Chapter 1 Chemical Bonding and Chemical Structure Organic
Chapter 1 Chemical Bonding and Chemical Structure
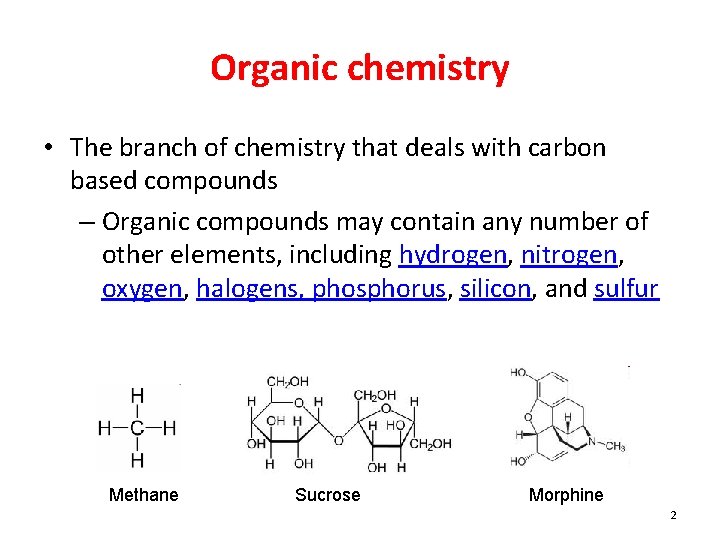
Organic chemistry • The branch of chemistry that deals with carbon based compounds – Organic compounds may contain any number of other elements, including hydrogen, nitrogen, oxygen, halogens, phosphorus, silicon, and sulfur Methane Sucrose Morphine 2

History • Vitalism: Only biological systems (e. g. , plants, animals) could produce organic compounds • Wohler’s synthesis of urea (1828), began to undermine vitalism

Why Study Organic Chemistry? • Organic chemistry lies at the heart of the modern chemical industry • Central to medicine and pharmacy • Interface of physical and biological sciences • Everyday applications: Plastics, textiles, communications, transportation, food, clothing, cosmetics, etc. 4
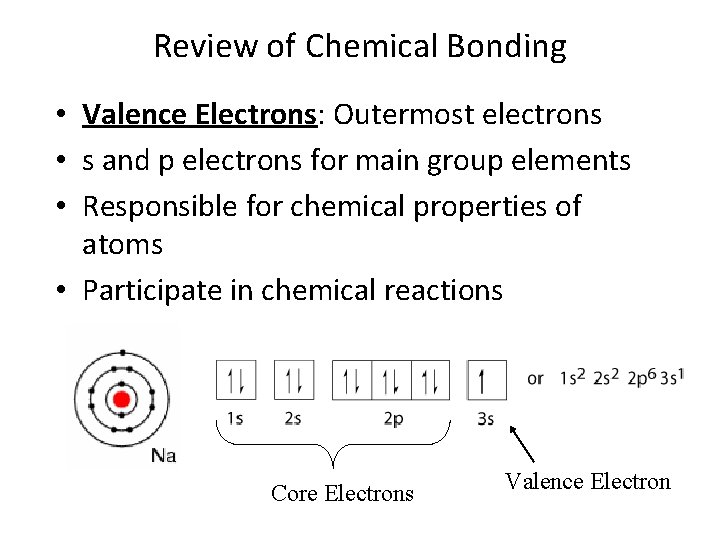
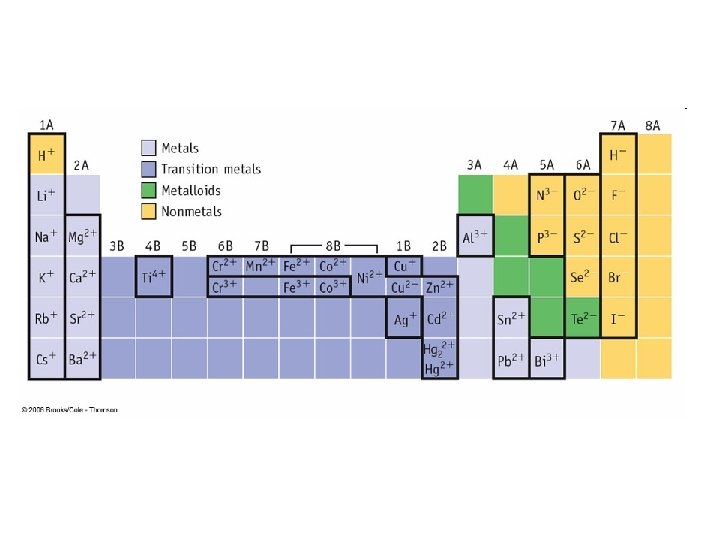
Review of Chemical Bonding • Valence Electrons: Outermost electrons • s and p electrons for main group elements • Responsible for chemical properties of atoms • Participate in chemical reactions Core Electrons Valence Electron

Octet Rule • Octet Rule: the tendency for atoms to seek 8 electrons in their outer shells – Natural electron configuration of the Noble Gases – Done by gaining, losing, or sharing electrons – Increases stability – H and He seek a “Duet”

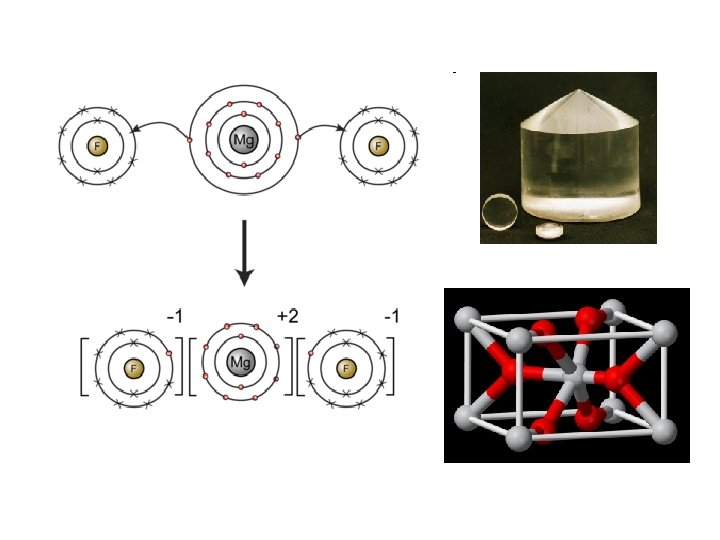
Ionic Bonding • Ions: atoms that have a charge due to gain or loss of electrons – Anion: (-) charged atom – Cation: (+) charged atom • Ionic Bond: a bond formed through the transfer of one or more electrons from one atom or group of atoms to another atom or group of atoms
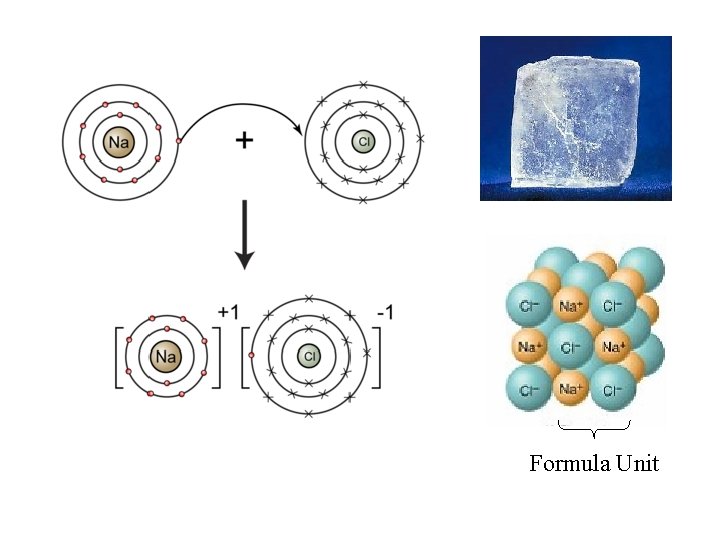
Formula Unit
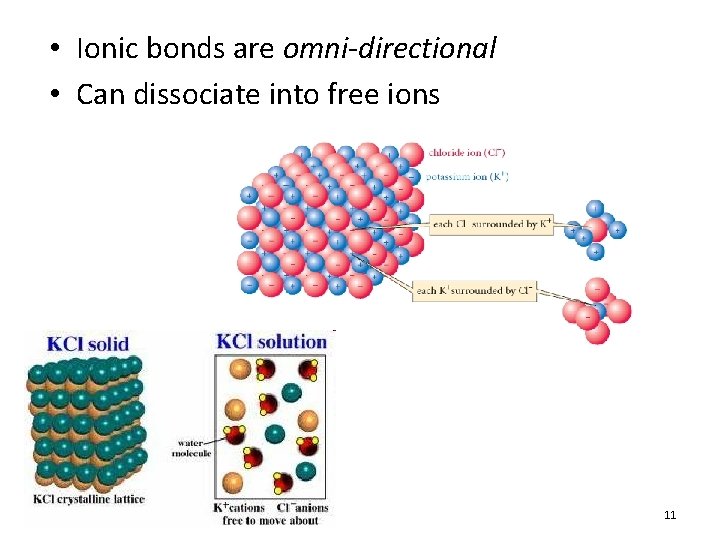
• Ionic bonds are omni-directional • Can dissociate into free ions 11

