Chapter 1 Introduction to Chemistry Dr Richard Malik















- Slides: 18

Chapter 1 Introduction to Chemistry Dr. Richard Malik 1

The Stories of Two Chemicals Ø Ozone is formed when oxygen gas is exposed to ultraviolet radiation in the stratosphere forming an ozone layer. Ø Ultraviolet radiation (UV) is harmful to plants and animals. Ø Importance of the Ozone Layer: – Ozone absorbs most of the harmful UV radiation before it reaches Earth’s surface. 2

The Stories of Two Chemicals Ø Chlorofluorocarbons (CFC) – Used as coolant for refrigerators – Used as propellants in spray cans – CFC’s escape into the atmosphere causing a thinning of the ozone layer. 3

What is Chemistry? Ø Chemistry is the study of the matter, its composition, properties, and the changes it undergoes. Ø Applied chemistry is the using of chemistry to attain certain goals, in fields like medicine, agriculture, and manufacturing Ø Pure chemistry gathers knowledge for knowledge’s sake 4

Which Comes First? Ø Pure Chemistry Usually comes first, applied later Called technology Or engineering Can’t be good or bad 5 ØApplied Chemistry Pure chemistry can explain behavior that has been used without knowing why Steel swords Can be good or bad depending on use

Mass and Matter? Ø Ø 6 Matter is anything that has mass and takes up space. (Ex. Book) Mass is the amount of matter in an object. Measured in grams (g) Weight- The weight of an object is a measure of the force of gravity on the object. Your weight will change if you move from the North pole to the equator but our mass will stay the same.

Branches of Chemistry Analytical Chemistry -studies composition of substances. Ø Organic Chemistry -compounds containing carbon Ø Inorganic Chemistry -substances without carbon Ø Biochemistry- Chemistry of living things Ø Physical Chemistry studies behavior of substances – rates and mechanisms of reactions – energy transfers Ø 7

Ø Material Applied Chemistry Design – Plastics – Paints – Nanotechnology Ø Scale – Macroscopic- Big enough to see – Microscopic- Too small to see unaided – Nanotechnology- manipulating individual atoms and molecules 8

Applied Chemistry Ø Agriculture – Production- fertilizers, soil tests – Protection – pesticide, herbicide Ø Medicine – Drugs – Materials- hips, artificial skin – Biotechnology- using organisms as a means of production 9

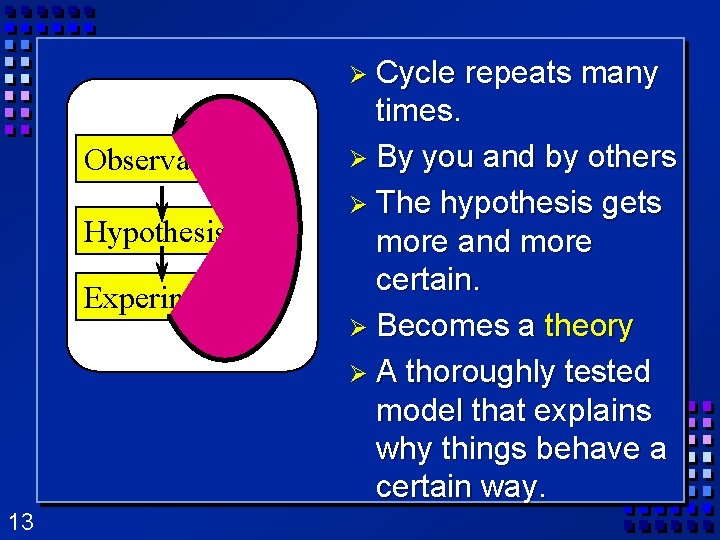
Scientific Method Ø Ø 10 A way of solving problems or answering questions. Step 1: Observation- the act of gathering information. Observations can be: – Qualitative data- Anything that relates to the five senses. (look, feel, sound, taste, smell) – Quantitative data- Numerical information that tells you how much, how little, etc.

Scientific Method (cont. ) 11 Ø Step 2: Hypothesis- a testable statement or prediction that explains the cause of the observation, based on research and previous knowledge Ø Step 3: Experiment- a process designed to test the hypothesis. – only two possible answers, hypothesis is right or wrong – Generates data -observations from experiments.

