Chapter 1 Statistical Basis of Thermodynamics Macroscopic Microscopic

Chapter 1: Statistical Basis of Thermodynamics

Macroscopic & Microscopic States • Start with a macroscopic system characterized by N identical particles confined to volume V. N 24 10 • We often will be considering the Thermodynamic Limit which is defined as N & V • These limits are taken so that the Particle Density n = (N/V) = constant.

• If the N particle system is such that there are no interactions between particles (unrealistic!) the total energy E of the system can be written: E i ni i, where i energy of particles of type i ni number of particles of type i. • Of course, it also must be true that N i ni. • If we model the microscopic physics of the system with Quantum Mechanics, the single particle energies i are discrete. If we use classical mechanics, they are continuous.

• For such a system, if we take specific values for The number of particles N, The volume V & The total energy E, we are said to have specified the system Macrostate.

• We are implicitly assuming 1 external parameter, the volume V. So it may seem like we are only talking about gases. But, most of what we say will be valid for systems with other external parameters. • For example, consider a system containing N magnetic moments In an external magnetic field B & with Total energy E. • The system Macrostate is specified by stating N, B & E. • B is an external parameter analogous to V

• In contrast to the system Macrostate, as discussed in the Review of Undergrad Statistical Physics that we did, the system Microstate in the Quantum picture is given by specifying every quantum number of every particle of the system! • In the Classical picture it is given by stating the system “location” in an abstract 6 N dimensional phase space.

• As we also discussed in the Review of Undergrad Stat. Physics: • There a HUGE number of Microstates that can be associated with a given Macrostate. • Then, to treat the system with probability, we must use The Fundamental Hypothesis: Each allowed microstate is equally probable.

• For such a system, lets define the function (N, V, E) the number of allowed microstates for a given macrostate. • In our review we showed some results for the functional form of (N, V, E). • However, for the following, lets assume that we know its functional form.

• From our review, we know that (N, V, E) is the primary connection between the macroscopic physics & the microscopic physics of our system!

Significance of (N, V, E) • Consider an isolated system A 0. It is divided into two sub-systems A 1 & A 2. 1(N 1, V 1, E 1) Number of accessible microstates of A 1 2(N 2, V 2, E 2) Number of accessible microstates for A 2 • Assume that A 1 & A 2 were initially separately in equilibrium. The isolated system A 0 is formed when A 1 & A 2 are brought together & allowed to interact. At first, they exchange energy but not particles.

• The situation is shown schematically in the figure: • Because it is isolated, the combined system A 0 has a constant energy E 0: E 0 = E 1 + E 2 = constant

E 0 = E 1 + E 2 = constant N 1 & N 2 fixed • Using The Fundamental Postulate, A 1 is equally likely to be in any one of its allowed microstates 1(E 1) & A 2 is equally likely to be in any one of its allowed microstates 2(E 2). (Focusing on the E dependences, suppressing the V dependences. ) • Further, the combined system A 0 is equally likely to be in any one its allowed microstates 1(E 1) 2(E 2) = 1(E 1) 2(E 0 – E 1) 0(E 0, E 1)

E 1 E 2 = E - E 1 E = E 1 + E 2 Let E 1 Most probable energy of system A 1 • An outline of a calculation of E 1 follows • The probability finding of A 1 with energy E 1 is proportional to the product of the number of accessible states of A 1 times the number of accessible states of A 2. This must be Consistent with Energy Conservation: E = E 1 + E 2.

E = E 1 + E 2 E 1 E 2 = E - E 1 • Stated another way: Each configuration is equally probable, but, the number of states having a particular energy E 1 is huge & unknown. Find E 1 by finding the energy where the left side of the above equation has a maximum. Note: Since entropy S = k. Bln , maximizing is the same as maximizing the entropy