Covalent Compounds • Covalent Compounds: compounds composed of atoms bonded to each other through the sharing of electrons • Electrons NOT transferred • No + or – charges on atoms • Non-metal + Non-metal • Also called “molecules” • Examples: – H 2 O – CO 2 – Cl 2 – CH 4
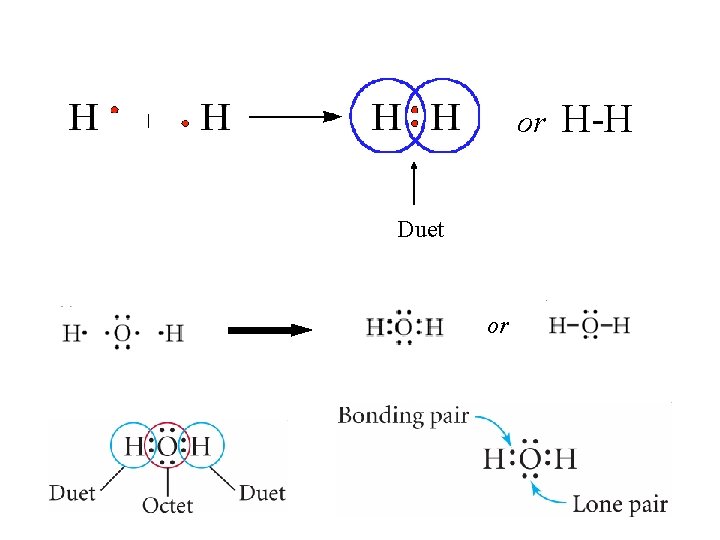
or Duet or H-H
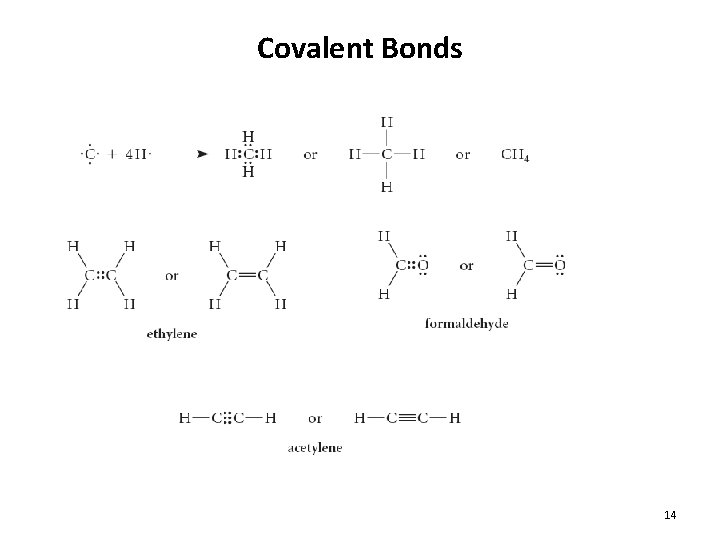
Covalent Bonds 14

Electronegativity • The measure of the ability of an atom to attract electrons to itself – Increases across period (left to right) and – Decreases down group (top to bottom) – fluorine is the most electronegative element – francium is the least electronegative element
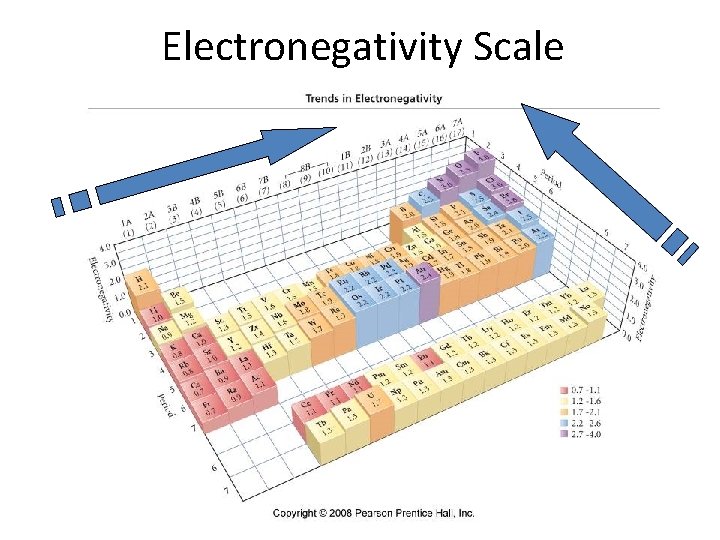
Electronegativity Scale

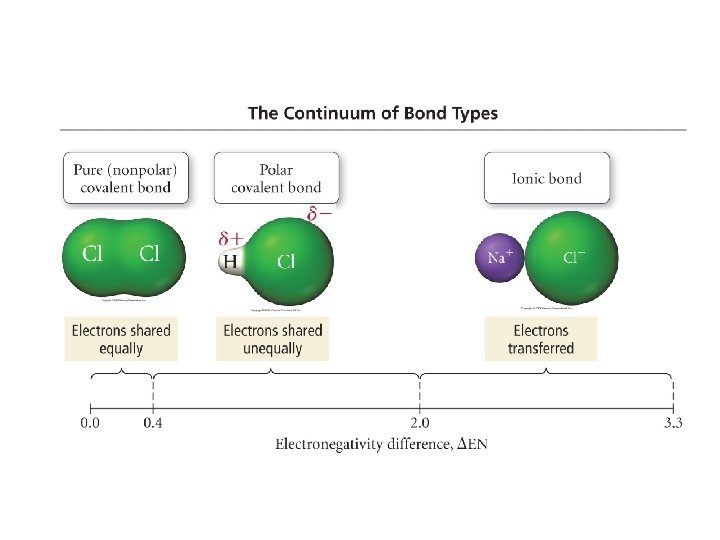
Types of Bonding 1) Non-Polar Covalent Bond: • Difference in electronegativity values of atoms is 0. 0 – 0. 4 • Electrons in molecule are equally shared • Examples: Cl 2, H 2, CH 4 ENCl = 3. 0 - 3. 0 = 0 Pure Covalent

2) Polar Covalent Bond: • Difference in electronegativity values of atoms is 0. 4 – 1. 7/2. 0 • Electrons in the molecule are not equally shared • • The atom with the higher EN value pulls the electron cloud towards itself Partial charges • Examples: HCl, Cl. F, NO ENCl = 3. 0 ENH = 2. 1 3. 0 – 2. 1 = 0. 9 Polar Covalent
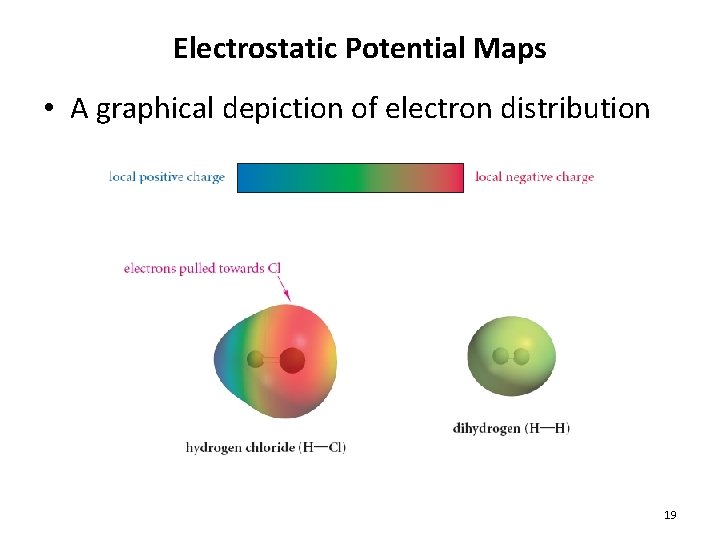
Electrostatic Potential Maps • A graphical depiction of electron distribution 19

