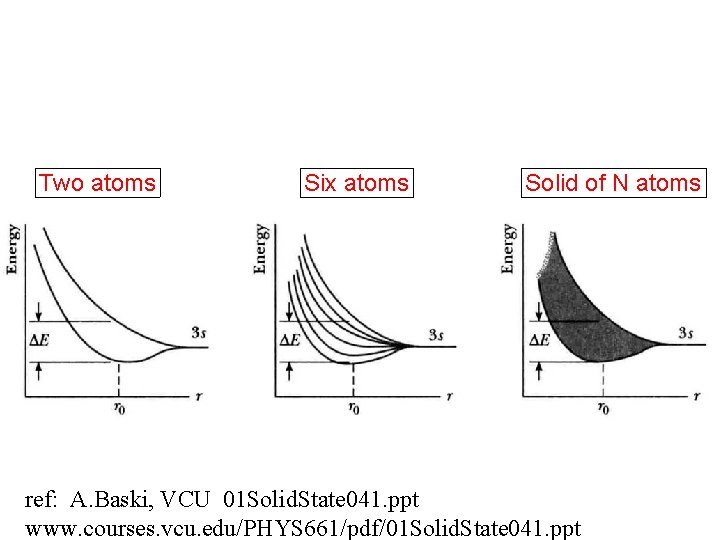
Two atoms Six atoms Solid of N atoms ref: A. Baski, VCU 01 Solid. State 041. ppt www. courses. vcu. edu/PHYS 661/pdf/01 Solid. State 041. ppt
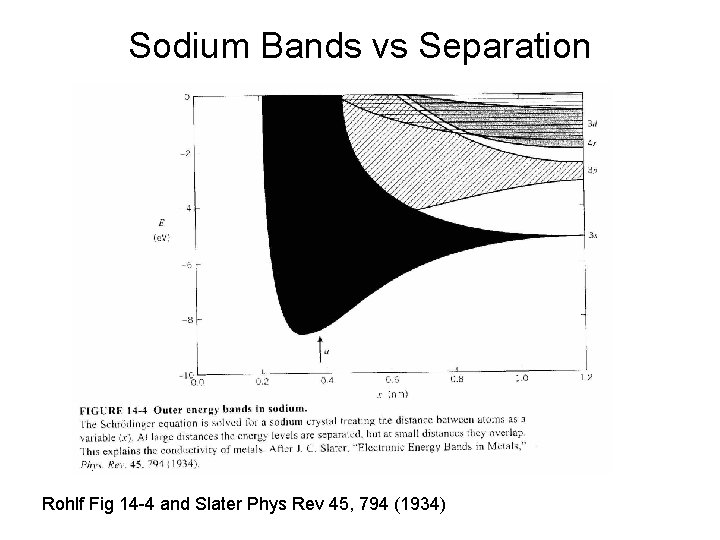
Sodium Bands vs Separation Rohlf Fig 14 -4 and Slater Phys Rev 45, 794 (1934)

Copper Bands vs Separation Rohlf Fig 14 -6 and Kutter Phys Rev 48, 664 (1935)

Differences down a column in the Periodic Table: IV-A Elements same valence config Sandin