Some Math Details Start with (1): Take the natural log of both sides: Find E 1 where this is max by setting the derivative = 0 (2): Combine (1) & (2):
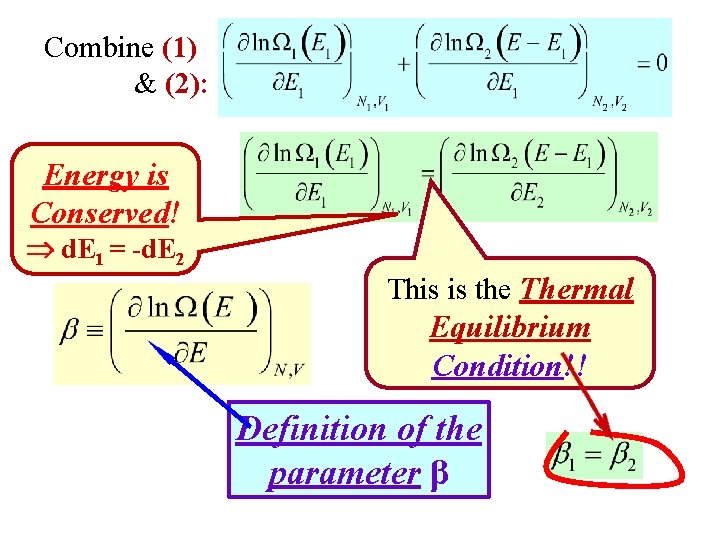
Combine (1) & (2): Energy is Conserved! d. E 1 = -d. E 2 This is the Thermal Equilibrium Condition!! Definition of the parameter β

So, at Equilibrium, it must be true that: Thermal Equilibrium Condition!! Definition of β A Fundamental Statistical Definition! • The physical interpretation of β is that it is a measure of A System’s Temperature!
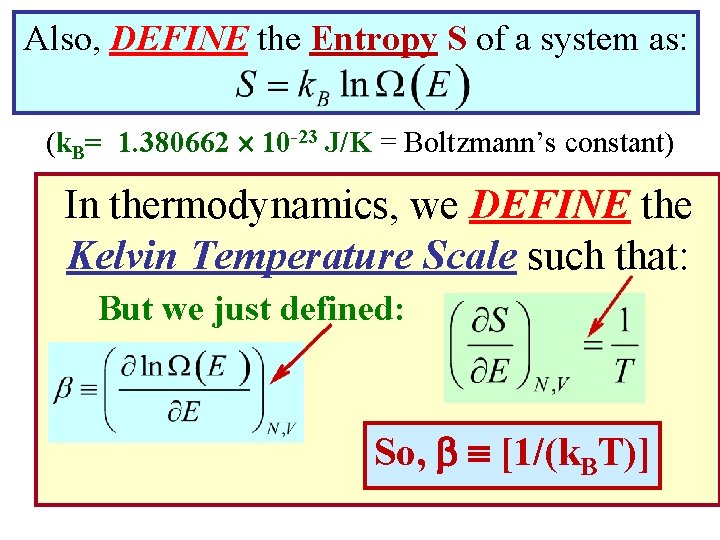
Also, DEFINE the Entropy S of a system as: (k. B= 1. 380662 10 -23 J/K = Boltzmann’s constant) In thermodynamics, we DEFINE the Kelvin Temperature Scale such that: But we just defined: So, [1/(k. BT)]

Entropy & Temperature are both related to the number of accessible states Ω(E). A “Statistical” Definition of Entropy: A “Statistical” Definition of Temperature:

The Fundamental Postulate leads to: 1. An Equilibrium Condition: Two systems are in thermal equilibrium when their temperatures are equal. (Obvious, but we proved it!) 2. Maximum Entropy for the coupled systems when they are at equilibrium. As we know, this is related to the The nd 2 Law of Thermodynamics. 3. The 3 rd Law of Thermodynamics also.

• Consider again the 2 macroscopic systems A 1 & A 2, interacting & in equilibrium. The combined system A 0 = A 1 + A 2, is isolated. Assume Thermal Interactions Only • Recall that in a pure Thermal Interaction, A 2 A 1 no mechanical work is done. So the energy exchange between A 1 & A 2, is a pure Heat Exchange!!

• As we said, the probability P(E 1) of finding the combined system A 0 = A 1 + A 2 in a state for which the system A 1 has energy E 1 is obtained by finding the energy E 1 at which the following function is maximized. P(E 1) is proportional to: • After a tedious derivation, it can be shown that an approximate form for P(E 1) is a Gaussian: P(E 1) [2π (ΔE)2]-½exp[-(E 1 -Ē 1)2/(2(ΔE)2)] • It can be shown that the width has the form = ΔE (Ē 1)/(f)½. • Note: if f = 1024, (f)½ = 1012 so ΔE (Ē 1)/(f)½ (10 -12)Ē 1. So the Gaussian with is, very tiny!