3) Ionic Bond: • Difference in EN above 1. 7 -2. 0 • Complete transfer of electron(s) • Whole charges ENCl = 3. 0 ENNa = 1. 0 3. 0 – 0. 9 = 2. 1 Ionic
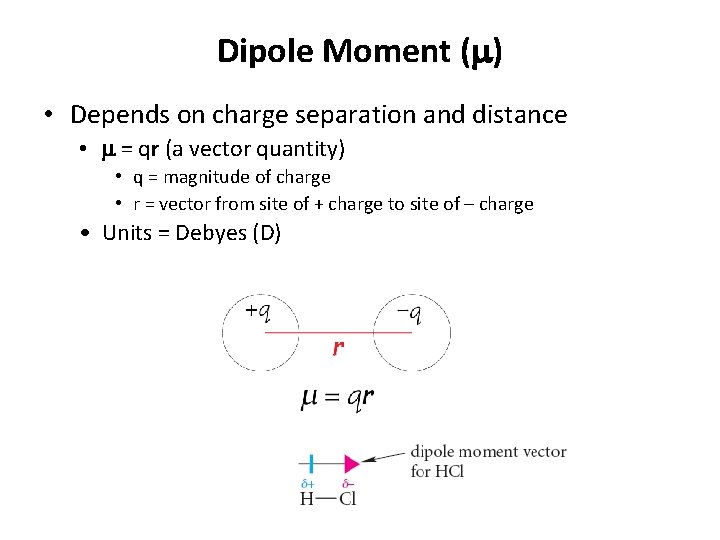
Dipole Moment (m) • Depends on charge separation and distance • m = qr (a vector quantity) • q = magnitude of charge • r = vector from site of + charge to site of – charge • Units = Debyes (D)
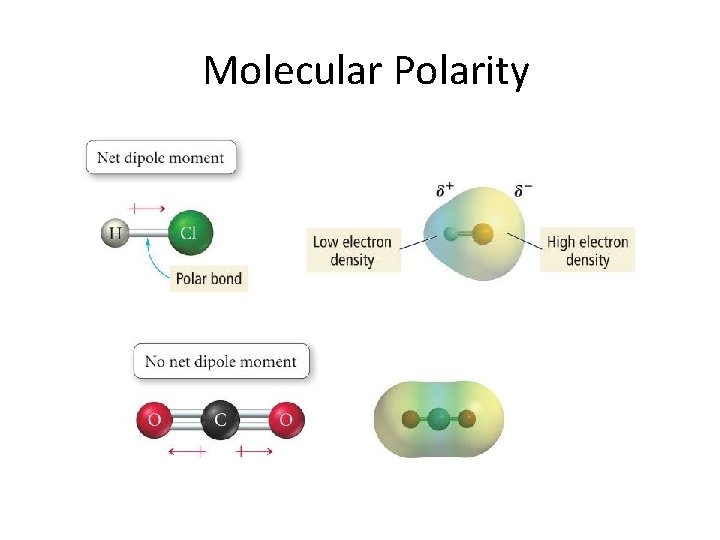
Molecular Polarity
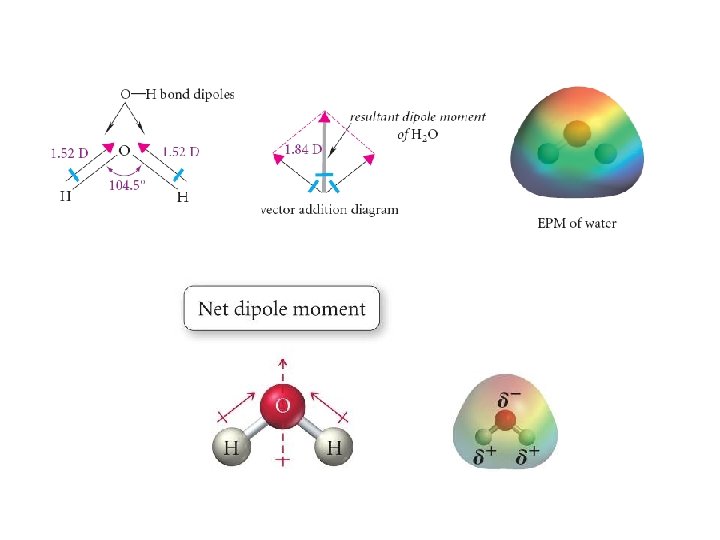
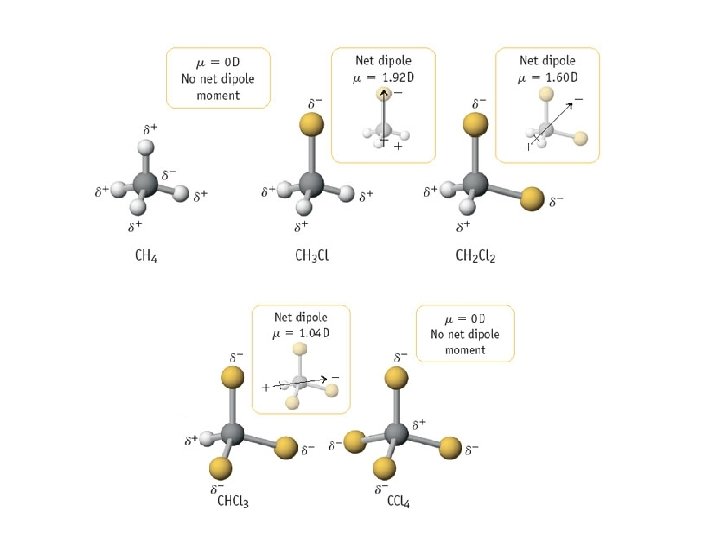

Lewis Dot Structures 1) Count the number of valence electrons present in the molecule 2) Determine the arrangement of atoms. Generally, the atom that occurs least often is central. Join the terminal atoms to the central atom(s) using shared pairs of electrons (bonds) 3) Place any remaining electrons around the terminal atoms to satisfy the octet rule • Exception: Hydrogen 4) Place any remaining electrons on the central atom(s) to satisfy the octet rule

5) Check to make sure: • You’ve used the correct number of valence electrons • Everyone has an octet (or duet) • Everyone is doing what they like to do 6) If the number of electrons around the central atom is less than 3, change the single bonds to multiple bonds
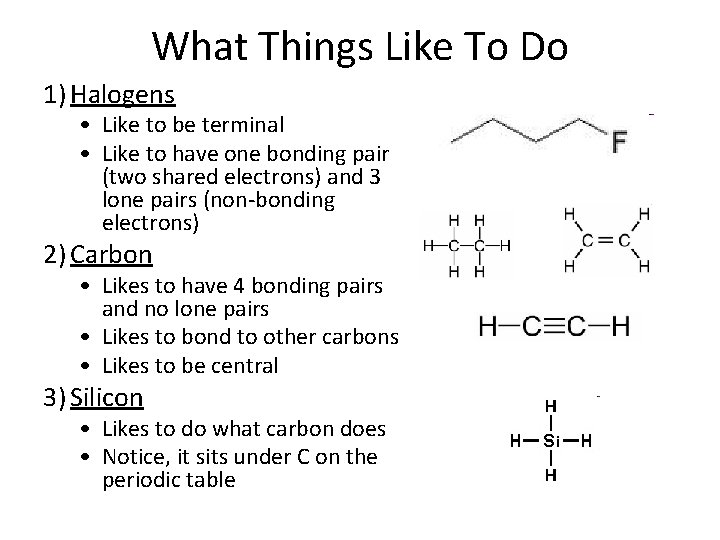
What Things Like To Do 1) Halogens • Like to be terminal • Like to have one bonding pair (two shared electrons) and 3 lone pairs (non-bonding electrons) 2) Carbon • Likes to have 4 bonding pairs and no lone pairs • Likes to bond to other carbons • Likes to be central 3) Silicon • Likes to do what carbon does • Notice, it sits under C on the periodic table
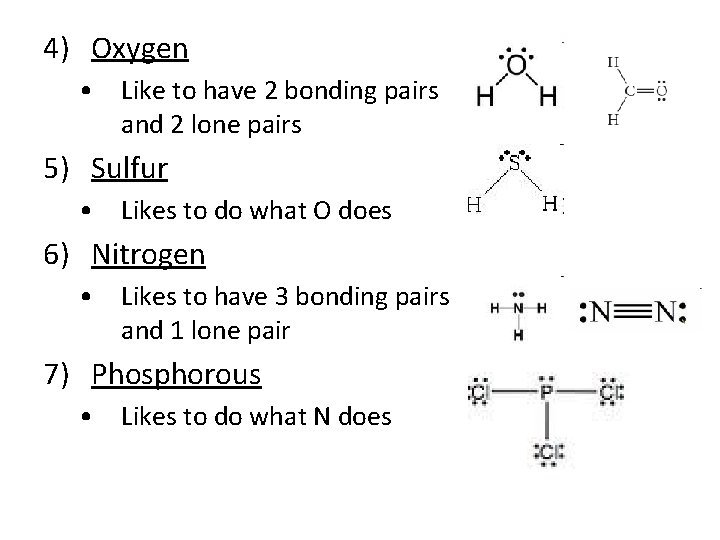
4) Oxygen • Like to have 2 bonding pairs and 2 lone pairs 5) Sulfur • Likes to do what O does 6) Nitrogen • Likes to have 3 bonding pairs and 1 lone pair 7) Phosphorous • Likes to do what N does
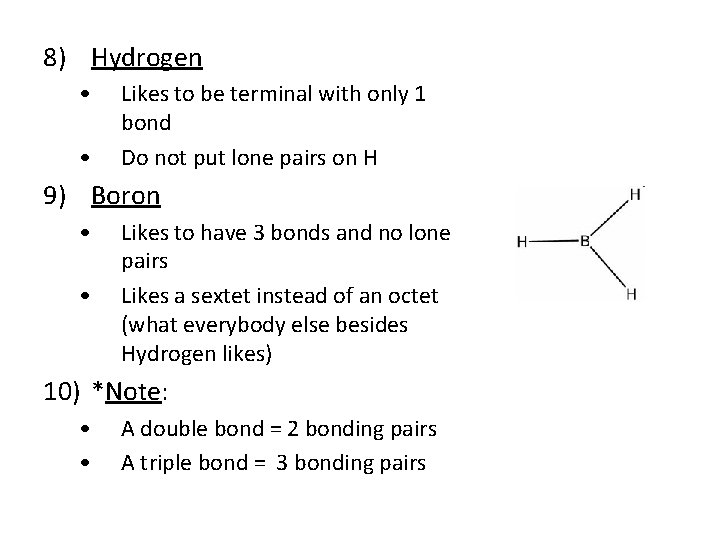
8) Hydrogen • • Likes to be terminal with only 1 bond Do not put lone pairs on H 9) Boron • • Likes to have 3 bonds and no lone pairs Likes a sextet instead of an octet (what everybody else besides Hydrogen likes) 10) *Note: • • A double bond = 2 bonding pairs A triple bond = 3 bonding pairs