Conductors vs insulators vs semiconductors
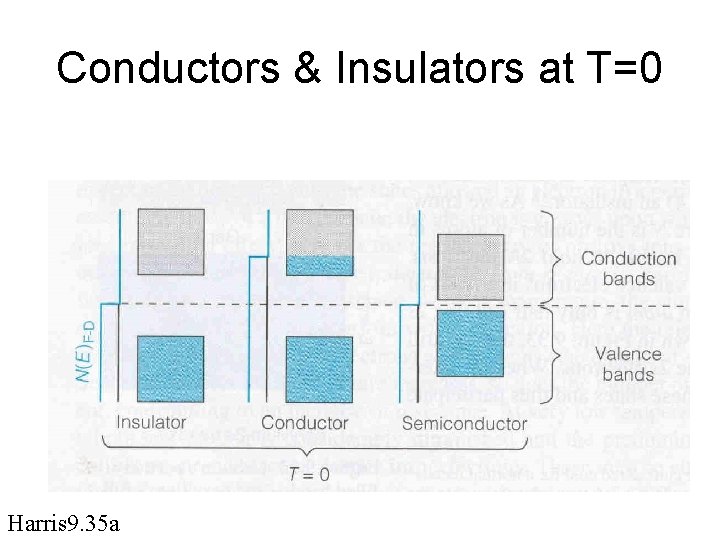
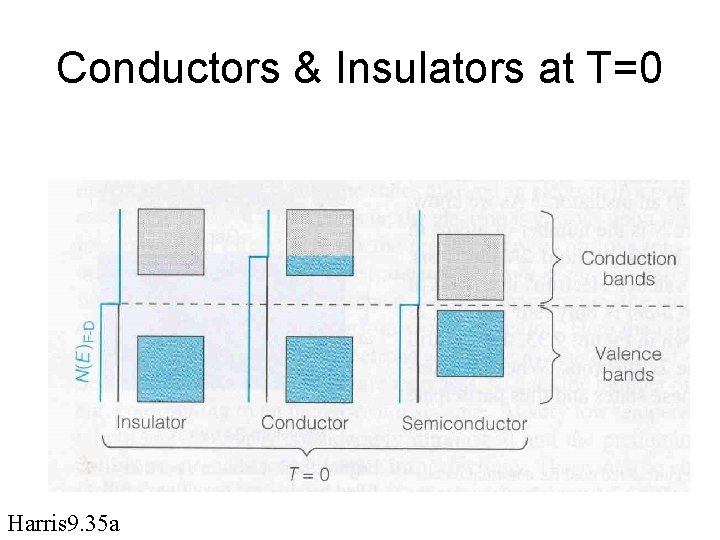
Conductors & Insulators at T=0 Harris 9. 35 a
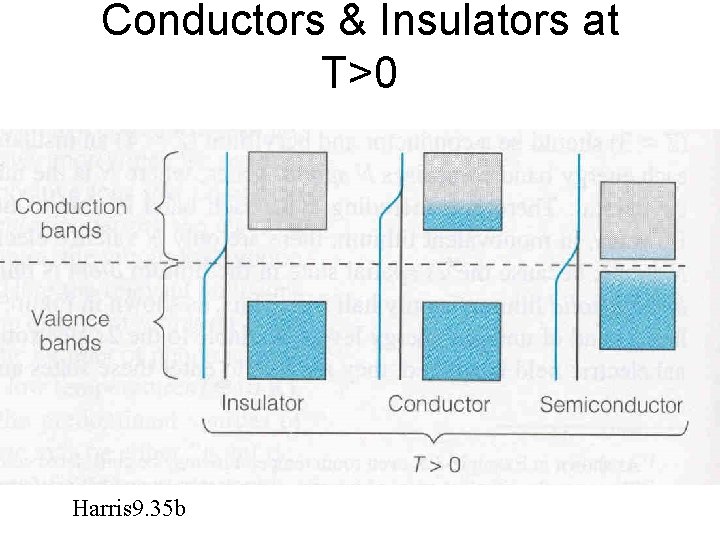
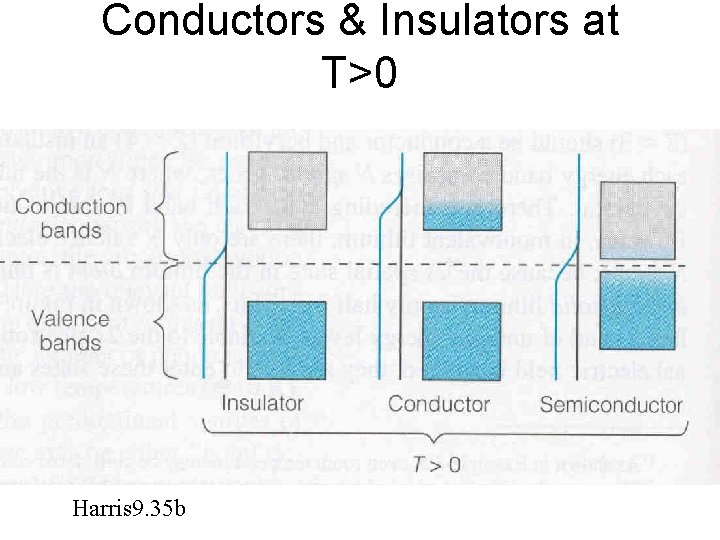
Conductors & Insulators at T>0 Harris 9. 35 b


Semiconductors & Superconductors Rex Thorton p 395 -398 p 362 -381
Two atoms Six atoms Solid of N atoms ref: A. Baski, VCU 01 Solid. State 041. ppt www. courses. vcu. edu/PHYS 661/pdf/01 Solid. State 041. ppt