• In other words, the probability P(E 1) of finding the combined system A 0 = A 1 + A 2 in a state for which the system A 1 has an energy E 1 looks like: P(E 1) So, P(E 1) Dirac delta function centered at Ē 1! ΔE (Ē 1 ΔE (10 -12)Ē 1 )/(f)½ Ē 1 E 1 For f = 1024, (f)½ = 1012, so ΔE (Ē 1)/(f)½ (10 -12)Ē 1

• Consider 2 macroscopic systems A 1 & A 2 interacting & in equilibrium. The combined system A 0 = A 1 + A 2 , is isolated. • Assume only A 2 Mechanical Interactions • Recall that in a pure A 1 Mechanical Interaction, mechanical work is done. • So, the energy exchange between A 1 & A 2 occurs because They Do Work on each other!

• With Mechanical Interactions A 1 & A 2 interact by Doing Work on each other • Recall also that doing mechanical work means that at least one external A 1 A 2 parameter (x) of A 1 changes. • Further, this change in x is characterized by a generalized force X -( E/ x)

V 1 V 2 = V - V 1 V = V 1 + V 2 • Now consider the volume dependences of the number of accessible states in a similar way to our earlier consideration of their energy dependences. Look for V 1 Most probable volume of system A 1 • Find V 1 by finding the volume where the left side of the following equation has a Maximum: (V 1, V – V 1) = 1(V 1) 2(V – V 1)

• This leads to the following condition for mechanical equilibrium between A 1 & A 2: [∂ln( 1)/∂V 1] = [∂ln( 2)/∂V 2] • After more manipulation, it is shown that the physical interpretation of the quantity [∂ln( )/∂V] is that it is related to the mean pressure p as p k. BT[∂ln( )/∂V] • More generally, the mean generalized force X has the form: X k. BT[∂ln( )/∂x]