Problems • Draw the Lewis Dot Structures for the following molecules 1) 2) 3) 4) CO 2 P 2 H 4 O 3 NO 3 -
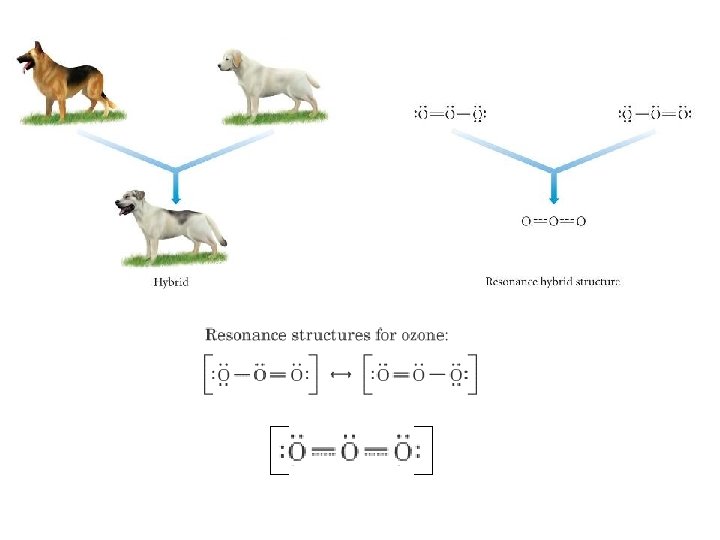
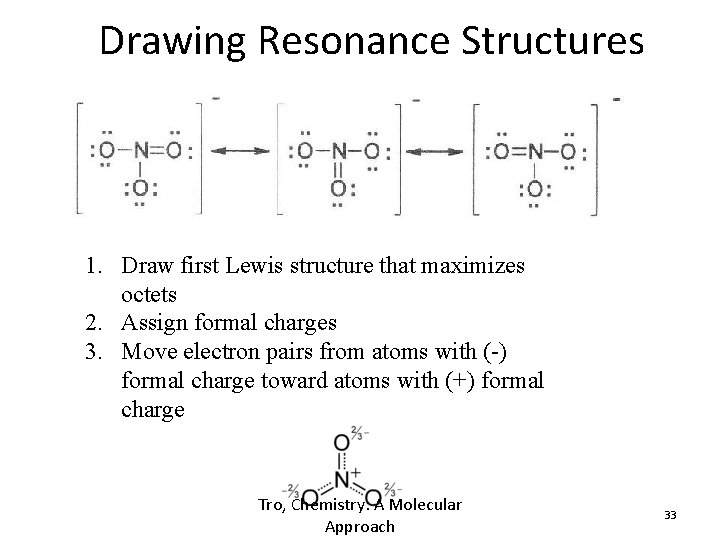
Drawing Resonance Structures -1 -1 1. Draw first Lewis structure that maximizes octets 2. Assign formal charges 3. Move electron pairs from atoms with (-) formal charge toward atoms with (+) formal charge Tro, Chemistry: A Molecular Approach 33