Temperature Dependence of Resistivity Ag 1. 5*10 -8 Wm Cu 1. 7*10 -8 C amorphous 10 -4 Rubber 1013 Air 1016
Conductors & Insulators at T=0 Harris 9. 35 a
Conductors & Insulators at T>0 Harris 9. 35 b
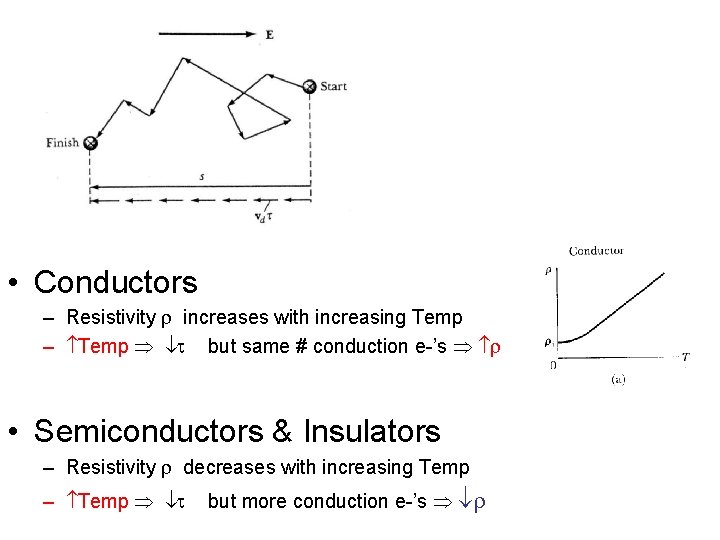
• Conductors – Resistivity r increases with increasing Temp – Temp t but same # conduction e-’s r • Semiconductors & Insulators – Resistivity r decreases with increasing Temp – Temp t but more conduction e-’s r

Semiconductors ~1/40 e. V gap ~1 e. V gap • Types – Intrinsic – by thermal excitation or high nrg photon – Photoconductive – excitation by VIS-red or IR – Extrinsic / Doped • n-type • p-type • ~1 -4 e. V gap ~0. 01 e. V gap with adjustable charge carrier density
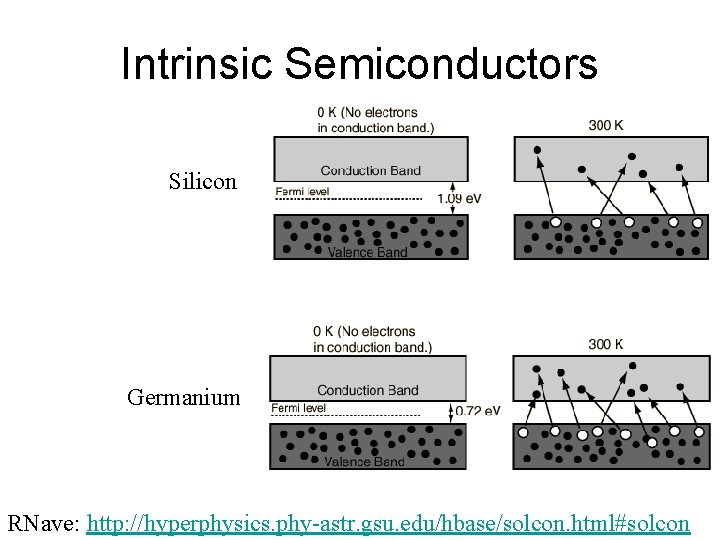
Intrinsic Semiconductors Silicon Germanium RNave: http: //hyperphysics. phy-astr. gsu. edu/hbase/solcon. html#solcon
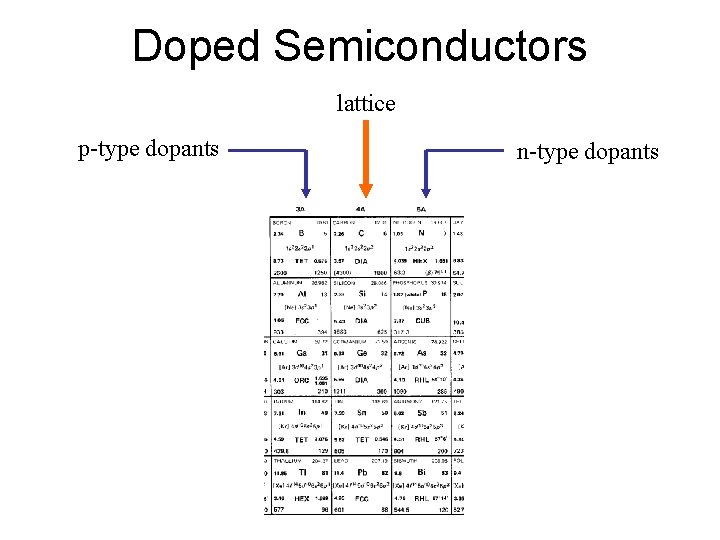
Doped Semiconductors lattice p-type dopants n-type dopants
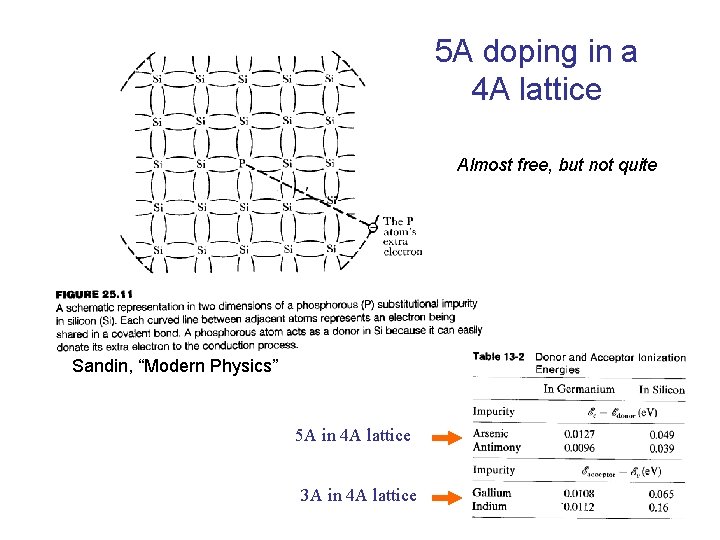
5 A doping in a 4 A lattice Almost free, but not quite Sandin, “Modern Physics” 5 A in 4 A lattice 3 A in 4 A lattice