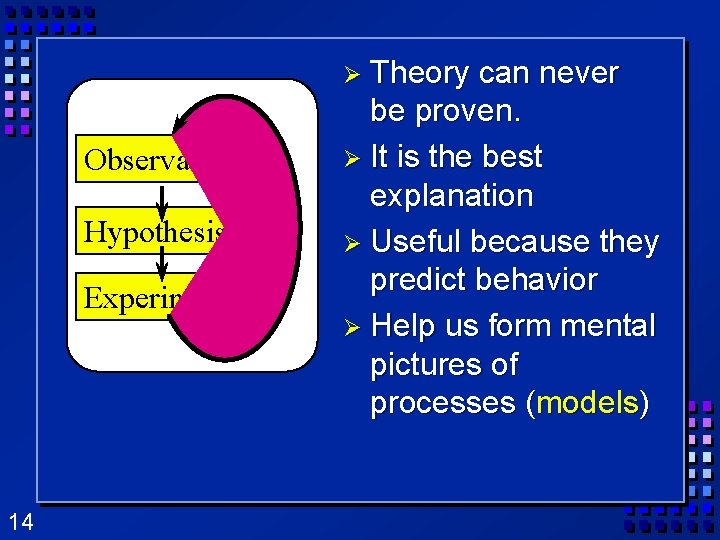
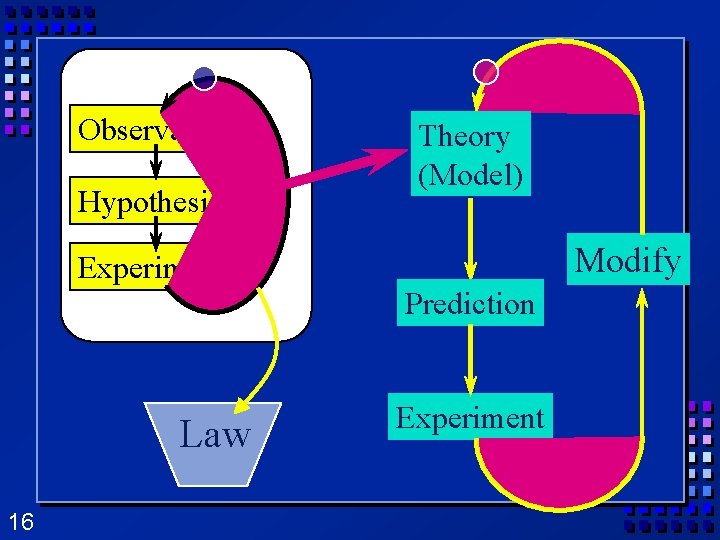
Scientific Method (cont. ) Ø Step 4 Conclusion: A judgment based on the information obtained. Can lead to a theory. – Theory - Hypothesis supported by many experiments. – Scientific Law- Summary of accepted facts of nature. Ø Step 5 Modify hypothesis - repeat the cycle. 12

Ø Cycle Observations Hypothesis Experiment 13 repeats many times. Ø By you and by others Ø The hypothesis gets more and more certain. Ø Becomes a theory Ø A thoroughly tested model that explains why things behave a certain way.

Ø Theory Observations Hypothesis Experiment 14 can never be proven. Ø It is the best explanation Ø Useful because they predict behavior Ø Help us form mental pictures of processes (models)

Ø Another Observations Hypothesis Experiment 15 outcome is that certain behavior is repeated many times Ø Scientific Law is developed Ø Description of how things behave Ø Usually an equation Ø Law - how Ø Theory- why

Observations Hypothesis Theory (Model) Modify Experiment Prediction Law 16 Experiment

Variables Ø Controlled experiment- Only want one thing to change at a time in a laboratory. Ø Independent variable- What you change or control directly. Ø Dependent variable – What changes as a result of changing the independent variable. No direct control. 17

References Ø Ø Dingranso, L, Gregg, K, Hainen, N, & Wistrom, C (2002). Chemistry: matter and change. Columbus, OH: Glencoe/Mc. Graw-Hill. Green, N. D. , Mr. Green’s Home Page. Retrieved October 17, 2008 from http: //www. tvgreen. com/index. htm 18