Formal Charge • Assigned charge for each atom in a molecule/ion – Electronic bookkeeping – may or may not correspond to a real charge – Sum of formal charges on each atom must equal the total charge on the molecule/ion • FC = Valence e-’s – Lone Pair e-’s – ½ bonding e-’s
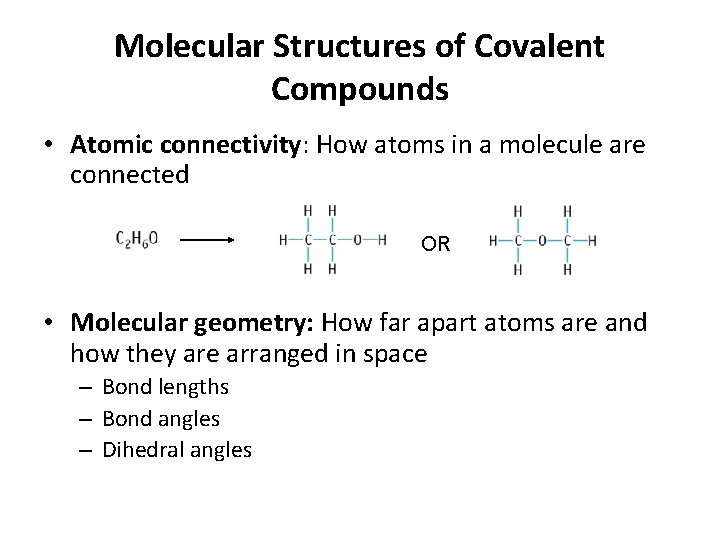
Molecular Structures of Covalent Compounds • Atomic connectivity: How atoms in a molecule are connected OR • Molecular geometry: How far apart atoms are and how they are arranged in space – Bond lengths – Bond angles – Dihedral angles
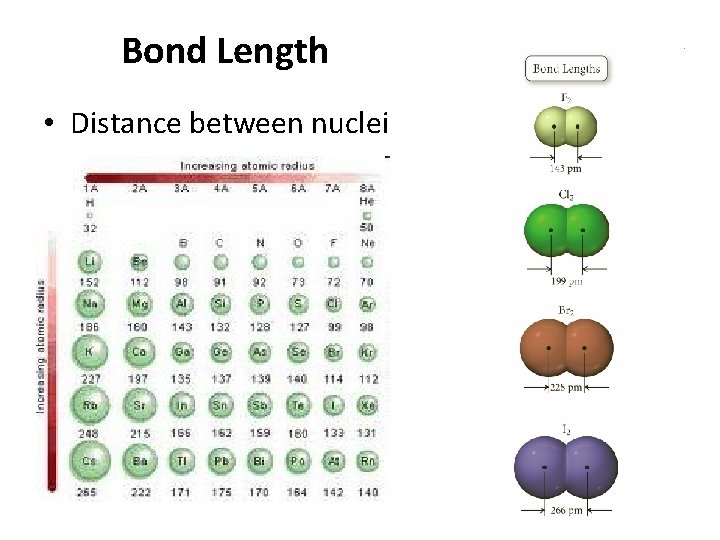
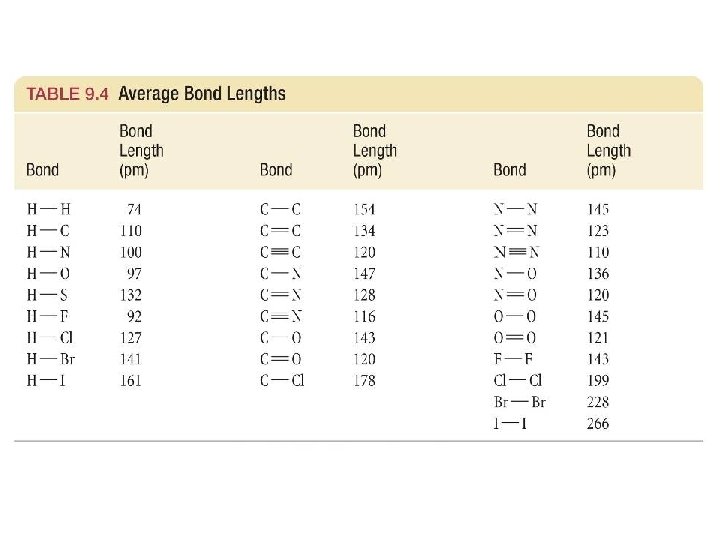
Bond Length • Distance between nuclei 36
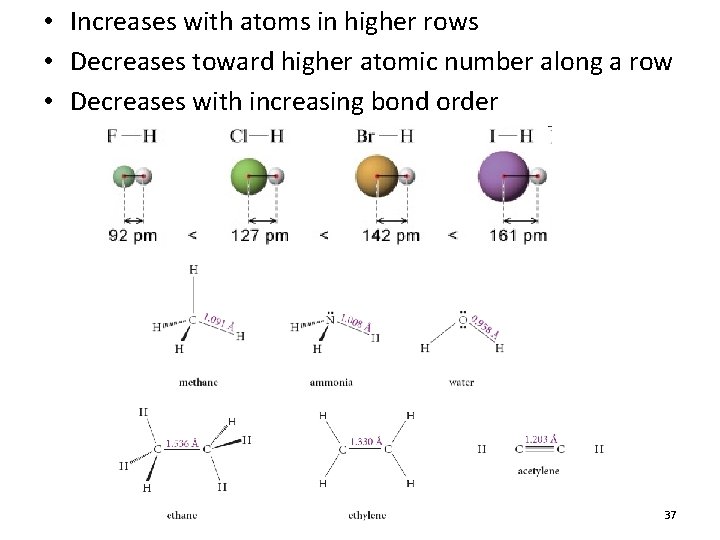
• Increases with atoms in higher rows • Decreases toward higher atomic number along a row • Decreases with increasing bond order 37
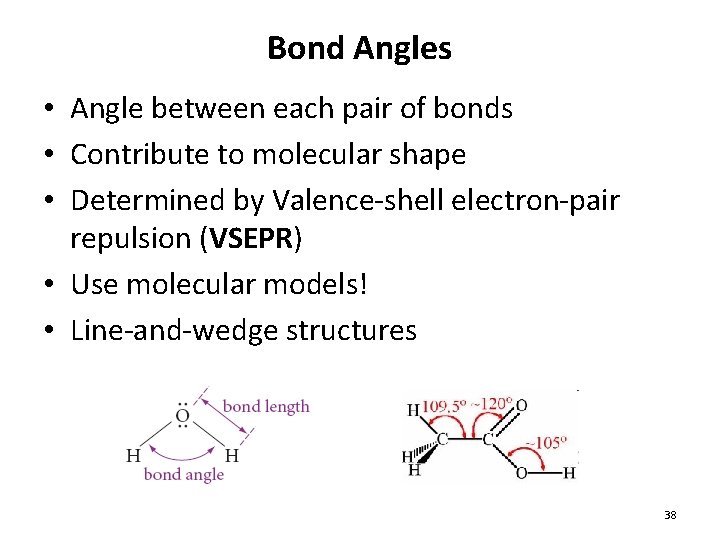
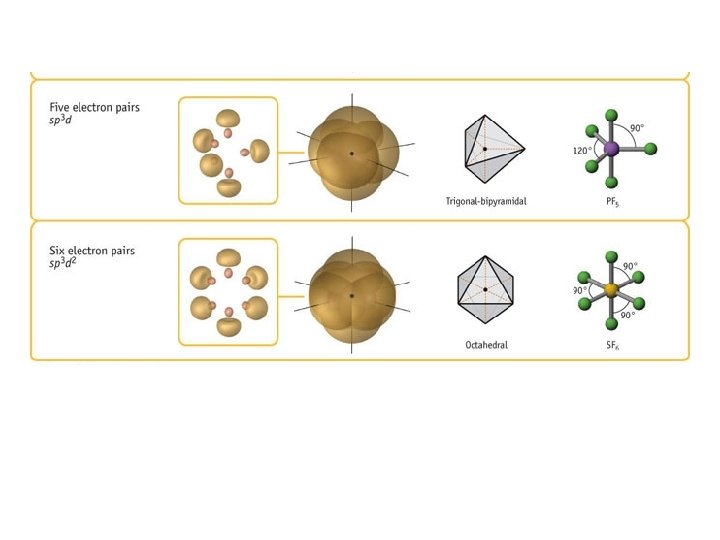
Bond Angles • Angle between each pair of bonds • Contribute to molecular shape • Determined by Valence-shell electron-pair repulsion (VSEPR) • Use molecular models! • Line-and-wedge structures 38
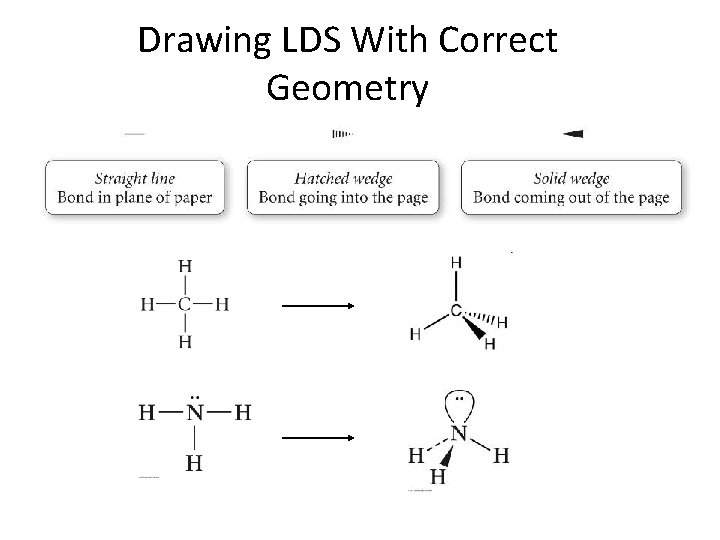
Drawing LDS With Correct Geometry