• The condition for mechanical equilibrium between A 1 & A 2 [∂ln( 1)/∂V 1] = [∂ln( 2)/∂V 2] therefore tells us (no surprise!) that, at equilibrium, the pressures are equal! p 1 = p 2
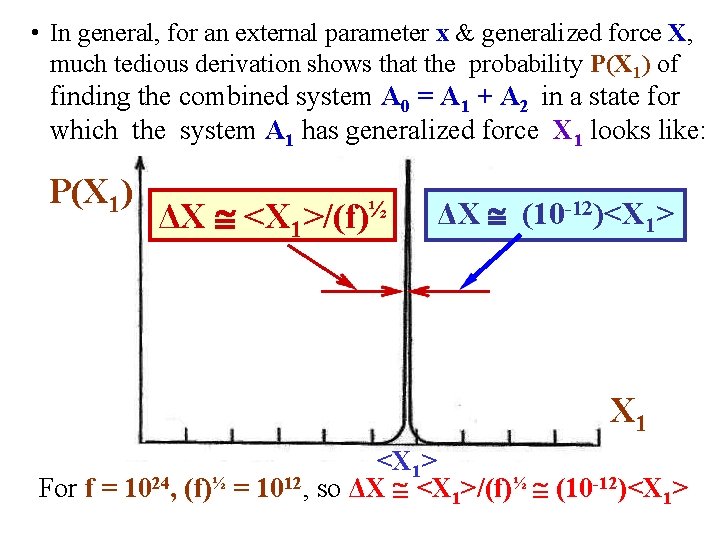
• In general, for an external parameter x & generalized force X, much tedious derivation shows that the probability P(X 1) of finding the combined system A 0 = A 1 + A 2 in a state for which the system A 1 has generalized force X 1 looks like: P(X 1) ΔX <X 1>/(f)½ ΔX (10 -12)<X 1> X 1 <X 1> For f = 1024, (f)½ = 1012, so ΔX <X 1>/(f)½ (10 -12)<X 1>
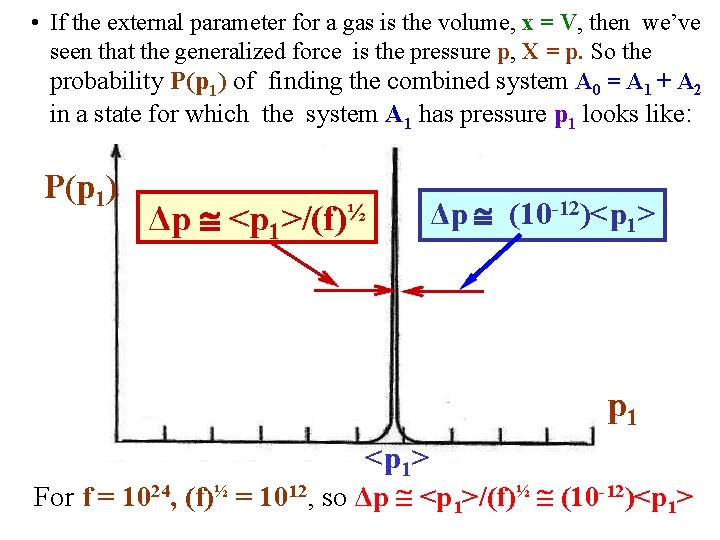
• If the external parameter for a gas is the volume, x = V, then we’ve seen that the generalized force is the pressure p, X = p. So the probability P(p 1) of finding the combined system A 0 = A 1 + A 2 in a state for which the system A 1 has pressure p 1 looks like: P(p 1) Δp <p 1 >/(f)½ Δp (10 -12)<p 1> p 1 <p 1> For f = 1024, (f)½ = 1012, so Δp <p 1>/(f)½ (10 -12)<p 1>
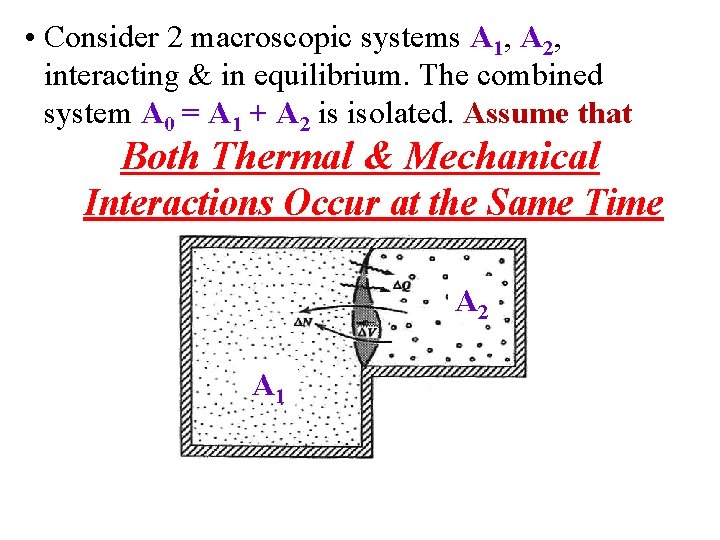
• Consider 2 macroscopic systems A 1, A 2, interacting & in equilibrium. The combined system A 0 = A 1 + A 2 is isolated. Assume that Both Thermal & Mechanical Interactions Occur at the Same Time A 2 A 1
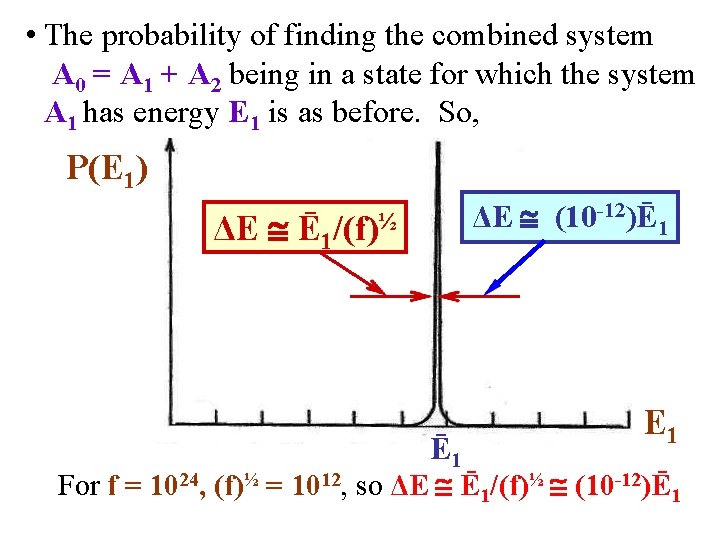
• The probability of finding the combined system A 0 = A 1 + A 2 being in a state for which the system A 1 has energy E 1 is as before. So, P(E 1) ΔE (10 -12)Ē 1 Ē 1/(f)½ Ē 1 E 1 For f = 1024, (f)½ = 1012, so ΔE Ē 1/(f)½ (10 -12)Ē 1
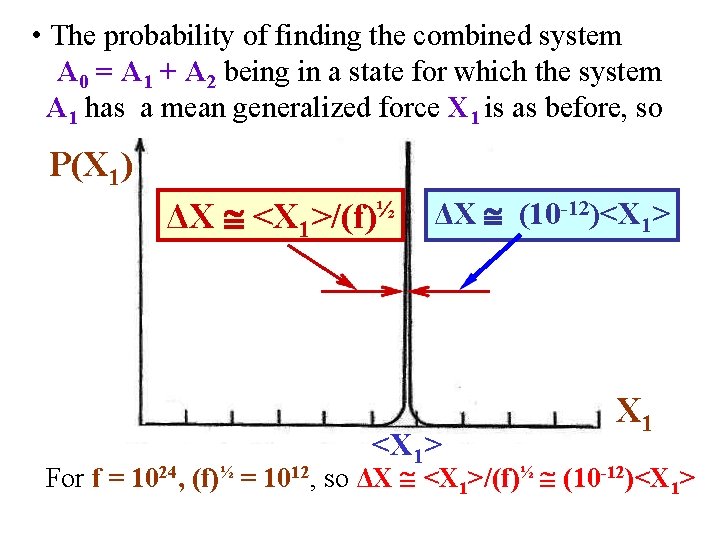
• The probability of finding the combined system A 0 = A 1 + A 2 being in a state for which the system A 1 has a mean generalized force X 1 is as before, so P(X 1) ΔX <X 1>/(f)½ ΔX (10 -12)<X 1> X 1 For f = 1024, (f)½ = 1012, so ΔX <X 1>/(f)½ (10 -12)<X 1>