Bands in n-doped Semiconductor 9. 44

Bands in p-doped Semiconductor 9. 45

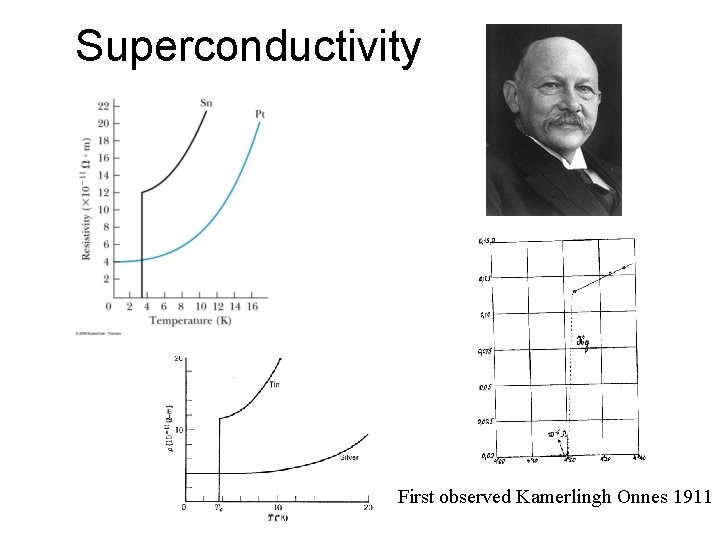
Superconductivity First observed Kamerlingh Onnes 1911
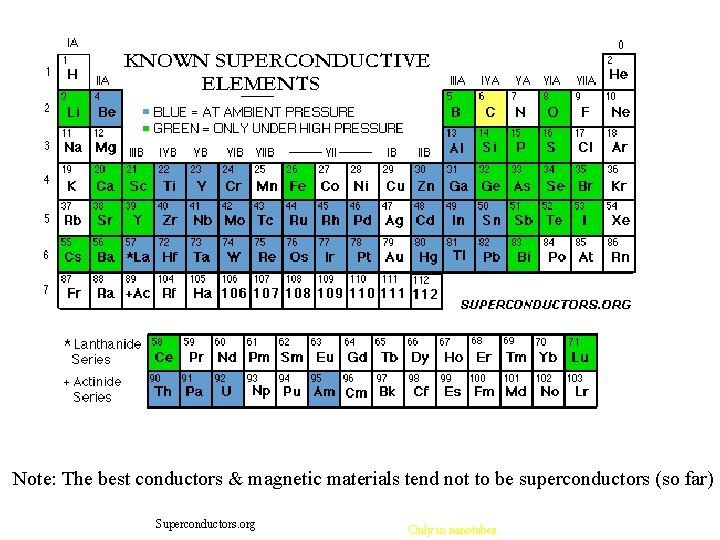
Note: The best conductors & magnetic materials tend not to be superconductors (so far) Superconductors. org Only in nanotubes
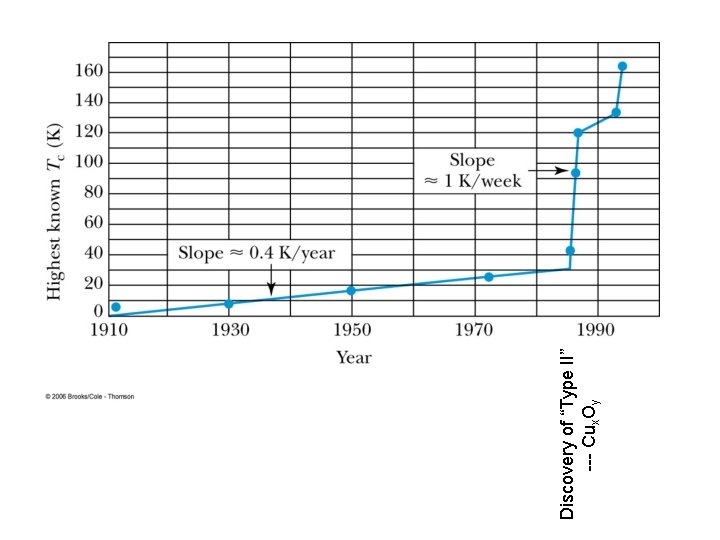
Discovery of “Type II” --- Cux. Oy
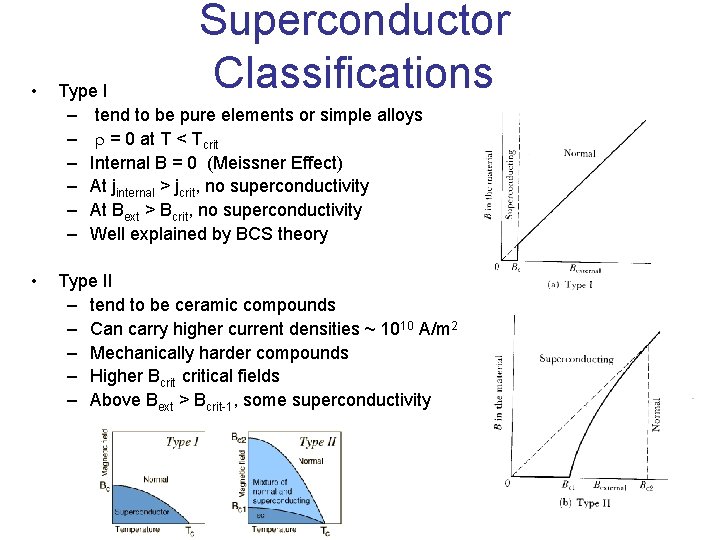
Superconductor Classifications • Type I – tend to be pure elements or simple alloys – r = 0 at T < Tcrit – Internal B = 0 (Meissner Effect) – At jinternal > jcrit, no superconductivity – At Bext > Bcrit, no superconductivity – Well explained by BCS theory • Type II – tend to be ceramic compounds – Can carry higher current densities ~ 1010 A/m 2 – Mechanically harder compounds – Higher Bcritical fields – Above Bext > Bcrit-1, some superconductivity
