Chapter 1 Chemistry Organic Analytical Biochem chemistry of

Chapter 1
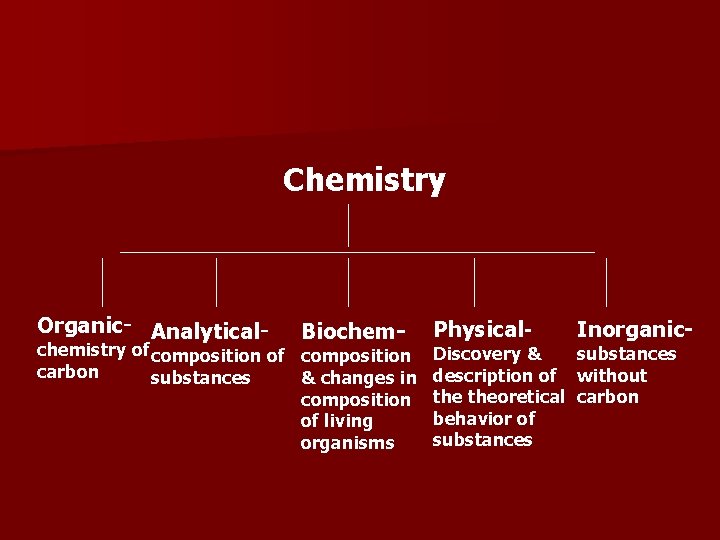
Chemistry Organic- Analytical- Biochem- chemistry of composition carbon substances & changes in composition of living organisms Physical- Inorganic- Discovery & substances description of without theoretical carbon behavior of substances
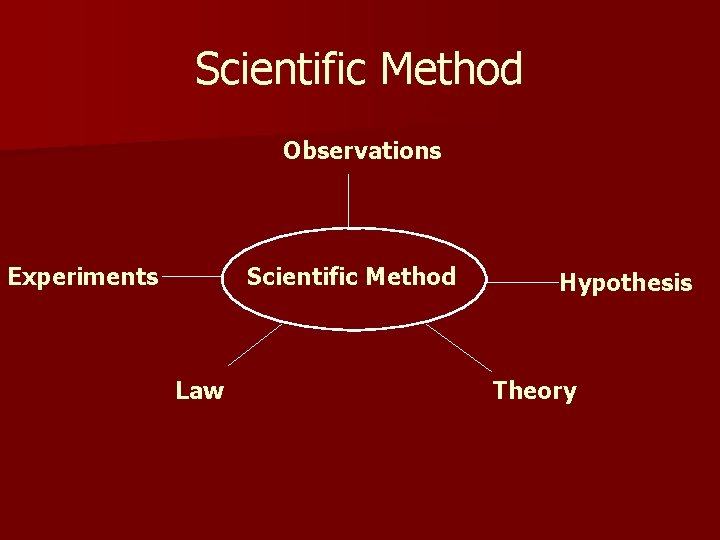
Scientific Method Observations Experiments Scientific Method Law Hypothesis Theory

Observations – recorded facts Hypothesis – descriptive models for observations Refine Experiments – controlled conditions to test hypothesis Theory - A thoroughly tested model that explains why experiments give certain results Observations Experiments Law – a concise statement that summarizes results of a variety of observations

Law vs. Theory Describes experimental observations Explains model – withstands test of time

• Matter- Takes up space and has mass • Substance- a kind of matter that has a uniform and definite composition Examples: sugar, salt 100% pure substance Is ice cream a substance?

Physical Property a quality or condition of a substance that can be observed without changing the substances condition Examples: melting point, boiling point, color, odor, density

Solid • has definite shape and volume; not compressible Liquid • flows, has a fixed volume and takes shape of the container; rarely compressible

Gas • takes shape and volume of its container; is compressible Examples: Nitrogen (N 2), Ammonia (NH 3)

Vapor • a substance, although in its gaseous state, is generally a liquid or solid at room temperature Is steam a vapor?

PHYSICAL CHANGE Will alter a substance without changing its composition

Example-H 2 O Solid, liquid, or gas Possibilities: boil, freeze, melt, condense, break, split, crack, grind, cut, crush, bend

Mixtures Physical blend of two or more substances Examples Meat Sauce -Ground beef & tomatoes Air -Mixture of gases Lemonade -Sugar, lemon juice & H 2 O

Heterogeneous Not uniform in composition Example Soil

Homogeneous Completely uniform in composition Examples Milk Sugar Water

Solutions A homogeneous mixture Gases- Air Liquids- Vinegar Solids- Sterling Silver