• So far, we’ve assumed that the 2 macroscopic A 2 systems A 1, A 2, can A 1 exchange energy (by doing work W on each other & exchanging heat Q), but that they cannot exchange particles. • Now, lets relax that assumption & allow them to exchange particles. • Follow the same type of procedure we used for thermal & mechanical interactions to

• So, consider 2 macroscopic systems A 1 & A 2 interacting & in equilibrium. The combined system A 0 = A 1 + A 2 , is isolated. A 1 A 2 Now Particle Exchange is Allowed. • So, consider the Energy Exchange between A 1 & A 2 due to the fact that particles are moving across the boundary between them. • To discuss this, we first need to discuss the concept of the Chemical Potential.

Chemical Potential • The Chemical Potential of an N particle system is defined as the energy required to remove one particle from the system & move it out to an infinite distance away.

N 1 N 2 = N - N 1 N = N 1 + N 2 • Consider the dependences of the number of accessible states on the particle number N in a similar way to our earlier considerations of the E & V dependences. Look for N 1 Most probable particle number for system A 1 • Find N 1 by finding the particle number where the left side of the following equation has a Maximum: (N 1, N – N 1) = 1(N 1) 2(N – N 1)

• This leads to the following condition for Chemical equilibrium between A 1 & A 2: [∂ln( 1)/∂N 1] = [∂ln( 2)/∂N 2] • After more manipulation, it is shown that the physical interpretation of the quantity [∂ln( )/∂N] is that it is related to the mean Chemical Potential as k. BT[∂ln( )/∂N]

• The condition for Chemical equilibrium between A 1 & A 2 [∂ln( 1)/∂N 1] = [∂ln( 2)/∂N 2] therefore tells us (no surprise!) that, at equilibrium, the Chemcial Potentials are equal! 1 = 2
- Slides: 39