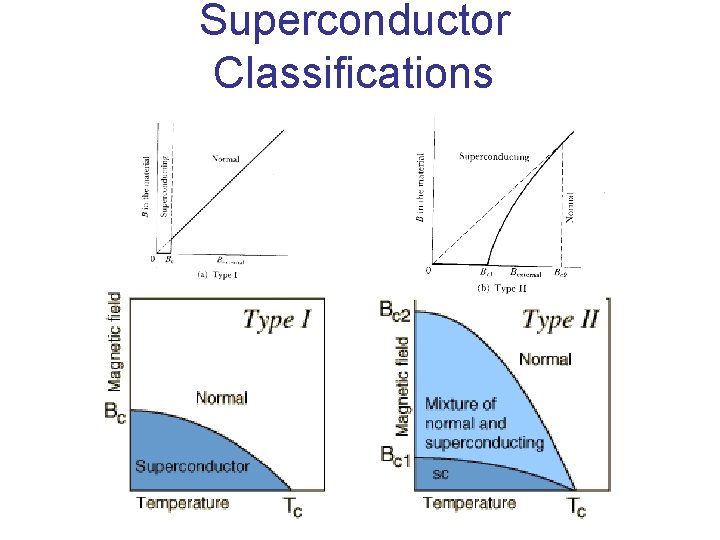
Superconductor Classifications

Type I Bardeen, Cooper, Schrieffer 1957, 1972 “Cooper Pairs” e- Q: Stot=0 or 1? L? J? e. Symmetry energy ~ -0. 01 e. V

Popular Bad Visualizations: correlation lengths Pairs are related by momentum ±p, NOT position. Sn 230 nm Al 1600 Pb 83 Nb 38 Best conductors best ‘free-electrons’ no e- – lattice interaction not superconducting

More realistic 1 -D billiard ball picture: Cooper Pairs are ±k sets Furthermore: “Pairs should not be thought of as independent particles” -- Ashcroft & Mermin Ch 34

• Experimental Support of BCS Theory – Isotope Effects – Measured Band Gaps corresponding to Tcrit predictions – Energy Gap decreases as Temp Tcrit – Heat Capacity Behavior