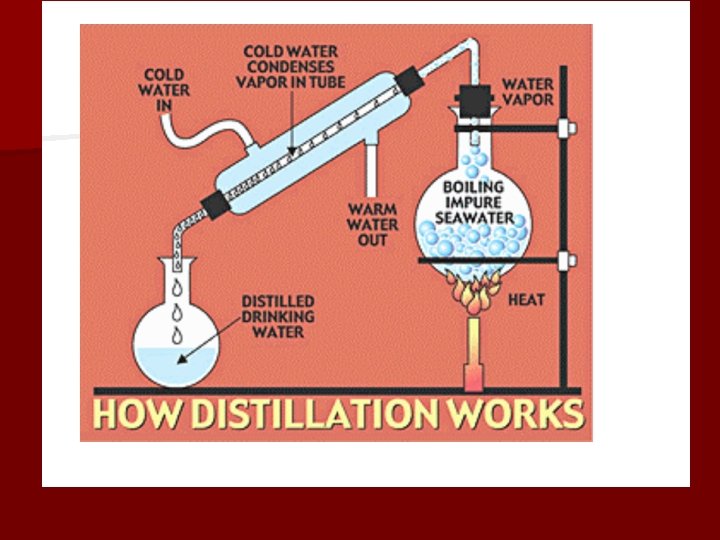
Distillation • A method of separating a mixture into its pure components • Used with liquids by producing a gas and then condensing it


Elements Simplest form of matter that can exist under normal laboratory conditions Example Ag Au H N

Compounds Substances that can be separated into simpler substances only by chemical reactions

Examples Na. Cl Na + Cl H 2 O H+O

Chemical Symbol Each element is represented by a chemical symbol Examples Ag-Silver Na-Sodium Ne-Neon

Energy-Capacity for doing work Forms of Energy Chemical Nuclear Radiant Mechanical Electrical Thermal

Potential Energy-Energy of Position (Stored) Kinetic Energy-Energy of Motion (Converted from PE) Heat-Energy that is transferred from one body to another because of a temperature difference

Law of Conservation of Energy In any chemical or physical process, energy is neither created nor destroyed

Chemical Reaction One or more substances are changed into new substances
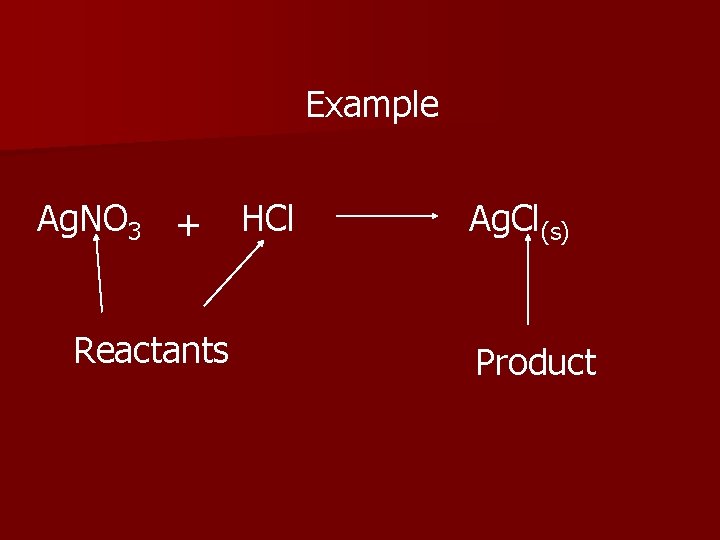
Example Ag. NO 3 + Reactants HCl Ag. Cl(s) Product

Chemical Properties Ability of a substance to undergo chemical reactions and form new substances
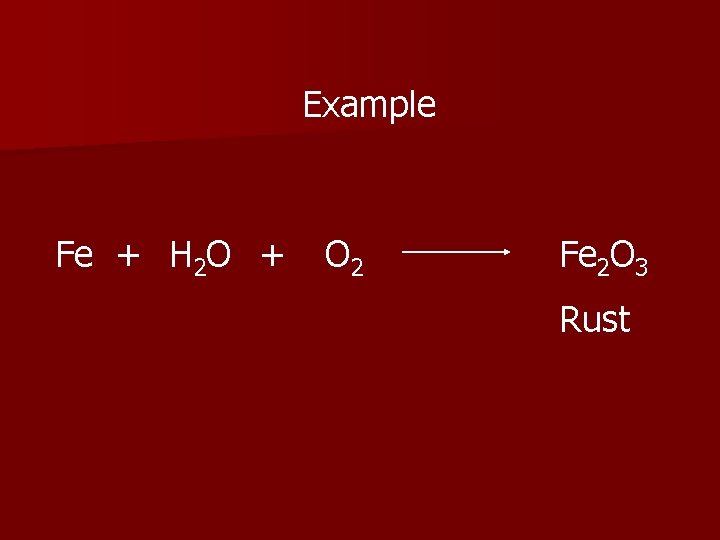
Example Fe + H 2 O + O 2 Fe 2 O 3 Rust

Law of Conservation of Mass In any physical or chemical reaction, mass is neither created nor destroyed Example 10 g of ice 10 g water
- Slides: 31