Chapter 1 Some Basic Concepts of Chemistry This
Chapter 1 : Some Basic Concepts of Chemistry This Photo by Unknown Author is licensed under CC BY-SA
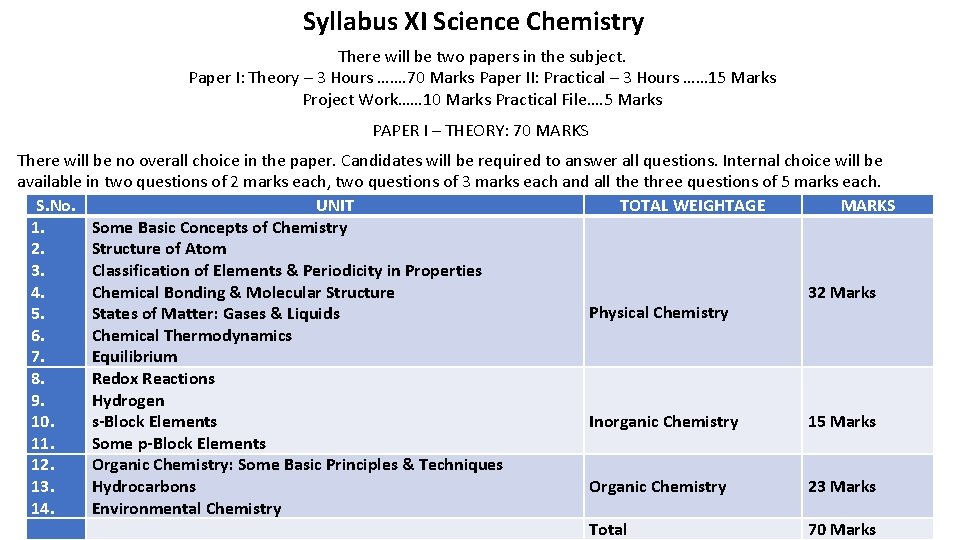
Syllabus XI Science Chemistry There will be two papers in the subject. Paper I: Theory – 3 Hours ……. 70 Marks Paper II: Practical – 3 Hours …… 15 Marks Project Work…… 10 Marks Practical File…. 5 Marks PAPER I – THEORY: 70 MARKS There will be no overall choice in the paper. Candidates will be required to answer all questions. Internal choice will be available in two questions of 2 marks each, two questions of 3 marks each and all the three questions of 5 marks each. S. No. UNIT TOTAL WEIGHTAGE MARKS 1. Some Basic Concepts of Chemistry 2. Structure of Atom 3. Classification of Elements & Periodicity in Properties 4. Chemical Bonding & Molecular Structure 32 Marks Physical Chemistry 5. States of Matter: Gases & Liquids 6. Chemical Thermodynamics 7. Equilibrium 8. Redox Reactions 9. Hydrogen 10. s-Block Elements Inorganic Chemistry 15 Marks 11. Some p-Block Elements 12. Organic Chemistry: Some Basic Principles & Techniques 13. Hydrocarbons Organic Chemistry 23 Marks 14. Environmental Chemistry Total 70 Marks

The candidate is to creatively execute one project/assignment on an aspect of Chemistry. Teachers may assign or students may select a topic of their choice. Following is only a suggestive list of projects. Suggested Evaluation criteria for Project Work: 1. Introduction / purpose 2. Contents 3. Analysis/ material aid (graph, data, structure, pie charts, histograms, diagrams, etc 4. Presentation 5. Bibliography Suggested Assignments: Chemistry Project Work – 10 Marks (Choose any one Topic) 1. Amino acids: Peptides, structure and classification, proteins structure and their role in the growth of living beings. 2. Nucleic Acid: DNA and RNA – their structure. Unique nature. Importance in evolution and their characteristic features. 3. Carbohydrates and their metabolism, Blood - haemoglobin and respiration. 4. Vitamins and hormones 5. Simple idea of chemical evolution. 6. Natural polymers (any five) - structure, characteristics, uses. Synthetic polymers (any five) - method of preparation, structure, characteristics and uses. 7. Types of Dyes - methods of preparation, characteristics and uses. 8. Chemicals in medicines: antiseptics, antibiotics, antacids, etc. and their uses. 9. Preparation of soap, nail polish, boot polish, varnish, nail polish remover, shampoo and perfumes. 10. Chemicals and chemical processes in forensic studies. 11. Insecticides, pesticides and chemical fertilisers. 12. Ancient Indian medicines and medicinal plants. 13. Organic Chemistry in Nutrition, Food Science and Biotechnology. 14. Effect of Green House Gases. 15. How Plastics have changed the world, both socially and economically.