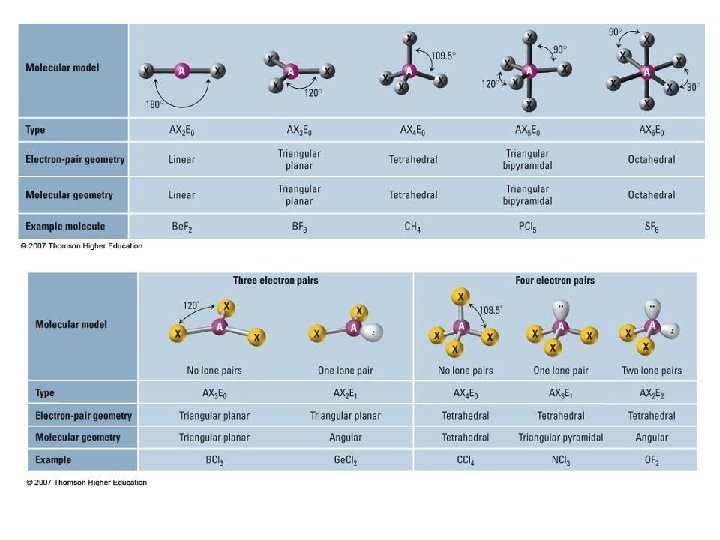
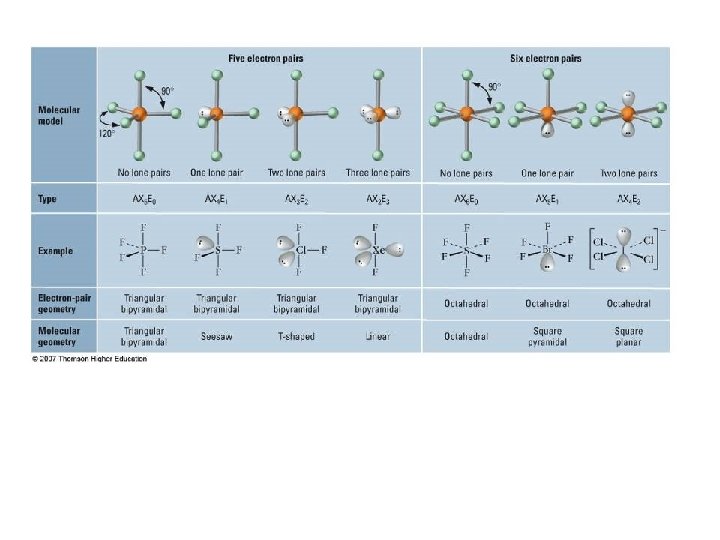
Valence Shell Electron Pair Repulsion Theory • VSEPR theory: – Electrons repel each other – Electrons arrange in a molecule themselves so as to be as far apart as possible • Minimize repulsion • Determines molecular geometry
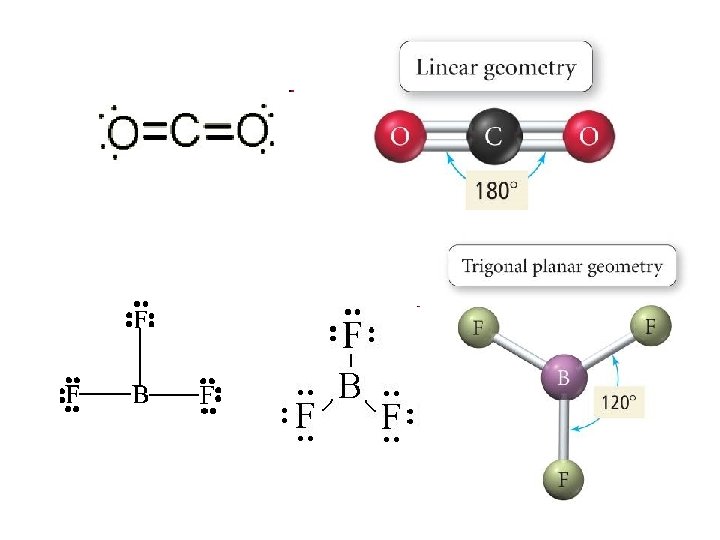
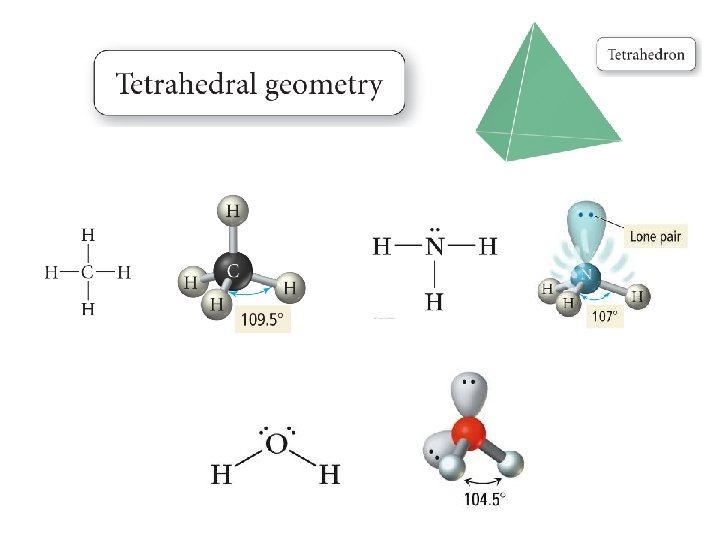
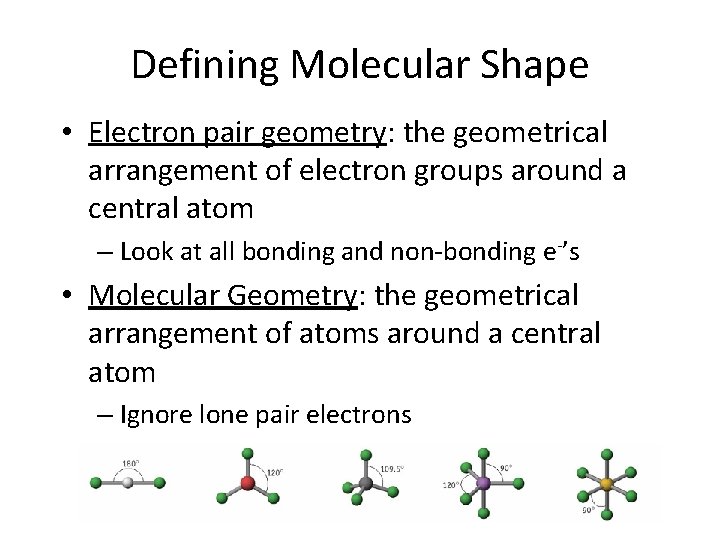
Defining Molecular Shape • Electron pair geometry: the geometrical arrangement of electron groups around a central atom – Look at all bonding and non-bonding e-’s • Molecular Geometry: the geometrical arrangement of atoms around a central atom – Ignore lone pair electrons
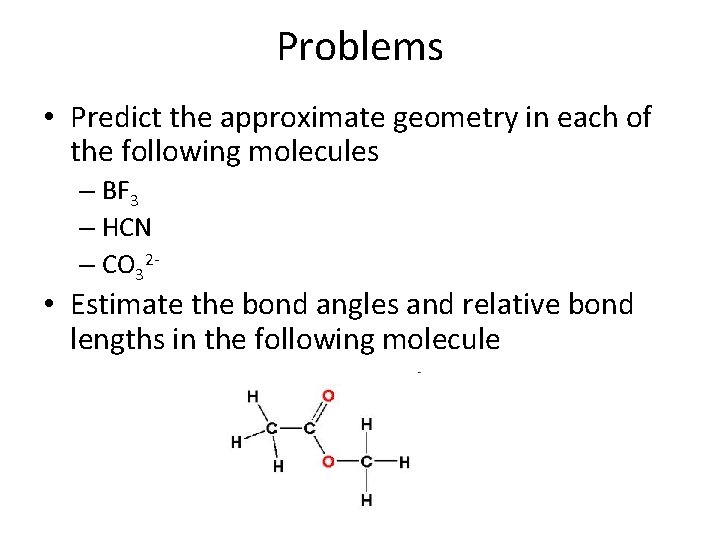
Problems • Predict the approximate geometry in each of the following molecules – BF 3 – HCN – CO 32 - • Estimate the bond angles and relative bond lengths in the following molecule
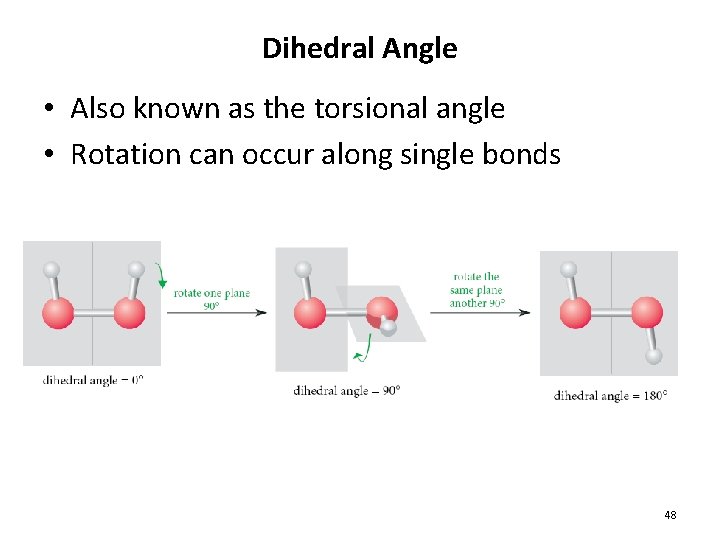
Dihedral Angle • Also known as the torsional angle • Rotation can occur along single bonds 48
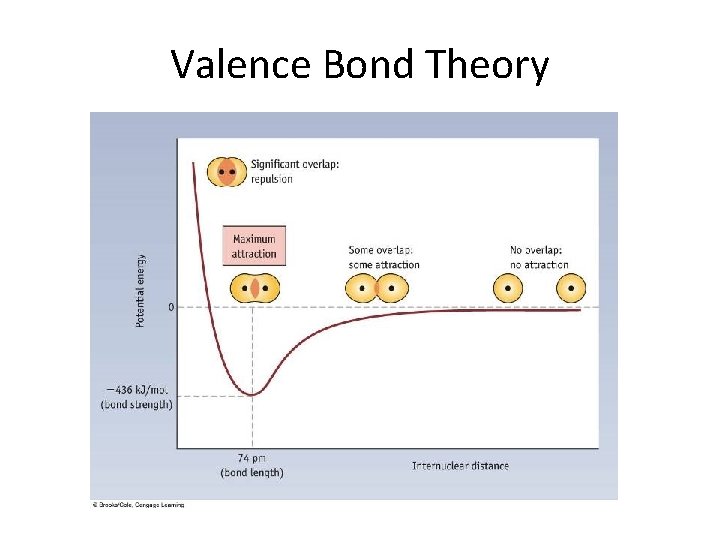
Valence Bond Theory
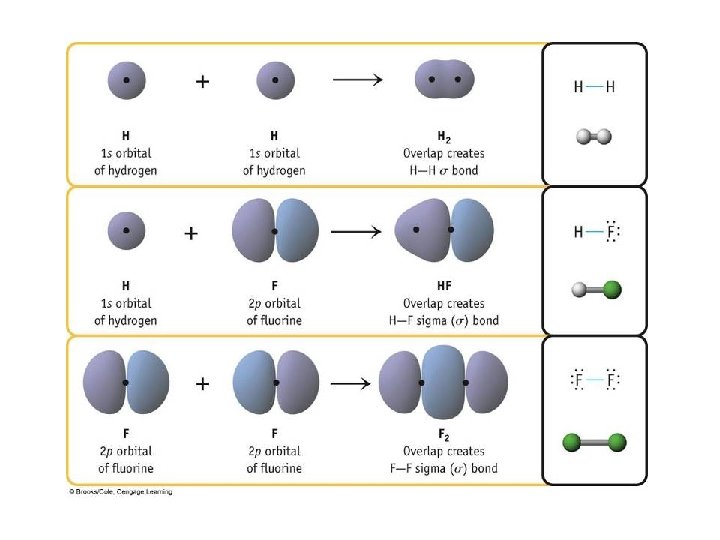
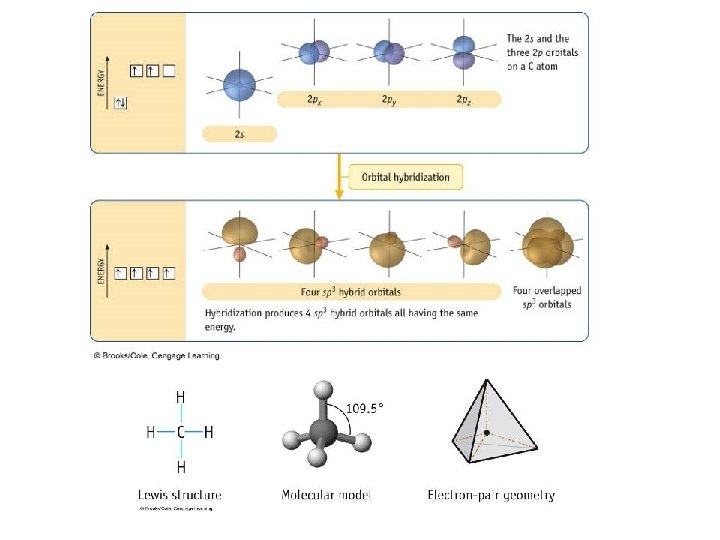
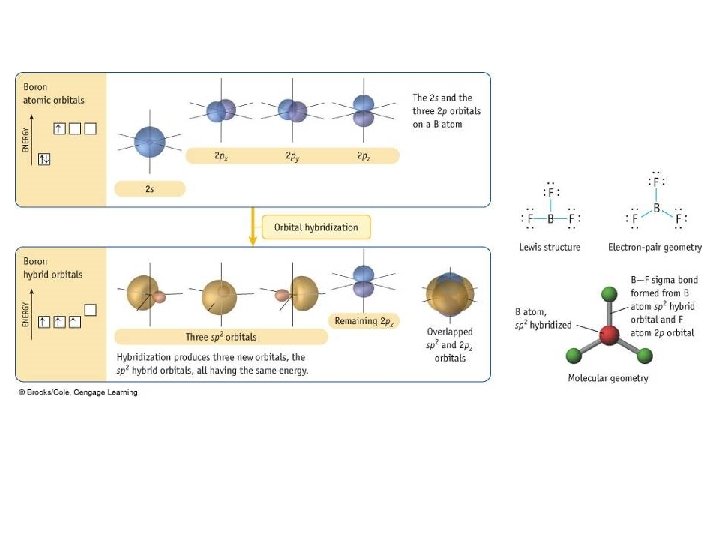
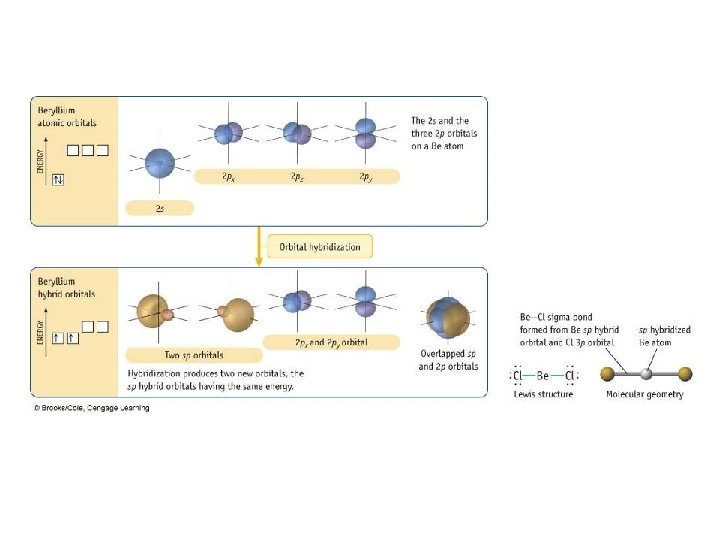
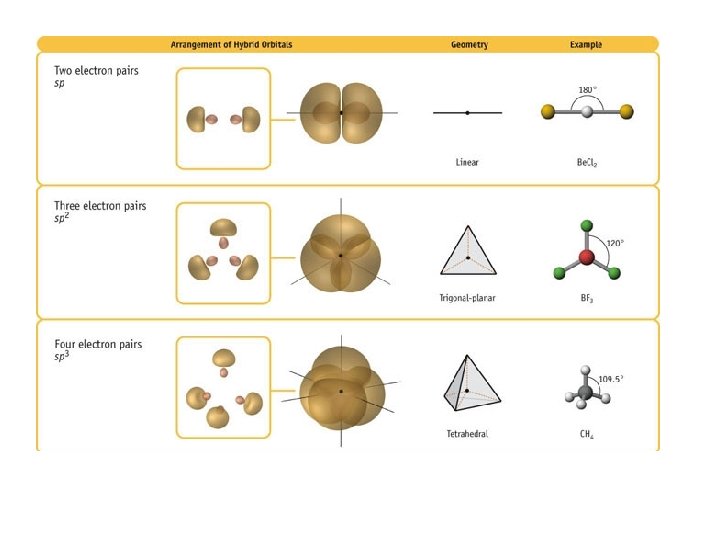