Normal Conductor Semiconductor or Superconductor

Superconductors and Semiconductors are the same animal from a band model viewpoint
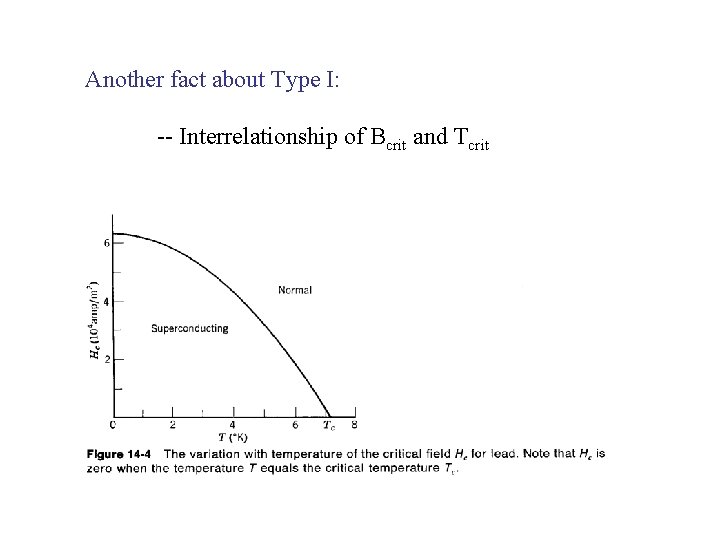
Another fact about Type I: -- Interrelationship of Bcrit and Tcrit

Type II Yr Mar 2011 Oct 2010 Q: does BCS apply ? Composition (Tl 5 Pb 2)Ba 2 Mg. Cu 10 O 17+ (Tl 4 Pb)Ba 2 Mg. Cu 8 O 13+ Tc 20 C 293 K 3 C 276 K May 2006 In. Sn. Ba 4 Tm 4 Cu 6 O 18+ 150 2004 Hg 0. 8 Tl 0. 2 Ba 2 Cu 3 O 8. 33 138 1987 YBa 2 Cu 3 O 7 93 1986 (La 1. 85 Ba. 15)Cu. O 4 30


actual ~ 8 mm Sandin
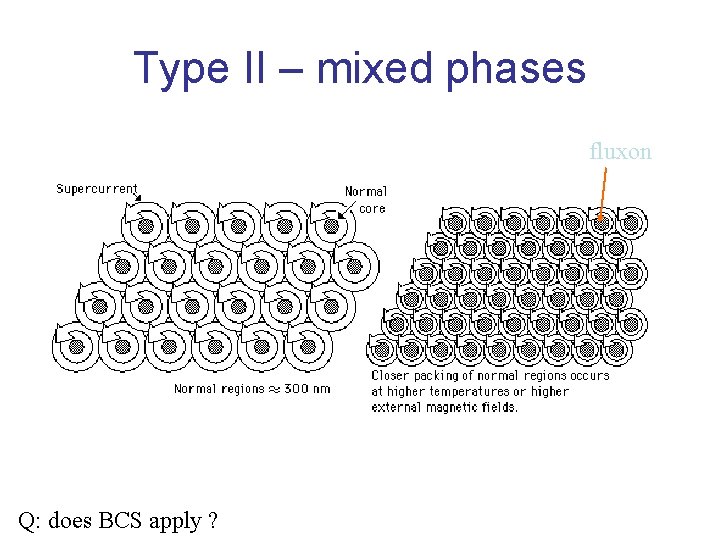
Type II – mixed phases fluxon Q: does BCS apply ?
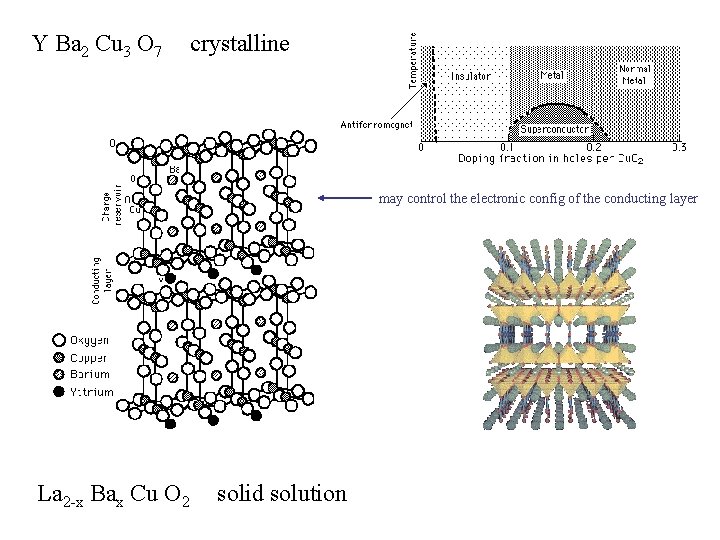
Y Ba 2 Cu 3 O 7 crystalline may control the electronic config of the conducting layer La 2 -x Bax Cu O 2 solid solution