Chemistry Class XI Science th Date: 18 May 2021 Time: 10: 40 A. M - 11: 20 A. M Chapter 1 Some Basic Concepts of Chemistry

(i) Precision and accuracy: Quantities and their measurements in Chemistry, significant figures, SI units. (ii)Dimensional analysis: Conversion of units, numericals and applications of units. (iii)The concept of atoms having fixed properties in explaining the laws of chemical combination. Study about atoms. Dalton’s atomic theory: Main postulates of theory; its limitations. Laws of chemical combinations: �Law of conservation of mass. �Law of definite proportions. �Law of multiple proportions. �Law of reciprocal proportions. �Gay Lussac’s law of gaseous volumes. Statement, explanation and simple problems based on these laws. (iv)Atomic (isotopic masses) and molecular mass. Relative molecular mass and mole: The atomic mass unit is one of the experimentally determined unit. It is equal to 1/12 of the mass of the carbon 12 isotope. Numerical problems based on mole concept, Avogadro’s number and gram molecular volume. (v) Empirical and molecular formula: Numericals based on the above. (vi)Chemical equivalents, volumetric calculations in terms of normality. C = 12. 00 should be taken as a standard for expressing atomic masses. Equivalent weight expressing the combining capacity of the elements with the standard elements such as H, Cl, O, Ag, etc. Variable equivalent weight. Gram equivalent weights, relationship between gram equivalent weight, gram molecular mass and valency. Determination of equivalent weight of acids, alkalis, salts, oxidising and reducing agents. (experimental details not required). Terms used in volumetric calculations such as percentage (w/w and w/v), normality, molarity, molality, mole fraction, etc. should be discussed. Students are required to know the formulae and normality and molarity equations. Simple calculations on the above topics. (vii)Chemical reactions – stoichiometric calculations based on mass-mass, mass-volume, volume-volume relationships and limiting reagent. 2

Some Basic Concepts of Chemistry Class XI Science Chemistry Accuracy determines the closeness of the measured value to the real or true value, but the precision decides how do the individual measurements agree with each other. For example: The true weight of salt is 20. 000 g. When the student ‘A’ weighs it , he takes two readings, i. e. , 19. 781 g and 19. 786 g. These values are very close to each other but not accurate because the true value is 20. 000 g. Now the student ‘B’ weighs it and takes two readings on analytical balance, i. e. , 20. 001 g and 20. 000 g. These values are very close to each other and are precise. These values are close to true value also. Therefore, the values are accurate also.


Significant Figures: The significant figures in a number are all the certain digits plus one doubtful digit. The following rules are followed in counting the significant numbers in a measured quantity: (i) All non-zero digits are significant. For example : 6. 9785 has 5 significant figures. (ii) A zero becomes significant if it appears between two non-zero digits. For example: 4. 6085 has five significant figures. (iii) The zeros placed to the left of the numbers are not significant. For example: 0. 038 has two significant figures. (iv) The zeros placed to the right of the decimal are significant. For example: 433. 00 has five significant figures. If a number ends in zeros but these zeros are not to the right of the decimal point , these zeros may or may not be significant. For example: 311800 may have four, five or six significant figures. Such a number is written in exponential form. i. e. , N x 10 n or N x 10 -n where N is a number with a single non-zero digit to the left of the decimal point or number of significant figures. Its value lies between 1. 000……. and 9. 999… and ‘n’ is the integer. Now , 311800 can be written as 3. 118 x 105 in exponential form and it has four significant figures.






Class XI Science Some Basic Concepts of Chemistry th 19 May 2021 5 th Period: Time : 11: 20 A. M – 12: 00 P. M Day 3





(vi). The non-significant figures in the measurements are rounded off. (a) If the most right digit is 5, then the preceding number is not changed if it is an even number but it is increased by one if it is an odd number. (b) If the most right digit is greater than 5, the preceding number is increased by one. (c) If the most right digit is less than 5, the preceding number is retained. Laws of Chemical Combination: The chemical reactions take place according to the laws of chemical combination. These are discussed below: 1. Law of Conservation of Mass: This law was given by Lavoisier. According to this law, “In a physical or chemical change, the total mass of the products is equal to the total mass of the reactants. ” This law does not hold good for nuclear reactions.



Class XI Science Some Basic Concepts of Chemistry 1 st 20 th May 2021 Period: Time : 08: 30 A. M – 09: 10 A. M Day 3










Class XI Science Some Basic Concepts of Chemistry 1 st 24 th May 2021 Period: Time : 08: 30 A. M – 09: 10 A. M Day 5

2. Law of Definite or Constant Proportions: This law was presented by Proust. It states that “ A chemical compound always contains the same elements combined together in fixed proportion by mass. ” For example: whatever sample of carbon dioxide is taken, it is observed that carbon and oxygen are always combined in the ratio of 3: 8.



