Chapter 1 Basic Concepts About Matter Chemistry the

Chapter 1 Basic Concepts About Matter • Chemistry = the study of the composition, structure, properties and reactions of matter. • Matter = anything that has mass and occupies space (all the substances that make up our world). • Chemistry is a science that requires us to deal with all the stuff that we see around us. Chemistry happens all around you every day.

States of Matter Shape: Volume: Particles: Interaction: Movement:

Properties of Matter: distinguishing characteristics that help us to identify a substance Physical Properties • can be determined without changing the composition of substances

Chemical Properties • Describe the way a substance undergoes change (or resists change) to form a new substance.

Physical Changes – the state or appearance may change, but the composition remains the same A Change in State is a Physical Change: • Melting & Freezing • Evaporation, Boiling, Condensation • Sublimation & Deposition

Other examples of Physical Change: • Cutting • Dissolving (creating a mixture? ) • Changing Shape

Chemical Changes – the original substance is converted into a new substance, which has different physical & chemical properties Examples of Chemical Change: • Rusting (Corrosion) • Combustion • Decompostion • Fermentation http: //www. youtube. com/watch? v=5 DKs. E 1 Ue 9 UM

Is it a Physical Change or a Chemical Change? An ice cube melts Paper is cut into pieces Sugar dissolves in water Silver becomes tarnished Wet clothes dry on a clothesline Milk turns sour and becomes curdled when heated • wood is burned in a campfire • • •

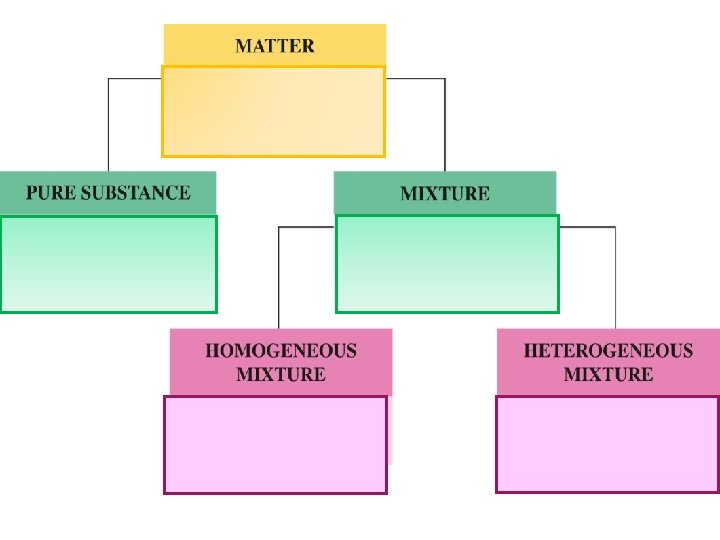
Classification of Matter 2 Types of Pure Substances Elements: The simplest type of pure substance Composed of only one kind of atom Cannot be broken down chemically Designated by symbols Compounds: Composed of 2 or more elements Atoms of different types Held together by bonds Can be broken down chemically Cannot be broken down physically Chemical formula

Some Elements can be found free in nature

Some Common Compounds

Mixtures: • • 2 or more pure substances Each substance retains its identity Variable composition 2 types of mixtures: • Homogeneous = 2 or more substances in one phase (also called a solution) • Heterogeneous = 2 or more substances in 2 or more phases

• Can be separated by physical means

Is it an Element? Compound? Homogeneous Heterogeneous Mixture? Dry ice Orange juice Diamond Brass Water Quartz Tap water Sucrose Fruit salad Room air Diet Coke on ice Blood Milk Table salt Salt water Gasoline Iron Baking soda
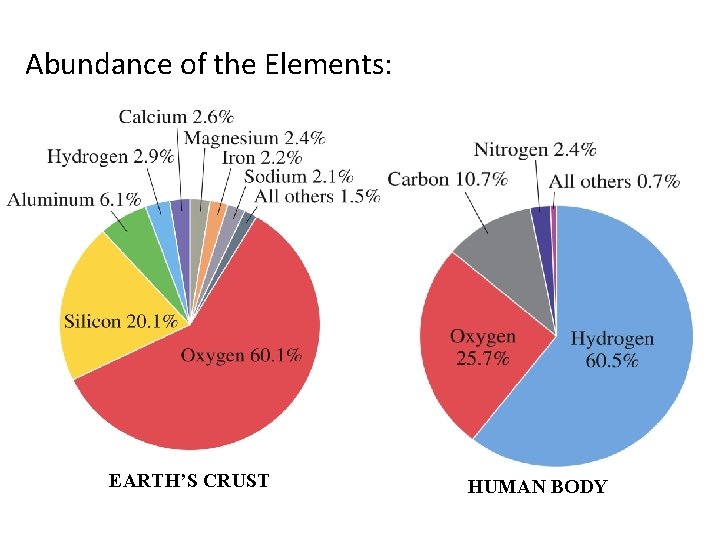
Abundance of the Elements: EARTH’S CRUST HUMAN BODY
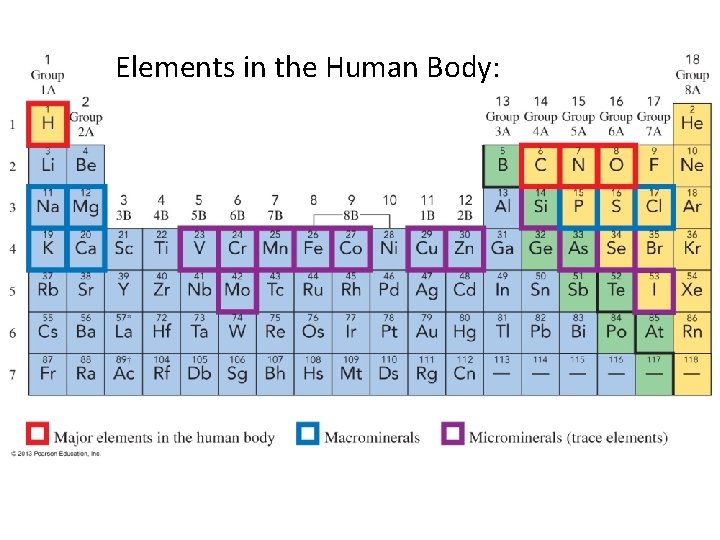
Elements in the Human Body:

Names and Symbols of the Elements: • Chemical Symbols: 1 - or 2 -letter designation for an element • Symbols are derived from the element’s name • a few are from Latin/German names • 118 known elements • 88 occur naturally—most as part of a compound • 30 have been made in the laboratory

Atoms and Molecules: Atom = the smallest particle of an element that can exist and still have the properties of the element. Pure Element = a substance composed of atoms of a single type. Molecule = a group of 2 or more atoms bound together to form a unit. Atoms in a molecule may be of the same element or of different elements. Compound = 2 or more different elements chemically combined.
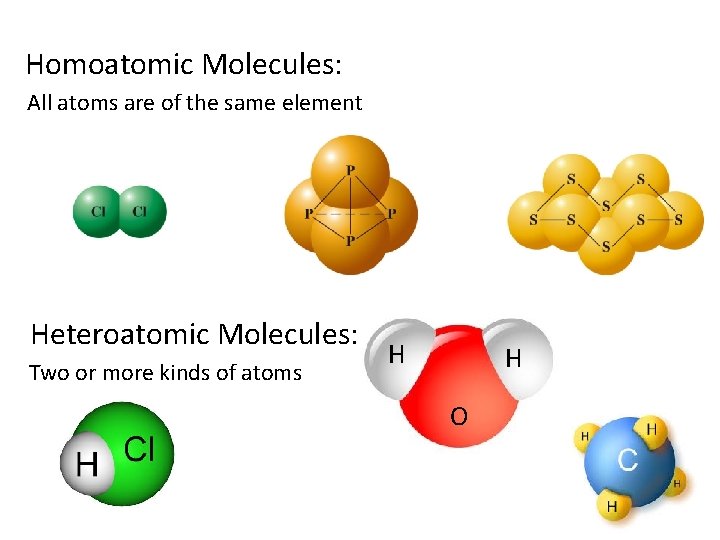
Homoatomic Molecules: All atoms are of the same element Heteroatomic Molecules: Two or more kinds of atoms H H O
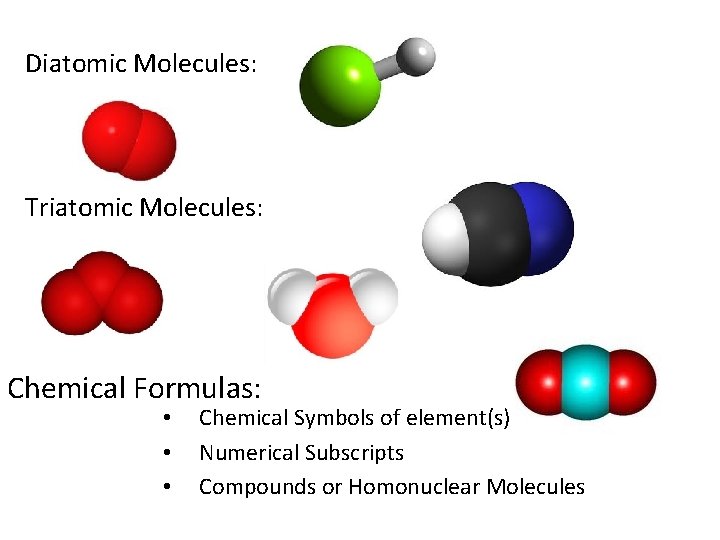
Diatomic Molecules: Triatomic Molecules: Chemical Formulas: • • • Chemical Symbols of element(s) Numerical Subscripts Compounds or Homonuclear Molecules
![[Not all compounds are “molecules”] Chemical Formulas of Ionic Compounds: • • Smallest whole [Not all compounds are “molecules”] Chemical Formulas of Ionic Compounds: • • Smallest whole](http://slidetodoc.com/presentation_image_h/741a9d02954c9da98c50391fbee7d8ee/image-22.jpg)
[Not all compounds are “molecules”] Chemical Formulas of Ionic Compounds: • • Smallest whole number ratio of elements in the compound “Formula Unit”

Interpretation of Chemical Formulas: How many atoms of each element are present in one molecule or one formula unit: C 9 H 8 O 4 Al 2(SO 4)3 COCl 2 Co. Cl 2 O 2
- Slides: 23