Applications OR Other Features of Superconductors http: //superconductors. org/Uses. htm

Meissner Effect
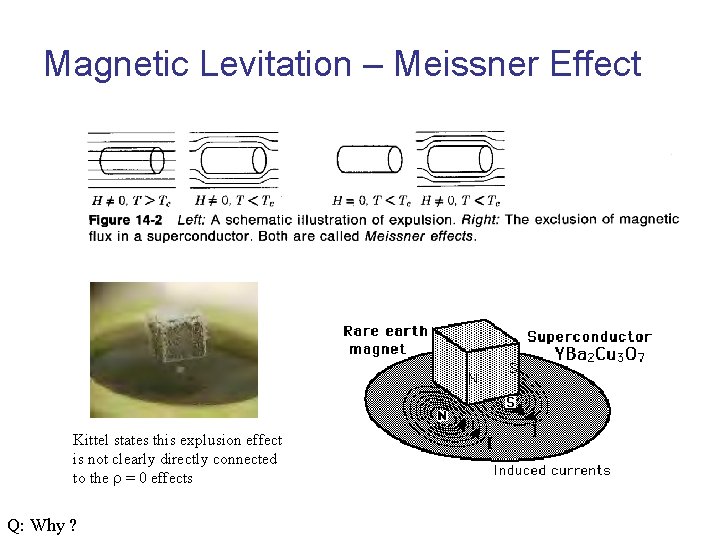
Magnetic Levitation – Meissner Effect Kittel states this explusion effect is not clearly directly connected to the r = 0 effects Q: Why ?

Magnetic Levitation – Meissner Effect MLX 01 Test Vehicle 2003 581 km/h 361 mph 2005 80, 000+ riders 2005 tested passing trains at relative 1026 km/h http: //www. rtri. or. jp/rd/maglev/html/english/maglev_frame_E. html

Mag. Lev in Shanghai

Maglev in Germany 32 km track 550, 000 km since 1984 Design speed 550 km/h Regularly operated at 420 km/h http: //en. wikipedia. org/wiki/2006_Lathen_maglev_train_accident NOTE(061204): I’m not so sure this track is superconducting. The Mag. Lev planned for the Munich area will be. France is also thinking about a sc maglev.

Maglev Frog A live frog levitates inside a 32 mm diameter vertical bore of a Bitter solenoid in a magnetic field of about 16 Tesla at the Nijmegen High Field Magnet Laboratory. http: //www. hfml. ru. nl/pics/Movies/frog. mpg
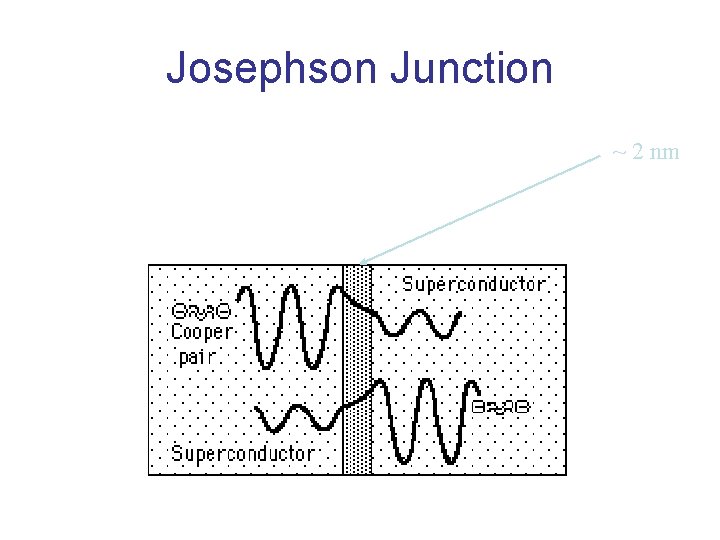
Josephson Junction ~ 2 nm

SQUID superconducting quantum interference device The phase of the wfn in left and right branches is different because of the penetrating flux.
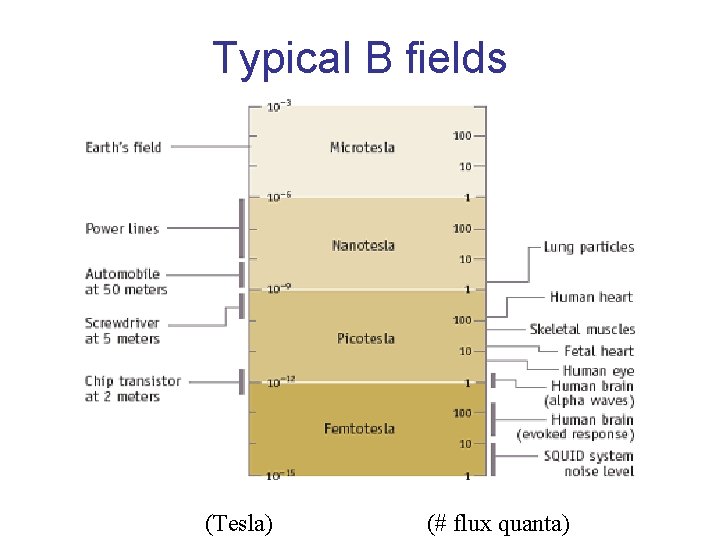
Typical B fields (Tesla) (# flux quanta)

