Chapter 1 Minerals Building Blocks of Rocks Minerals

Chapter 1 Minerals: Building Blocks of Rocks

Minerals: Building Blocks of Rocks n By definition a mineral is/has Naturally occurring n Inorganic solid n Ordered internal molecular structure n Definite chemical composition n n Rock n A solid aggregate of minerals

Composition and Structure of Minerals n Elements Basic building blocks of minerals n Over 100 are known (92 naturally occurring) n n Atoms Smallest particles of matter n Retain all the characteristics of an element n

Composition and Structure of Minerals n Atomic structure n Central region called the nucleus n n Consists of protons (+ charges) and neutrons (0 charges) Electrons n n Negatively charged particles that surround the nucleus Located in discrete energy levels called shells
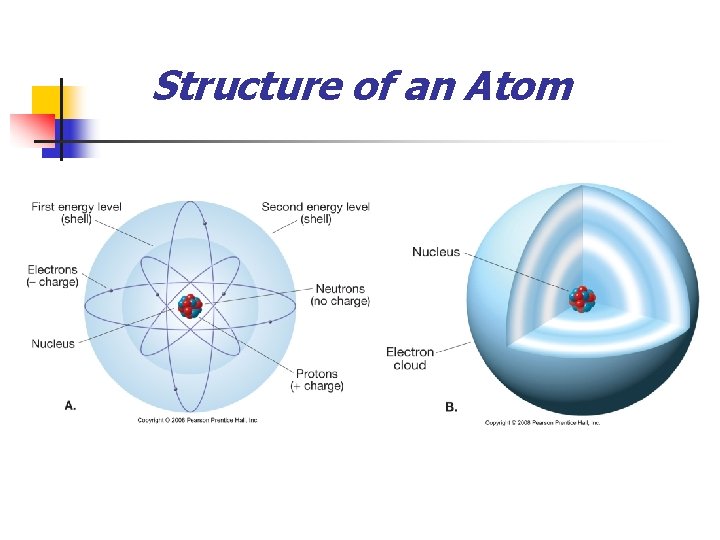
Structure of an Atom

Composition and Structure of Minerals n Chemical bonding Formation of a compound by combining two or more elements n Atoms gain or lose outermost electrons to form ions n Oppositely charged ions attract one another to produce a neutral chemical compound n

Ionic Bonding
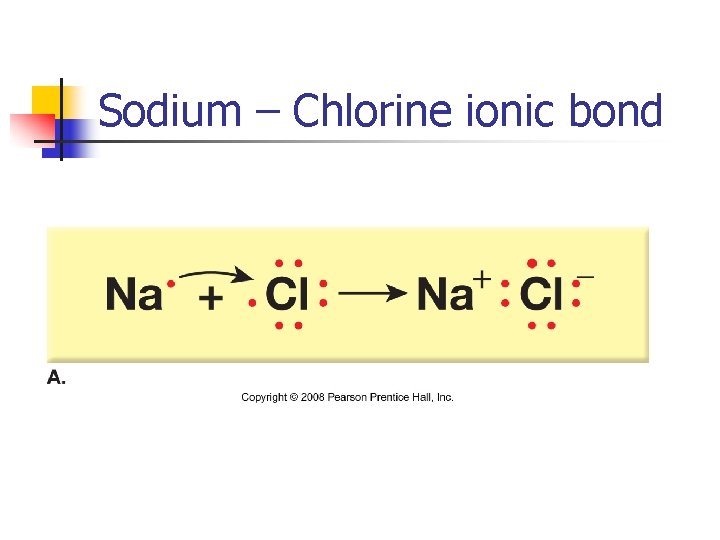
Sodium – Chlorine ionic bond

Halite mineral

Composition and Structure of Minerals n Isotopes and radioactive decay n Mass number = sum of neutrons + protons in an atom n Isotope = atom that exhibits variation in its mass number n Unstable isotopes emit particles and energy in a process known as radioactive decay