Types of Bonds • A sigma (s) bond results when the bonding orbitals point along the axis connecting the two bonding nuclei – either standard atomic orbitals or hybrids • s-to-s, p-to-p, hybrid-to-hybrid, s-to-hybrid, etc. • A pi (p) bond results when the bonding orbitals are parallel to each other and perpendicular to the axis connecting the two bonding nuclei – between unhybridized parallel p orbitals • the interaction between parallel orbitals is not as strong as between orbitals that point at each other; therefore s bonds are stronger than p bonds
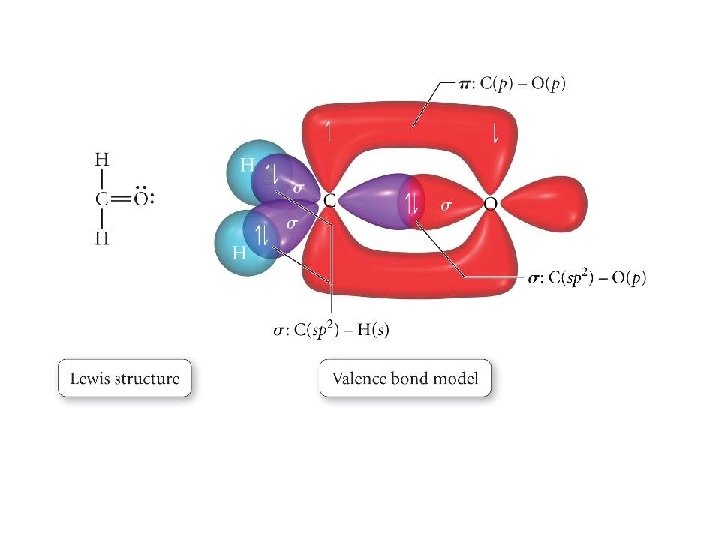
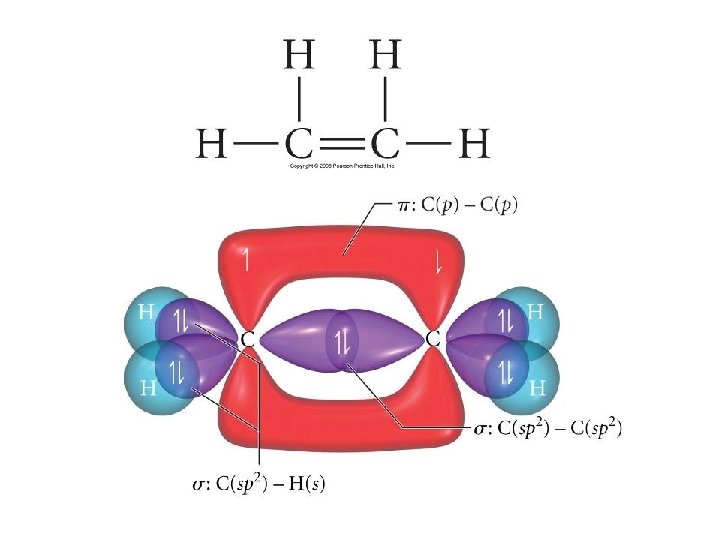