http: //www. csiro. au/science/magsafe. html Finding 'objects of interest' at sea with MAGSAFE is a new system for locating and identifying submarines. Operators of MAGSAFE should be able to tell the range, depth and bearing of a target, as well as where it’s heading, how fast it’s going and if it’s diving. Building on our extensive experience using highly sensitive magnetic sensors known as Superconducting QUantum Interference Devices (SQUIDs) for minerals exploration, MAGSAFE harnesses the power of three SQUIDs to measure slight variations in the local magnetic field. MAGSAFE will be able to locate targets without flying close to the surface. Image courtesy Department of Defence. MAGSAFE has higher sensitivity and greater immunity to external noise than conventional Magnetic Anomaly Detector (MAD) systems. This is especially relevant to operation over shallow seawater where the background noise may 100 times greater than the noise floor of a MAD instrument.

http: //www. csiro. au/science/magsafe. html Phillip Schmidt etal. Exploration Geophysics 35, 297 (2004).
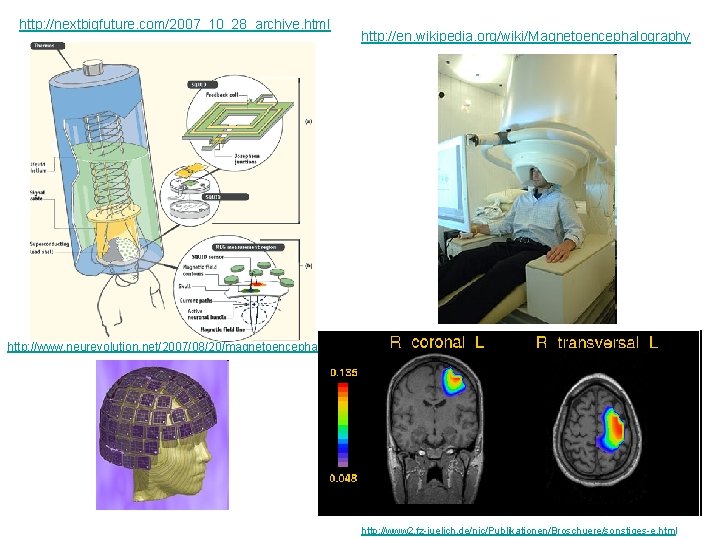
http: //nextbigfuture. com/2007_10_28_archive. html http: //en. wikipedia. org/wiki/Magnetoencephalography http: //www. neurevolution. net/2007/08/20/magnetoencephalography/ http: //www 2. fz-juelich. de/nic/Publikationen/Broschuere/sonstiges-e. html

SQUID 2 nm 10 -14 T SQUID threshold Heart signals 10 -10 T Brain signals 10 -13 T

• • Fundamentals of superconductors: – http: //www. physnet. uni-hamburg. de/home/vms/reimer/htc/pt 3. html Basic Introduction to SQUIDs: – http: //www. abdn. ac. uk/physics/case/squids. html Detection of Submarines – http: //www. csiro. au/science/magsafe. html Fancy cross-referenced site for Josephson Junctions/Josephson: – http: //en. wikipedia. org/wiki/Josephson_junction – http: //en. wikipedia. org/wiki/B. _D. _Josephson SQUID sensitivity and other ramifications of Josephson’s work: – http: //hyperphysics. phy-astr. gsu. edu/hbase/solids/squid 2. html Understanding a SQUID magnetometer: – http: //hyperphysics. phy-astr. gsu. edu/hbase/solids/squid. html#c 1 Some exciting applications of SQUIDs: – http: //www. lanl. gov/quarterly/q_spring 03/squid_text. shtml

• • • Relative strengths of pertinent magnetic fields – http: //www. physics. union. edu/newmanj/2000/SQUIDs. htm The 1973 Nobel Prize in physics – http: //nobelprize. org/physics/laureates/1973/ Critical overview of SQUIDs – http: //homepages. nildram. co. uk/~phekda/richdawe/squid/popular/ Research Applications – http: //boojum. hut. fi/triennial/neuromagnetic. html Technical overview of SQUIDs: – http: //www. finoag. com/fitm/squid. html – http: //www. cmp. liv. ac. uk/frink/thesis/node 47. html
- Slides: 76