Physical Properties of Minerals n Crystal form External expression of a mineral’s internal structure n Often interrupted due to competition for space and rapid loss of heat n

A Garnet Crystal

Quartz Crystals – Hot Springs, AR

Physical Properties of Minerals n Luster Appearance of a mineral in reflected light n Two basic categories n n Metallic Nonmetallic Other descriptive terms include vitreous, silky, or earthy

Galena (Pb. S) Displays Metallic Luster Figure 1. 13

Physical Properties of Minerals n Color Generally unreliable for mineral identification n Often highly variable due to slight changes in mineral chemistry n Exotic colorations of certain minerals produce gemstones n

Quartz (Si. O 2) Exhibits a Variety of Colors

Physical Properties of Minerals n Streak n n Color of a mineral in its powdered form Hardness Resistance of a mineral to abrasion or scratching n All minerals are compared to a standard scale called the Mohs n scale of hardness

Streak Is Obtained on an Unglazed Porcelain Plate Figure 1. 8
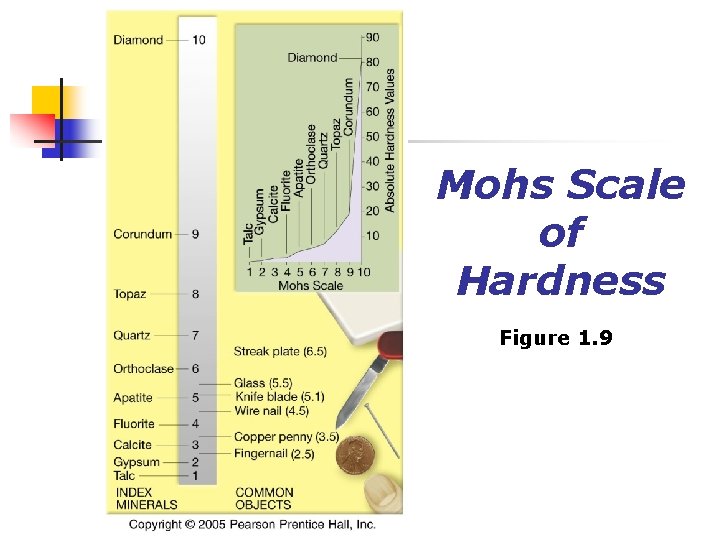
Mohs Scale of Hardness Figure 1. 9

Physical Properties of Minerals n Cleavage Tendency to break along planes of weak bonding n Produces flat, shiny surfaces n Described by resulting geometric shapes n n n Number of planes Angles between adjacent planes

Fluorite, Halite, and Calcite All Exhibit Perfect Cleavage Figure 1. 11

Physical Properties of Minerals n Fracture n n Absence of cleavage when a mineral is broken Specific Gravity Weight of a mineral/weight of an equal volume of water n Average value = 2. 5 – 3. 0 n

Conchoidal Fracture Figure 1. 12

Physical Properties of Minerals n Other properties Magnetism n Reaction to hydrochloric acid n Malleability n Double refraction n Taste n Smell n Elasticity n

Mineral Groups n n Nearly 4000 minerals have been named Rock-forming minerals Common minerals that make up most of the rocks of Earth’s crust n Only a few dozen members n Composed mainly of the 8 elements that make up over 98% of the continental crust n
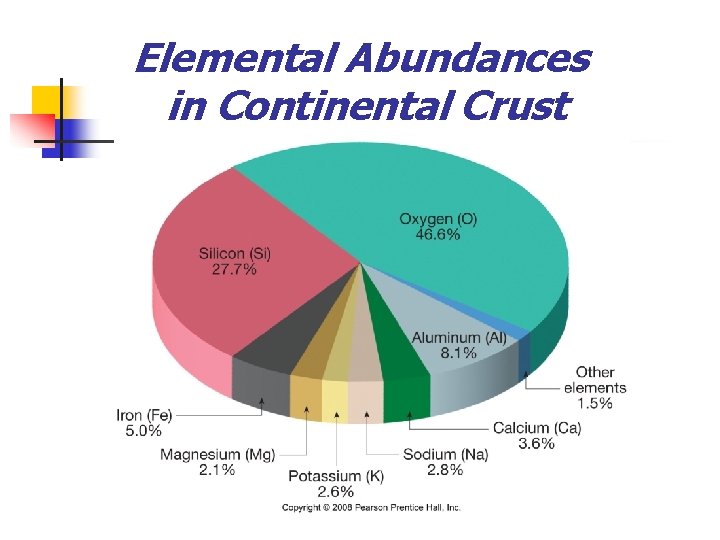
Elemental Abundances in Continental Crust