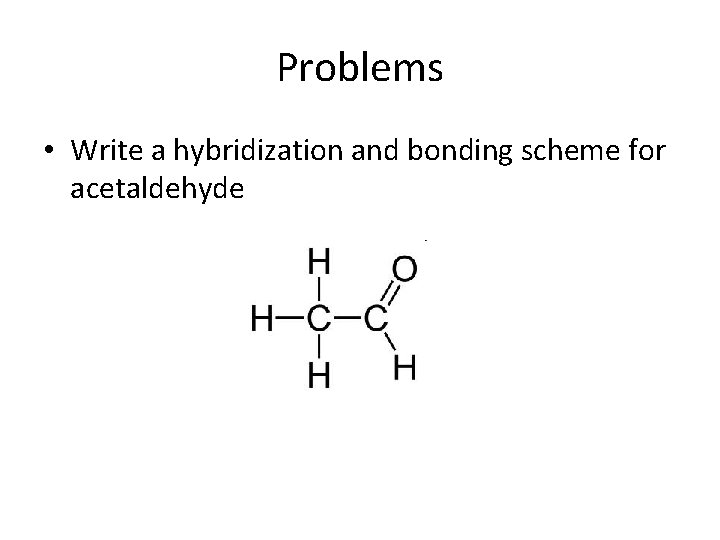
Problems • Write a hybridization and bonding scheme for acetaldehyde
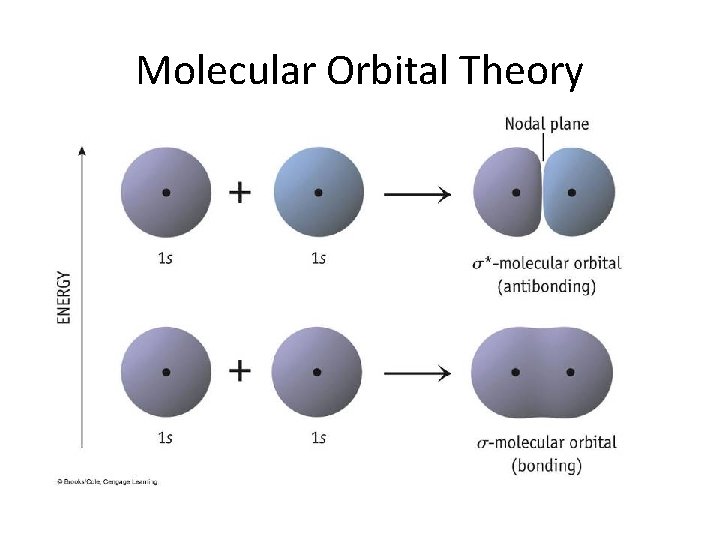
Molecular Orbital Theory
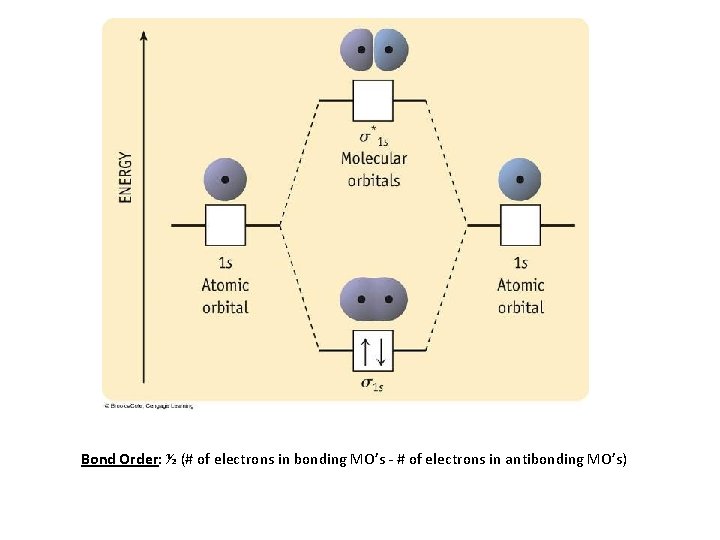
Bond Order: ½ (# of electrons in bonding MO’s - # of electrons in antibonding MO’s)
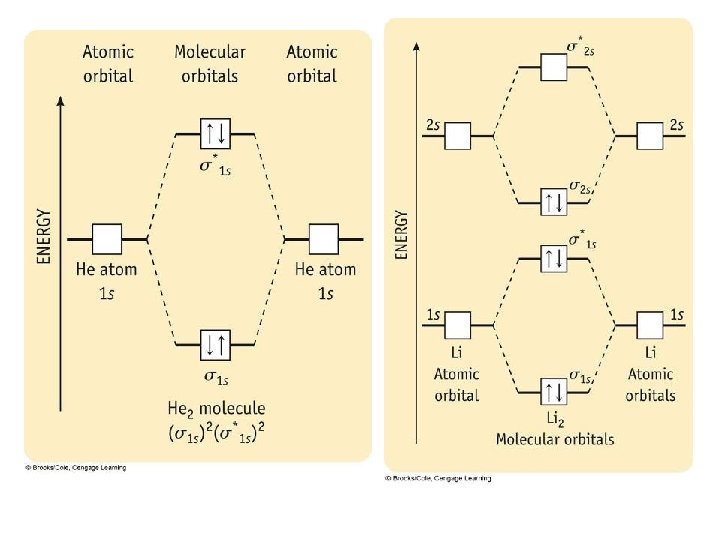
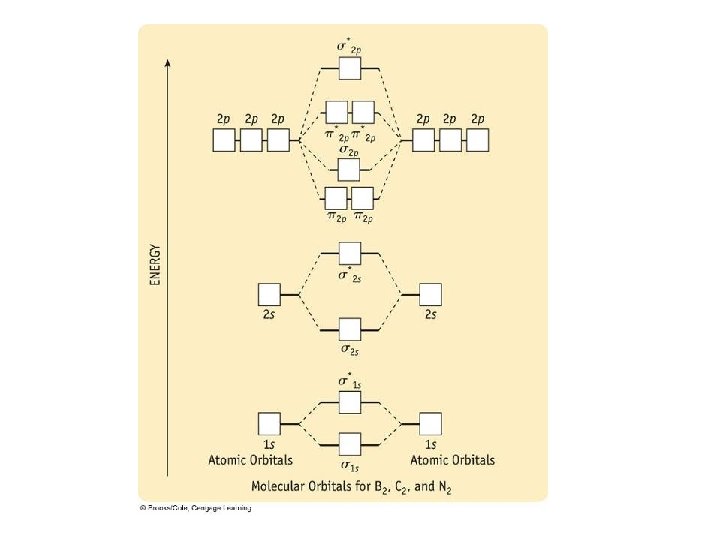

Problems 1) Draw an MO diagram to predict the bond order of N 2 2) Draw an MO diagram to predict the bond order of CN 3) Use MO theory to determine the bond order of Ne 2
- Slides: 65