3. Law of Multiple Proportions: This law was given by Dalton. According to this law, “if two elements combine to form more than one compound, the different masses of one element which combine with the same mass of the other element are in simple ratio to one another. ” For example: hydrogen and oxygen combine to form two compounds, H 2 O water and H 2 O 2 hydrogen peroxide. In water, hydrogen 2 parts combine with 16 parts of oxygen and in hydrogen peroxide, 2 parts of hydrogen combine with 32 parts of oxygen. The masses of oxygen that combine with the same mass of hydrogen in these two compounds bear a simple ratio of 1: 2. Non-stoichiometric compounds e. g. , Fe 0. 93 O 1 do not follow this law.


4. Law of Reciprocal Proportions: This law was given by Richter. According to this law, “Two elements A and B would combine in the same ratio of masses or in simple multiple ratios of masses in which they separately combine with a fixed mass of the third element C. ” For Example: In CH 4, 1 g of H reacts with 3 g of C. In H 2 O, 1 g of H reacts with 8 g of O. The mass of C and O which combine with a constant mass 1 g of third element H are in the ratio 3: 8. Now when C and O combine together to form CO 2 the ratio of the mass of C and O is 12: 32 or 3: 8 which is same as above.

Class XI Science Some Basic Concepts of Chemistry 25 th May 2021 4 th Period: Time : 10: 40 A. M – 11: 20 A. M Day 6
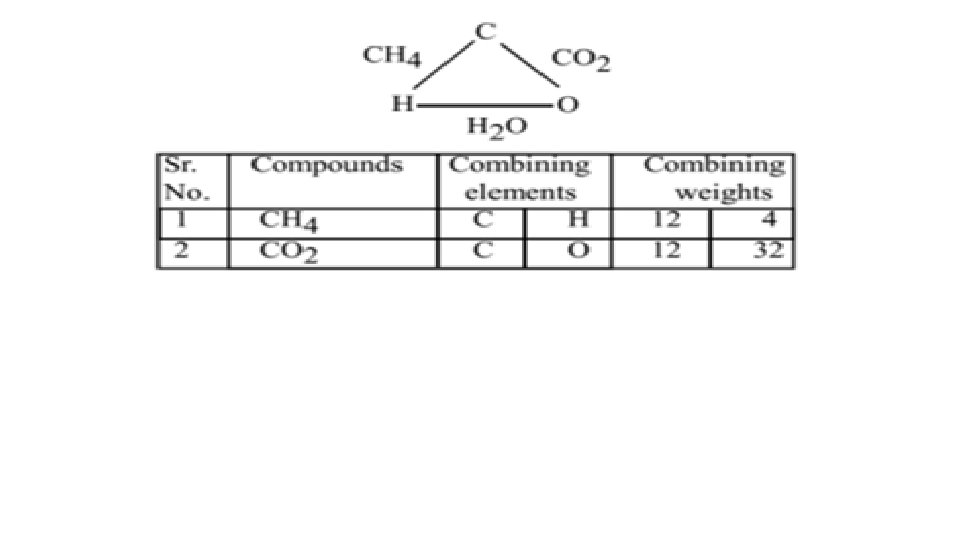





5. Law of Gaseous Volumes; This law was enunciated by Gay-Lussac and is applicable to gaseous reactions. According to this law, “ Under the same conditions of temperature and pressure the volumes of gases used and produced in a chemical reaction are in ratios of simple whole numbers. ” For example: H 2 (g) + Cl 2(g) → 2 HCl(g) 1 vol 2 vol The ratio of volumes of H 2, Cl 2 and HCl is 1: 1: 2 which is a simple ratio.


Dalton’s Atomic Theory: 1. Matter is made up of extremely small particles called atoms. 2. Atoms of same element are identical in mass, shape, size etc. 3. Atoms of different elements vary in mass, shape, size etc. 4. Atom is the smallest particle that participates in chemical reactions. 5. Atoms of same or different elements combine to form molecules and they do so in simple ratio. 6. Atoms of elements may combine in different simple ratio to form different compounds (molecules). Limitations: 1. It does not explain the cause of chemical combination. 2. It fails to explain the idea of isotopes and isobars. 3. It does not explain the law of gaseous volumes. 4. It could not explain the nature of binding forces between atoms and molecules.


Class XI Science Some Basic Concepts of Chemistry 26 th May 2021 5 th Period: Time : 11: 20 A. M – 12: 00 P. M Day 7






Chemical Equivalents: Equivalent mass is the number of parts by mass of the substance which combine or displace directly or indirectly 1. 008 parts by mass of hydrogen or 8 parts by mass of oxygen, 35. 5 parts by mass of chlorine or 108 parts by mass of silver. When equivalent mass is represented in grams, it is called gram equivalent mass. The equivalent mass of a substance may have different values under different conditions. Generally it does not have any units. Relation between Atomic Mass, Equivalent Mass and Valency: Atomic Mass = Equivalent mass x Valency


5. Double decomposition method





- Slides: 62