Mineral Groups n Silicates n Most important mineral group n n n Comprise most rock-forming minerals Very abundant due to large % of silicon and oxygen in Earth’s crust Silicon-oxygen tetrahedron n n Fundamental building block Four oxygen ions surrounding a much smaller silicon ion
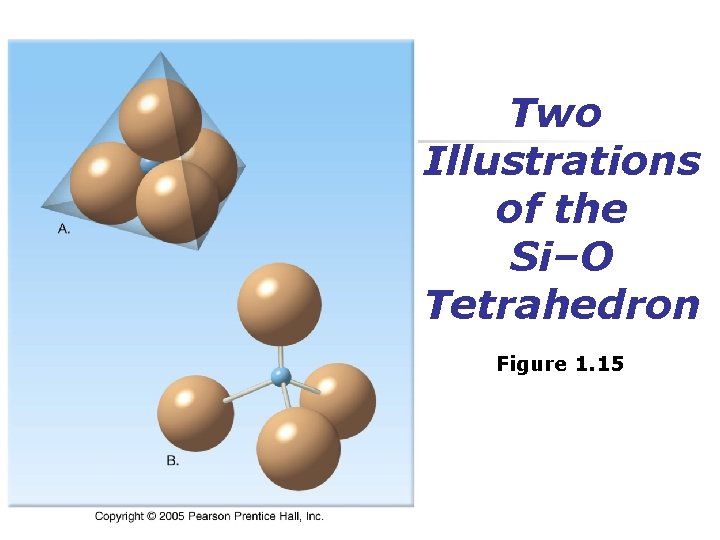
Two Illustrations of the Si–O Tetrahedron Figure 1. 15

Silicate Mineral Groups

Silicate Mineral Groups

Potassium Feldspar Figure 1. 17

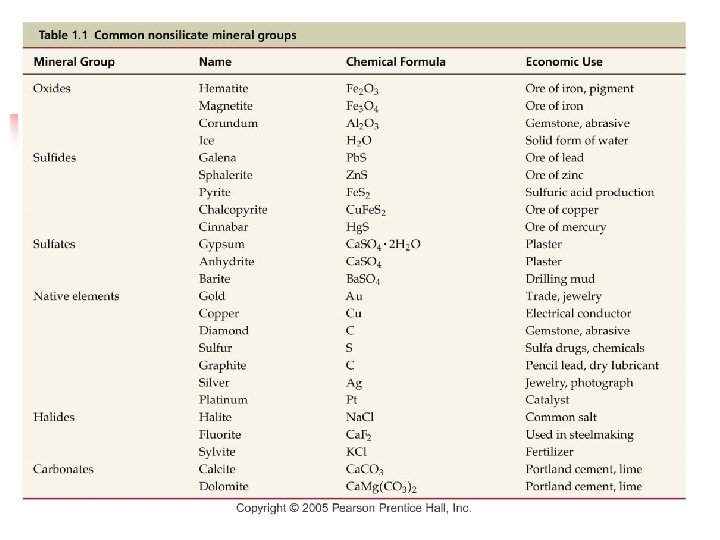
Nonsilicate Mineral Groups n Important nonsilicate minerals Comprise only 8% of Earth’s crust n Often occur as constituents in sedimentary rocks n

End of Chapter 1
- Slides: 35